Abstract
Background:
Kleefstra syndrome (KLEFS1) is a rare neurodevelopmental genetic condition caused by the loss of function of EHMT1. Currently, there are no approved targeted therapies for KLEFS1. Disease Concept Models (DCMs) identify aspects of a condition that may serve as meaningful interventional targets via patient and/or caregiver interviews. This study aimed to produce a KLEFS1-specific DCM for the development of condition-specific interventions.
Methodology:
We conducted semi-structured interviews with 16 caregivers of individuals under 18 years of age with KLEFS1 and 2 healthcare providers (HCPs). Interviews were coded using NVivo and emergent themes were grouped into 4 categories: (1) Defining features of KLEFS1, (2) Individual impacts, (3) Caregiver impacts, and (4) Developmental age groups.
Results:
Defining pediatric KLEFS1 features referenced by the greatest number of caregivers included gross motor skills delayed or impaired (n = 15, 93.8%). Both caregivers and HCPs selected communication as an aspect of health which, if treated, would have a significant, positive impact on quality of life. The most referenced condition-specific impacts on individuals with KLEFS1 included therapies (n = 16, 100%). The most referenced KLEFS1-specific impacts on caregivers included time and organization needed for care (n = 12, 75%). Challenges with self-care skills and socializing were referenced more frequently in older children, while features of physical health were emphasized in younger children.
Conclusions:
The developed pediatric KLEFS1 DCM is a valuable patient-centered conceptualization, providing insight into the lived experiences of affected individuals and caregivers. Challenges with communication, physical mobility, as well as emotional and behavioral regulation were identified as having the most impact on daily life and may serve as targets for KLEFS1-specific interventions. Endpoint measures that assess the success of interventions should prioritize independence with activities of daily living and self-care tasks, as well as social behaviors and skills.
Keywords
Introduction
Kleefstra syndrome (KLEFS1) is a neurodevelopmental disorder (NDD) estimated to impact between 1:25,000 and 1:35 000 individuals globally1. KLEFS1 is caused by a loss of function of euchromatin histone-lysine methyltransferase 1 (EHMT1) due to a subtelomeric chromosomal deletion at 9q34.3 or a pathogenic intragenic EHMT1 variant. 1 Some phenotypic differences have been reported in individuals with deletions versus intragenic variants.2 -6 The majority of KLEFS1 cases are de novo, with a small number of reported inherited cases. 1
Published literature indicates that individuals with KLEFS1 present with a variety of multisystemic clinical phenotypes. Core features include global developmental delay (GDD) or intellectual disability (ID), autism spectrum disorder (ASD), hypotonia, and distinctive facial features.1,2,6 Additional features include speech disorders, epilepsy, neurobehavioral disorders, neuropsychiatric conditions, and congenital defects including cardiac and renal abnormalities. Additionally, a subset of the KLEFS1 population may experience a post-pubertal regression characterized by a triad of significant sleep disruption, emergence/worsening of psychiatric features such as psychosis or catatonia, and loss of self-help and other developmental skills.6 -10 Interventions, such as speech/language therapy and Applied Behavior Analysis (ABA) therapy, can be utilized to manage some KLEFS1 features; however, there are no approved targeted treatments.
The Food and Drug Administration (FDA) guidelines11,12 encourage the necessity of including patient perspectives on disease to define meaningful endpoints for interventional clinical trials. Systematic assessments of drug trials revealed that specific disease treatments do not always prioritize outcomes that are most valuable to affected individuals, and as a result, patients often report treatment outcomes to be of little value to them. 13 To justify the significant investment required to develop new treatments, therapeutic efficacy must be optimized by incorporating patient perspectives in drug development. 14
To date, there is one large survey-based study that utilized reports from 172 KLEFS1 caregivers across 28 countries. 15 The study aimed to provide estimations of the prevalence of KLEFS1 symptoms and perceptions of their severity. This survey identified behavior problems, vision problems, problems during the newborn period, walking problems, and diagnosis of ID as the groups of clinical symptoms reported by the greatest number of KLEFS1 caregivers. Within these symptom groups, caregivers were able to categorize specific symptoms, as present, as “mild, moderate, or severe,” thus providing insight into caregiver-perceived ranges of symptom severity.
In addition, one survey-based study has focused on the impacts of KLEFS1 on parents of affected individuals. 16 Results indicated that most caregivers perceived communication deficits, learning difficulties, and GDD to be the symptoms of highest concern. Caregivers reported complex and nuanced experiences of how their relationships with others and daily life were impacted, including strain on their romantic partnership, difficulty giving attention to other children, a sense of isolation and a loss of friendships, and gain of a unique and supportive community. Many described significant changes to their perspectives, attitudes, and values surrounding disability, patience, and gratitude. This research focused on quantifying dimensions of parental functioning and comparing these metrics to those of parents of individuals with Prader Willi syndrome, another rare neurogenetic condition. Further research is required to better understand the full impact of KLEFS1 on the lived experiences of affected individuals and their caregivers.
Disease concept models (DCMs) are condition-specific representations of the lived experiences of a rare disease community generated using qualitative interviews to elicit thorough summations of a patient’s perspective. 17 DCMs aim to provide an overview of the both the breadth of clinical features of a condition which are most salient to the community, and their community’s perception of how the condition impacts individuals and their caregivers. They also frequently enable direct comparison between perspectives of caregivers and that of healthcare providers. Notable rare disease DCMs include those developed on STXBP1-related disorders, 18 Angelman syndrome, 19 and Dravet syndrome. 20
While the phenotype and genotype of KLEFS1 continues to be investigated, a review of the literature failed to reveal additional initiatives that explore the perspectives of affected individuals and their caregivers on the impact of the disorder more globally. The purpose of this study was to interview a diverse sample of caregivers of individuals with KLEFS1 to construct a DCM. A small set of HCPs were also interviewed to further facilitate the comparison of clinical features previously reported in medical literature and novel concepts observed within KLEFS1 healthcare. The resulting DCM can become a foundation for patient-centered research and condition-specific therapeutic interventions for the KLEFS1 community.
Methods
A grounded theory approach informed design of this qualitative study, with an emphasis on determining a relationship between domains of KLEFS1 features and domains of impact on lived experiences.
Literature Review
We conducted a targeted review of published literature, including interventional studies, observational studies, and case reports, to draft a preliminary model of KLEFS1 clinical features and impacts. We conducted this search via the Boston University electronic library and included publications made available between March 4, 2009 and January 26, 2023. All publications including the search terms “Kleefstra syndrome” and “EHMT1” were reviewed for relevance. A draft pediatric KLEFS1 DCM was developed on the basis of this review and was utilized to both inform development of caregiver and HCP interview guides and build a coding tree. A 2024 study by Zdolšek Draksler et al. 15 which expands on reported clinical features of KLEFS1 was published following completion of this study, but given its relevance, data was utilized to modify the present KLEFS1 DCM.
Study Sample and Recruitment
We recruited caregivers of individuals with KLEFS1 through the KLEFS1 patient advocacy foundation, IDefine. Participants were required to be over the age of 18, self-identify as a caregiver of an individual with KLEFS1 between the age of 2 and 18 years, and be able to conduct a 1-hour long interview in English. To ensure a representative participant population, purposive sampling was used to ensure that the individuals with KLEFS1 included a range of ages, geographic locations, and ancestral/racial backgrounds. Additionally, we prioritized sampling caregivers whose children had reached 13 years of age to capture data regarding post-pubertal regression. HCPs associated with the Kleefstra Clinic at Boston Children’s Hospital were identified and recruited using convenience sampling.
Interview Conceptualization and Implementation
The KLEFS1 DCM caregiver interview guide was modified from a series of NDD DCM interview guides developed by COMBINEDBrain, a non-profit organization that conducts research and supports collaborations aimed to develop clinical treatments for rare NDDs. An interview guide for HCPs developed by COMBINEDBrain was adapted similarly. This interview guide included additional questions about clinical features seen across the lifespan and HCP-perceived family experiences. The semi-structured inductive interview format was utilized to elicit narratives of the symptoms of KLEFS1 and their impacts on lived experiences of caregivers and affected individuals. 21 Follow-up questions were used to probe significant concepts that did not arise spontaneously, and to facilitate greater consistency in the data obtained across interviews. Interviews were conducted virtually via Zoom between October and December of 2023. Interview recordings were transcribed using transcription software Rev.com. Transcripts were manually edited for accuracy and any identifiable participant information was removed.
Data Analysis
Interview transcripts were analyzed using NVivo, a HIPAA-compliant qualitative data analysis software, and Microsoft Excel. Data was analyzed descriptively and assigned to a priori and emergent codes from a coding tree compiled by COMBINEDBrain for neurodevelopmental disorders. The coding structure was built from previous DCMs,18,19 with new codes added for novel concepts based on Human Phenotype Ontology (HPO) and World Health Organizations International Classification of Functioning, Disability and Health (ICF). 22 Additional concepts from existing literature about the defining features of KLEFS1 were added following the same guidelines.
Codes were grouped into 4 a priori categories. The most significant challenges and concerns described by caregivers were also identified and quantified. We recorded the number of caregiver interviews in which a concept was referenced, and the number of times within all interviews that a concept was referenced. KLEFS1-specific symptoms or symptom impacts were co-coded with age group to reveal age-specific impacts of KLEFS1. Symptoms were co-coded within 4 defined pediatric developmental age groups: under 2 years old (infancy), 2 to 4 years (early childhood), 5 to 11 years (late childhood), and 12 to 17 years (adolescent and teenage years).
Interviews were coded in an iterative approach, with additional interviews conducted until new codes no longer emerged. Based on the literature on data saturation in patient-centered qualitative research and existing DCMs,17 -20 this was expected to be at approximately 12 to 20 interviews. Our analysis reached saturation after 13 interviews. Three additional interviews were conducted, as interview invitations had been extended before the study team determined saturation. In addition to coding concept references, exemplar quotes for each concept were extracted to support dimensionality of the KLEFS1 DCM. A member of the IDefine board of directors, who was also a research participant, reviewed the results and provided feedback as a form of member-checking. 23
Ethical Approval and Informed Consent
This qualitative study was approved by the Institutional Review Board at Boston University (H-43810) in October 2023. Verbal consent for audio-recording of interviews was obtained for each participant.
Results
Demographics
All 16 respondents were caregivers of a child with KLEFS1, including biological, adoptive, and step-parents. Caregivers represented a wide demographic spectrum including 5 (31.3%) who did not identify as being of solely White European ancestry (Table 1). A variety of educational backgrounds, employment statuses, and geographic locations were represented.
Demographics of Caregivers (n = 16).

Caregivers described the experiences of caring for 16 children with KLEFS1 aged 3 to 17 years (Table 2). Nine of these individuals with KLEFS1 identified as female (56.3%) and 7 identified as male (43.8%). Age at diagnosis ranged from infancy (<1 year) to 16 years, with the majority receiving their diagnosis before 5 years of age.
Demographics of Individuals with Kleefstra Syndrome (n = 16).

Two HCPs, specializing in genetics and pediatric neurology, were interviewed. Each HCP had more than 3 years of clinical experience treating patients with KLEFS1.
Generation of the DCM
The analysis of concepts from caregiver interviews was used to generate a KLEFS1 DCM comprised of the following 4 categories: (1) defining features of KLEFS1, (2) individual impacts, (3) caregiver impacts, and (4) developmental age groups. These 4 categories contain 27 domains with a total of 159 concepts, excluding modifying factors which are described but not incorporated into the final disease concept model. Caregivers referenced these concepts a total of 3191 times throughout all interviews. The frequency of concept references within domains is shown in Figure 1. Exemplar quotes are included in Supplemental Table 1, parts A to C.

Defining domains of Kleefstra syndrome. The total number of concept references for each domain across all caregiver interviews in each of the 4 categories.
Our DCM presents the number of interviews in which a concept arose as well as the total number of times a concept was described—which demonstrates that a concept may have been described multiple times by 1 participant over the course of a single interview. This provides context for the relative importance of a concept to our sample of the KLEFS1 community, while also preventing misrepresentation of any single concept due to variation in frequency of expression within an interview.
The most frequently referenced of the defining features of KLEFS1 were within the communication domain, with concepts described 110 times (16.0% of the 688 references of defining features of KLEFS1). The most referenced individual impacts were within the health impact domain, with concepts described 304 times (51.4% of the 592 references of individual impacts). The most referenced caregiver impacts were within the caregiver requirements and adjustment domain, with concepts described 123 times (27.6% of the 445 references of caregiver impacts).
Category 1: Defining features of Kleefstra syndrome
All symptoms and physical features referenced by caregivers were used to construct a table of the defining features of KLEFS1 (Table 3). This table is organized into 14 domains with 91 concepts. Defining features referenced by the greatest number of caregivers were gross motor skills delayed or impaired (n = 15, 93.8%), expressive communication decreased (n = 14, 87.5%), autism spectrum disorder (n = 13, 81.3%), global developmental delays (n = 12, 75%), and maintenance difficulties of sleep (n = 11, 68.8%). When caregivers were prompted to describe the KLEFS1-related health challenges that have the most significant impact on their daily lives, 3 domains were most frequently reported: communication (n = 9, 56.3%), mobility (n = 7, 43.8%), and emotional and behavioral regulation challenges (n = 7, 43.8%; Figure 2).
Category 1: Defining Features of KLEFS1. Table of Defining Features was Generated from Caregiver-Reported Concepts. Bolded Concepts were Described by 50% or more of Caregivers. Italicized Concepts were Those that had not Previously been Reported in the Literature. Number of Interviews Referenced in and Total Number of References Across Interviews, Respectively, is Described as (N1, N2) Following Each Concept.

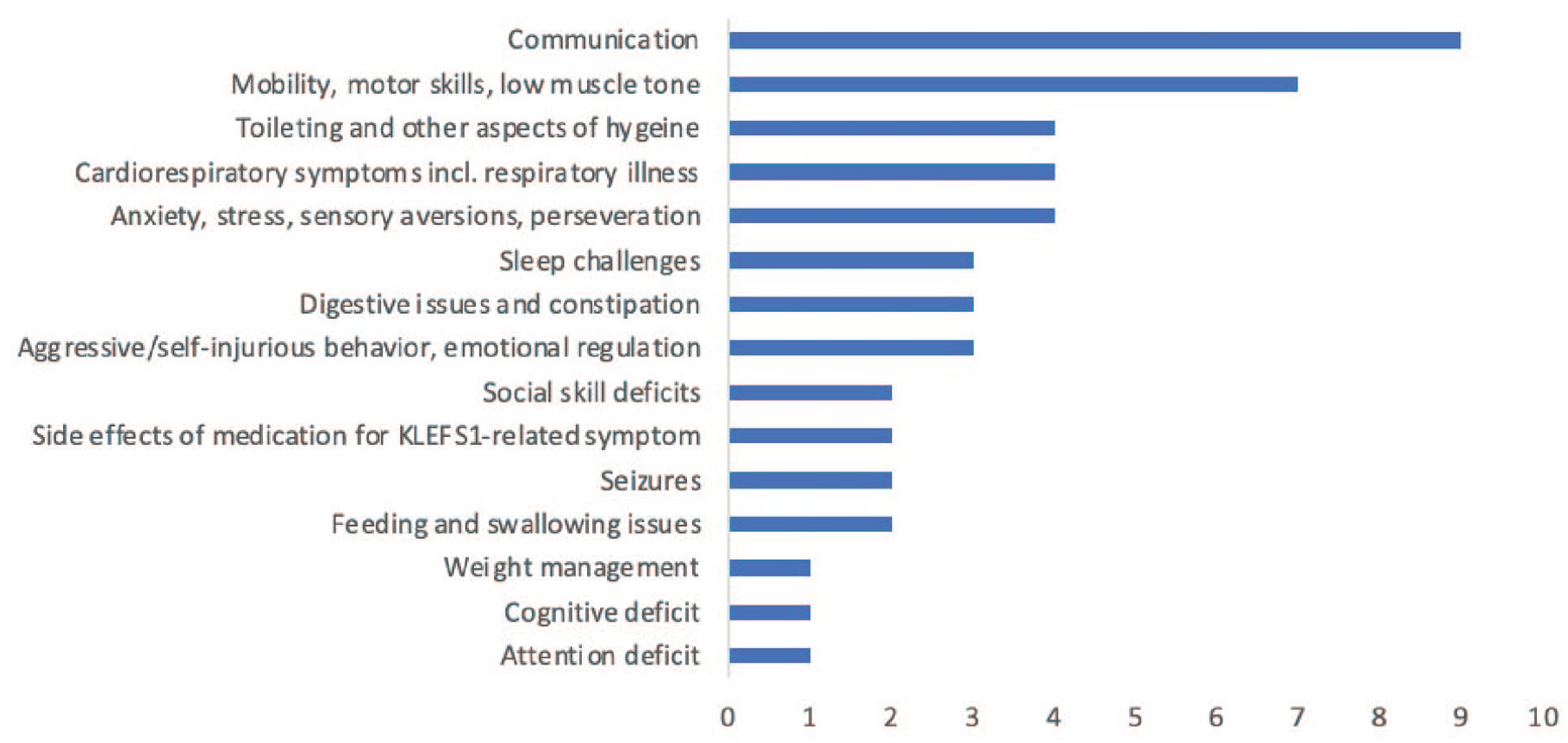
Symptoms reported by caregivers of children with KLEFS1 as having the greatest impact on their child’s daily life (each caregiver reported 3 features).
Defining features of KLEFS1 were also elucidated during HCP interviews. The following concepts were described by at least 1 HCP, but not by any caregiver: atrial flutter, kidney defects, structural genitourinary differences, compulsive overeating, obsessive and compulsive disorder-like (OCD) behaviors, and epigastric hernias. These concepts include both features that may be seen in children with KLEFS1 from birth and behavioral features that develop in some individuals during childhood and adolescence. When asked what they felt were the most important features of KLEFS1 to target with treatments, HCPs described communication, intellectual disability, developmental delay, and post-pubertal regression. Of the defining features described by caregivers that were not also referenced by HCPs, fatigue, drowsiness, and nighttime incontinence or toileting issues were most frequently described (n = 4, 25%).
Novel defining features of KLEFS1 that had not previously been previously reported in the literature included atypical regression, defiant behavior, perseverating or fixating, laryngeal cleft, autonomic changes, distinctive cry, and migraines surrounding post-pubertal regression.
Category 2: Individual impacts
The 31 concepts coded within individual impacts were organized into proximal impacts (ie, direct impacts of KLEFS1) comprised of the health impact domain and the daily living impact domain, and distal impacts (ie, indirect impacts of KLEFS1) comprised of education impact domain and social impact domain (Table 4).
Category 2: Individual Impacts of KLEFS1. Table of Individual Impacts was Generated from Caregiver-Reported Concepts. Bolded Concepts were Described by 50% or More of Caregivers. Italicized Concepts were Those that had not Previously been Reported in the Literature. Number of Interviews Referenced in and Total Number of References Across Interviews, Respectively, is Described as (N1, N2) Following Each Concept.

Proximal Individual impacts referenced by the greatest number of caregivers were therapies (n = 16, 100%), non-medication medical interventions (n = 14, 87.5%), medications (n = 12, 75%), bathing or hygiene assistance needed (n = 11, 68.8%), and toileting assistance needed (n = 11, 68.8%). Of types of therapies described by caregivers, occupational therapy (n = 16, 100%), speech therapy (n = 15, 93.8%), and physical therapy (n = 15, 93.8%) arose in the greatest number of interviews. Specific types of medication and non-medication interventions as well as therapies are detailed in Supplemental Table 2.
Non-medication medical interventions included dietary changes (a concept which was referenced by 5 caregivers (31.3%)), such as adoption of the ketogenic diet as described by 2 caregivers (12.5%) whose children had seizures. This concept also included equipment and devices, and surgeries or procedures. The most frequently described type of equipment was an assisted mobility device (n = 6, 37.5%), such as an adapted stroller. The medication most often described by caregivers was sleep medication (n = 7, 43.8%).
Distal individual impacts referenced by the greatest number of caregivers were adaptive classrooms or student support plan needed (n = 14, 87.5%), engaged socially (n = 14, 87.5%), communication with non-family members challenging (n = 10, 62.5%), behaviors are challenging or inappropriate in social situations (n = 8, 50%), and social isolation (n = 8, 50%).
A variety of adaptations to learning environ-ments were made for children including Individual Education Programs (IEPs), integrated classrooms, and specialized schools. Cognitive, communicative, neurodevelopmental, and behavioral symptoms were cited by caregivers as reasons for additional support needs. Regional differences in access to services was a trend noted throughout caregiver interviews. Often, specialized services and educational interventions were more readily available to families in urban communities. Some families reported having moved to be closer to medical or educational resources. Modifying factors to KLEFS1 symptoms, which are factors that impact the child’s presentation but are not a direct result of their underlying EHMT1 variant, included side effects of behavioral, seizure and other medications, and co-occurring medical diagnoses and unrelated history of injury (Supplemental Table 3).
Descriptions of the concept behaviors are challenging or inappropriate in social settings included screaming and hitting objects or people. These behaviors were often co-coded with the KLEFS1 defining features of anxiety, emotional regulation challenges, and sensory defensive behaviors or overstimulation, as well as the caregiver social impacts of leaving home more challenging, and socialization or leisure activities reduced or modified. The social isolation of children with KLEFS1 had a multitude of symptom-based co-codes, including challenges interpreting social cues or lacking confidence with social skills, communication, and aspects of daily living impact which reduced autonomy and independence. The majority of individual impact concepts identified in caregiver interviews had not previously been reported in the literature.
Category 3: Caregiver impacts
The 33 concepts of the caregiver impacts category were organized into domains of caregiver requirement and adjustment, emotional impact, financial and professional impact, health impact, and social impact (Table 5). Modifiers to caregiver impact were also identified and grouped into the domains of family and community support, knowledge and experience, medical and therapeutic care access, and other (Supplemental Table 4). The caregiver impacts referenced by the greatest number of caregivers were time and organization needed for care (n = 12, 75%), physical aid (n = 12, 75%), and sleep disrupted (n = 12, 75%).
Category 3: Caregiver Impacts of KLEFS1. Table of Caregiver Impacts was Generated from Caregiver-Reported Concepts. Bolded Concepts were Described by 50% or More of Caregivers. Italicized Concepts were Those that had not Previously been Reported in the Literature. Number of Interviews Referenced in and Total Number of References Across Interviews, Respectively, is Described as (N1, N2) Following Each Concept.

Numerous intersecting factors modified the lived experiences of caregivers of children with KLEFS1. The medical and therapeutic care access modifier described by the greatest number of caregivers was regional differences in access to services, insurance, and medical care (n = 8, 50%). The family and community support modifier described by the greatest number of caregivers was supportive family including spouse or co-parent (n = 7, 43.8%). The knowledge and experience modifier described by the greatest number of caregivers was lack of information on disorder or healthcare provider awareness (n = 6, 37.5%). In particular, caregivers described 2 common phenomena: (1) isolation due to the rarity of KLEFS1 and (2) the simultaneous burden of the parent serving as the dual KLEFS1 expert and advocate within the medical field while often also having their knowledge disregarded by HCPs.
Many caregiver impact concepts identified in caregiver interviews had not previously been reported in the literature. In particular, most novel concepts described by the DCM are within the caregiver requirement and adjustment domain.
Category 4: Developmental age groups
In order to understand the variable ways in which KLEFS1 impacts individuals and caregivers across different stages of development, concepts were co-coded with pre-defined age groups (Figure 3). All 16 caregivers described symptoms or symptom impacts when their child was in infancy, with a total of 183 references, 12.5% of all concept references that were co-coded with an age group. Fifteen caregivers described symptoms or symptom impacts when their child was in early childhood, with a total of 258 references, 17.6% of all references of age. The concepts and impacts of global developmental delays and gross motor delays were described within these age groups more frequently than in older children.

Category 4: Developmental age groups. The heatmap indicates the frequency interviews referenced concepts in each domain relative to all references of the domain within each age group, with darker colors representing higher frequency.
Thirteen caregivers described symptoms or symptom impacts of their child with KLEFS1 during late childhood years, with a total of 548 references, 37.4% of all references of age. Six caregivers described symptoms or symptom impacts of their child with KLEFS1 during adolescent and teenage years, with a total of 477 references, 32.5% of all references of age. Concepts co-coded with decreased expressive communication were described within these age groups more frequently than in younger children, having been mentioned by all 6 (100%) caregivers of individuals during adolescent and teenage years and by 8 (61.5%) of the caregivers whose children were in or had passed their late childhood years. Concepts related to daily living impact were also described in a greater proportion of these older age groups, especially descriptions of self-care tasks that could be done independently versus with assistance such as bathing or hygiene, toileting, and dressing.
The defining concepts need for rigid routine and anxiety were referenced proportionally more often within the adolescent and teenage years group, as were individual social impact concepts related to challenging, inappropriate, or aggressive social behaviors. The greatest proportion of caregiver social impact concepts were also co-coded with this older age group. Finally, concepts related to the post-pubertal regression domain were principal points of discussion during caregiver interviews of children in this oldest age group.
Exploration of Intersecting Themes Across KLEFS1 Defining Concepts and Impacts
In addition to elucidating concepts across 4 categories, several notable themes were revealed from the intersection of KLEFS1 defining concepts and their impacts (Table 6). The themes identified represent a sample of the spectrum of complex experiences that KLEFS1 features influence and exemplify the interconnectedness of the 4 KLEFS1 DCM categories.
Six Selected Themes Identified Across KLEFS1 Defining Concepts and Impacts, with Examples from Caregiver Interviews.

Discussion
KLEFS1 is a rare neurodevelopmental condition that has previously been characterized principally by physician reports. Although there are not yet FDA approved KLEFS1-specific therapeutics, the research and patient community are striving to develop such interventions. As advised by regulating entities, the development of a pediatric KLEFS1 patient-centered framework would further promote the development of pertinent outcome measures when designing interventions, resources, and assessments. 24
We present the first KLEFS1-specific pediatric DCM. Our framework represents a patient-centered conceptualization of pediatric KLEFS1 and its impact on the lived experiences of affected children and their caregivers. Each domain of this DCM provides insight into the relative significance of concepts related to the lived experience with KLEFS1. The resulting data serves to augment physician-reported and caregiver survey-based clinical descriptions of KLEFS1 in children and help guide relevant clinical interventions in a variety of domains. Some features that are well-established as part of the KLEFS1 spectrum, such as genitourinary defects and talipes equinovarus, did not arise spontaneously during interviews. Rather than serving as a comprehensive reflection of all possible features and impacts, this patient and caregiver-centered model reflects those disease concepts that our study participants chose to discuss. Our DCM provides a distinct, comprehensive portrayal of defining features KLEFS1 as compared to that of Zdolšek Draksler et al., 15 which prompted caregivers to indicate whether features from a pre-selected set of clinical symptoms were present in their child with KLEFS1.
Our DCM highlights the experiences of families of children affected by KLEFS1 within a constellation of concepts across 4 major categories: features of the condition, how the condition impacts the affected individual’s life, how the condition impacts the caregiver’s life, and how features shift throughout the affected child’s development. Our results supported the previously reported findings in the literature that gross motor delays and expressive communication challenges are considered by caregivers to be principal symptoms within the spectrum of KLEFS1. 16
Caregiver descriptions of KLEFS1 largely aligned with descriptions existing in the literature. However, 9 KLEFS1 concepts spanning various domains that had not previously been described were identified in our DCM. Two novel concepts, defiant behavior and perseverating or fixating, refine and expand the disease concepts in the neurodevelopmental and behavioral domain, and are closely related to social stressors that individuals and their caregivers experience. Similar neurodevelopmental and behavioral concepts including aggressive behavior and repetitive behaviors have been described in KLEFS1 literature,3,5 -7,9 but the inclusion of these 2 concepts enables further clarification of phenotypes that were described by caregivers as distinct.
A novel cognitive feature, atypical regression, was described by a caregiver as a distinctly different neuropsychiatric presentation compared to the well-established post-pubertal regression. While the definition and etiology of atypical regression was not well understood in 1 caregiver-reported case, it appears similar to the acute/episodic loss of skills with behavioral changes that has been reported in individuals with neurodevelopmental disorders. 25 This presentation is a product of a variety of identified and proposed mechanisms including autoimmune responses. As our DCM indicates, KLEFS1 caregivers face significant worry about the future possibility of a post-pubertal regression in their child. More research is needed to further define the presentation and contributory factors of typical and atypical regression within the KLEFS1 community.
In addition to newly described KLEFS1 features, our DCM provides the first comprehensive description of individual impacts of KLEFS1 and expands on caregiver impacts first described by Haseley et al. 16 Multiple caregivers described their child’s need for assistance with activities of daily living (ADLs), such as bathing, dressing and toileting, as a major impact of the symptoms of KLEFS1, which in turn impacted the parents and caregivers in time and organization needed for care. These findings indicate that the development of quantifiable ADL metrics would be a valuable way to measure treatment effectiveness and track longitudinal periods of progress or regression.
The domain of sleep was also a major concern throughout the categories, with concepts described by the majority of caregivers. Significant sleep disruption is a hallmark sign of the highly concerning post-pubertal regressive periods observed in a subset of the KLEFS1 patient population. 10 Furthermore, the concept of sleep disrupted, was 1 of the novel and principal caregiver impacts, extending the impacts of the well-characterized sleep challenges experienced by individuals with KLEFS1 to the whole family.
Our study also revealed a range of impacts of KLEFS1 on the daily lives of children with the disorder and their caregivers in the domain of social impacts. There was significant variability, and often the degree of expressed caregiver concerns was proportional to their child’s level of need. Caregivers with children who required constant medical care, or were nonverbal, less frequently described concerns regarding interactions with extended community compared to caregivers with children who have greater levels of independence. Previous research regarding the priorities of caregivers of individuals with NDDs further supports our finding that clinical severity influences perceptions of how a disorder impacts daily life.17,26
Differences between caregiver and HCP reports of KLEFS1 manifestations were identified during our analysis. HCPs more often referenced entire impacted systems, whereas caregivers referenced specific symptoms (e.g., providers reported “gastrointestinal issues” whereas caregivers described instances of reflux), indicating what disease concepts were of most importance to families. This also indicated that caregiver reports may offer more precise insights on salient features of KLEFS1. While there were differences in the frequency and specificity with which KLEFS1 features were described, both caregivers and HCPs selected communication as the domain that, if targeted with treatments, would have the most significant impact on patient quality of life.
Finally, the KLEFS1 DCM highlights variations in symptom importance across the pediatric lifespan. Caregivers of adolescents and teenagers more frequently reported challenges with self-care skills, such as toileting or maintaining hygiene, and difficulties in socializing. This emphasis in later developmental stages may not imply a lack of pervasiveness earlier in childhood but rather, could stem from both parental adaptations to physical KLEFS1 health features and an increased focus on independence and social fulfillment in older years. Caregivers also described significant challenges with public spaces and social acceptance of disabled or neurodivergent older children. As child development is associated with increased independence and autonomy, a lack of public supports often forces caregivers to adapt. Similar findings in studies on NDDs suggest that caregivers tend to identify social, behavioral, and independence-related aspects as more significant in later childhood, while focusing on physical health features during infancy and early childhood.18,19,27
Practical Implications
Importantly, the present KLEFS1 DCM captures the domains of pediatric KLEFS1 which caregivers report that, if improved, would have the greatest impact in improving the daily life of their affected children. These domains include communication challenges, issues with physical mobility, and challenges with emotional and behavioral regulation, which differed from 2 of the treatment targets identified by HCPs (cognitive disability and developmental delay). If prioritized as treatment targets for future KLEFS1 clinical trials in children, the interventional impact on these domains would likely be interpreted as meaningful to families. 28 The impact of these KLEFS1 disease domains on independence with ADLs, self-care tasks (especially in bathing, safety, dressing and toileting), and social engagement was described in the caregiver interviews. This KLEFS1 DCM indicates that design of clinical trials and development of standards of care should utilize instruments that measure improvement of ADLs, as well as the underlying symptoms. Novel individual impacts in the social impact domain including behaviors are challenging or inappropriate in social situations, and decreased recognition of danger or concerns of unsafe behavior also indicate the importance highly individualized IEPs that prioritize best adaptations to support safety, social inclusion, and skill-building.
By highlighting salient concepts across age groups, the present KLEFS1 DCM also provides insight into resources that might be most useful to families based on the age of their child with KLEFS1. For caregivers of children approaching their teenage years, our DCM suggests that resources specific to post-pubertal regression are imperative for supporting awareness and preparedness. Further, this research highlights gaps in access to critical supports for families across childhood. Resources such as respite care networks, which provides caregivers with critical time away from caregiving responsibilities, is often impeded by barriers including geography and non-standardized eligibility criteria. 29 While these supports are needed by many other families caring for children with disabilities, narratives from KLEFS1 families demonstrate the barriers that healthcare systems create in accessing care across different demographics.
Study Limitations and Future Directions
A limitation of this study was that our inclusion criteria restricted interviewee representation compared to the world-wide population with KLEFS1. The experiences of families who did not speak English or lived outside of the United States and Canada were not captured within the study. Future iterations of this work may include recruiting in different countries. Resulting interviews may be conducted in a variety of languages to capture a greater breadth of the impact of KLEFS1 in different sociocultural contexts. The sample size (n = 2) of healthcare providers interviewed similarly limits generalizability, as only 2 of the many subspecialties that may support individuals with KLEFS1 were represented.
Further, this research focused on childhood development and therefore did not capture the experiences of adults with KLEFS1, nor the full impacts of regression which likely unfold later in life. Future research may delve into the perspectives of caregivers of adults with KLEFS1, thus capturing a larger portion of the population who are at an increased risk for post-pubertal regression and providing insights on shifting needs in adulthood.
Finally, by using caregiver reports of symptoms and impacts, the underlying mechanism of a phenotype may not be captured, resulting in redundant or possibly inaccurate coding. For example, global developmental delay may be influenced by varying degree of cognitive deficits, but these root features become underrepresented if the etiology of the delay was not clarified in caregiver descriptions. Given the variability in how features and impacts may be characterized and coded, it is also possible that symptoms and impacts considered to be “novel” have been reported previously but attributed to a different organ system or under a descriptor not recognized as comparable.
Conclusion
The pediatric KLEFS1 DCM developed here stands as a valuable patient-centered conceptualization, providing insight into the lived experiences of families. Unlike traditional clinical reports, the DCM emphasizes caregiver perspectives, indicating the relative significance of concepts in their daily lives. Key takeaways from the study underscore the prominence of gross motor skill delay and expressive communication challenges as principal pediatric symptoms, echoing previous research. The DCM also highlights the relationship between defining features of KLEFS1 and spheres of impact, with the importance of certain symptoms varying across dimensions such as age. Notably, the identified potential treatment targets: communication challenges, physical mobility, emotional and behavioral regulation, ADLs, and sleep, serve as crucial objectives for clinical interventions aimed at improving the daily lives of individuals affected by KLEFS1. Success of such interventions may be best measured with scales that center on ADLs and social behavior skills.
Supplemental Material
sj-docx-1-gph-10.1177_30502225251336880 – Supplemental material for Development of a Patient and Caregiver-Centered Pediatric Disease Concept Model for Kleefstra Syndrome
Supplemental material, sj-docx-1-gph-10.1177_30502225251336880 for Development of a Patient and Caregiver-Centered Pediatric Disease Concept Model for Kleefstra Syndrome by Kristen L. Connors, Nikkola E. Carmichael, Terry Jo Bichell, Kira A. Dies and Zoë J. Frazier in Global Pediatric Health
Footnotes
Acknowledgements
The authors thank the families and individuals with Kleefstra syndrome for their invaluable contributions to this study. This work was done on behalf of and with guidance from IDefine, with exceptional efforts from Eric Scheeff, CSO, and Geoff Rhyne, CEO. Further, Dr. Siddharth Srivastava, a core leader of the Kleefstra Clinic of Boston Children’s Hospital provided clinical expertise to refine codes as well as reviewing the final manuscript. Additionally, we thank COMBINEDBrain for providing essential materials guiding study design, development of materials, and coding support. This study was funded by IDefine – The Kleefstra Syndrome Foundation, the Warren Alpert Foundation, and the Rosamund Stone Zander Translational Neuroscience Center. Finally, we thank the Boston University MSSP statistical consulting service, specifically Wuge Li, Amie Thomas, and Maysen Pagan under the guidance of Professor Masanao Yajima and teaching fellow Minh Thu Bui, for their data analysis support.
Ethical Considerations
This research study was reviewed by the Boston University Medical Campus. Institutional Review Board and was granted approval as an exempt study in October of 2023 (Protocol H-43810).
Consent to Participate
All interview participants were read a research information sheet detailing the purpose of the study and storage and protection of data. Participants verbally agreed to participate in the interview and be audio-recorded.
Consent for Publication
Not applicable.
Author Contributions
Conception and design: KC, NC, KD, ZF. Data aquisition: KC. Data analysis and interpretation: KC, TB, ZF. Drafting of the manuscript: KC. Critical revision: NC, KD, TB, ZF. All authors agreed to be acountable for all aspects of work ensuring integrity and accuracy.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: First author was supported by the Warren Alpert Foundation. Additionally, funding for participant compensation, and AI transcription services were provided by IDefine. Funding for publication fees were provided by the Rosamund Stone Zander Translational Neuroscience Center.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
