Abstract
Advances in biomaterials and drug delivery systems are redefining modern dentistry, moving beyond traditional restorative approaches toward biologically guided regeneration. This review highlights the development of smart, multifunctional platforms such as nanoparticles, hydrogels, scaffolds, and additive manufacturing (AM) technologies that enable localized, controlled therapeutic delivery while promoting tissue repair. These innovations are transforming treatments across periodontics, oral oncology, endodontics, and pediatric care by offering site-specific action and enhanced biocompatibility. Stimuli-responsive materials and dual-function scaffolds are engineered to respond to environmental cues such as pH or enzymatic activity, providing on-demand drug release and supporting healing in dynamic oral environments. AM technologies further allow for the fabrication of patient-specific constructs with optimized architecture and mechanical properties.
Despite promising preclinical and early clinical outcomes, several challenges persist, including concerns regarding long-term safety, biocompatibility, and regulatory complexities, particularly for nano-enabled and multifunctional systems. Standardized toxicity assessments and harmonized approval pathways are needed to facilitate clinical translation. This review critically examines current material strategies, technological platforms, and their therapeutic applications in dental medicine and addresses emerging safety considerations and regulatory frameworks. Collectively, these innovations represent a shift toward more personalized, regenerative, and minimally invasive oral healthcare solutions.
Keywords
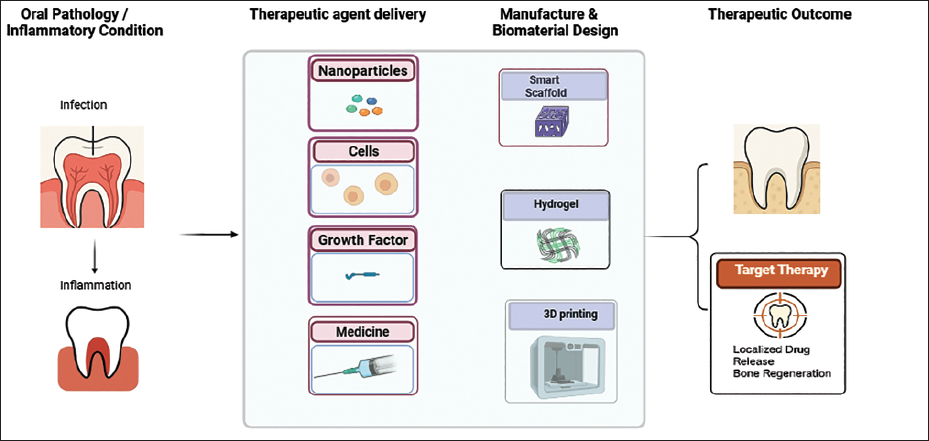
Introduction
The landscape of dentistry is being reshaped by the rapid evolution of biomaterials and drug delivery technologies. Traditionally focused on mechanical restorations, the field now increasingly embraces regenerative strategies, shifting the paradigm from repair to regeneration. 1 In this transformation, biomaterials are no longer passive structural components but functional tools that actively guide healing, control infections, and modulate immune responses. Bone regeneration plays a central role in this shift, particularly in addressing conditions like infections, tumors, and trauma. 2 However, natural healing is often hindered by large defects, poor vascularization, and systemic diseases. While autografts are still considered the gold standard due to their biocompatibility and osteoinductive potential, they come with drawbacks such as limited availability and donor site morbidity. Synthetic grafts, though structurally supportive, often lack the biological signaling needed for effective regeneration. 2 These challenges have fueled the development of tissue engineering approaches that integrate biodegradable scaffolds with cell recruitment and bioactive cues to enhance bone repair and regeneration. 3 Recent advances in bioengineering have enabled the development of smart, multifunctional materials that respond to environmental stimuli, support tissue regeneration, and deliver therapeutic agents with precision. These include nanoparticles, hydrogels, composite scaffolds, and stimuli-responsive systems that release drugs in a controlled and site-specific manner. Such innovations are particularly crucial in the oral cavity, where complex anatomical structures, constant mechanical stress, and a dynamic microbial environment present unique challenges for both treatment efficacy and patient compliance. 4 In parallel, the application of advanced therapy medicinal products, including cell-based and gene therapies, has opened new frontiers in regenerative dentistry. Stem cells sourced from dental pulp, periodontal ligaments, and apical papilla are now being explored for their potential to regenerate complex oral tissues such as dentin-pulp complexes, periodontal structures, and even entire teeth. However, while preclinical results are promising, clinical translation remains hindered by safety, regulatory, and cost-related challenges. 5
The integration of biocompatible materials into drug delivery systems offers several advantages over conventional therapeutic strategies. Natural polymers like chitosan and alginate, and synthetic polymers such as PLGA and polycaprolactone (PCL), have demonstrated the ability to prolong drug release, enhance bioavailability, and minimize systemic side effects. These materials can be formulated into hydrogels, microspheres, or nanocarriers tailored for the unique demands of oral and dental applications, including infection control, pain management, cancer therapy, and post-surgical healing. 6
Moreover, additive manufacturing (AM) (3D printing) and biofabrication technologies are facilitating the design of patient-specific scaffolds with biomimetic architecture, paving the way for personalized regenerative therapies. Incorporating drug-loaded nanoparticles into these constructs enables dual functionality, simultaneous structural support and therapeutic delivery.7, 8
The aim of this review is to explore and critically evaluate recent innovations in biomaterials and drug delivery systems tailored for dental applications, with a particular focus on regenerative medicine. By examining a wide range of platforms, including nanoparticles, hydrogels, scaffolds, and AM approaches, the study seeks to highlight how these technologies are transforming traditional dental treatments into more personalized, site-specific, and biologically integrated solutions. Special attention is given to the dual functionality of these materials in promoting tissue regeneration while providing therapeutic delivery, as well as the regulatory and safety considerations that accompany their clinical translation.
Methods and Materials
A narrative literature search was conducted using PubMed, Scopus, Web of Science, and Google Scholar, covering publications from 2009 to 2024. Key search terms included “dental biomaterials,” “drug delivery systems,” “oral tissue engineering,” “regenerative dentistry,” “nanoparticles,” “hydrogels,” “scaffolds,” “additive manufacturing,” and “oral drug delivery.” Peer-reviewed articles, including one relevant systematic review, were included based on their relevance to dental and oral applications. This review synthesizes current evidence and emerging trends but does not follow a systematic review protocol.
Smart Drug Delivery Platforms in Dentistry
Modern drug delivery platforms in dentistry are rapidly evolving to incorporate “smart” or stimulus-responsive materials that react to local environmental conditions such as pH, oxidative stress, or enzymatic activity. 9 These adaptive systems enable precise, on-demand release of therapeutics, especially important in infected or inflamed periodontal and endodontic sites, where traditional systems often fail to maintain effective concentrations. 10
For instance, MMP-sensitive hydrogels and reactive oxygen species (ROS)-responsive polymer matrixes have demonstrated success in selectively releasing antimicrobials like chlorhexidine or ciprofloxacin when triggered by inflammation-associated enzymes or oxidative stress. These approaches reduce unnecessary drug exposure and improve treatment outcomes. 11
Functional Scaffolds for Regeneration and Targeted Therapy
Scaffolds used in oral tissue regeneration are no longer passive structures. They are now engineered as multifunctional systems capable of supporting tissue architecture, delivering drugs, and promoting biological healing.
Composite scaffolds made from PLGA, β-tricalcium phosphate (β-TCP), HA, and silk fibroin have been widely explored as carriers for osteogenic agents like bone morphogenetic proteins (BMPs) and simvastatin. These bioactive constructs stimulate angiogenesis, osteogenesis, and control infection simultaneously. Notably, PLGA scaffolds loaded with simvastatin enhanced BMP-2 expression and vascularization in periodontal defects, while remaining biodegradable and biocompatible. 12
Natural Compounds and Nanoformulations
Natural therapeutic agents such as curcumin, known for its antioxidant and anti-inflammatory properties, have been successfully encapsulated in chitosan hydrogels, HA-polylactic acid (PLA) nanoparticles, or PLGA nanospheres to improve their stability and bioavailability. These nanoformulations enable sustained drug release and deeper tissue penetration, making them suitable for post-extraction socket healing, periodontitis, and oral mucositis (OM) treatment. 13
Multifunctional Scaffolds for Cancer and Regeneration
Recent innovations have yielded dual drug-loaded nanofibrous scaffolds that provide both anticancer activity and support for tissue regeneration. For instance, electrospun polyhydroxybutyrate/gelatin nanofibers co-loaded with chemotherapeutics enabled spatiotemporal drug delivery in oral squamous cell carcinoma (OSCC), followed by scaffold degradation and tissue repair.14, 15
Treatment of cancers such as OSCC often results in significant hard and soft tissue loss, challenging both function and aesthetics. To address this, dual-function scaffolds are emerging as powerful tools that combine localized cancer therapy with regenerative healing, tailored to the unique anatomical and physiological demands of the oral cavity. 14
One of the most promising platforms is the injectable, self-assembling peptide-based hydrogel, which allows for the localized, prolonged release of anticancer agents directly within the oral lesion. 16 These systems offer enhanced mucosal retention, reduced systemic toxicity, and are engineered to degrade into bioactive matrixes that support the healing of surrounding oral tissues, such as gingiva, alveolar bone, and mucosa. 17 From recent studies, multifunctional scaffolds have been developed that simultaneously release chemotherapeutics and osteogenic or angiogenic factors, such as BMP-2 or VEGF, enabling both tumor suppression and periodontal or alveolar bone regeneration. 18 This is particularly beneficial in cases of post-surgical defects or in peri-implant sites compromised by malignancy.
Moreover, scaffold surfaces functionalized with immune-modulatory or cell-adhesive peptides (e.g., RGD) help shift the microenvironment from an inflammatory, tumor-supportive state to one that promotes Mesenchymal Stem Cells (MSC) recruitment, angiogenesis, and tissue regeneration. These smart systems can also be stimuli-responsive, designed to release anticancer agents in acidic or enzyme-rich tumor environments, and subsequently support regenerative processes as the local conditions stabilize.
In the context of oral cancers and post-excisional wound management, such dual-function materials represent a crucial advancement offering a single-stage approach to both eradicate residual malignancy and guide tissue repair, especially in functionally critical zones like the mandible, palate, or gingiva.2, 19
Surface-functionalized Biomaterials for Enhanced Bioactivity
Surface functionalization is a cornerstone strategy for improving scaffold performance in dental and periodontal regeneration. By modifying material surfaces with bioactive molecules, these systems enhance cell–material interactions and promote superior regenerative outcomes. 20
Incorporation of short bioactive peptides such as RGD (Arg-Gly-Asp) facilitates cell adhesion, migration, and differentiation. When combined with osteogenic or angiogenic growth factors like VEGF and BMP-2, these surfaces accelerate tissue healing, especially in periodontal and peri-implant regions. 21
Nanoparticle coatings of gold, silver, or calcium-based on titanium implants further amplify bioactivity. These nanoscale modifications enhance osseointegration, stimulate angiogenesis and osteogenesis, and modulate immune responses toward a regenerative phenotype by promoting M2 macrophage polarization.22, 23
Electrospun membranes functionalized with antimicrobial, anti-inflammatory, and osteoinductive agents closely mimic the extracellular matrix (ECM), acting as localized drug delivery systems for controlled release at defect sites. 24 Advanced approaches such as VEGF-loaded nanoparticles and siRNA-modified coatings have demonstrated improved vascularization and periodontal ligament formation.25, 26
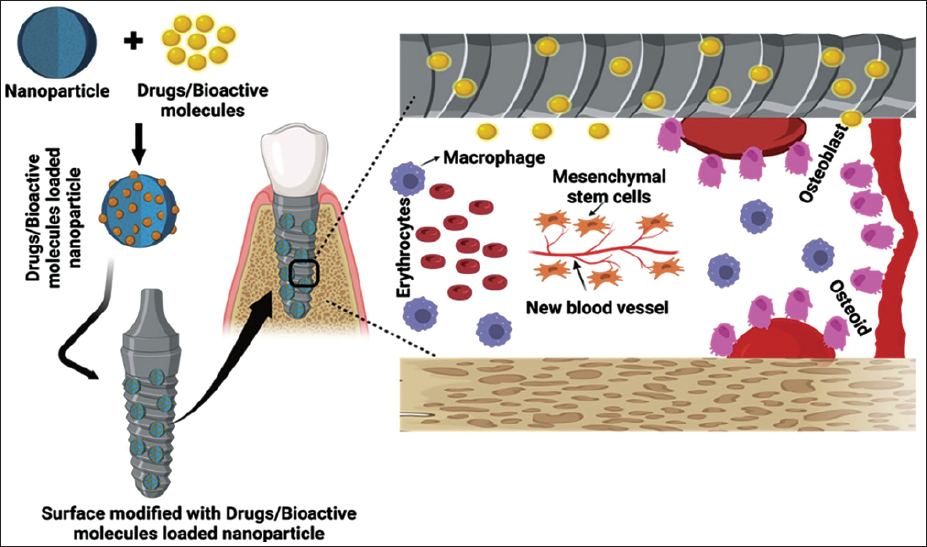
As illustrated in Figure 1, nanoparticles embedded on implant surfaces enable localized release of osteogenic and angiogenic factors, promoting cell adhesion, stem cell recruitment, and bone regeneration at the implant–bone interface, hallmarks of successful osseointegration. 27
Collectively, these surface-engineering strategies are redefining regenerative dentistry by creating site-specific, biologically responsive scaffolds that integrate structural stability with cellular signaling, ultimately enhancing clinical predictability and success.
Schematic Illustration of Drug- and Bioactive Molecule-loaded Nanoparticles Integrated on the Surface of a Dental Implant. These Surface-functionalized Platforms Enable Localized, Controlled Release of Osteogenic and Angiogenic Factors, Enhancing Cell Recruitment, Osseointegration, and Bone Regeneration at the Implant–Bone Interface. Adopted From Ref. 27.
Photodynamic and Mucoadhesive Therapies
Photodynamic therapy (PDT) is a promising non-invasive strategy for treating oral mucosal tumors. It employs nanoparticle carriers loaded with light-sensitive agents such as methylene blue or porphyrins, which generate ROS upon light activation to induce tumor cell apoptosis. Methylene blue-loaded PLGA nanoparticles have shown strong antibacterial effects in root canals and notable potential for oral cancer therapy.28–30
Complementary to PDT, mucoadhesive drug delivery systems extend the residence time of therapeutics on mucosal surfaces. Liposomal films containing chemotherapeutics like methotrexate enable sustained, localized release with minimal systemic exposure, ideal for superficial oral lesions and precancerous conditions. These systems adhere to moist oral tissues, maintaining effective local concentrations and improving bioavailability and patient compliance. 31
Together, photodynamic and mucoadhesive therapies exemplify site-specific, minimally invasive, and patient-friendly approaches that integrate nanoparticle technology with precision drug delivery, advancing personalized and regenerative dental care.
AM and Scaffold Design for Personalized Regeneration
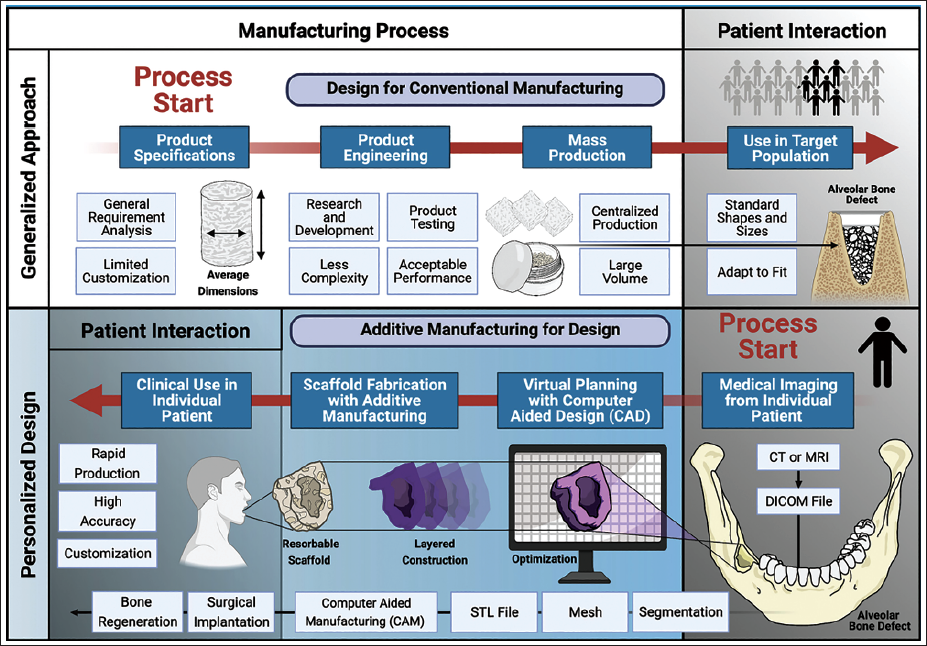
AM, widely known as 3D printing, has transformed scaffold fabrication in oral regenerative medicine. By using imaging data from CT or MRI, patient-specific constructs can be designed in CAD software and printed layer by layer through technologies such as fused deposition modeling (FDM), stereolithography (SLA), or selective laser sintering (SLS). This enables the production of scaffolds with high spatial accuracy, controlled porosity, and mechanical properties tailored to mimic native tissues.7, 8, 32
Biocompatible materials such as PCL, PLGA, and hydroxyapatite (HA) composites are commonly used in AM to fabricate scaffolds with adjustable degradation rates and mechanical strength. These scaffolds can incorporate advanced features like pore channels, microgrooves, and gradient stiffness zones to guide cell orientation and differentiation, particularly useful for complex interfaces like the periodontal ligament or alveolar ridge (Figure 2). 32
Paradigm Shift in Scaffold Production. Scaffold Fabrication Has Shifted From Standardized Manufacturing to Patient-specific Design Through AM. Traditional Methods Limit Complexity and Customization, Whereas AM Uses CAD/CAM and Patient Imaging to Create Personalized Scaffolds for Bone Regeneration. This Patient-centered Approach Enhances Precision and Accessibility in Clinical Dentistry. Adopted From Ref. 32.
Mechanical cues embedded within the scaffolds, such as stiffness and viscoelasticity, also influence stem cell behavior, enhancing osteogenesis and matrix deposition. AM’s capability to fabricate multiphasic or biphasic scaffolds supports both soft and hard tissue regeneration in a single construct. Furthermore, these scaffolds can be loaded with bioactive agents such as antibiotics (e.g., doxycycline), growth factors (e.g., BMP-2, VEGF), or gene vectors to enable simultaneous regeneration and therapeutic delivery.19, 33
Clinical prototypes have already shown success in alveolar ridge reconstruction and complex periodontal defects. While reproducibility, bioresorption timing, and in vivo integration remain as challenges, AM-based scaffolds are quickly advancing toward clinical application in large bone defects, trauma care, and post-cancer reconstruction. 34
Material Considerations for Dental Scaffolds
The success of scaffold-based dental regeneration relies heavily on selecting suitable biomaterials, particularly polymers, that meet both biological and engineering requirements. These materials must balance biocompatibility, printability, mechanical stability, and degradation behavior to ensure safe and effective performance in the complex oral environment.32, 35
Polycaprolactone
PCL is one of the most used polymers in dental scaffold fabrication due to its mechanical flexibility, slow degradation rate, and compatibility with FDM-based 3D printing. Its low melting point preserves the activity of temperature-sensitive biofactors, making it ideal for bioactive, cell-laden constructions. PCL is particularly well-suited for long-term support in load-bearing or large defects.32, 36
PLA and PLGA
PLA and PLGA offer faster degradation rates and robust mechanical strength. PLGA is widely used in both scaffolds and drug delivery due to its tunability and biocompatibility. However, its acidic degradation products may cause local irritation, especially in larger volumes. These polymers are ideal for applications requiring short-term scaffolds, such as microspheres in endodontic or periodontal therapies.37, 38
Composite Polymers
To address the limitations of single-polymer systems, composite scaffolds blend polymers like PCL or PLGA with bioactive ceramics such as HA, β-TCP, or bioactive glass. These composites enhance osteoconductivity, mechanical strength, and mimic the hybrid structure of native bone. They are especially effective in distributing mechanical stress under complex oral loads. 39
Tailoring Polymer Properties for Clinical Success
Optimizing scaffold properties for specific clinical needs involves tuning factors like pore size, elasticity, degradation rate, and viscoelasticity. For example, softer scaffolds may be used for gingival regeneration, while stiffer ones better support bone growth. Surface modifications, such as peptide conjugation or nanoparticle coating, enhance cell adhesion, immune response modulation, and overall clinical performance. 20
Innovations in Drug Delivery Systems for Oral Applications
Innovations in biomaterials have opened exciting possibilities for targeted therapy across a wide range of dental challenges, from managing peri-implant infections and oral cancers to promoting bone regeneration and pediatric care. These advanced systems are not just about delivering drugs; they are designed to do so intelligently, responding to local conditions, staying put where needed, and minimizing systemic side effects.40–42 Table 1 summarizes some of the most promising platforms currently under investigation or in clinical use. It highlights how nanoparticles, hydrogels, mucoadhesive films, and other delivery vehicles are tailored to specific dental needs, each selected for its material type, therapeutic goal, and clinical benefit.
Innovations in Drug Delivery and Regenerative Medicine in Dentistry.
Local Therapeutics for Site-specific Challenges in Dentistry
The oral cavity’s dynamic environment, including salivary enzymes and chewing forces significant obstacles to localized drug delivery. 40 Traditional approaches, such as mouthwashes and gels, while convenient, are often rapidly cleared and fail to maintain therapeutic concentration at the target site. This has led to the development of local drug delivery systems that provide prolonged, site-specific action while minimizing systemic exposure. 41
Mucoadhesive formulations, such as tablets, films, and in situ gelling systems, enhance drug residence by adhering to the mucosal surface. 61 For instance, thermoresponsive chitosan/β-glycerophosphate gels have been engineered to transition from liquid to semi-solid at body temperature, enabling sustained drug retention within periodontal pockets. 42 In “Zhao et al.” study showed that minocycline-loaded chitosan gel significantly reduced probing depth (PD) and gingival index in rats with induced periodontitis compared to untreated controls showed that minocycline-loaded chitosan gel significantly reduced PD and gingival index in rats with induced periodontitis compared to untreated controls. 62
Beyond gels, biodegradable polymer-based inserts, such as PLGA strips impregnated with tetracycline, have shown excellent results in clinical trials. 63 Maze et al. demonstrated that these films released antibiotics over a 10-day period and led to significant reductions in bleeding on probing and microbial load in patients with persistent periodontitis. 64
Table 2 summarizes representative examples of LDDS technologies applied in dental therapy, including microparticles, nanoparticles, and scaffolds across endodontic and regenerative contexts and in Figure 3, we present the most recent advancements in biomaterials used for dental drug delivery and regenerative medicine. These include a diverse range of systems from nanoparticles and hydrogels to electrospun scaffolds and mucoadhesive films designed to target everything from pediatric caries to oral cancers and peri-implant infections. Together, these innovations represent the foundation for a new era in dental care, one that is more personalized, predictive, and regenerative.
Summary of Local Drug Delivery Systems in Dentistry.
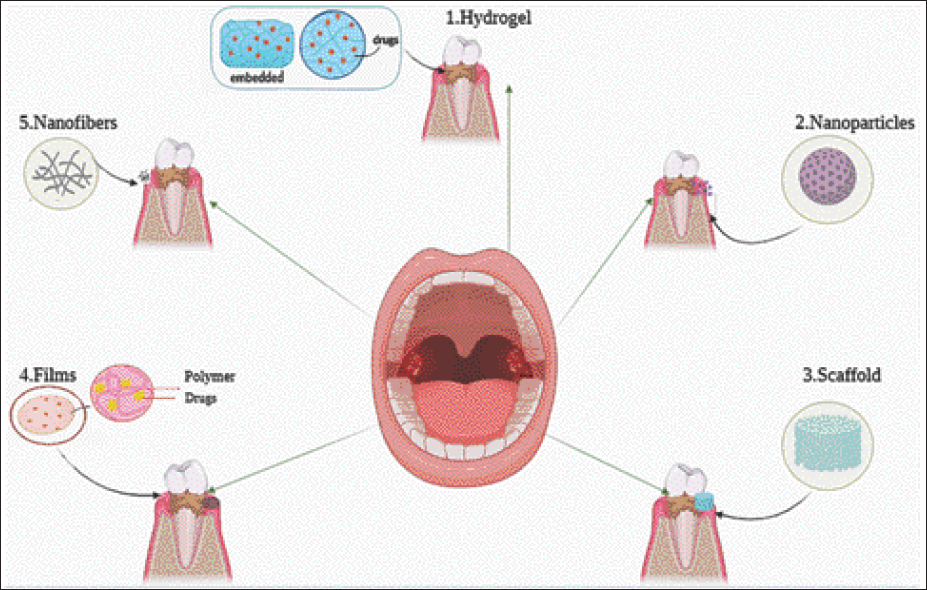
Models of Different Material Forms Used in Dental Drug Delivery and Regenerative Applications. (1) Hydrogels, Composed of 3D Polymer Networks, Are Capable of Carrying Drugs or Nanoparticles. (2) Nanoparticles, Which Can Be Inorganic or Organic, Are Exemplified Here as Mesoporous Structures. (3) Scaffolds, Typically Loaded with Growth Factors, Are Primarily Used for Supporting Tissue Regeneration. (4) Films Are Commonly Applied for Oral Mucosal Drug Delivery. (5) Nanofibers, Approximately 100 nm in Diameter, Serve as Drug Carriers.
Among the commercially available LDDS, Periochip, a gelatin-based matrix releasing chlorhexidine gluconate, remains a gold standard. 65 Likewise, Arestin, a PLGA microsphere formulation containing minocycline, provides sustained antibacterial activity and has received FDA approval for use in periodontal therapy. 66
More recently, polymeric and metallic nanoparticles have emerged as advanced delivery platforms capable of penetrating periodontal biofilms and releasing antimicrobials in response to environmental triggers. 67 In the study by Lecio et al., doxycycline-loaded nanoparticles achieved drug release for up to 21 days and effectively suppressed Porphyromonas gingivalis, significantly improving clinical parameters in beagle dogs. 68 Similarly, liposomal delivery systems have demonstrated improved antimicrobial retention and reduced osteoclast activity in rat models of bone loss. 54
Importantly, LDDS are also being explored for immunomodulatory purposes. For example, chitosan nanoparticles loaded with statins or N-phenacylthiazolium bromide have shown the ability to reduce inflammation and promote bone formation in periodontal defects, highlighting a dual role in both infection control and tissue regeneration. 69
These innovations mark a shift toward personalized, non-invasive therapies, where drug–biomaterial composites are customized to the anatomical site, disease condition, and patient profile. This precision-targeted approach, especially beneficial for older or immunocompromised patients, enhances outcomes while minimizing systemic effects. Nanofiber scaffolds, bioresorbable hydrogels, and polymer-based carriers have proven effective for localized delivery in periodontal and craniofacial applications, particularly within complex areas such as periodontal pockets and alveolar bone defects. 3
Targeted Endodontics: Microsphere-based Delivery Systems
The root canal’s complex structure and persistent infections make drug delivery challenging. Microsphere-based systems have emerged to support both disinfection and regenerative endodontic therapy. 96 A novel approach uses PLGA-coated biphasic ceramic microparticles loaded with a modified antibiotic mix of penicillin G, metronidazole, and ciprofloxacin chosen to be effective against key pathogens while avoiding tooth discoloration. These particles show ideal size for canal navigation, high drug loading efficiency, and a sustained release over 21 days while maintaining a neutral pH. 70 They exhibit strong antibacterial effects, low cytotoxicity to human gingival fibroblasts, and offer an added osteoconductive benefit from the HA/β-TCP core, making them promising for dual-function endodontic treatments. 97 Moreover, calcium hydroxide-loaded PLGA microspheres have demonstrated sustained Ca²⁺ ion release for up to 30 days, improving outcomes in apexification and pulp capping by stimulating mineralization. 98 Additional studies have successfully incorporated amoxicillin, minocycline, and ciprofloxacin into microsphere matrixes for targeted root canal disinfection, maintaining both antimicrobial potency and the mechanical integrity of sealing materials. 99
Beyond antimicrobial action, microspheres fabricated from PLGA, PLLA, and PCL are increasingly used as injectable scaffolds for stem cell support and tissue engineering. 100 For instance, PLGA microspheres loaded with VEGF have significantly improved angiogenic responses, essential for pulp revascularization. 101 Nanofibrous PLLA microspheres that mimic the ECM have enhanced odontogenic differentiation of dental pulp stem cells (DPSCs), especially when combined with BMP-2. Meanwhile, GelMA-based microspheres have enabled both cryopreservation of DPSCs and enhanced cell spreading, ECM formation, and in vivo integration when compared to bulk hydrogel scaffolds. 102
Crucially, the injectability and moldability of these microspheres allow for minimally invasive delivery, making them especially effective in treating irregular, curved, or narrow canal spaces that are difficult to access with bulk materials. 103 Their surface functionalization with biomolecules such as RGD peptides, collagen, and fibronectin has also been shown to promote cell adhesion, migration, and signal transduction, which are pivotal for initiating and sustaining regenerative processes within the root canal environment. 104
Altogether, microsphere-based delivery systems represent a dual-function platform in endodontics, combining localized, sustained antimicrobial therapy with a biomimetic scaffold that fosters tissue regeneration. These systems address the unmet need for targeted, biologically active therapies in endodontics and are poised to redefine clinical strategies in both infection management and regenerative treatment.
Nanotechnology and Nanosizing in Dental Applications
The complexity of periodontal pockets inflamed mucosal tissues, and cancerous lesions in the oral cavity demand nanocarrier systems capable of deep tissue penetration and targeted payload release in response to specific stimuli such as acidic pH, enzymatic activity, or oxidative stress. 105
Nanoparticles for Periodontitis and Endodontics
In periodontitis treatment, nanoparticles formulated with antimicrobial agents like doxycycline, chlorhexidine, or augmentin have been embedded into injectable gels or directly applied to periodontal pockets. 106 These systems provide sustained antimicrobial activity along with a supportive matrix for healing. For instance, chitosan/gelatin nanoparticles loaded with augmentin and modified triple antibiotic paste (mTAP) exhibited strong antibacterial effects while promoting fibroblast adhesion and viability, both essential for effective endodontic disinfection and tissue repair. 78 Similarly, simvastatin-loaded PLGA microparticles have been shown to promote BMP-2 expression and angiogenesis in periodontal defects, linking infection control with regenerative outcomes. 107
Nanoparticle-based Strategies for Mucositis
OM, a common and debilitating side effect of cancer therapies, has been targeted using gold nanoparticles (AuNPs) due to their anti-inflammatory and antioxidant properties. 108 In a 5-FU-induced hamster model of OM, PVP-stabilized AuNPs significantly reduced ulceration and inflammatory mediators such as NF-κB and COX-2, while enhancing Nrf2-mediated antioxidant responses, including HO-1 and GSH. These findings highlight the potential of AuNPs to both prevent and manage OM by modulating oxidative and inflammatory pathways. 109
Advanced Nanocarriers in Oral Cancer Therapy
In OSCC, nanoparticles such as liposomes, polymeric nanoparticles (e.g., PLGA, PEGylated systems), and core-shell nanospheres are being developed for tumor-targeted chemotherapeutic delivery, minimizing damage to healthy tissues and enhancing co-delivery of immune modulators. 110 These platforms can be fine-tuned with targeting ligands or stimuli-responsive coatings to improve tumor specificity. For example, AuNPs conjugated with cetuximab and cisplatin have been shown to significantly reduce the survival of radioresistant oral cancer cell colonies compared to conventional radiotherapy protocols. 111 In another approach, PLGA-based nanocarriers modified with anti-PD-L1 antibodies and loaded with all-trans retinoic acid (ATRA) demonstrated improved targeting capabilities and therapeutic outcomes in both CAL-27 cell lines and SCC-7 xenograft mouse models. 112
Controlled Drug Delivery: Localized and Targeted Strategies
Scaffolds fabricated through AM are increasingly being used as localized drug delivery systems, allowing for both structural regeneration and targeted therapeutic action. By incorporating antibiotics such as doxycycline or metronidazole directly into biodegradable polymers like PCL or PLGA, these scaffolds can achieve sustained release over weeks, reducing infection risk while supporting tissue healing.6, 100 For instance, doxycycline-loaded PCL scaffolds not only promote osteogenesis but also inhibit osteoclast activity and collagenase production. 113
Newer approaches include nanofiber delivery systems and nanocoatings, which enable the delivery of anti-inflammatory agents, antibiotics, and even gene vectors. 114 However, high processing temperatures can degrade certain drugs, highlighting the importance of post-printing drug loading strategies or using nanofiber surface modifications. 115 To minimize cytotoxicity, precise dosing and release control are critical, particularly in sensitive oral environments. Additionally, anti-fouling coatings like zwitterionic polymers and antimicrobial peptides are being investigated to prevent microbial colonization of scaffolds, ensuring better integration and regenerative success. 116
Toxicity and Regulatory Considerations of Nanocarriers
Although nanocarriers are transforming dental therapeutics, their nanoscale properties pose potential risks requiring precise evaluation. NPs smaller than 100 nm can cross the blood–brain barrier and olfactory pathways, accumulating in the brain and other organs with unpredictable biodistribution. 117 For instance, cationic gold and polystyrene NPs can induce hemolysis and coagulation, while carbon nanotubes promote platelet aggregation and ROS generation. 118
Toxicity depends on factors such as particle size, shape, surface charge, aggregation, and coating. Both cationic and anionic NPs may disrupt the blood–brain barrier at high concentrations, and dendrimers, despite their potential for drug conjugation—raise immunological safety concerns. 119 In the oral cavity, TiO₂, ZnO, and AgNPs found in toothpastes and restoratives interact with salivary proteins (e.g., lactoferrin, lysozyme), forming a protein corona that can alter antimicrobial activity and trigger immune responses. Incorporating nanosilver into non-degradable scaffolds may cause tissue accumulation and persistent inflammation.120, 121 Importantly, these risks are further influenced by oral-specific exposures, including constant saliva flow, biofilm interactions, and dynamic protein corona formation unique to the oral environment. Standardized, context-specific testing frameworks are emerging, but they remain fragmented and not yet harmonized across regulatory bodies.122, 123
Toxicity testing remains limited, as most studies rely on immortalized cell lines that fail to mimic the complex oral environment (saliva, mucosa, biofilms). 122 Artifacts, such as nonspecific binding of fluorescent dyes used in NP tracking, further confound results. 123 Occupational exposure is another concern for dental professionals who may inhale or contact airborne NPs during implant polishing, milling, or laser ablation. Long-term effects depend on exposure route, duration, and health status, yet these factors are inconsistently assessed.124, 125
Regulatory gaps add complexity. Nanomedicine lacks a unified global classification, leading to inconsistencies in toxicity thresholds and testing standards. 126 The BIORIMA Decision Support System, based on Integrated Approaches to Testing and Assessment (IATA), provides a life cycle–oriented framework for risk evaluation, integrating material-specific testing, exposure modeling, and scenario simulations to guide safe design and occupational risk management. 127
Ensuring the safe use of nanocarriers in clinical dentistry requires a multifaceted approach: developing realistic in vivo models that replicate the oral environment, assessing protein corona formation and its immunogenic effects, conducting comprehensive hazard and exposure analyses, and adopting globally harmonized regulations. Protective measures for dental professionals are equally crucial. 123
In summary, while nano biomaterials hold great promise for targeted drug delivery and regenerative care, their complex toxicity profiles demand robust, standardized, and context-specific safety evaluation to protect both patients and practitioners.
Discussion
Advances in biomaterials have accelerated the transition from traditional restorative approaches toward biologically driven and regenerative therapies. 128 Current innovations in scaffolds, nanocarriers, and smart delivery systems enable localized and controlled therapeutic effects, improving outcomes for conditions such as periodontitis, pulp disease, and oral cancers.114, 129, 130 Biodegradable polymers, nanocomposites, and hydrogels provide structural support while simultaneously acting as delivery platforms that modulate cellular activity and promote tissue healing.131–133
In regenerative dentistry, bioengineered scaffolds composed of PCL, PLA, chitosan, and HA have demonstrated significant potential in enhancing osteogenesis and periodontal regeneration.6, 131 Coupling these materials with bioactive molecules or growth factors further improves integration and accelerates functional recovery. 132 Recent interest in natural-derived cargoes such as curcumin-loaded nanoparticles illustrates the shift toward multifunctional systems that combine mechanical reinforcement with immunomodulatory and antimicrobial activity. 134
Smart drug delivery systems represent another important advance. Stimuli-responsive carriers activated by changes in pH, temperature, or magnetic fields are designed to respond dynamically to the oral microenvironment, enabling precise spatiotemporal drug release.133, 135, 136 Complementary developments in mucoadhesive polymers and injectable hydrogels improve retention, penetration, and controlled release of therapeutics in the oral cavity. 137
Digital fabrication and AM technologies, including 3D printing and bioprinting, further expand personalization and functionality. These platforms allow precise control of scaffold architecture and enable fabrication of patient-specific constructs and drug-loaded devices. 138 Emerging bioprinting strategies are increasingly capable of generating complex dental tissues such as pulp-dentin interfaces by co-delivering cells, biomaterials, and bioactive factors in spatially organized patterns. 139
Despite notable progress, significant translational barriers remain. Material biocompatibility, degradation kinetics, and mechanical stability must be optimized to balance safe degradation with adequate support during tissue regeneration.140, 141 Moreover, multifunctional biomaterials combining structural scaffolds with active drug delivery pose regulatory complexities that are not yet fully harmonized across agencies. 1 Cost-effective manufacturing, long-term in vivo validation, and standardization of testing frameworks will be essential for clinical translation. 142
A particularly innovative direction involves biomaterial-guided organoid development. Recent work by Zhang et al. (2024) demonstrated that bio-orthogonally cross-linked gelatin hydrogels can support epithelial-mesenchymal interactions required for tooth organoid formation, emphasizing how the physico-mechanical environment influences stem cell behavior and morphogenesis. 143 This highlights a future in which engineered biomaterials may enable complete dental organ regeneration rather than repair alone.
Conclusion
The convergence of advanced biomaterials, nanotechnology, and precision drug-delivery platforms is redefining the future of dentistry, shifting the field from mechanical repair toward biologically guided regeneration. 144 Smart scaffolds, injectable hydrogels, bioactive nanoparticles, and AM strategies now offer targeted, site-specific interventions that integrate structural support with controlled therapeutic release. Among these, polymers such as PLGA, PCL, and chitosan combined with growth factors or antimicrobial agents serve as versatile, multifunctional systems improving clinical outcomes across periodontics, endodontics, implantology, and oral oncology. Over the next decade, technologies with the strongest translational potential include stimuli-responsive hydrogels, personalized 3D-printed constructs incorporating biological cues, immunomodulatory nanomaterials, and emerging gene-activated scaffolds. These innovations align with global trends toward patient-specific, minimally invasive, and biologically informed treatment modalities. Despite notable progress, key challenges persist, including the complexity of the oral environment, inter-patient variability, long-term safety considerations, and regulatory uncertainty. Continued interdisciplinary collaboration supported by standardized testing frameworks and harmonized clinical evaluation pathways will be essential to translate these emerging biomaterial technologies into predictable and widely applicable clinical solutions, ultimately advancing dentistry toward a fully regenerative and patient-centered paradigm.
Footnotes
Authors’ Contributions
The authors contributed equally.
Data Availability Statement
No datasets were generated or analyzed during the current study.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval Institutional Statement
Not applicable.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
Not applicable.
