Abstract
Background
Vertebral artery injury is a rare but potentially catastrophic complication of cervical spine surgery. Its low incidence belies its importance, given the risk of severe neurological outcomes or death. As surgical techniques evolve and the complexity of cervical procedures increases, a comprehensive understanding of vertebral artery injury is essential for safe practice.
Objective
To provide an in-depth review of vertebral artery anatomy, radiologic evaluation, preoperative planning, and intra and postoperative management of vertebral artery injuries, with a focus on current techniques to mitigate complications and improve patient outcomes.
Methods
A narrative literature review was conducted with articles selected based on relevance to cervical spine surgery, including anatomical considerations, incidence, mechanisms of injury, clinical consequences, diagnostic approaches, treatment strategies, and preventive measures.
Results
Vertebral artery injury most commonly occurs at high-risk anatomical locations, particularly at C1–C2. Prompt recognition and appropriate imaging are critical for diagnosis. Management strategies vary from conservative anticoagulation to endovascular repair or surgical ligation, depending on severity and risk for developing complications. Preoperative planning with imaging is important to reducing the risk of iatrogenic vertebral artery injury.
Conclusion
Vertebral artery injury remains a significant concern in cervical spine surgery. Early identification, appropriate imaging, and individualized management are essential to minimizing morbidity and improving surgical outcomes.
Keywords
Introduction
The vertebral artery (VA) plays a crucial role in the blood supply to the brain, accounting for approximately 28% of total cerebral circulation. 1 Its anatomical complexity and variability make it a significant point of concern for cervical spinal surgery, where vertebral artery injury (VAI) can result in catastrophic outcomes. Several recent reviews have covered various aspects of VAI, such as detailed segmental anatomy, 2 outcomes, 3 and epidemiological patterns. 4 The current review, however, offers distinct contributions to existing literature. Specifically, the emphasis on practical clinical insights through illustrative case examples demonstrate how anatomic variations directly impact surgical decision-making. Additionally, this review offers a discussion on updated trauma screening criteria, including the recent algorithm from Fourman et al, highlighting its potential advantages in assessing posterior circulation stroke risk following cervical spine injuries. 5 Finally, this preview presents a detailed synthesis of contemporary intraoperative management approaches and endovascular interventions, with particular attention to the challenges posed by dominant vertebral arteries. Through these elements, this review aims to serve as a comprehensive and clinically relevant guide for surgeons managing VAI.
Anatomy of the Cervical Spine and Vertebral Arteries
The vertebral arteries originate from the subclavian arteries just above the first rib, ascending through distinct anatomical segments before supplying critical regions of the brain and spinal cord.1,6 Anatomically, the VA is traditionally divided into four segments: pre-foraminal (V1), foraminal (V2), atlantic or extraspinal (V3), and intracranial (V4). The V1 segment extends from the subclavian artery to the transverse foramen of C6. The V2 segment then ascends vertically through the transverse foramina from C6 to C2. After exiting the transverse foramen of C2, the V3 segment curves posterolaterally around the atlas, traversing the suboccipital triangle before penetrating the dura at the foramen magnum. Finally, the V4 segment travels intradurally along the ventral surface of the medulla, joining the contralateral artery to form the basilar artery. In addition to supplying posterior cerebral circulation, the vertebral artery provides essential branches including the anterior spinal artery, muscular branches to cervical musculature, and the posterior inferior cerebellar artery supplying the cerebellum and fourth ventricle.
Anatomic Variants
The vertebral artery is well known for several anatomical variations which pose challenges for surgical intervention and management of traumatic vertebral artery injury (VAI).7,8 Certain anatomical variations have been identified as a major risk factor for iatrogenic VAI, with Akinduro et al reporting that 10% of VAI cases exhibited anomalous anatomy.
9
The most common variation in VA anatomy is the existence of a dominant VA, defined as having one artery with larger diameter than the other, more often the left VA (∼45%).
10
VA dominance has been implicated as a risk factor in pontine and PICA infarcts.
10
The degree of dominance is an important factor in considering approach laterality and risk of iatrogenic ligation of the dominant side. Hypoplasia of the VA, seen in 3.2% of patients and defined as a VA diameter <2 mm, can be seen as an extreme form of dominance which carries the same risks and considerations.
11
High-riding VAs, defined as having C2 isthmus heights of <=5 mm or <2 mm if measured 3 mm from the spinal canal, are found in 25.3% of the population with notably higher incidence in rheumatoid arthritis patients.
12
Extraforaminal VA anomalies involve entry of the VA into the course of the transverse foramina at a level other than C6, with the most common variant having the VA run anterior to the transverse foramina at C6 before entering at C5 (Figure 1).
7
Other variations of the V2 segment are possible, including looping out of the foramina after entry (Figure 2), and may increase risk of iatrogenic injury if the VA is unexpectedly in the surgical approach. Extraforaminal VA anomaly determined through MRI imaging. Extraforaminal VA anomaly whereby the VA courses anterior to the transverse foramen of C6 before entering its typical course at C5. MRI slice above the level of C6 shows the left VA running anterior to its typical course while the right VA shows no displacement. Created in BioRender. Bilodeau, R. (2025) https://BioRender.com/d3933at. Case of V2 segment variation determined through MRI imaging. In this patient, the right V2 segment at C4 loops around the transverse foramen. The left VA remains in its typical course. Created in BioRender. Bilodeau, R. (2025) https://BioRender.com/gkqoi0f.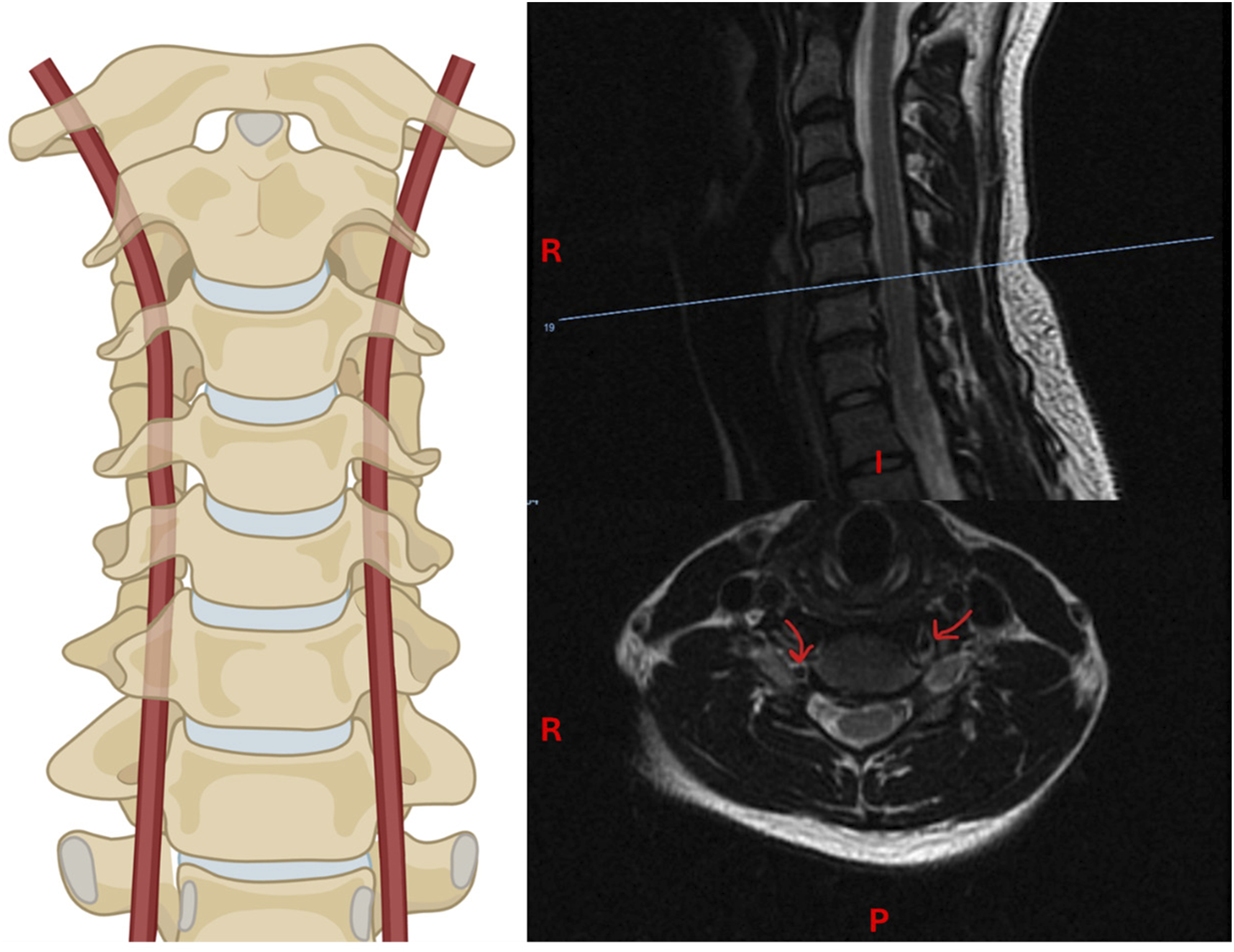

There are other less common anatomical anomalies of the VA which are still highly relevant to cervical spine procedures as they are associated with higher risk of iatrogenic VAI.
8
Persistent first intersegmental artery, where the V3 segment of the VA courses through the spinal canal beginning above the C2 level, is observed in 1.8% of patients.8,13 Fenestrated VA, where the artery divides into two segments which later rejoin, is observed in the V3 segment for 0.7% of patients and early emergence of the posterior inferior cerebellar artery from the V3 segment of the VA is seen in 1.6%.8,13 Figure 3A depicts a possible anomaly with features of both persistent first intersegmental artery and fenestrated artery (Figure 3). Prevalence of these anomalies is reported to be higher in Asian populations.
14
Patients may also have anomalous origin of the VA, with the artery arising directly from the aorta rather than the subclavian artery. An occurrence of this anomaly from the left side is depicted in Figure 3B (Figure 3B). Ponticulus posticulus (PP) is a variant of the atlas whereby a bony bridge connects the superior articular process to the posterolateral posterior arch, forming a foramen through which the VA and other structures may course.
15
The prevalence of PP is 16.7% in the overall population and in some cases, it can cause headache and symptoms of vertebrobasilar insufficiency via compression of the VA.
16
In surgical cases involving C1 lateral mass screw placement, an unidentified PP can obscure the VA and be mistaken for the superior portion of the posterior arch, resulting in drilling or placing a screw into the PP and damaging the VA. Preoperative knowledge of PP and planning of screw trajectories is essential for avoiding this injury.
15
Other possible vertebral artery anomalies. (A) Depicts a persistent first intersegmental artery with concurrent with fenestrated VA at the level of C1. (B) Depicts instance of anomalous left VA origin from the aorta. Created in BioRender. Bilodeau, R. (2025) https://BioRender.com/97lpbge, https://BioRender.com/g8cnkcw.
Case Example: VA Anatomic Variants
Case derived from Abtahi et al: 13 A 55-year-old man sustained an unstable fracture of the anterior ring of C1 and synchondrosis of the posterior arch of C1 after falling 15 feet from a ladder. 13 CT angiography revealed the presence of a dominant right vertebral artery characterized as a persistent first intersegmental artery. As the anomalous artery was positioned posterior to the caudal C1 lateral mass, placement of a C1 lateral mass screw was not possible on the right side. Knowing this, the surgical team planned to perform a C3 to occiput fusion instead of C1–C2 fusion. An occiput plate was placed, affixed to C2 using pars screws, and affixed to C3 using lateral mass screws. The operation was successful, and a 6-month post-operative evaluation revealed no injury to the VA.
Diagnostic Evaluation of Vertebral Arteries
Imaging Modalities
Digital subtraction angiography (DSA) has historically been considered the gold standard for vertebral artery evaluation and screening for trauma-related VAI due to its temporal resolution and high sensitivity. However, DSA presents issues in its cost and invasiveness, limiting its use in smaller institutions, risk of stroke and vessel injury, and risk of missing certain cases of non-stenotic intramural hemorrhage as it only images the vascular lumen. 17 With the development of fine-cut computed tomography (CT), use of DSA for diagnosis of VAI has now been largely replaced by CTA, which is less invasive, carries lower risk complications, and has reported sensitivity of up to 98% compared to DSA.17,18 However, in common practice, the sensitivity of CTA varies depending on facility equipment and operator training, and DSA is used for patients with inconclusive or negative CTA where VAI is still suspected or to assess collateral circulation.17,19 CTA may also be complicated by metal hardware, atherosclerotic plaques, or anomalous anatomy, which may lead to image artifact or be mistaken for VAI. 17 Magnetic resonance angiography is not commonly used to screen for VAI as it is less sensitive than both DSA and CTA, though non-contrast MRI can be used for patients with contraindications to contrast such as in the setting of severe renal disease. 20 It can also be run simultaneously with magnetic resonance imaging for the detection of cerebral infarction, for which VAI is a significant risk factor, if infarction is suspected. 21
Screening Recommendation Criteria after Traumatic Injury
Following blunt cervical trauma, patients with VAI may be asymptomatic until neurological deficits appear, making early screening of at-risk patients vital. For patients undergoing cervical surgery after traumatic injury, identifying factors that predict VA damage can help surgical teams minimize the risk of intraoperative injury and hemorrhage. Many guidelines have been developed to determine which patients are most at risk for VAI following blunt cervical trauma. The American College of Surgeons Committee on Trauma Advanced Trauma and Life Support (ATLS) has published guidelines which recommend CTA screening for C1–C3 cervical fractures, cervical fractures involving the foramen transversarium, and cervical subluxation. 22 The Denver criteria considers the mechanism of injury in conjunction with risk factors, including cervical spine fracture or subluxation, complex skull fracture, Le Fort II or III (midface) fracture, traumatic brain injury with Glasgow coma scale score <6, and hanging as the mechanism of injury. 23 Most institutions currently use one of the many iterations of the Denver criteria that have since been developed, expanding the risk factors to include seat belt injury, thoracic vascular injury, upper rib fracture, scalp degloving, or blunt cardiac rupture. 23 However, the effectiveness of these additions in improving the identification of vessel injury remains debated. Wagner et al recently reported that cerebrovascular injury was significantly associated only with the original Denver criteria, whereas the expanded and modified criterion sets did not demonstrate a significant correlation. 24 Fourman et al developed an alternative criterion which aims only to identify cervical trauma patients based on their likelihood of developing posterior circulation stroke and found their criteria superior to the Denver criteria for the detection of stroke risk and prevention of unneeded CTA. 5 Their screening protocol included major selection criteria: high energy trauma, loss of consciousness at time of trauma, and altered mental state or inability to protect the airway. 5 If one of these criteria was positive, CTA was only recommended if the patient had fractures involving the transverse foramen, bilateral facets, combined C1 and C2 fractures, or facet dislocations.
In addition to blunt cervical trauma, vertebral artery injury or occlusion can also be associated with degenerative spine conditions such as spondylosis, where dissection or damage to the intima from repeated compression can lead to thrombus formation and posterior circulation strokes. Compression of the VA by spondylotic inflammation, osteophytes, or disk displacement can also lead to vertebrobasilar insufficiency and cerebellar infarction when the head is kept rotated for a long period, such as while sleeping, though this is rare.25,26 In many cases, slow progression of degenerative disease allows for the formation of collateral blood supply from the anterior spinal, vertebrobasilar, or cervical vessels to reconstitute the VA above the level of compression or stenosis.27,28 During a posterior surgical approach, it is possible that cervical collaterals are disrupted, leading to vertebrobasilar insufficiency or infarction. For patients presenting with posterior circulation stroke with a history of cervical spine degeneration, MR angiography allows screening for VA compression, though DSA or dynamic CT angiography in different head positions is needed for confirmation. 26
Preoperative Imaging
Mapping a patient’s VA anatomy with preoperative imaging is crucial to reducing the risk of iatrogenic VAI. 29 Generally, the presence of VA anomaly at the V3 level predisposes patients to increased risk of VAI during drilling or insertion of lateral mass screws; therefore, preoperative CTA is recommended when anomalous anatomy is suspected. 9 With high-riding VAs, a shortened C2 ithsmus and proximity of the VA increases the risk of VAI during placement of C1–C2 transarticular or C2 pedicle screws, and it is recommended to use ipsilateral translaminar screws when possible.12,30
Patients with dominant VA or hypoplasia of the VA on one side are at increased risk of neurological complications with injury to the dominant side, as the non-dominant or hypoplastic side is not able to provide sufficient collateral flow.29,31 Similarly, these anomalies complicate ligation and embolization of the dominant VA if injured intraoperatively, and understanding of the patient’s specific anatomy becomes critical in adjusting treatment. Since injury to the dominant VA is more consequential and the left VA is often dominant, the right-sided anterior approach is technically safer when arterial anatomy is unknown or if appropriate imaging is inconclusive.10,32 In practice, most surgeons will not change their approach due to dominance due to the low overall risk of injury. However, knowledge of VA positioning may inform surgeons to be more cautious during certain parts of the approach. For example, if the V2 segment of the VA lies completely outside of the transverse foramina, care must be taken during retraction of the longus colli on anterior approach. 33
For patients with cervical degeneration, preoperative MRI can reveal changes to landmarks or bone structure which offset the VA from its usual course. For example, it was found that the distance between the VA and uncinate process was shortened in cases of cervical disc degeneration, exposing the VA to risk of injury during corpectomy or lateral decompression techniques. 34
Surgical Approaches to the Cervical Spine and Comparative Risk of Vertebral Artery Injury
Iatrogenic vertebral artery injury is exceedingly rare in most cervical spine procedures, with an overall reported incidence of 0.07%. 35 However, the risk of VAI is highly dependent on the type of procedure, approach used, and, most significantly, the experience of the surgeon. 35
Anterior Approach
The anterior approach is commonly used in subaxial cervical spine surgery and is often the preferred procedure for management of cervical disc disease and other degenerative conditions which lead to cervical myelopathy at C3–C7 levels. 32 A left-sided anterior approach is typically used due to the anatomical consideration of the recurrent laryngeal nerve. The right recurrent laryngeal nerve is more superficial, shorter, and less predictable compared to the left side. However, right-sided VAs are more likely to be non-dominant, potentially reducing the risk of posterior circulation stroke in the event of right-side VAI. 10 The difference in recurrent laryngeal nerve injury for right-sided approach has not been significant, and many right-handed surgeons have preferred right-sided approach. Many studies have analyzed the risk of VAI for the anterior surgical approach and the reported rates are typically <0.5%.32,36 For anterior procedures, occurrence of VAI is commonly attributed to drilling off midline, excessive lateral bone disk removal, excessive lateral placement of instrumentation, instrumentation failure, loss of midline landmarks, or abnormal VA anatomy. 29 The reported rate of VAI is higher for cervical corpectomy compared to ACDF, likely due to the higher complexity, morbidity, and greater exposure required of the corpectomy procedure. 36
Posterior Approach
The posterior approach is useful for surgery in the C1–C2 region and for certain indications including injury to the posterior ligamentous structures and for direct decompression options. 32 For approach to C1–C2, a dissection extending at most 2 cm lateral to midline should be sufficient for the placement of C1 pedicle screws and avoids risk to VA and other structures.37,38 The risk of VAI during the posterior approach varies across studies and procedures. For posterior screw fixation in the subaxial region, there are exceedingly few cases of VAI, although screw fixation at the C1–C2 level is associated with a 1.3% to 8.2% risk of VAI. 8 One study linked individual transarticular, C1 lateral mass, or C2 pedicle screw placement to increased risk of VAI, reporting increased risk of 1% for every screw placed. 39 The heightened risk of VAI during C1–C2 screw placement is partially attributable to the increased incidence of anatomical variants of the VA in this area, with one study reporting the presence of high-riding VA as the most significant risk factor for VAI. 40 Because the screw trajectory can be elusive during posterior placement, preoperative understanding of VA anatomical variants is especially important for this approach.
Current Management Strategies
Intraoperative Management
Intraoperative VAI typically presents with pulsatile, high-pressure bleeding, most often during drilling or screw placement. Following initial identification of an intraoperative VAI, bleeding is controlled using direct tamponade while hemostatic agents and fluid replacement are judiciously used to prevent hypoperfusion of the spinal cord or posterior circulation. 4 The anesthesiologist should be informed immediately to begin preparing for transfusion and/or chemical resuscitation and vascular surgery should be consulted if available. If no vascular surgeon or endovascular team is available, transport to a level 1 center with these teams should be considered. Use of electrocautery for control of bleeding has been found to be ineffective and can potentially damage cervical nerve roots. 41 In cases of VAI while drilling for C1–C2 transarticular screw placement, hemostasis is often achieved by screw placement or application of bone wax. 42 In practice, direct tamponade is the most common method of primary management, having been used by 76% of surgeons, followed by ligation (29%), direct repair (13%), embolization (12%), screw placement (7%), and stenting (6%). 35
Once hemostasis is achieved, management focuses on direct repair, anastomosis, clipping, ligation, or tamponade with staged endovascular repair or embolization. Primary repair of the VA minimizes the risk of delayed hemorrhage and vertebrobasilar ischemia but is often technically difficult due to the course of the VA and proximity to venous structures. 41 Temporary clipping of the proximal and distal VA followed by anastomosis with artificial vessel is another option to preserve vertebrobasilar flow. 43 If tamponade, screw or bone wax placement, direct repair, or anastomosis are used, immediate postoperative angiography should be used to confirm patency of the repaired vessel and detect any vascular complications. 42 Immediate postoperative imaging helps mitigate the risk of delayed hemorrhage or arteriovenous (AV) fistula formation; however, pseudoaneurysm formation may manifest sub acutely or long after surgery. Follow-up CT angiography is recommended, even in asymptomatic patients.42,44,45
Management strategies for intraoperative VAI are summarized in Figure 4. A preoperative or intraoperative angiogram is crucial for determining the optimal treatment approach by assessing the adequacy of blood flow from the contralateral VA. Permanent occlusion via ligation or clipping can lead to cerebellar infarction and is associated with high mortality, hemiplegia, and cranial nerve paresis, especially for ligation of a dominant VA.
42
If an angiogram confirms that the contralateral vertebral artery (VA) is not hypoplastic and can provide sufficient collateral circulation, clipping or ligation of the injured VA can effectively control uncontrolled bleeding and prevent delayed hemorrhage or pseudoaneurysm formation when repair is not feasible.
42
To minimize risk of steal from collateral circulation, it is essential to clip/ligate both the distal and proximal portions of the injured vessel. At this point, the decision to proceed with the planned surgery or to abort depends on the success of hemostatic measures and the patient’s condition. Consulting with available vascular or ENT surgeons to evaluate the success of hemostasis and assess the risk of further injury can be beneficial. If hemostasis cannot be achieved or patient stability is compromised, the procedure should be halted, and surgical intervention should be reassessed after managing the vascular injury. Flowchart detailing process for management of intraoperative vertebral artery injury.
If endovascular staff is available, intraoperative angiogram can be used to evaluate the status of the injured vessel and contralateral flow in real time. If contralateral flow is sufficient, coil embolization can be performed by endovascular staff to completely occlude flow to the injured site, thereby achieving hemostasis and preventing recurrent hemorrhage.4,46 Ensuring embolization of both the proximal and distal sides of the injury is crucial to prevent hemorrhage from retrograde flow. The placement of a covered stent at the site of injury is another option to control bleeding while maintaining flow through the VA. 47 Stents may also be placed to treat gross dissections or aneurysm formations but are generally more useful in the V1 and V2 sections as the vessel curves sharply at V3 and placement may be anatomically challenging.
Case Example
Case derived from Jung et al: 48 A patient with cervical spondylosis and central stenosis at C4–C7 and right foraminal disc herniation at C5–C6 underwent successful decompressive laminectomy but had persistent right arm pain. He then underwent ACDF of C5–C6 from a right-sided anterior approach, during which the VA was injured, and pulsatile high-pressure bleeding was encountered while drilling in the right medial uncinate process of C5–C6. Hemostatic agents were used to temporarily attain hemostasis while the procedure was completed, and the wound closed. The patient was transferred to the interventional radiology suite for angiography, which revealed a small pseudoaneurysm in the V2 segment of the right VA. The collateral flow from the left VA was assessed and found to be sufficient, and the interventional team decided to perform coil embolization of the injured VA. An additional angiogram was performed to assess the success of the embolization and to ensure collateral flow. CT scans of the neck and brain were performed immediately after embolization and showed no evidence of hemorrhage or infarction. However, at 2-week follow up, CT and MRI showed a right cerebellar infarction. The patient was given anti-coagulation therapy and had no neurological deficits at 3 months postoperatively.
Postoperative Care and Follow-Up
Vascular complications, including delayed hemorrhage, pseudoaneurysm, AV fistula, and vertebrobasilar thromboembolism, can occur regardless of VAI management protocol.3,44,45,48,49 Pseudoaneurysm formation or dissection can be asymptomatic or lead to delayed hemorrhage with hypotension, neck swelling, dyspnea, or cervical bruit from arteriovenous fistula. 29 Postoperative angiography is recommended to promptly identify dissection, pseudoaneurysm, or persistent extravasation, particularly when primary management involved direct tamponade or repair, and intraoperative angiography was not performed.41,42,50 Patients should be admitted to the intensive care unit postoperatively so that neurologic function and hemodynamics can be closely monitored. Long-term follow-up with CT or MR angiography is recommended, even with a normal initial scan, as hemorrhage or pseudoaneurysm may develop days to weeks after initial injury.51-53 If continued vascular complications are detected, availability of collateral flow can be assessed. If needed, endovascular stent placement, embolization, or reoperation can be performed based on injury characteristics. Due to risk of thromboembolism, the patient’s neurological function should be closely monitored, and anticoagulant/antiplatelet therapy should be started if deficits are detected or if high risk thromboembolism is suspected.42,49 Anticoagulation therapy is contraindicated for patients with major intracranial infarctions or intraspinal lesions due to the elevated risk of exacerbation.
Outcomes for patients with intraoperative VAI are varied due to severity of injury and management strategy. One report of 110 VAI cases found that 90% of patients recovered with no lasting deficits, 5.5% had cerebellar infarcts, and 4.5% did not survive. 35 Another reporting of 69 patients found a mortality rate of 11.6% and that 73.9 % of patients improved neurologically from their initial deficits. 3
Case Example
Case derived from Prabhu et al: 49 31-year-old male presenting with trauma-related, unstable os odontoideum for which C1–C2 transarticular screw fixation was indicated. 47 During drilling for the left screw placement, copious, pulsatile welling of bright red blood indicated damage to the left VA. The screw hole was packed with bone wax and gelatin sponge powder leading to hemostasis. An angiography was performed immediately postoperatively which showed a 5 mm sharply marginated pseudoaneurysm. IV heparin was administered 6 h postoperatively and was bridged to long-term anticoagulation therapy with oral warfarin. At 4-week follow-up, repeat angiography showed the aneurysm had increased to 7 mm. Subsequently, endovascular coiling was attempted but failed due to the location of the pseudoaneurysm. Collateral circulation was evaluated, revealing a co-dominant right vertebral artery (VA), which enabled the team to safely sacrifice the left VA through coil embolization both distal and proximal to the aneurysm. A follow-up angiogram confirmed successful embolization and sufficient collateral flow from the right VA.
Conclusion
Vertebral artery injury (VAI) poses a rare but serious risk during cervical spine surgery due to the critical role of the vertebral arteries. Preoperative imaging (particularly CTA) to assess anatomical variations and VA condition reduces risk of iatrogenic injury and helps with intraoperative decision-making. The posterior cervical approach, particularly during instrumentation of C1 and C2, carries a higher VAI risk. In the event of an injury, hemostasis should be achieved first, followed by the appropriate repair technique. Postoperative imaging should be performed promptly, with plans in place for any necessary secondary interventions. When primary repair and hemorrhage control become problematic for the spine surgeon, the availability of an endovascular team becomes paramount, as endovascular techniques are effective in controlling hemorrhage and reducing the incidence of major complications. Due to the variability in the anatomical course and dominance of the vertebral arteries, injury prevention and management strategies must be tailored to each patient. Careful preoperative planning, intraoperative mitigation techniques, and close follow-up practices are essential for achieving optimal outcomes.
Footnotes
Acknowledgments
We acknowledge Robert E. Bilodeau, BS, for creating the depictions of vertebral artery anomalies included in this work.
Author Contributions
Conceptualization: NK, RR, CG, CN, RW, MJS, YC, JYL; Investigation: NK, VG, RZ, MS; Project administration: RR, CG; Supervision: RW, MJS, YC, JYL; Writing—original draft: NK, VG, RZ; Writing—review & editing: NK, RR, CG, CN, MS, RW, MJS, YC, JYL.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
