Abstract
Frontal fibrosing alopecia (FFA) is a progressive cicatricial alopecia primarily affecting the frontal and temporal hairlines, often accompanied by eyebrow loss. First described by Steven Kossard in 1994, FFA has since emerged as a globally recognised condition, predominantly affecting postmenopausal women, though cases in men, premenopausal women, and adolescents are reported. Its pathogenesis remains unclear, involving autoimmune, hormonal, genetic, and environmental factors. Histopathological similarities to lichen planopilaris (LPP) suggest a potential overlap. FFA progresses slowly and is challenging to diagnose early, with symptoms such as hairline recession, perifollicular erythema, and scalp fibrosis. The condition is often associated with thyroid disorders, rosacea, and other autoimmune diseases. FFA’s multifactorial aetiology includes immune dysfunction, PPAR-γ dysregulation, and hormonal influences, with oestrogen depletion being significant in postmenopausal women. Environmental factors, such as certain skincare products and surgeries, may trigger the disease in genetically predisposed individuals. Treatments aim to halt disease progression and alleviate symptoms, as hair regrowth is typically unattainable once scarring occurs. Topical corticosteroids, calcineurin inhibitors, 5α-reductase inhibitors, and systemic agents like hydroxychloroquine are commonly used, but no standardised treatment exists. Ongoing research seeks to clarify its pathogenesis and improve therapeutic options. Early diagnosis and tailored management are essential to mitigate FFA’s impact.
Keywords
Introduction
Frontal fibrosing alopecia (FFA) is a type of cicatricial alopecia characterised by hairline recession at the frontal, temporal, and sometimes temporoparietal regions, often accompanied by partial or complete eyebrow loss.1-5 First described in 1994 by Steven Kossard, 1 FFA cases are increasingly reported across all genders, races, and even among family members.4-9 It is considered a potential variant of lichen planopilaris (LPP) due to shared histopathological features, though whether it is a distinct disorder remains uncertain. 4
The condition’s cause is unclear, with autoimmune, hormonal, environmental, and genetic factors implicated.5,10 FFA progresses slowly, often delaying diagnosis and treatment, and stabilising the disease can take months to years with frequent recurrences. 11 Despite ongoing research, no standard treatment has yet been established.
Epidemiology and Demographic Data
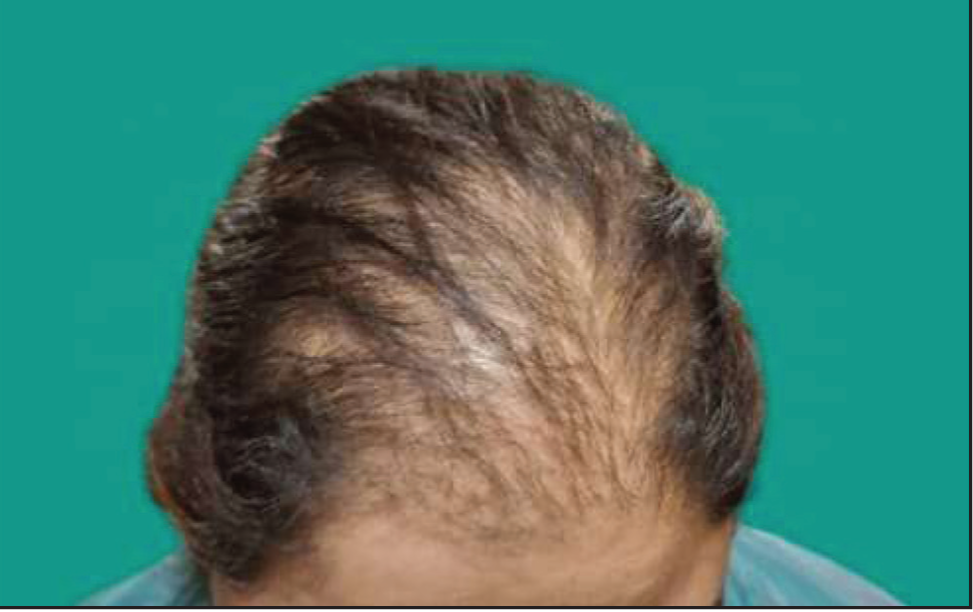
FFA predominantly affects postmenopausal women, accounting for 83%-95% of cases among Caucasian and Asian women and 74% among South African Black women.5,6,8,10,12 However, it also occurs in premenopausal women (Figure 1), men, and even adolescents, with a reported case in a 15-year-old girl.5,13,14 Men typically experience earlier onset (mean age 47.3 years) 15 but are often misdiagnosed due to overlapping features with androgenetic alopecia (AGA). Most male patients seek consultation for eyebrow loss rather than alopecia. 16
Frontal Fibrosing Alopecia in a 45-year-old Female.
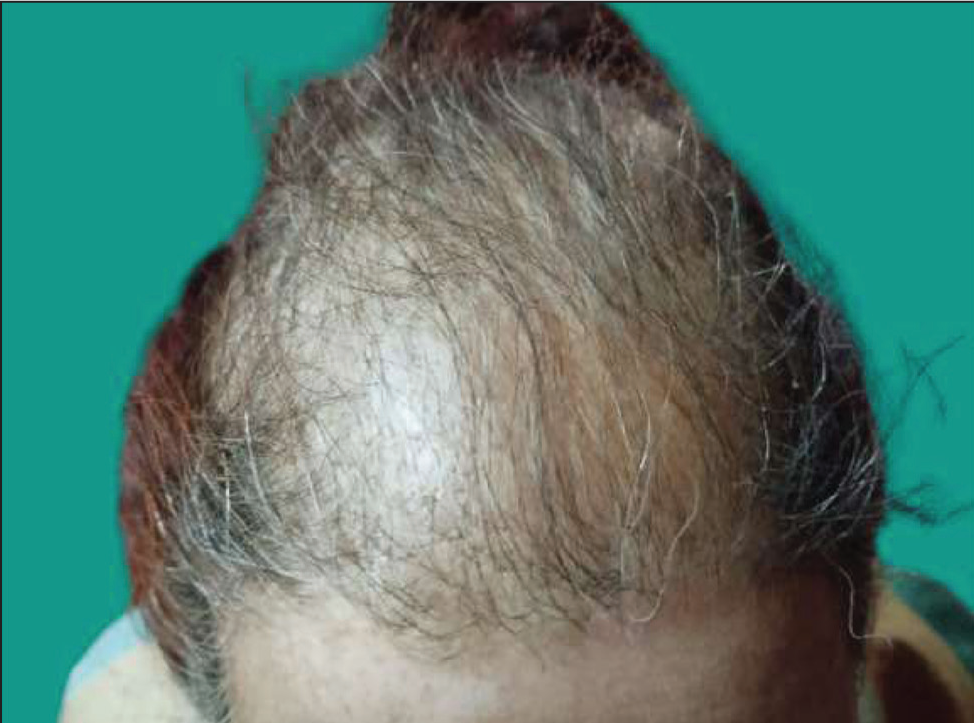
The mean onset age for postmenopausal women (Figure 2) is 56-63 years.5,7,12 Due to the condition’s slow progression, patients often struggle to recall the onset, with an average diagnostic delay of 3.4-5.3 years.5,6,17 While FFA is the leading cause of cicatricial alopecia globally, most cases are reported in Europe and North America, predominantly among Caucasians, 8 with fewer cases from Africa and even fewer from Asia,9,18 likely due to under recognition or underreporting.
Frontal Fibrosing Alopecia in a 70-year-old Female.
Aetiopathogenesis
It is a complex condition with uncertain origins, but autoimmune, genetic, and hormonal factors are thought to contribute.
10
Key mechanisms include:
Autoimmune dysfunction: A Th1-based immune response targets hair follicles, destroying stem cells and immune privilege in the bulge region. This leads to fibrosis and hair follicle loss.
19
PPAR-γ dysfunction: Downregulation of PPAR-γ, a regulator of lipid metabolism, triggers inflammation. Environmental toxins or genetic variations may impair its function, and mitochondrial dysfunction may play a role.
20
TGF-β and fibrosis: Transforming growth factor-beta (TGF-β) drives fibrosis through epithelial-mesenchymal transition, further influenced by Treg and Snail1 activity. PPAR-γ suppresses TGF-β-driven fibrosis.
18
JAK-STAT pathway: Increased JAK1/3 expression in inflammatory cells heightens IFN-mediated hair follicle destruction.
21
Neurogenic inflammation: Reduced nerve density, altered neuropeptides (substance P, cGRP), and increased mast cells contribute to inflammation.
22
AhR/KP axis and environmental triggers: Excessive photoprotection and exposure to chemicals activate the aryl hydrocarbon receptor (AhR), which suppresses PPAR-γ and affects hair follicle immunity.
23
Emerging hypotheses involve circulating microRNAs and mitochondrial dysfunction, though further research is needed to clarify these pathways. 24
Hormones
FFA is influenced by hormonal factors, particularly in postmenopausal women, due to oestrogen depletion. 5 Oestrogen regulates hair cycles and has anti-fibrotic and immunomodulatory roles, and its reduction during menopause can trigger inflammation in susceptible individuals. However, the disease often progresses despite hormone replacement therapy. Early menopause and tamoxifen (which lowers oestrogen) are linked to an increased risk, while intrauterine contraceptive devices (IUCDs) may offer protection. 25
In men, FFA has been reported after hormonal treatments for prostate cancer, and familial cases suggest hormonal influences. 4 Hormonal studies reveal mixed findings: androgen deficiency is associated with FFA, and lower levels of DHEAS, androstenedione, and FSH have been observed in women with FFA compared to controls.14,17 Additionally, abnormal oestrogen and testosterone levels may correlate with reduced disease activity. 14
Associated Diseases and Autoimmunity
FFA is associated with various autoimmune disorders in approximately 30% of cases, predominantly in females. 6 The most common association is with thyroid disorders, especially hypothyroidism (8%-44.6%).5,6,10,11 Other autoimmune conditions are depicted in (Table 1). Other linked conditions include systemic lupus erythematosus (SLE), vitiligo, atopy (40%), and various forms of lichen planus, particularly LPP in up to 25% of cases. 10 Rare associations include vulvar lichen sclerosis, lichen planus pigmentosus, and common variable immunodeficiency (CVID). 11
Autoimmune Diseases Associated with Frontal Fibrosing Alopecia.

FFA is frequently associated with skin conditions like rosacea (15%-61% prevalence) and AGA, which is more common in men (67%-83%) than women (16%-57%). 5 Cases of overlap between AGA and FFA with frontal fibrosing pattern distribution (FAPD) have also been described. 26 A unique case supporting the Renbök phenomenon highlights the suppression of FFA in a vascular nevus area. 27
Genetic Factors
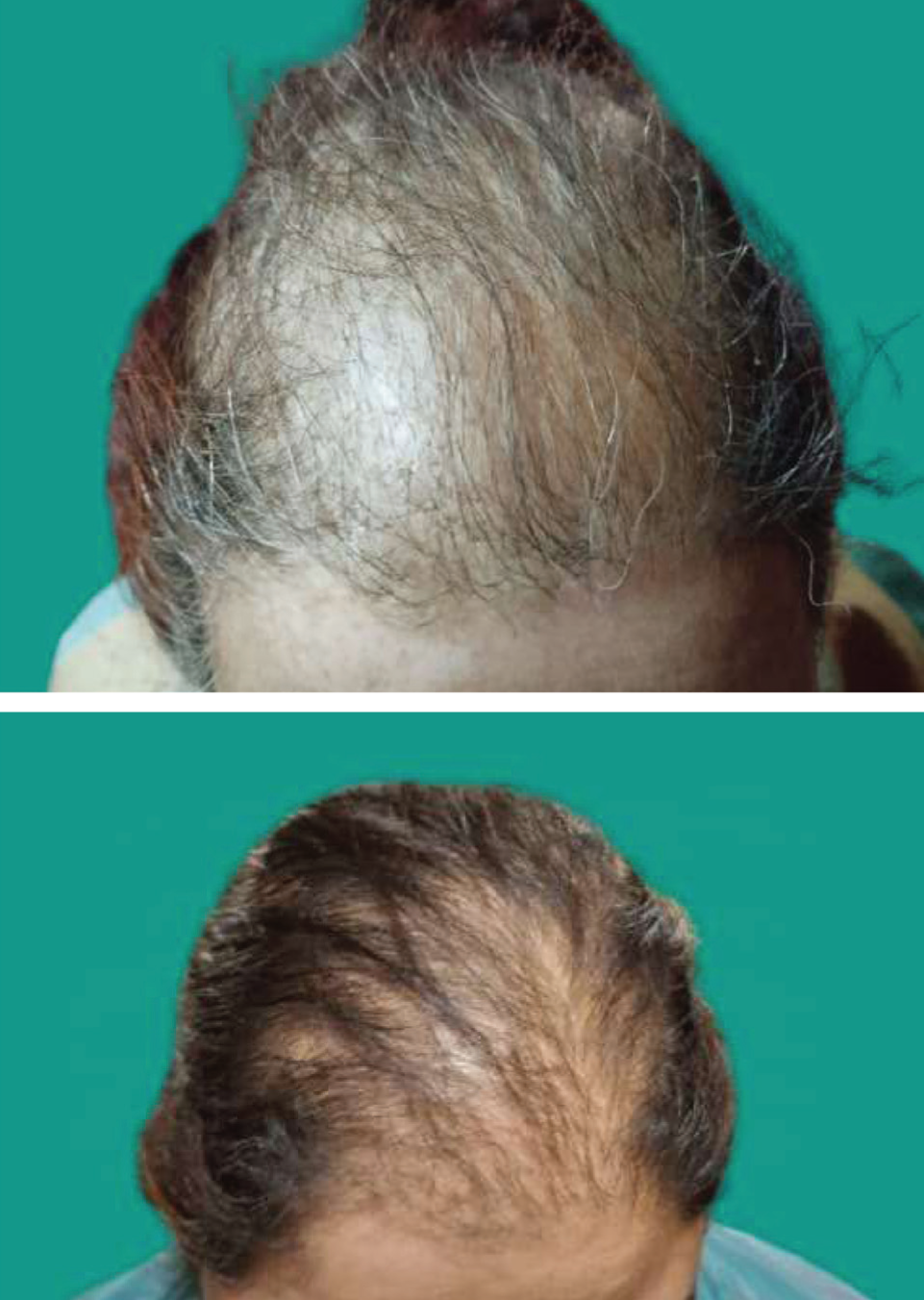
The genetic predisposition for FFA is linked to specific human leukocyte antigen (HLA) class I alleles, such as HLA-DR1 and HLA-B7. 28 Environmental factors, alongside genetic susceptibility, contribute to the disease’s development. Familial FFA cases have been reported since 2010, with a 5%-8% familial occurrence in new cases.5,8 The disease tends to start earlier in daughters of postmenopausal mothers (Figure 3), with daughters having milder symptoms. 29 An autosomal dominant inheritance pattern with reduced penetrance is proposed. Genetic markers such as the CYP21A2 mutation and specific HLA haplotypes (e.g., C07:02:01:03/B07:02:01:01) have been linked to increased FFA susceptibility. 30 Additionally, epigenetic factors are thought to play a key role in its pathogenesis, supported by studies on monozygotic twins. 31
Frontal Fibrosing Alopecia in a Mother and Daughter.
Surgical Procedures and Hair and Skin Care Products
The association of FFA with products like soaps, moisturisers, and hair straightening treatments using formalin suggests that external agents may trigger the condition. Interestingly, studies show that people with FFA tend to shampoo their hair less frequently, suggesting that regular shampooing could help remove foreign particles that might enter the hair follicle and trigger inflammation in genetically predisposed individuals. Additionally, cases of FFA have been reported after surgeries, such as hair transplants and facelifts. 32 This could be due to the Koebner phenomenon, where immune responses or inflammation triggered by the surgery contribute to the condition. 32 Similarly, FFA has been observed in areas where wigs are attached, potentially caused by this same phenomenon. 33
Sunscreen Controversy
Recent studies have increasingly linked environmental factors, including sunscreens, to the development of FFA. 34 However, no conclusive evidence establishes a direct role for sunscreens in FFA’s development. A retrospective study found higher sunscreen use in FFA patients compared to healthy controls, but some cases of FFA occurred without sunscreen use, making a causal link unclear. Sunscreens are thought to potentially trigger FFA through lichenoid reactions that disrupt follicular immunity, especially when titanium dioxide nanoparticles, commonly found in sunscreens, penetrate the skin. However, further studies have not consistently supported this theory. Other hypotheses include endocrine disruptions from chemical UV filters and exacerbation of scalp inflammation. 35 Despite some associations, no clear evidence links sunscreen use to FFA, and more research is needed to explore this potential relationship.
Medications, Drugs and Behavioural Factors
The connection between smoking and FFA is debated, with some studies suggesting smokers are less likely to develop FFA, and that quitting smoking can reduce its severity.5,6 There is no clear link between specific medications and the onset of FFA, though one study found controls used oral contraceptives more than FFA patients, 6 potentially due to selection bias. FFA has been reported in a woman treated with nilotinib for chronic myeloid leukaemia, showing symptoms like hairline regression and alopecia. 36 Diets high in buckwheat and millet may be linked to FFA, but other studies have found no connection with phytoestrogens or natural PPAR-agonists. Exposure to alkyl phenolic chemicals at work, which affect PPAR-γ and DHEA conversion, is also noted. Additionally, many FFA patients report a stressful event before its onset. 6
Clinical Features/Clinical Presentation
FFA is characterised by scarring alopecia at the frontal and/or temporoparietal hairlines, with a pale, shiny, atrophic area of hair thinning. The hairline can be identified by raising the eyebrows, revealing a sharp demarcation. In challenging cases, Wood’s light may help identify the hairline. A ‘lonely hair sign’ can also assist in diagnosis. Typically, the recession is symmetrical, 12 though asymmetry can occur. Based on hairline recession, various clinical patterns are described in the literature (Table 2). 16 The ‘clown alopecic pattern’ appears in advanced cases, with complete frontoparietal alopecia. 12 Other signs include the pseudo-fringe sign, eyebrow and eyelash loss (63%-83% and 3%-14%, respectively), and possible loss of body hair. Occipital involvement is present in 15%-30% of cases, and males may experience more body hair loss and facial papules.2,5,6 Symptoms may include itching, pain, and even anxiety and depression in older patients. 11
Typical and Unusual Patterns of Frontal Fibrosing Alopecia.

Clinical Classification and Severity Scores
The severity of FFA is classified based on the size of scar alopecia from frontotemporal hairline regression into five levels:
I: Less than 1 cm II: 1-2.99 cm III: 3-4.99 cm IV: 5-6.99 cm V: More than 7 cm (also called ‘clown alopecia’).
5
There are two main scoring systems for FFA:
FFASS (FFA Severity Score): Includes local inflammation indicators and patients’ complaints 38 (Table 4).
The Frontal Fibrosing Alopecia Severity Score (FFASS).

Clinical Course and Prognostic Factors
FFA is typically a gradual and subtle condition but can progress rapidly or be self-limited. It may stabilise or remain unchanged for varying periods, though the extent of progression before stabilisation is unpredictable.2,4,5 Hairline recession is estimated at 2–21 mm annually, 5 and erythema at the hairline suggests active disease. 4 Factors like older age at onset, higher BMI, and the presence of rosacea are linked to more severe forms. 17 Clinically, pattern III has the best prognosis, pattern II the worst, and pattern I has an intermediate prognosis. 8
Diagnosis
The criteria established for diagnosing FFA are enumerated in Table 5.16,39 It is diagnosed based on specific clinical, histopathological, and dermoscopic criteria. Histologically, it is characterised by lymphocytic infiltration around hair follicles and perifollicular fibrosis, features similar to LPP, though they can be differentiated by specific clinical manifestations.1,2
Diagnostic Criteria for Frontal Fibrosing Alopecia.

Dermoscopic findings include perifollicular erythema, follicular hyperkeratosis, 9 and broken hairs, with darker skin types showing hyperpigmentation. Imaging techniques such as optical coherence tomography (OCT) and reflectance confocal microscopy (RCM) reveal changes in the skin’s structure, including increased epidermal thickness and vascularity in active areas.40,41 Histopathologically, FFA features T-lymphocytic inflammation and destruction of sebaceous glands, with advanced stages showing fibrosis and follicular loss.1,12,42
Laboratory tests typically show normal results, though some patients may have elevated antinuclear antibodies. Further testing is usually unnecessary unless systemic illness or hyperandrogenism is suspected. 12
Differential Diagnosis
FFA is a primary cicatricial alopecia that must be differentiated from other alopecia types, such as the Ophiasis pattern of alopecia areata (AA) and traction alopecia. 12 Trichoscopy and histopathology play crucial roles in distinguishing FFA. AA may present with similar symptoms, like eyebrow loss, but lacks features such as hyperkeratosis and perifollicular erythema, which are common in FFA. 2 Traction alopecia, caused by tight hairstyles, usually does not affect the eyebrows. 2 Other scarring alopecias, like pseudopelade of Brocq and discoid lupus erythematosus, can also mimic FFA but often show multifocal alopecic areas. 2
Regarding FFA and LPP, there is debate over whether FFA is a variant of LPP or a separate condition. 5 Both share similar histopathological features, but FFA generally has lower inflammatory infiltrates and more pronounced eosinophilic necrosis at the isthmus. FFA also differs from LPP in its inflammatory response, follicular destruction, and presence of foreign body reactions. Additionally, FFA patients have less basal layer damage and more terminal catagen-telogen hairs, whereas LPP shows concentric lamellar fibroplasia and more significant epidermal damage. 43
Methods of Treatment
Hair loss typically progresses gradually, though it can sometimes advance rapidly or stabilise temporarily.3,6 The extent to which it will progress before stabilising remains unclear, and the effectiveness of treatments varies. It is uncertain whether treatments induce stabilisation or if it occurs spontaneously.2,3,5 Early treatment is essential to prevent severe follicle loss, and various therapies are used, including topical treatments, systemic agents like 5-α-reductase inhibitors, intralesional steroids, hydroxychloroquine, and others. Despite numerous therapies, there is no clear ‘gold standard’ treatment. Once follicles are destroyed, hair restoration is impossible, and treatment aims to halt hair loss and alleviate symptoms. 3 The existing data on treatments primarily comes from case reports and cohort studies.6,12 Treatment options are divided into local and systemic therapies.
Local Treatments/Topical Agents
Topical Corticosteroids (tCS)
Topical corticosteroids (tCS) are commonly used as first-line treatment for FFA but are more effective when combined with other therapies. They are best used in early inflammation but can lead to relapses once discontinued. While they lower inflammation, they do not delay alopecia. When combined with other treatments, they stabilise the condition, though prolonged use may cause side effects like skin atrophy and telangiectasia. Alternating with other topical agents like calcineurin inhibitors can help reduce these effects.2,4,6,10-13
Intralesional Corticosteroids (ILC)
ILCs are a key treatment for FFA, effectively reducing inflammation and stabilising the disease.4,5 They are particularly helpful for long-standing cases. Studies show good results with regular ILC injections, often combined with other treatments like finasteride or minoxidil. 13 While ILCs are effective, they can lead to skin atrophy, and they are less helpful once the disease progresses to the fibrotic phase. 12
Topical Calcineurin Inhibitors (tCI)
tCIs, such as tacrolimus and pimecrolimus, are effective in treating FFA by inhibiting T-cell activation.3,6 They can reduce inflammation and may work better than tCS when combined with other therapies.5,6 However, their effectiveness in halting disease progression is limited, and they can cause side effects like erythema and scaling.
Topical Minoxidil
Minoxidil is commonly used for pattern baldness and can be beneficial in cases where AGA coexists with FFA.1,2,13,44 Although minoxidil alone is not effective in halting FFA progression, it has shown promise when combined with other treatments like oral finasteride, corticosteroid injections, and hydroxychloroquine. 45
Prostaglandin Analogue
Prostaglandin analogues, such as bimatoprost, are useful for eyebrow alopecia in FFA. A study found that most patients treated with bimatoprost experienced eyebrow regrowth, and it may also be effective for eyelash loss. 33
Systemic Therapeutic Agents
5α-Reductase Inhibitors (5ARi)
5α-Reductase inhibitors, such as finasteride and dutasteride, are effective treatments for FFA. 6 These drugs inhibit the enzyme 5α-reductase, which converts testosterone into dihydrotestosterone (DHT), a hormone linked to hair loss. Finasteride, a type II inhibitor, works primarily in hair follicles, while dutasteride, a type I and II inhibitor, affects hair follicles, sweat glands, and sebaceous glands. 6 Both have been shown to stabilise or improve hair loss in FFA patients, with dutasteride being more potent.5,44 Combination therapies with corticosteroids or minoxidil can enhance results, and studies show 5ARi can stabilise hair loss in 70% of FFA cases.3,13
Hydroxychloroquine
Hydroxychloroquine, an anti-malarial drug, has shown promise in treating FFA by reducing inflammation and stabilising the condition.1,2,7 It is considered a first-line treatment for its anti-inflammatory effects, though results vary. Many studies report partial improvements, especially in the first six months, but complete regrowth is rare.5,11 Its safety profile during pregnancy adds to its appeal, though it requires regular ophthalmologic monitoring due to potential retinal toxicity. 34
Oral Tetracyclines
Tetracycline antibiotics like doxycycline and minocycline have anti-inflammatory properties that may help manage FFA symptoms.3,4 However, their effectiveness is unpredictable, 35 with some patients showing stabilisation11,44 and others experiencing side effects like gastrointestinal issues and photosensitivity. 7 They are often used in combination with other therapies, such as corticosteroids. 44
Oral Retinoids
Retinoids, like isotretinoin, are used for FFA due to their ability to reduce inflammation and promote hair follicle regeneration. 46 Studies have shown that retinoids, especially when combined with other treatments like finasteride, can improve symptoms, though they have significant side effects, including teratogenicity, and are contraindicated in pregnancy. 6
PPAR-γ Agonists (Pioglitazone)
Pioglitazone, a drug that regulates lipid metabolism and impacts hair follicle function, has been explored for FFA treatment. 47 Though early results are promising, more research is needed to confirm its effectiveness.3,47 Adverse effects like reduced bone mineral density and heart-related issues are concerns.
Other Oral Agents
Naltrexone: An opioid receptor antagonist with potential immunomodulatory effects. Limited research suggests it might help with FFA, but more studies are needed. 34
Oral Minoxidil: Low-dose minoxidil has shown potential in treating various forms of alopecia, including FFA, with promising results in improving hair thickness. 48
Overall, while there are various treatment options for FFA, 5ARi remains the most commonly prescribed and effective treatment, with other therapies used to complement or enhance results.
Systemic Immunosuppressant Agents
Methotrexate (MTX)
Methotrexate has been used in a few FFA cases with mixed results. In a Duke University trial, only one out of three patients experienced stabilisation. However, a Mayo Clinic study showed stabilisation in both of the two patients treated.11,44 MTX is generally reserved for refractory conditions due to its potentially severe side effects, including liver and kidney toxicity, gastrointestinal issues, and infections. It is also contraindicated in pregnancy (Category X). More research is needed before recommending MTX for FFA treatment.
Mycophenolate Mofetil (MMF)
MMF has limited research in FFA treatment. In one study, three out of five patients showed improvement. Another study found five out of eight patients had disease stabilisation, though three developed undesirable drug reactions.2,7,11,34 More extensive research is needed to determine MMF’s effectiveness in FFA. 48
Oral Prednisone
Oral prednisone has been tested in FFA with some success, particularly in preventing hairline recession in about 43% of patients. However, relapses often occur once treatment stops.1,2,12 A short-term dose of 25-50 mg/day for four weeks has shown temporary effectiveness, especially in rapidly progressing cases. No benefits were observed for slowly progressing cases. 2 Despite its limited benefits, intramuscular triamcinolone acetonide (40 mg every three weeks) has also been used, and improvement in facial papules has been noted with oral prednisone combined with antimalarials. 49
Other Therapies
Platelet-rich Plasma (PRP)
PRP, a blood-derived product rich in growth factors, has been explored in treating hair loss, including and AGA and LPP. 50 While data supporting PRP’s effectiveness is limited, it has shown promise when combined with other treatments for AGA and LPP, particularly in improving hair thickness. 50 However, for FFA, PRP’s benefits remain unclear, and its use should be discussed carefully with patients due to cost and limited evidence of effectiveness.
Low-level Lasers
Excimer laser: This UV-B laser has been used to treat inflammatory conditions like AA and psoriasis. 51 It may be helpful for active inflammatory stages of FFA,8,25 although more randomised trials are needed to confirm its effectiveness.
Light-emitting diodes (LEDs): Superluminescent diodes are a form of photobiomodulation showing potential in treating cicatricial alopecia. 52 A small study suggested benefits for FFA patients, including reduced symptoms and increased hair growth, 52 but more research is needed.
Hair Transplantation
Hair transplants in FFA are controversial and typically only considered after the disease is stable for at least one to five years. 53 Transplanted hair often fails due to disease progression, and graft survival rates drop over time.34,53 Some studies report short-term success, but long-term results are poor, with many grafts lost within three to four years.34,53 Careful patient selection and realistic expectations are essential, as long-term success is uncommon. 3
Other Treatments
The effectiveness of therapies like azathioprine and griseofulvin for FFA has been inconsistent or absent.1,2,7,13,44,49 Limited anecdotal evidence suggests that treatments such as cyclosporine, griseofulvin, and others did not slow alopecia progression. 12 However, two FFA patients showed clinical improvement with oral tofacitinib (a pan-JAK inhibitor) over 2-19 months. 54 Additionally, a woman with refractory FFA and LPP showed improvement after receiving tildrakizumab (an anti-IL23 monoclonal antibody). 55 A case report also noted hair regrowth in an LPP-affected area following adalimumab treatment for rheumatoid arthritis and hidradenitis suppurativa. 56
Conclusions
In recent years, there has been growing interest in FFA, focusing on its characterisation, causes, and treatment. Most studies so far have been observational, and more research is needed to explore environmental triggers versus genetic factors. It is suggested that FFA may develop in genetically predisposed individuals due to factors like face creams, sunscreens, environmental pollutants, surgery, or stress. Hormonal and immune factors likely play a role as well. Research into cosmetics and environmental influences on FFA, particularly in different geographical areas, could offer new insights. Although some patients have responded well to 5ARi, further investigation into novel treatments, such as biological therapies or oral minoxidil, is needed.
Footnotes
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author received no financial support for the research, authorship and/or publication of this article.

