Abstract
Background:
In aesthetics and cosmetic therapeutics, cells in the epidermis, dermis and hypodermis are stimulated, repaired, regenerated and/or replaced to alter skin quality or facial appearance. Conventional cosmetic aesthetic therapies have limitations either in the administration procedure, required technical skill, the lack of sustained efficacy or risk of side effects. Over the last decade, exosomes have revolutionised and challenged conventional cosmetic aesthetic therapies due to their inherent biocompatibility, regenerative potential and ability to deliver paracrine substances, including proteins, lipids, nucleic acids and metabolites, thereby reducing inflammation and promoting skin regeneration. This review outlines recent advances in exosome research for aesthetic and cosmetic applications. It focuses on various types of studied exosomes and explores their mechanistic pathways in treating skin disorders, including hyperpigmentation, ageing, wound healing, scar removal and immune-related dermatoses.
Method:
A comprehensive literature review was conducted using PubMed and ScienceDirect search engines. Relevant studies, including research articles, review articles and clinical trials focusing on exosome-based therapies in aesthetic and cosmetic applications, were screened and evaluated.
Result:
Findings indicate that milk-derived, phyto-derived and stem cell-derived exosomes demonstrate significant potential for above-skin disorders. Mechanistic studies suggested that exosomes improve skin quality and enhance aesthetic outcomes by inhibiting melanogenesis, promoting collagen production, reducing proinflammatory cytokines and stimulating regeneration.
Conclusion:
Despite promising results of exosomes in aesthetic and cosmetic therapeutics, standardised procedures for purification, isolation, standardisation and regulatory approval must be established. Nevertheless, to ensure long-term safety and efficacy, more extensive research and clinical trials are still required.
Keywords
Background
Exosomes are phospholipid bilayer-enclosed, nano-sized (40–160 nm) vesicles. These cell-derived vesicles are involved in intercellular signalling by carrying contents such as proteins, metabolites, lipids and nucleic acids from the parent cell to the recipient cell.1-3 The biosynthesis of exosomes begins with the maturation of early endosomes into late endosomes and further to multivesicular bodies. Intraluminal vesicles are produced through the inward infolding of the membrane of multivesicular bodies. These intraluminal vesicles later fuse with the plasma membrane, releasing their content by exocytosis into the extracellular environment. 4 Exosomes’ characteristic biomarkers include enriched proteins such as biogenesis proteins (endosomal sorting complex required for transport complex), membrane transport proteins (GTPase and annexins), tetraspanins (CD9, CD63, CD81 and CD82), heat shock proteins (HSP60, HSP70 and HSP90), cytoskeletal proteins and growth factors. 5
Due to these enriched cellular biomolecules, exosomes can govern critical cellular processes such as cell proliferation, differentiation and apoptosis depending upon their origin, pathophysiological condition and target sites. Similar to liposomes and niosomes, exosomes possess the ability to encapsulate drugs and deliver them to target sites. The reduced immunogenicity of exosomes enhances their advantages over artificial nanovesicles. 6 These distinctive features of exosomes can be exploited in nanotechnology-based drug delivery to treat complicated diseases, including cardiovascular and neurodegenerative disorders. Researchers are now engineering exosomes to carry specific therapeutic agents and targeting ligands, significantly enhancing drug delivery efficiency even across challenging biological barriers such as the blood-brain barrier. 7 Moreover, recent studies have identified exosomes as promising diagnostic biomarkers; their molecular contents often reflect the physiological state of the parent cell, making them valuable in early disease detection. 8
Emerging research confirms the potential of exosomes in both the diagnosis and therapeutic intervention of genetic, immune-mediated and even neuro-oncological diseases. 9 Cancer cell-derived exosomes have been found to carry tumour-promoting cellular molecules to normal cells, contributing to progression, invasion and metastasis.10-14 Consequently, exosomes have emerged as a potential targeted drug delivery system for cancer diagnosis and therapies. 15 Similarly, immune-derived exosomes carry cargo involving miRNA, immunomodulators, cytokines and other chemical mediators from the immune cells to normal cells, resulting in triggering immune reactions. 16
Theoretically, exosomes might facilitate the repair and restoration of cellular processes or regenerate human cells to improve skin quality, such as tone, texture and appearance. Innovative applications now leverage the natural regenerative properties of exosomes to enhance skin rejuvenation and healing. Current limitations in medical aesthetics include difficulties in controlling dosage in fillers and injections, abnormal fat contouring in fat transfer therapy, the need for repeated sessions, inconsistent results in platelet-rich plasma therapy, skin sensitivity and the requirement for multiple sessions in laser or energy-based therapies. 17 In recent years, the use of stem cells has attracted interest due to their pluripotency, self-renewal potential and ability to regulate cellular processes.17,18 Exosomes have been shown to hasten wound healing while promoting natural regeneration. 19 Extensively, medical aesthetics is significantly advanced with the inclusion of exosomes. 20 Stem cell-derived exosomes potentially secrete and transfer paracrine factors to recipient cells and promote cellular events in skin rejuvenation. In addition to mesenchymal stem cell-derived exosomes (MSCE) derived from the umbilical cord, bone marrow and adipose tissue, bovine milk, colostrum and phyto-derived exosomes are also utilised in medical aesthetics, demonstrating the possibility of cross-kingdom communication, thereby interacting and influencing mammalian cellular processes (Figure 1). 21
Sources and Biosynthesis of Exosomes.
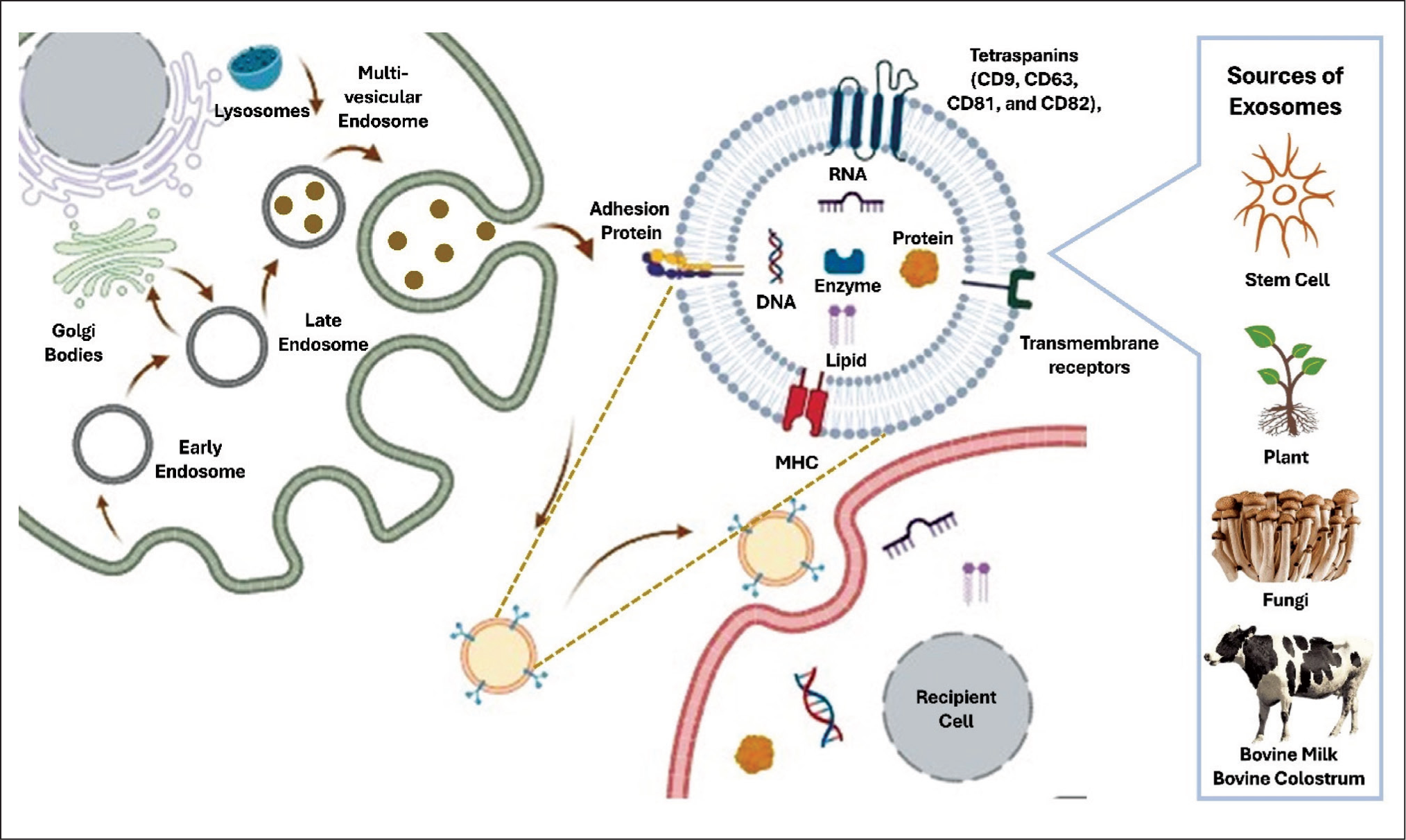
The presented review focuses on the sources of derived exosomes and their therapeutic mechanisms for skin disorders (Figure 2), outlining recent advances in exosome research for cosmetic and dermatological applications, including hyperpigmentation, ageing, wound healing, scar removal and immune-related dermatoses. Furthermore, the review discusses how the unique ability of exosomes to cross biological barriers and be bioengineered for targeted delivery could pave the way for next-generation diagnostic tools and treatment modalities in medical aesthetics.
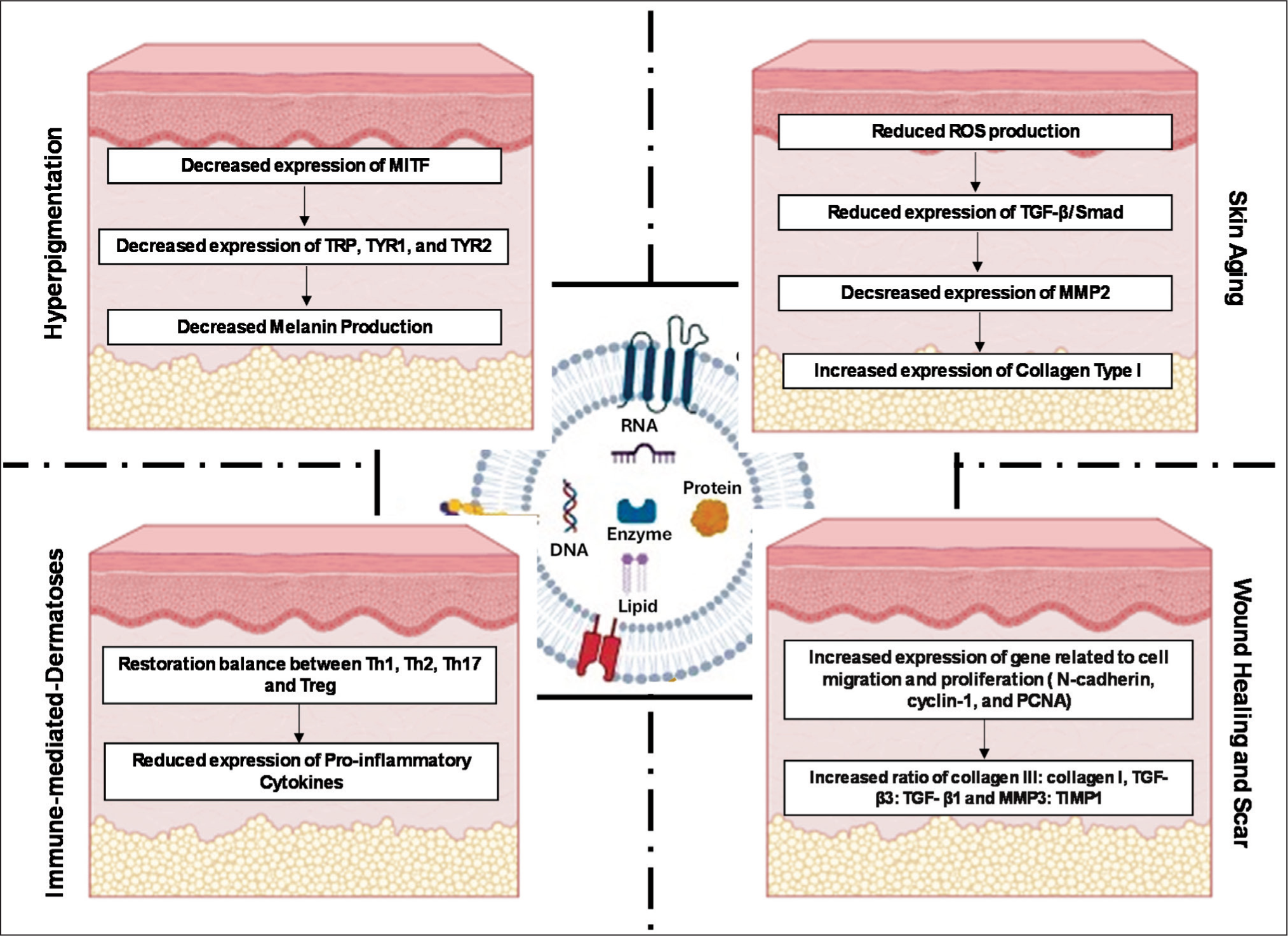
The Mechanistic Pathway Involved in the Application of Exosomes for Skin Disorders.
Methodology
Using the search engines Science Direct and PubMed, a systematic literature search was executed using the terms ‘exosomes’, ‘skin’, ‘cutaneous’, ‘cosmetic’, ‘aesthetic’ and ‘dermatology’. Different combinations of keywords such as ‘exosomes and skin’, ‘exosomes and cutaneous’, ‘exosomes and aesthetic’, ‘exosomes and cosmetic’ and ‘exosomes and dermatology’ were pursued. Research articles that were published in the last 10 years were screened according to relevance to the topic (involving the application of exosomes in in-vitro, pre-clinical and clinical studies in the field of aesthetic and dermatology). Duplicate research articles were excluded. Cross-references and review articles were also perused to assess additional information.
Exosomes for Aesthetic and Cosmetic Therapeutics
Exosomes for Hyperpigmentation
Various cell types, including keratinocytes, fibroblasts and stem cells, regulate cutaneous pigmentation through exosome-mediated intercellular communication in response to physiological, pathological conditions and environmental stimuli such as UVB radiation. 22 Exosomes derived from Codium fragile and Sargassum fusiform (Korean seaweeds) significantly suppressed melanogenesis in MNT-1 human melanoma cells through downregulating MITF, tyrosinase and TRP-1 expression. 23 Likewise, milk-derived exosomes demonstrated potent anti-melanogenic effects, particularly attributed to their bioactive protein cargo (casein, β-lactoglobulin and α-lactalbumin) that exhibit intrinsic tyrosinase-inhibitory activity.24-26 Microarray profiling identified a high abundance of miR-2478, miR-1777b, miR-1777a, let-7b and miR-1224 within milk-derived exosomes.24,27 Among these, only miR-2478 exerts a more pronounced inhibitory effect on tyrosinase. Mechanistically, miR-2478 targets Rap1a, promoting AKT phosphorylation and subsequent GSKβ activation, suppressing MITF and TYR expression. This signalling cascade attenuates melanin synthesis, highlighting miR-2478 as a key mediator of tyrosinase-inhibitory activity of milk-derived exosomes. 24
Keratinocytes, together with melanocytes, constitute the epidermal melanin unit and play a crucial role in cutaneous pigmentation regulation. 28 Exosomes derived from UVB-irradiated keratinocytes significantly activate melanocytes, suggesting that quantitative variations in exosomes secreted from keratinocytes may regulate skin pigmentation. 29 Cicero et al. demonstrated that under UVB irradiation, keratinocyte-derived exosomes modulate melanogenesis via exosomal miR-3196 in an MITF-dependent manner or through miR-203 independently of MITF. 30 Keratinocyte-derived exosomes also deliver miR-675 to melanocytes, directly targeting MITF and downregulating tyrosinase, TRP-1 and TRP-2 through the inhibition of the CREB/ERK/AKT pathway. 31
Dermal fibroblasts also influence melanogenesis through the secretion of exosomes. Exosomes from UVB-induced human dermal fibroblasts inhibit melanin production and downregulate TYR and MITF expression, potentially via the miR-25-5p-TSC2 axis. 32 In patients with hypertrophic scars, fibroblast-derived exosomes decrease melanocyte proliferation and melanin secretion, leading to epidermal hypopigmentation. 33
In regenerative medicine, various stem cell-derived exosomes demonstrate anti-melanogenesis properties. Cho et al. developed adipose-derived mesenchymal stem cell exosomes that significantly suppressed melanin production in B16F10 melanoma cells. Translational studies showed a significant reduction in hyperpigmentation after 4 weeks compared to placebo. 34 Human umbilical cord blood-derived exosomes inhibit melanin synthesis by modulating the expression of MITF. 35 Wang et al. demonstrated that miR-181a-5p and miR-199a, derived from human amniotic stem cell-derived exosomes, suppressed melanogenesis by inhibiting MITF expression and promoted melanosome degradation through autophagy activation, respectively. 36 A prospective clinical trial by Proffer et al. preliminarily demonstrated the efficacy of platelet-derived exosomes in skin whitening. 37
Exosomes for Skin Ageing
Keratinocytes and fibroblasts, the primary structural cells of the skin, are crucial for maintaining hydration and preventing wrinkle formation. Skin ageing is primarily driven by the deterioration of the extracellular matrix, particularly the loss of type I collagen. UV radiation is responsible for ~80% of facial ageing, accelerating the ageing process by promoting collagen degradation. Lu et al. demonstrated that milk-derived exosomes enhanced cutaneous hydration through transcriptionally upregulating FLG and CD44 in keratinocytes and HAS2 in fibroblasts, thereby augmenting hyaluronic acid retention. Upon UV irradiation, pretreatment with milk-derived exosomes facilitated fibroblast migratory activity and reinstated type I collagen. Clinically, topical application of milk-derived exosomes increased skin hydration by ~4.64% in female volunteers aged 36-45 years. 38 Colostrum, compared to mature milk, is richer in immunomodulatory and bioactive constituents. Colostrum-derived exosomes markedly reduced intracellular ROS in keratinocytes through enhanced expression of glutathionylated proteins. In fibroblasts, colostrum-derived exosomes suppressed MMP-2 while upregulating collagen type I. The combined antioxidant and matrix-preserving effects underscore their enhanced anti-ageing potential. 39
Skin ageing predominantly arises from dermal fibroblast senescence. Adipose MSCEs exert an anti-senescence effect in fibroblasts through attenuated ROS generation, upregulated collagen type I and decreased senescence-associated β-galactosidases (SA-β-Gal) expression. Expression of key cell-cycle regulators p16, p21 and p53 was also significantly downregulated. 40 Similarly, exosomes derived from human induced pluripotent stem cells prevented UVB-induced cellular senescence by inhibiting SA-β-Gal, MMP-1 and MMP-3, while maintaining type I collagen expression. 41 Bae et al. engineered murine embryonic stem cell exosomes overexpressing mmu-miR-291a-3p, which delayed fibroblast senescence through influencing TGF-β receptor 2 signalling pathway. 42 Exosomes isolated from Phellinus linteus inhibited MMP-1 expression, reduced ROS accumulation and SA-β-Gal levels, while enhancing antioxidant defence via superoxide dismutase upregulation. Denovo miRNA sequencing showed high expression of 5 miRNAs (miR-CM1, miR-CM2, miR-CM3, miR-CM4 and miR-CM5). These 5 miRNAs (miR-CM1, miR-CM2, miR-CM3, miR-CM4 and miR-CM5) were transfected into HaCaT keratinocyte cells. Of these 5 miRNAs transfected to HaCaT cells, HaCaT cells transfected with miR-CM1 inhibited Mical2 expression through cross-kingdom regulation. Synthetic exosomes encapsulating miR-CM1 further promoted type I collagen expression, suppressed MMP-I, reduced oxidative stress markers (ROS and MDA) and enhanced SOD activity in the murine photoaging model. 43 In parallel, apple-derived exosomes exerted anti-ageing activity by inhibiting the NF-κB signalling pathway, resulting in decreased expression of MMP-1, MMP-8 and MMP-9. 44
The culture microenvironment significantly influences exosome bioactivity. Human dermal fibroblast-derived exosomes obtained from 3D spheroid culture systems demonstrated superior anti-ageing potential relative to conventional monolayer-derived human dermal fibroblast-derived exosomes. Specifically, 3D-human dermal fibroblast-derived exosomes upregulated collagen type I, downregulated MMP-I, suppressed TNF-α and upregulated the TGF-β signalling pathway. 45 Overall, exosomes demonstrated significant anti-ageing potential by modulating oxidative stress, inhibiting senescence pathways and restoring extracellular matrix homeostasis.
Exosomes for Immune-mediated-dermatoses
Psoriasis
Psoriasis is a chronic autoimmune skin disorder characterised by localised lesions, scaling, inflammation and pruritus, with the IL-23/IL-17 axis central to its pathogenesis. Exosomes enriched with immunomodulatory cargo have emerged as promising modulators of the hyperactive immune response. Topical application of immortalised E1-MYC 16.3 human embryonic stem cell-derived mesenchymal stem cell exosomes reduced psoriatic-related cytokines (IL-7, IL-23 and C5b-9) in imiquimod-induced murine psoriasis models. However, retention was largely confined to the stratum corneum.46,47
Dysregulated PD-1 signalling in psoriasis amplifies T-cell activation and cytokine release; intriguingly, Pristimerin-loaded tumour-derived PD-L1-positive exosomes enhanced drug uptake in CD4+ T-cells and keratinocytes, suppressed Th17 proliferation and reversed imiquimod-induced psoriatic lesions. 48 Similarly, tofacitinib-loaded exosomes markedly downregulated TNF, IL-23, IL-6 and IL-15 transcripts in imiquimod-induced BALB/c psoriasis models. 49
Atopic Dermatitis
Atopic dermatitis is a chronic, relapsing and inflammatory skin disorder characterised by impaired epidermal barrier function, erythematous and exudative lesions and severe pruritus. Its pathogenesis is closely linked to dysregulated Th1/Th2 immune responses.50,51 Exosomes derived from adipose mesenchymal stem cells demonstrated reduced serum IgE, eosinophils, mast cells and CD86+/CD206+ cells in house dust mite-induced NC/Nga mice skin lesions with concurrent suppression of IL-4, IL-23 and TNF-α. 52 In the oxazolone-induced dermatitis mouse model, subcutaneous adipose MSCE further suppressed the production of IL-4, IL-5, IL-13, IL-17 and TNF-α. Improved stratum corneum hydration and decreased trans-epidermal water loss were also observed, indicating restoration of epidermal barrier function through enhanced de novo synthesis of ceramides. 53
Clinically, Han et al. reported that topical adipose MSCE reduces dupilumab-associated facial erythema in 19 of 20 patients. Results also demonstrated significantly reduced expression of IL-α and human thymic stromal lymphopoietin and significantly increased expression of filaggrin and vascular endothelial growth factor. 54 A case report of 2 clinical cases was also aligned in the same direction as previously cited studies. The obtained findings showed that the treatment with electroporation-assisted topical application of adipose MSCE for dupilumab-related facial redness patients showed a significant reduction in erythema skin lesion score. 55
Systemic Lupus Erythematosus
Systemic lupus erythematosus is a chronic autoimmune disorder characterised by autoantibody-mediated inflammation that affects multiple organs, including the skin, kidneys, heart, lungs and nervous tissue. Its aetiology involves genetic predisposition (HLA-DRA, DR3 and C1q/C2 mutations), environmental triggers (UV, viral infections and hormones) and immune dysregulation (nuclear autoantibodies and elevated IFN-α). 56 Circulating exosomes in systemic lupus erythematosus patients exhibit increased levels of IFN-α, TNF-α, IL-1β and IL-6, as well as overexpression of NEA1, S100A4, ApoE, BPI and Has_circ_0000479, along with reduced levels of GAS5 and miR-124, distinguishing them from healthy controls and highlighting their potential as biomarkers for diagnosis and disease monitoring.57–59
Liu and his coresearchers infused MSCE into the MRL/lpr mice lupus model, leading to significantly improved osteopenia by modulating the miR-29B/Dnmt1/Notch epigenetic cascade. 60
Systemic Sclerosis
Systemic sclerosis is a complex autoimmune disorder characterised by fibrosis, immune dysregulation and vascular abnormalities. Exosomes isolated from systemic sclerosis patients were implicated in the upregulation of miR-17, miR-23b, miR-29a and Let-7g and the downregulation of Let-7a, miR-125b, miR-140 and miR-146a. These exosomal biomarkers are being explored for theragnostic purposes, as they may help in both diagnosing the disease and guiding targeted therapy.61-66
Rozier et al. demonstrated that murine and human adipose MSCE engineered with antagomiR-29a-3p attenuated skin fibrosis in an HOCl-induced systemic sclerosis model by downregulating Dnmt3a, PDGFRBB, BCL2 and BCL-XL, thereby reducing profibrotic, remodelling and anti-apoptotic signalling. 67 Xie et al. studied bone marrow MSCE with overexpressed miR-214 in a bleomycin-induced skin fibrosis mouse model. Mechanistically, formulated exosome alleviates skin fibrosis by targeting IL-33 and inhibiting the IL-33/ST2 axis. 68
Exosomes for Wound Healing, Scar and Tissue Remodelling
Exosomes play a crucial role in wound healing and scar formation by promoting the activation, proliferation, migration, angiogenesis, extracellular matrix production and re-epithelialisation of fibroblasts. Adipose tissue-derived mesenchymal stem cell exosomes increased the translational expression of genes related to cell migration and proliferation, such as N-cadherin, cyclin-1 and PCNA. Notably, intravenous exosome injection in a full-thickness skin incision mouse model achieved 90% wound closure by 21 days of treatment, compared to the control group with 60% wound closure, along with increased expression of collagen, thereby hastening cutaneous wound healing. 69
Fibroblasts are activated in the early phase of wound healing and further differentiated into myofibroblasts. Myofibroblasts synthesise extracellular matrix components and promote wound contraction. Adipose tissue-derived mesenchymal stem cell exosomes facilitate scarless wound healing by downregulating α-SMA expression, inhibiting the differentiation of fibroblasts into myofibroblasts and upregulating MMP-3 via ERK/MAPK signalling. A higher collagen III:collagen I ratio, TGF-β3:TGF-β1 and MMP3:TIMP1 ratio support extracellular matrix regeneration.70-72 Zhang and his colleagues formulated that human adipose tissue-derived mesenchymal stem cell exosomes also facilitate wound healing via regulation of the PI3K/AKT signalling pathway. 73
Scarring has been associated with elevated TGF-β levels near the wound site. TGF-β activates the membrane receptor serine/threonine kinase complex and phosphorylates SMAD proteins. Phosphorylated p-SMAD2, a recognised marker for myofibroblast differentiation, contributes to scar formation.74,75 Umbilical cord blood-derived mesenchymal stem cell exosomes enriched with microRNAs (miRNA-21, miRNA-23a, miRNA-125b and miRNA-145) attenuate α-smooth muscle actin and collagen deposition through the TGF-β/SMAD2 signalling pathway, reducing visible scarring. 76
Wang et al. fabricated an injectable adhesive polysaccharide-based dressing with sustained pH-responsive exosomes for diabetic wound healing. This innovative dressing significantly increased cell proliferation, migration, angiogenesis, collagen deposition and reepithelisation in diabetic full-thickness cutaneous wound models. 77 Despite exosomes being a potential cell-free therapy for wound healing and scar remodelling, synergetic outcomes were observed when combined with MSC 78 and other medical aesthetic treatment interventions.79,80
However, despite their immense potential, several limitations remain. Challenges such as isolation and purification difficulties, lack of standardisation, safety concerns and regulatory issues must be addressed. Exosomes derived from different parent cells exhibit varying compositions and yields. This varying composition makes it challenging to standardise production and isolation procedures. During the isolation and production of exosomes, they may get contaminated with other extracellular vesicles or proteins; therefore, purification also remains another noteworthy challenge in the preparation of exosomes. Currently, there are no established procedures for the isolation, purification and production of exomes, which makes reproducibility challenging. Moreover, physical factors such as temperature and pH significantly affect exosome stability, necessitating the development of methods to ensure long-term stability. A stringent regulatory framework covering isolation, purification, production, approval and commercialisation is essential to ensure the reproducibility and safety of exosome-based products.
Conclusion
Exosomes are challenging the mainstay treatments in the fields of aesthetics and dermatology due to their richness in proteins, lipids and cellular biomolecule content, which endow them with the potential to improve skin tone, texture and appearance. They function as effective nano-carriers that deliver bioactive substances deep within the skin, including peptides, ceramides, antioxidants and hyaluronic acid. To preserve the integrity of the skin and achieve a youthful, radiant appearance, this site-specific delivery enhances the efficacy of bioactive ingredients, working synergistically with exosomes. In addition to being easily formulated into topical creams, serums and masks, exosomes can be utilised in conjunction with minimally invasive skin care techniques, such as laser treatment or microneedling, to enhance skin health further. Moreover, genetic modification of exosomes can augment the stability and potency, paving the way for more advanced therapeutic applications. Extensive preclinical and clinical research is required to thoroughly evaluate the safety and effectiveness of exosomes in both the dermatological and cosmetic fields.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest concerning the research, authorship and publication of this article.
Funding
This work received no funding from any public, commercial or not-for-profit agency.
