Abstract
Introduction
The disadvantages of chemical mouthwashes have prompted the search for safer and more effective anti-plaque alternatives, leading to the use of probiotics. This study evaluated and compared the efficacy of probiotic toothpaste and probiotic mouthwash in reducing Streptococcus mutans levels in plaque around orthodontic brackets.
Materials and Methods
Fifty-four patients were randomly allocated into three groups (n = 18 each) using a computer-generated random number table prepared by an independent researcher. Plaque samples were collected at baseline (Day 0) and after 30 days (Day 30) from the labial surfaces adjacent to maxillary lateral incisor brackets using the 4-pass technique. The presence of S. mutans was quantified as colony-forming units (CFU/mL) through culture methods, and statistical analysis was performed.
Results
The probiotic toothpaste group showed a mean reduction from 2.23 ± 0.68 to 1.51 ± 0.54 CFU/mL (∇ = 0.72, p = .002), and the probiotic mouthwash group from 2.06 ± 0.76 to 1.70 ± 0.55 CFU/mL (∇ = 0.36, p = .015). The control group exhibited a minimal, non-significant reduction (∇ = 0.08, p = .756). Although the reduction was greater in the toothpaste group, the intergroup difference was not statistically significant (p = .051).
Conclusion
Both probiotic toothpaste and mouthwash significantly reduced S. mutans levels over 30 days, with toothpaste showing a slightly greater, though not statistically significant, effect. Probiotic-based oral care products may be a viable adjunct for microbial control in orthodontic patients.
Introduction
Recent advancements in orthodontics have enhanced patient care by refining both treatment approaches and appliance quality. However, white spot lesions continue to pose a significant challenge for patients and clinicians alike, as enamel demineralization is still a common side effect of orthodontic procedures. 1
White spot lesions may form due to orthodontic appliances creating plaque-retentive areas—especially near brackets, bands, wires, and other attachments—when oral hygiene is inadequate. 1 Individuals undergoing orthodontic treatment typically exhibit increased levels of acid-producing bacteria in their plaque, particularly Streptococcus mutans, compared to those not receiving such treatment.2–4 These lesions arise from localized enamel demineralization around orthodontic components.
While various anti-plaque products have been employed as supplementary tools, tooth brushing remains the most commonly recommended and practiced method for mechanical plaque removal. Mouthwashes are widely used due to their safety and effectiveness in delivering antimicrobial agents. These agents can disrupt bacterial processes by preventing adhesion, colonization, and metabolic activity, thereby reducing bacterial proliferation. 5
Although antimicrobial mouthwashes can effectively reduce S. mutans levels in saliva, their frequent use may lead to undesirable side effects, including tooth staining and the potential development of antimicrobial resistance. 6 As an alternative, probiotic therapy offers a promising approach to oral care that may help mitigate these drawbacks. In today’s antibiotic-centric landscape, interest in probiotics has grown. Probiotics consist of live microorganisms—or formulations containing them—that can provide health benefits to the host. Several studies suggest that consuming products with probiotic strains such as Lactobacillus or Bifidobacterium may help reduce the salivary concentration of mutans streptococci.7, 8
Limited research has explored the effects of locally administered probiotic formulations. Therefore, this study was undertaken to evaluate the efficacy of two different probiotic agents in preventing the accumulation of S. mutans in plaque around orthodontic brackets.
Materials and Methods
Study Design and Registration
This was a prospective, randomized, controlled clinical study conducted in the Department of Orthodontics and Dentofacial Orthopedics. The study was prospectively registered with the Clinical Trial Registry – India (CTRI/2025/04/084988).
Ethical Approval and Consent
Approval for the study was obtained from the Institutional Ethics Committee (IEC/MPDC_302/ORTHO-63/24). Written informed consent was obtained from all patients prior to enrollment, after explaining the study objectives, procedures, potential risks, and benefits.
Participants and Sample Size
A total of 54 patients undergoing fixed orthodontic treatment for at least three months were enrolled. Participants were randomly assigned to three groups (n = 18 per group) using a computer-generated random number table. The allocation sequence was prepared by an independent researcher who was not involved in recruitment or intervention delivery, thereby minimizing selection bias and maintaining the integrity of the randomization process.
Group 1 (Control): Non-probiotic toothpaste combined with saltwater gargles.
Group 2 (Probiotic toothpaste): Probiotic toothpaste (Purexa) along with saltwater gargles.
Group 3 (Probiotic mouthwash): Probiotic mouthwash (Perfora) with non-probiotic toothpaste.
Inclusion Criteria
Patients undergoing fixed orthodontic treatment for a minimum of 3 months with MBT 0.022 bracket system (Ormco) and round nickel-titanium archwires (G&H Orthodontics).
Use of identical elastomeric modules and absence of e-chains, elastics, or auxiliary appliances during the study.
Age between 12 and 35 years with permanent dentition and intact anterior teeth.
Clinically healthy periodontal tissues.
No systemic diseases such as diabetes or immunocompromised conditions.
Exclusion Criteria
Composite restorations or crowns on anterior teeth.
Oral Hygiene Index-Simplified (OHI-S) score greater than 3.
Developmental defects or fluorosis.
Current use of phenytoin, immunosuppressants, or other interfering medications.
Requirement for antibiotics or anti-inflammatory drugs during the study.
Pregnancy or tobacco use.
History of frequent vomiting.
Baseline Examination and Prophylaxis
At baseline, oral hygiene status was assessed using the Simplified OHI-S of Greene and Vermilion. 9 Patients with scores >3 were excluded. All eligible participants underwent professional dental prophylaxis prior to starting the intervention
Interventions
Group 1 (Control): Brushed twice daily with non-medicated toothpaste and performed routine saltwater gargles.
Group 2 (Probiotic toothpaste): Brushed twice daily using probiotic toothpaste (Purexa) and performed regular saltwater gargles.
Group 3 (Probiotic mouthwash): Continued brushing with non-medicated toothpaste and rinsing with 10 mL of undiluted probiotic mouthwash (Perfora) three times daily for 3–4 minutes after meals, avoiding food/drink for 30 minutes post-rinse.
In the probiotic toothpaste group, participants were additionally instructed to perform routine saltwater gargles twice daily. This adjunct was included to provide non-chemical mechanical cleansing support and to potentially enhance the effect of the probiotic toothpaste. The control group was prescribed non-probiotic toothpaste only, without gargles, to isolate its effect. The probiotic mouthwash group did not include saltwater gargles in order to evaluate the mouthwash’s efficacy independently. The intervention period lasted 30 days.
Sample Collection
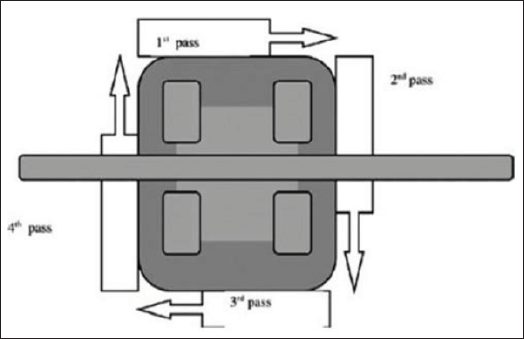
Plaque samples were collected at baseline (Day 0) and after 30 days (Day 30) from the labial surfaces adjacent to orthodontic brackets on the maxillary lateral incisors (Figures 1 to 3). A sterilized scaler and the 4-pass technique of Pellegrini et al. 10 were used (Figure 4). Samples were transferred to sterile containers and sent immediately to the microbiology laboratory.
Control Group.
Probiotic Toothpaste Group.
Probiotic Mouthwash Group.
Microbiological Analysis
Samples were inoculated into Luria broth for isolation of S. mutans.
Grown cultures underwent serial dilution and plating on chromogenic agar.
Colonies were further inoculated in Luria broth and incubated overnight at 37°C.
DNA extraction was performed using a genomic DNA kit, followed by PCR amplification of the 16S rDNA region using universal primers.
PCR conditions: 94°C for 3 min; 94°C for 45 s, 50°C for 60 s, 72°C for 90 s (35 cycles); final extension at 72°C for 10 min.
Products were confirmed by 0.8% agarose gel electrophoresis, purified with a PCR cleanup kit, and sequenced.
Outcome Measures
The primary outcome was the change in S. mutans colony-forming units (CFU/mL) between baseline and Day 30 across the three groups.
Statistical Analysis
The collected data was entered into a Microsoft Excel spreadsheet. Descriptive and Inferential statistics were analyzed using Statistical Package for Social Sciences (SPSS version 26.0) for MS Windows. The Shapiro-Wilk test was used to check the normality of the data. The Wilcoxon Signed Ranks Test is used to compare two related samples. The Kruskal–Wallis Test is used to compare three independent samples.
Results
After one month of application of normal toothpaste and salt gargles (Group 1), probiotic toothpaste (Group 2) and probiotic mouthwash with normal toothpaste (Group 3), the level of S. mutans significantly reduced in Group 2 and Group 3, but minimal reduction was shown in Group 1, as shown in Table 1.
The Wilcoxon Signed Ranks Test for Pre-post Comparison of Groups.
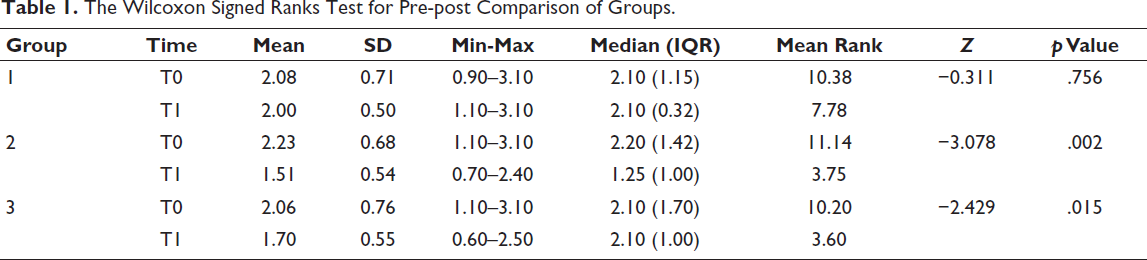
The Wilcoxon Signed Ranks (Table 1) Test showed that there was a statistically significant difference between T0 and T1 S. mutans of Group 2 (p = .002) and Group 3 (p = .015), while no significance in Group 1.
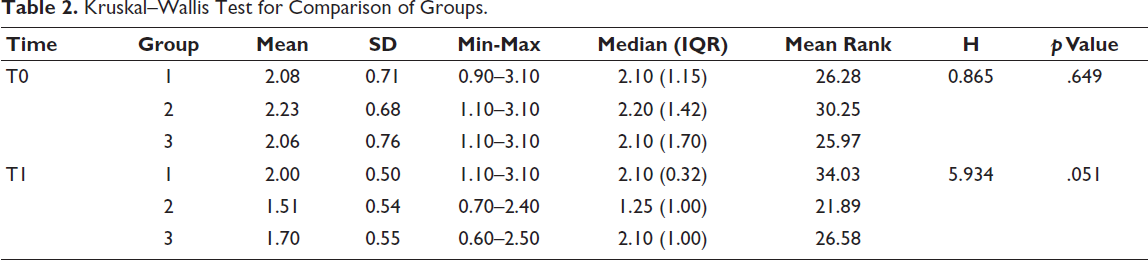
The Kruskal–Wallis Test (Table 2) showed that there was no statistically significant difference in S. mutans between the three groups at T0 and T1.
Kruskal–Wallis Test for Comparison of Groups.
Discussion
Effective oral hygiene is essential for preventing dental diseases, but it becomes challenging in orthodontic patients because fixed appliances create plaque-retentive sites that increase the risk of enamel demineralization and white spot lesions. While toothbrushing is the most common method of plaque removal, adjunctive approaches such as mouth rinses can provide synergistic benefits. 7 However, the disadvantages of chemical antimicrobials, including staining and resistance, have stimulated interest in safer alternatives such as probiotics. 7
Probiotics can reduce cariogenic bacteria by competitive inhibition, production of antimicrobial metabolites, and modulation of host immune responses. This is particularly relevant for orthodontic patients, in whom S. mutans levels are elevated and associated with white spot lesion development.11, 12
The complex design of orthodontic brackets makes effective tooth brushing challenging, often leading to the accumulation of food particles and dental plaque. As a result, brackets can significantly contribute to enamel demineralization. Notably, metal braces—due to their higher surface tension—pose an increased risk for enamel demineralization, which can ultimately lead to the development of white spot lesions and early enamel caries. This is a crucial consideration for clinicians, dentists, and patients alike.13, 14
Although limited studies have been conducted so far, existing evidence suggests that probiotics are effective in managing and preventing various oral infections, including dental caries, gingival inflammation, and halitosis. 15 Research indicates that probiotic consumption can lead to a reduction in S. mutans levels in the oral cavity. This study was therefore undertaken to evaluate the effectiveness of different probiotic agents in inhibiting the growth of S. mutans in plaque surrounding orthodontic brackets.
In the anterior region, maxillary lateral incisors are particularly vulnerable to white spot lesions due to reduced salivary flow and the limited space between the brackets and the gingival margin, which hinders effective cleaning. These factors contribute to poor accessibility for oral hygiene measures. Since white spot lesions in the anterior teeth are highly visible, they present a significant aesthetic concern. 10 Therefore, plaque samples were collected from the labial surfaces adjacent to the orthodontic brackets on the maxillary lateral incisors using a sterilized scaler.
Saltwater gargles were included in the probiotic toothpaste group as a simple, non-chemical adjunct. Saltwater provides mechanical cleansing and osmotic effects that can reduce microbial load and gingival inflammation. However, the mouthwash and control groups did not receive gargles, allowing their outcomes to reflect their respective interventions alone. Although gargles may have provided supportive short-term benefits, the sustained reduction in S. mutans in the toothpaste group indicates that the probiotic activity was the primary driver of the effect. The complementary actions of the two interventions may explain the slightly greater numerical reduction observed in the toothpaste group compared to the mouthwash group, although the difference was not statistically significant.
Both probiotic toothpaste and probiotic mouthwash significantly reduced S. mutans levels after 30 days, while no meaningful change was observed in the control group. The probiotic toothpaste group showed a greater numerical reduction compared with mouthwash, though the difference narrowly missed statistical significance (p = .051). One possible explanation is that brushing with probiotic toothpaste combines the probiotic effect with the mechanical disruption of plaque, ensuring closer contact between probiotic bacteria and tooth surfaces. In contrast, mouthwash relies primarily on the rinsing action, which may result in less sustained contact. Although this finding suggests a potential advantage for toothpaste, confirmation in larger and longer-term studies is necessary. Ultimately, the success of either intervention depends largely on patient compliance and willingness to consistently use the product.
Recent evidence corroborates the beneficial impact of probiotics during orthodontic treatment. A multicenter randomized trial found that probiotic mouthwash achieved similar effectiveness in reducing plaque, gingival inflammation and white spot lesions as chlorhexidine or fluoride over 12 weeks. 16 Furthermore, a 2024 systematic review and meta-analysis concluded that probiotics significantly reduce mutans streptococci levels in patients with fixed appliances, although the evidence for their effect on enamel lesion prevention remains inconclusive. These contemporary findings strengthen the external validity of our results while also highlighting that further targeted research is warranted. 17 Our findings align with the recent systematic review and meta-analysis by Chen et al., 18 which concluded that probiotic interventions can significantly reduce cariogenic bacterial counts and improve oral health in orthodontic patients, although the magnitude of benefit varies depending on strain, dosage and delivery method. Their analysis highlighted the potential for probiotics as an adjunctive strategy in orthodontic care, supporting the rationale for our investigation.
It is important to note that the antimicrobial efficacy of probiotics may vary depending on the specific strains, their concentrations, and the formulation used for delivery. 12 Different strains of Lactobacillus, Bifidobacterium, or other beneficial microorganisms may possess unique adhesion capabilities, competitive inhibition mechanisms, and metabolic activities against S. mutans. Likewise, the delivery form—whether toothpaste, mouthwash, lozenge, or varnish—can influence the duration of contact with tooth surfaces and the ability of the probiotic to colonize the oral cavity. These factors may account for variations observed between studies and emphasize the need for strain-specific and formulation-specific evaluations. 12
Limitations
This study has certain limitations that should be acknowledged. The intervention period was limited to 30 days, which may not reflect the long-term effects of probiotic use on S. mutans levels or the prevention of white spot lesions. Being a single-center study with a relatively small sample size (n = 18 per group) may limit the generalizability of the findings to broader populations. Only S. mutans counts were assessed, whereas orthodontic plaque harbors a complex microbiota that may also influence demineralization risk. Participant compliance with the prescribed toothpaste or mouthwash regimen was self-reported and not objectively verified, potentially introducing variability. Additionally, factors such as dietary sugar intake, brushing technique and other oral hygiene practices could have affected bacterial levels. Furthermore, the study did not evaluate the colonization or persistence of probiotic strains in the oral cavity after discontinuation, and the absence of placebo-controlled formulations restricts the ability to attribute the observed effects exclusively to probiotic activity.
Conclusion
Topical use of both probiotic toothpaste and probiotic mouthwash led to a significant reduction in S. mutans levels in plaque surrounding various orthodontic brackets.
However, as this was a short-term study, an extended follow-up is necessary to better understand the long-term benefits of probiotic use in orthodontic patients. Further research is also needed to compare the effectiveness of systemic versus local probiotic administration, as well as the targeted application of probiotic toothpaste around different types of fixed and functional orthodontic appliances.
Footnotes
Authors’ Contribution
Jainil Patel: Conceptualization, methodology, investigation, resources, data curation, writing - original draft, visualization, supervision, and project administration.
Padmaja Arora: Conceptualization, methodology, writing - review and editing draft, visualization, supervision, and project administration.
Kalpesh Patel: Conceptualization, methodology, writing - review and editing draft, visualization, supervision, and project administration.
CTRI Registration
This clinical study was prospectively registered with the Clinical Trial Registry – India (CTRI/2025/04/084988; Registered on 16/04/2025). The full trial details are accessible at:
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Declaration of Patient Consent
The authors certify that they have obtained all appropriate patient consent forms. In the form, the patient(s) has/have given his/her/their consent for his/her/their images and other clinical information to be reported in the journal. The patients understand that their names and initials will not be published and due efforts will be made to conceal their identity, but anonymity cannot be guaranteed.
Ethical Approval
The research/study was approved by the Institutional Review Board at the Institutional Ethics Committee (IEC) for Research, number IEC/MPDC_302/ORTHO-63/24, dated December 26, 2024.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
