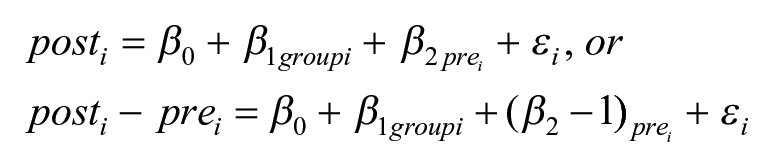Abstract
This study examined the combined effects of StrongerMemory program (brain exercises involving reading, writing, and math) and weekly social engagement on cognitive, behavioral, and emotional outcomes in older adults with subjective cognitive decline (SCD). A 12-week randomized controlled trial was conducted with 50 participants, who were randomly assigned to either a control group (StrongerMemory only) or an intervention group (StrongerMemory plus weekly social engagement). Cognitive function (MoCA), perceived cognitive decline (SCD-Q), health behaviors (GHPS), and emotional well-being (SWEMWBS) were assessed at baseline and post-intervention. Both groups showed significant cognitive improvements (increased MoCA, decreased SCD-Q) post-intervention. ANCOVA revealed significantly better cognitive function in the intervention group, demonstrating the synergistic benefits of social engagement. The intervention group also experienced enhanced emotional well-being. These findings suggest that incorporating social engagement into cognitive training programs enhances their effectiveness in improving cognitive function and emotional well-being in older adults with SCD, potentially mitigating further decline. While the findings are promising, this exploratory study’s small sample size resulted in modest achieved power (0.64), which may limit the generalizability of the results.
Keywords
Introduction
Cognitive decline remains one of the most pressing challenges in the evolving field of aging (Murman, 2015). A substantial portion of older adults experience Subjective Cognitive Decline (SCD), self-perceived decrease in cognitive function that can be an early indicator of dementia (Centers for Disease Control and Prevention [CDC], 2019; Smid et al., 2022). Pike et al.’s (2022) meta-analysis found that individuals with SCD had nearly double the risk of developing dementia compared to those without. In the United States, approximately 11.1% of adults aged 45 and older report having SCD (CDC, 2019). Of these individuals, 45.3% experience functional difficulties that adversely affect their daily activities, social lives, and work. Furthermore, 29.3% of those with SCD live alone (CDC, 2019), a circumstance that may exacerbate the challenges associated with cognitive decline.
SCD is now widely recognized a preclinical stage of Alzheimer’s and a potential target for early prevention efforts (Wang et al., 2020). Early diagnosis and intervention of SCD present a crucial opportunity to implement preventive measures, potentially delaying or halting the onset of more severe cognitive deficits. This study aims to contribute to these efforts by examining the potential synergistic effects of integrating social engagement with the StrongerMemory program, a non-pharmacological cognitive intervention.
The StrongerMemory program, developed by Goodwin Living, targets the prefrontal cortex—a key area for memory retrieval—to enhance cognitive performance through daily brain-stimulating activities (Collins, 2024). The program requires participants to read aloud for 20 to 30 min, write in a notebook, and solve simple math questions each day. Phase I of the StrongerMemory study, involving 102 older adults in Northern Virginia, reported improvements in cognitive function (Ihara et al., 2025). Furthermore, a qualitative analysis of focus groups from Phase I revealed a recurring theme: the optional group meetings were a highly valued aspect of the program (Tompkins et al., 2024). Participants emphasized that social interaction from the optional group meetings enhanced their motivation, satisfaction, and overall sense of well-being (Tompkins et al., 2024). Following this unexpected but enlightening discovery, we considered adding social engagement to the StrongerMemory program to see if it would have any synergistic effects.
Building on Phase I, which demonstrated the cognitive benefits of the StrongerMemory program (Ihara et al., 2025), the present study (Phase II) expands the research to examine the collaborate and synergistic effects of integrating weekly group social engagement with these daily brain exercises. Social engagement, particularly in group settings, has been suggested to play a protective role for cognitive health in older adults, potentially lowering the risk of cognitive decline and preventing Alzheimer’s Disease and Related Dementias (ADRD; Haslam et al., 2014; Lydon et al., 2022).
This study adopts a multi-domain approach that acknowledges cognitive decline as not solely a cognitive issue, but also one involving behavioral and emotional changes (Tsukii, 2022). This broader perspective is crucial for understanding the mechanisms underlying both risk and opportunity in early intervention along the trajectory of Alzheimer’s disease and related dementias (ADRD). SCD is increasingly viewed as a potential precursor to Mild Cognitive Impairment (MCI), a transitional stage in cognitive decline representing a state between normal aging and dementia that can lead to ADRD (Smid et al., 2022). MCI is frequently accompanied by behavioral symptoms such as disinhibition and apathy, and lower levels of emotional well-being in older adults (Apostolova et al., 2014; Fu et al., 2021). These non-cognitive symptoms often manifest subtly but can significantly affect an individual’s daily functioning and quality of life. By integrating behavioral and emotional dimensions alongside cognitive measures, this study shifts beyond a narrow focus on memory loss to adopt a more holistic understanding of risk and resilience. In short, by examining a broader range of outcomes, this study seeks to contribute to a more comprehensive understanding of ADRD risk and protection strategies.
The goal of this study is to identify effective non-pharmacological interventions that address multiple risk factors for ADRD. To this end, we investigate whether adding social engagement to the StrongerMemory program enhances its cognitive, behavioral, and emotional benefits. We hypothesize that after 12 weeks, participants in the intervention group (StrongerMemory with social engagement) will show greater improvements in cognitive function, positive behavioral outcomes, and emotional well-being than those in the control group (StrongerMemory alone). Ultimately, this research seeks to advance our understanding of ADRD and inform the development of innovative, holistic strategies that support cognitive health and overall well-being in older adults.
Theoretical Framework
This research is grounded in the biopsychosocial model, which highlights the interdependence of psychological, social, and biological elements in determining health over the lifespan (Lugg, 2022). From this perspective, cognitive health and dementia risk are not exclusively determined by biological and physical factors, but also by psychological and social determinants (Hopper et al., 2023). Many studies have used this model to investigate the complex relationships that exist between biological aging processes, social isolation, depression, and education, as well as how these factors change with age and affect cognitive trajectories (Bolton & Gillett, 2019; Zahodne, 2021). In this study, the biopsychosocial model provides a framework for understanding how the interplay of brain activities and social engagement can influence psychological and behavioral processes, ultimately impacting cognitive function in older adults.
This study also draws upon the Cognitive Reserve hypothesis, which holds that people’s ability to adjust to age-related and pathological brain changes varies (Stern, 2012). These individual differences in neural networks and cognitive processes allow some people to retain cognitive function in spite of underlying brain pathology (Hindle et al., 2014). According to Stern (2012), lifelong participation in cognitively and physically demanding activities might develop a cognitive reserve that serves as a barrier against cognitive deterioration. As people age, those with higher cognitive reserves typically see less cognitive decline (Yang et al., 2024).
Improving cognitive reserve could be a crucial strategy for preventing or postponing cognitive aging. Amanollahi et al. (2021) suggest that various socio-behavioral characteristics, including more schooling, better jobs, frequent social connections, and brain-challenging activities, may strengthen one’s cognitive reserve and be associated with improved cognitive performance in later life. Cheng’s (2016) research provides additional evidence that a variety of experiences, including leisure activities, cardiovascular exercise, and cognitive training, can enhance cognitive reserve. These results emphasize the possible advantages of mentally and socially engaging activities for maintaining cognitive health.
In this research, we posit that social engagement plays a critical role in developing and preserving cognitive reserve. Opportunities for social learning, emotional control, positive health-related behaviors, and social stimulation can be provided by social interaction through social engagement, and these activities all support cognitive resilience (Dause & Kirby, 2019; Grotz et al., 2018; Watt et al., 2014). We hypothesize that a multi-component intervention combining cognitive training (such as the StrongerMemory program) and social interaction can more effectively mitigate age-related cognitive decline by enhancing cognitive reserve.
Increasingly, research points to the importance of social engagement as a protective factor for cognitive decline (Lydon et al., 2021). According to Zhou et al. (2018), older adults who participate in social activities more frequently and have robust social networks have superior cognitive function and a lower risk of dementia and cognitive decline. While social interaction alone has been shown to be effective in protecting against dementia, pairing it with other interventions can provide greater overall benefits as part of a multi-domain protection strategy (Ngandu et al., 2015). Given that cognitive decline can be attributed to a variety of factors, such as physical health, cognitive stimulation, and emotional well-being (Dinius et al., 2023), a multi-domain approach that incorporates social, cognitive, and physical components can maximize the effectiveness of an intervention and provide a more complete solution (Rosenberg et al., 2020).
Multi-domain interventions can produce synergistic effects, yielding a greater impact than the sum of their individual parts (Salzman et al., 2022). This strategy enables a more efficient and potentially more impactful method of preventing dementia. In addition, implementing a multi-domain strategy guarantees that all the important risk factors for cognitive decline are tackled concurrently. It is essential to consider all factors to maintain the overall effectiveness of the intervention, since disregarding any one component might lead to a weakening of its impact (Hafdi et al., 2021). For example, focusing solely on cognitive training while neglecting physical health or emotional well-being (Salzman et al., 2022) may not be as effective as addressing all these factors in a coordinated manner.
Social engagement is crucial in multi-domain programs to enhance cognitive health and overall well-being (Murukesu et al., 2020). Engaging in group activities, participating in voluntary work, and utilizing online platforms offer chances for pleasure, inspiration, and mental stimulation. More precisely, active engagement in social activities might enhance the pleasure and commitment to interventions, potentially leading to increased participation and positive outcomes (Davis et al., 2021). Social interaction through social engagement has numerous advantages, such as influencing others, actively involving oneself in social activities, and gaining access to valuable resources and material possessions. Regular social engagement inherently involves cognitive challenges, such as participating in conversations, learning new skills, and engaging in mental exercises, all of which contribute to the preservation of cognitive function (Zhou et al., 2018). Additionally, the support system created through social engagement helps manage stress and mood (Zhang et al., 2022), promotes healthy behaviors, and ultimately protects cognition, with recent evidence particularly emphasizing the role of active social engagement and social connections for cognitive benefits (Joshi et al., 2024).
Integrating social engagement into multi-domain programs can be achieved through various methods. Group-based cognitive training and exercise activities foster social interaction and a sense of community (Haslam et al., 2014). Promoting volunteerism encourages individuals to engage in meaningful social interactions, fostering both a sense of purpose and opportunities for mental stimulation, which can contribute to cognitive health (Stukas et al., 2016). Furthermore, technology-based platforms facilitate communication and social interaction, which is especially beneficial for individuals who are socially isolated due to their location or other constraints (Heins et al., 2021).
Figure 1 presents the conceptual model for this study. It illustrates how the core components of cognitive stimulation and social engagement are expected to work through key mechanisms to produce positive cognitive, emotional, and behavioral outcomes. We believe that a multi-domain strategy that incorporates social interaction with other cognitive interventions offers a comprehensive and promising approach to supporting cognitive health and protecting against dementia. Research shows that engaging cognitive activities builds brain “reserve,” helping older adults resist age-related decline (Lee et al., 2020), and structured social activities enhance social connections and interaction can boost mental function (Wei et al., 2024).
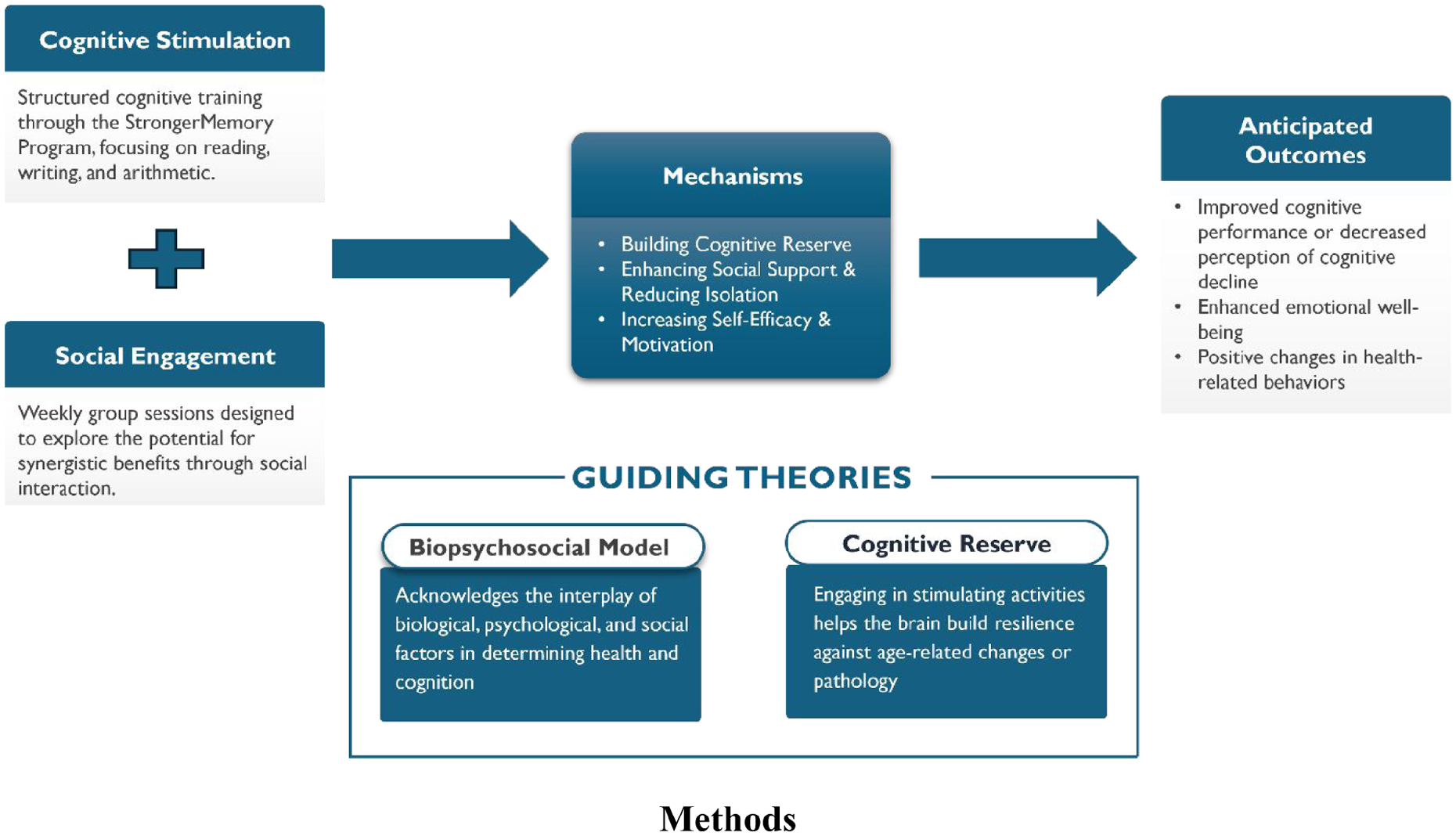
Conceptual framework: Synergistic effects of cognitive stimulation and social engagement on well-being.
The combination of cognitive training and social engagement is anticipated to increase participants’ self-efficacy, or their confidence in their ability to succeed. Previous research supports this connection. For example, individualized brain-training has been shown to significantly improve cognitive self-efficacy in older adults (Goghari & Lawlor-Savage, 2018). Similarly, a structured cognitive stimulation program led to a doubling of average self-efficacy scores in the treatment group (AbdElsalam & ElKholy, 2024). These studies also indicate that older adults with higher self-efficacy tend to demonstrate better memory performance.
Although the direct effects of brain-training and social engagement programs on health behavior change remain underexplored, substantial research highlights the central role of self-efficacy in influencing health behaviors (Warner & Schwarzer, 2020). This is particularly important because self-efficacy determines whether individuals initiate behavioral change, how much effort they invest, and how persistently they pursue these changes despite challenges (Warner & Schwarzer, 2020). Furthermore, combining cognitive stimulation with social engagement (Woolf et al., 2024) has been associated not only with cognitive maintenance or gains, but also with improved emotional well-being in older adults.
Positive experiences with the StrongerMemory program and social interactions can boost self-efficacy, which in turn may enhance motivation to sustain these activities and potentially yield cognitive, emotional, and overall health benefits. Multi-domain programs are expected to play an increasingly significant role in addressing dementia, offering hope for a future where cognitive decline can be effectively managed and mitigated.
Methods
Study Design
This study investigated how social engagement affected older adults’ StrongerMemory program results. A 12-week, single-blind, randomized controlled study (RCT) including two groups (intervention and control) was utilized to assess the collaborative impact of the program and social interaction on behavioral outcomes, emotional well-being, and cognitive performance. The intervention group (StrongerMemory with social engagement) and the control group (StrongerMemory alone) were assigned to participants at random. The study employed a single-blind design. Participants were not made aware of the specific research hypotheses distinguishing the groups or that the social engagement component itself was the primary aspect under investigation. All participants were informed that they were part of a study evaluating the StrongerMemory program. Those in the intervention group were aware they were attending weekly group sessions in addition to the StrongerMemory program, while the control group completed the StrongerMemory program with minimal check-ins.
The intervention group involved weekly group sessions and completion of the 12-week StrongerMemory program. All weekly social engagement sessions were conducted by the same moderator to ensure consistency in delivery. The moderator utilized the “StrongerMemory Facilitator Manual” developed by Goodwin Living as a guiding framework. However, as an exploratory study, this research did not include a formal quantitative assessment of intervention fidelity for the social component. This manual provided thematic content linked to emotional wellness, cognitive performance, and brain health, which the moderator used to encourage participants to share their thoughts and experiences. The 12-week StrongerMemory program was completed by the control group without weekly group sessions. Standardized assessments were given to participants at the beginning and the end of the intervention to evaluate improvements in their emotional well-being, healthy behavior, and cognitive performance.
Sample and Recruitment
This study targeted individuals aged 60 and over residing in Northern Virginia. Participants were eligible if they displayed signs of Subjective Cognitive Decline (SCD), as assessed by the Subjective Cognitive Decline Questionnaire (SCD-Q) but had not received a formal dementia diagnosis. Eligibility required scoring “yes” to more than seven out of 24 questions on the SCD-Q. Individuals who had previously participated in any phase of the StrongerMemory program were also excluded.
The flow of participants through the study is detailed in Figure 2. A comprehensive recruitment strategy was implemented from October to December 2022 to engage potential participants. Initial outreach targeted senior centers in Northern Virginia and was later expanded to include senior housing facilities, adult day health centers, and other community organizations for older adults. In response to the limited racial/ethnic diversity in Phase I, active recruitment was also conducted within the Korean community through partnership with local community centers. All research materials for this effort were professionally translated into Korean to ensure cultural sensitivity and accessibility. From an initial 150 individuals screened, 65 were deemed eligible, and 62 were subsequently enrolled and provided consent.

Participant flow diagram.
Intervention Procedures
Participants were enrolled across two 12-week intervention sessions. The first session focused on the Korean-speaking community and included 34 participants, while the second session targeted the broader Northern Virginia community and included 28 participants. Within each session, participants were randomly assigned to either the intervention or control group. Participants assigned to the intervention group completed the daily StrongerMemory program exercises (reading, writing, and simple math). In addition, they attended weekly, in-person social engagement meetings led by a moderator. These sessions lasted approximately 70 min and were designed to foster peer interaction and discussion. Participants assigned to the control group completed the same daily StrongerMemory program exercises. To control for program engagement, this group received biweekly check-in messages via email or text and were invited to two informational webinars about the StrongerMemory program. These contact points were purely informational and did not involve peer-to-peer social interaction.
In the first session, 17 participants were assigned to the intervention group and 17 to the control group. Following participant withdrawals, the final sample for this session consisted of 30 individuals, with 15 in each group. The 12-week StrongerMemory program was conducted from December 14, 2022, to March 1, 2023.
The second session, conducted from March 24, 2023, to June 9, 2023, began with 14 participants in both the intervention and control groups. However, several participants did not complete the program, resulting in a final count of 11 in the intervention group and 9 in the control group. Across both sessions, the final analyzed sample consisted of 50 participants: 26 in the intervention group and 24 in the control group.
Ethical Considerations
The study protocol was approved by the Institutional Review Board (IRB) at George Mason University. Informed consent was obtained from all participants before their participation, ensuring they understood the research aims and procedures. Confidentiality and anonymity were maintained throughout the study. All collected information was de-identified after screening.
While this study involved individuals with subjective cognitive decline (SCD), not clinical cognitive impairment, we recognize the ethical complexities that may arise when engaging populations whose cognitive capacities could fluctuate. As highlighted by Saleem et al. (2024), ethical research in this context requires particular attention to ensuring truly informed consent, safeguarding participant dignity, and respecting autonomy. These principles were central to our research design and implementation, reflecting our commitment to protecting the rights and well-being of all participants.
Measures
Demographic information was collected at baseline through a self-administered questionnaire, gathering data on age, gender, race/ethnicity, marital status, education level, employment status, household income, and residence type. Weekly logs were submitted by participants detailing time spent on each of the exercises (reading aloud, simple math and writing). The extent and patterns of social engagement among participants in the intervention group were assessed following the 12-week intervention period. A structured survey instrument was used to evaluate participants’ social engagement, including the frequency and type of social interactions, involvement in group activities, and use of technology for communication. This tool provided both quantitative and qualitative insights into their social behaviors.
Subjective Cognitive Decline Questionnaire (SCD-Q)
This 24-item self-report questionnaire, used for both screening and outcome assessment, measured perceived changes in memory, language, and executive functioning over the past 2 years (Rami et al., 2014). Participants responded with “yes” or “no” to each item, with higher scores indicating greater perceived cognitive decline. The SCD-Q demonstrated excellent internal consistency (Cronbach’s alpha = .90 pre-intervention, 0.92 post-intervention).
Montreal Cognitive Assessment (MoCA) Blind/Telephone Version 8.1
Telephone-administered assessment evaluating various cognitive domains including attention/concentration, memory, language, conceptual thinking, calculations, and orientation was used (Nasreddine et al., 2005). The K-MoCA, a culturally adapted Korean version, was used for Korean participants. Assessment time was approximately 5 to 10 min, with a total possible score of 22 points. A score of 18 or above was considered normal. The MoCA Blind/Telephone demonstrated very good internal consistency (Cronbach’s alpha = .85 pre-intervention, .82 post-intervention).
Good Health Practices Scale (GHP)
This 16-item questionnaire, a shortened version of the Health Behavior Checklist, assessed healthy behaviors across various domains, including diet, physical activity, sleep, and smoking habits (Hampson et al., 2019). The GHP showed very good internal consistency (Cronbach’s alpha = .85 pre-intervention, .82 post-intervention).
The Short Warwick-Edinburgh Mental Well-Being Scale (SWEMWBS)
The SWEMWBS, 7-item measure of mental well-being, assessed positive feelings and thoughts over the past 2 weeks (Shah et al., 2021). Higher scores indicate higher levels of mental well-being. The SWEMWBS demonstrated very good internal consistency (Cronbach’s alpha = .80 pre-intervention, .76 post-intervention).
Data Analysis Procedures
This quantitative study evaluated the impact of social engagement in the StrongerMemory program using a pretest-posttest control group design. Data analysis was conducted using SPSS 27.0. For each variable, measures of central tendency (mean, median) and variability (standard deviation, range) were calculated. To assess deviations from normality, skewness and kurtosis were also evaluated. The underlying factor structure of the scales was examined using exploratory factor analysis, and internal consistency was assessed using Cronbach’s alpha. Paired-samples t-tests were conducted to compare pre- and post-test scores on the MoCA Blind/Telephone, SCD-Q, SEAQ, GHP, and SWEMWBS scales within each group, in order to assess the effectiveness of the intervention. The homogeneity of variances was tested using Levene’s test (p > .05) and data normality was verified using the Shapiro-Wilk test (p > .05) before the t-tests. The amount and significance of any observed changes were measured using Cohen’s d effect sizes, t-statistics, mean differences, and standard errors of the mean. Lastly, a one-way ANCOVA was conducted to investigate the impact of social engagement on outcomes. To compare post-test results on cognitive, behavioral, and emotional outcomes between the intervention and control groups, baseline (pre-test) scores were considered as covariates. By accounting for pre-existing group differences, this method improves statistical power and allows for a more accurate assessment of the intervention’s effect (Jennings & Cribbie, 2021).
With this approach, we examined, based on the pre-test scores, whether there is a difference between the post-test scores for the intervention and the control groups. Following this, to further contextualize our findings and address the study’s sample size, post-hoc power analyses were conducted using G*Power (Version 3.1.9.7) to determine the achieved power for detecting the observed effect sizes for each ANCOVA outcome (Faul et al., 2007).
Results
Descriptive Statistics Results
Participant Demographics
Table 1 presents the demographic characteristics of the study participants. The participants had an average age of 73.6 years, ranging from 60 to 88 years. The majority were female (76.0%) and Asian American (60.0%). Most participants were married (71.4%) and lived in a home with other individuals (70.0%). The majority were retired (83.3%), with a bachelor’s degree being the most common level of education (38.0%). Annual household incomes varied; however, the most common income range was $50,000 to $100,000 (38.0%). The demographic characteristics of the intervention and control groups were largely similar, although some differences were noted. The intervention group was slightly older, more likely to have been male, to have lived alone, and to have had higher socioeconomic status.
Demographic Characteristics.

Social Engagement in Weekly Meetings (Intervention Group, n = 26)
Participants in the intervention group demonstrated a high level of engagement throughout the 12-week period. As detailed in Table 2, participants attended an average of 9.6 out of 12 sessions, each lasting approximately 71 min. The majority (70.8%) reported strong commitment to participation. Most attendees frequently responded to the moderator (70.8%), asked questions (50%), provided feedback to peers (79.2%), and shared their own experiences (98%). Overall, 82.6% of participants rated the group sessions positively, suggesting the experience was both worthwhile and rewarding.
Social Engagement in Weekly Meetings.

Paired t-Test Results
Both the control and intervention groups showed a statistically significant improvement in MoCA scores, with mean scores increasing from 18.3 to 18.9 (t(23) = −2.61, p = .016) for the control group and 17.5 to 19.4 (t(25) = −4.27, p < .001) for the intervention group. The intervention group exhibited significantly greater mean improvements from pre- to post-intervention than the control group. Additionally, both groups reported a significant decrease in perceived cognitive decline (SCD-Q scores; control group: Mean = 8.5–6.4, t(23) = 2.90, p = .008; intervention group: Mean = 11.6–7.2, t(25) = 3.60, p < .001), with a greater reduction observed in the intervention group.
The control group showed a significant improvement in self-reported health behaviors after the intervention (t(23) = −3.44, p = .002). The intervention group showed a small increase in GHPS scores, but this change was not statistically significant (t(25) = −0.93, p = .361). The control group showed no significant change in emotional well-being, whereas the intervention group showed a significant increase in SWEMWBS scores, suggesting a potential positive impact on mental well-being (t(25) = −1.98, p < .05; Table 3).
Results of Paired T-Tests for Control and Intervention Groups.

p < .05. **p < .01. ***p < .001.
ANCOVA Results
To determine the actual influence of social engagement, an Analysis of Covariance (ANCOVA) was conducted. ANCOVA adjusts pre-intervention scores, allowing for a more precise evaluation of treatment effectiveness by reducing the potential bias caused by initial group differences. Although the t-test method compares the mean between groups, it may not completely account for pre-existing differences that could impact the results. By considering the possible influence of the pre-test scores, ANCOVA offers adjusted outcome means.
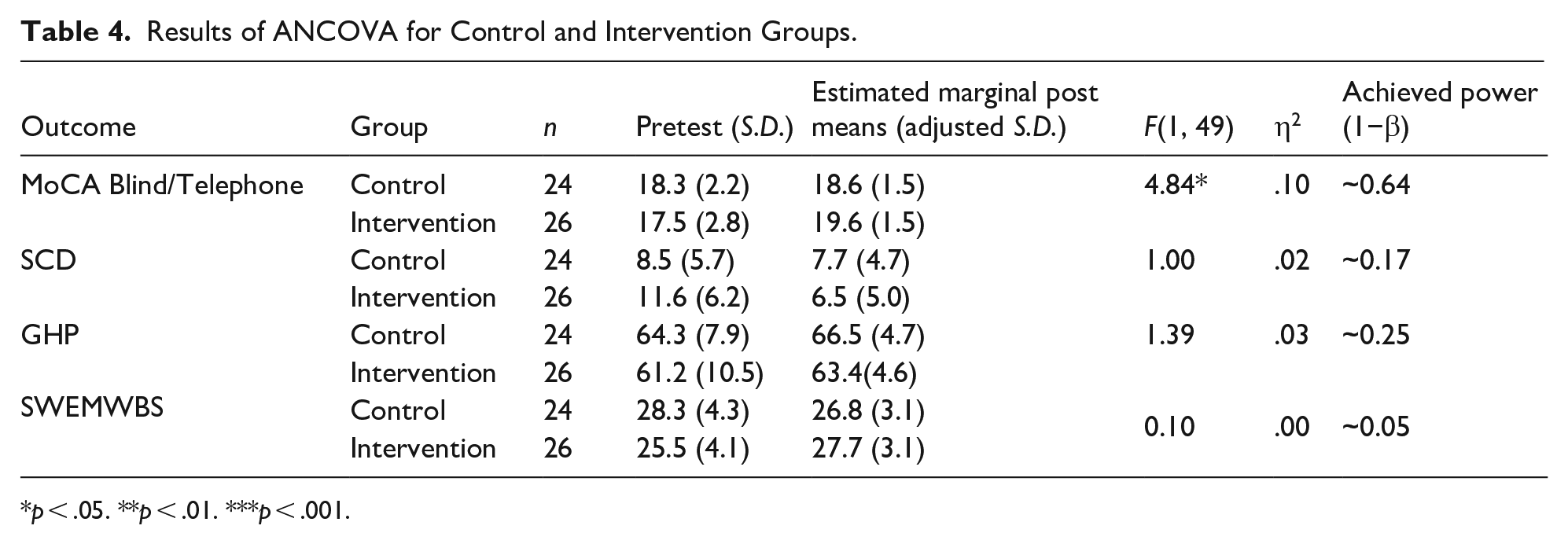
Focusing on the MoCA outcome, Levene’s test results (F(1, 49) = 3.42, p > .05) confirmed the assumption of homogeneity of regression, indicating a consistent relationship between pre- and post-test MoCA scores across both groups. After controlling for pre-test MoCA scores, a significant main effect for the intervention was observed (F(1, 49) = 4.84, p < .01, partial η2 = .10). This medium to large effect size suggests that the StrongerMemory program with weekly social meetings significantly enhanced cognitive performance in the intervention group compared to the control group. A post-hoc power analysis conducted using G*Power indicated an achieved power of approximately 0.64 for this outcome (Faul et al., 2007). While the effect was statistically significant, this power level is below the conventional target of .80 (Cohen, 1988), suggesting the study was somewhat underpowered to detect an effect of this magnitude.
ANCOVA results did not reveal a significant effect of social engagement on the program’s effectiveness regarding Subjective Cognitive Decline (SCD), Good Health Practices (GHP), or satisfaction with emotional well-being (SWEMWBS). While slight differences in estimated marginal post means were observed between the intervention and control groups for these outcomes, they were not statistically significant and showed small effect sizes, suggesting no meaningful impact of the program on these aspects (Table 4).
Results of ANCOVA for Control and Intervention Groups.
p < .05. **p < .01. ***p < .001.
Discussion
The results of the paired t-test indicated that both the control and intervention groups showed significant improvements in MoCA scores. Decreases in SCD scores were consistent with the findings of Phase I of our research (Kang et al., 2022), which confirmed that the StrongerMemory program has a positive impact on cognitive function in older adults. This is consistent with previous research showing the positive impacts of non-pharmacological cognitive interventions on cognitive functions (Chang et al., 2021; Li et al., 2017).
Our findings also highlighted an interesting, albeit unexpected, development in health behaviors. Specifically, the control group exhibited substantial enhancements in self-reported health practices (GHP). One possible explanation for this observation is that engagement in the structured StrongerMemory program, which was common to both groups, may have fostered a general increase in self-monitoring or discipline that carried over to health behaviors. This interpretation aligns with previous research indicating that engagement in structured activities can support healthy eating behaviors (Yang et al., 2019) and healthy daily functioning (Willis et al., 2006).
In addition, the significant improvement in SWEMWBS scores observed in the intervention group implies that including social engagement into the StrongerMemory program could potentially have a positive effect on emotional well-being. This enhancement in emotional well-being might be attributed to the supportive peer interactions and shared experiences fostered by the weekly group meetings (Kotwal et al., 2021), which can create a sense of community and mitigate feelings of isolation sometimes associated with subjective cognitive decline.
The influence of social engagement on the effectiveness of the StrongerMemory can be more accurately determined through our ANCOVA analysis results. Overall, the results showed a promise for improving older adults’ cognitive outcomes by combining the StrongerMemory program with social engagement. Once baseline scores were taken into consideration, the intervention group showed significantly higher improvements in cognitive function as measured by the MoCA when compared to controls. The ANCOVA results can contribute to the growing body of evidence supporting the efficacy of multi-domain interventions for cognitive health.
An important example is the Finnish Geriatric Intervention Study to Prevent Cognitive Impairment and Disability (FINGER) program, which shows that physical activity, cognitive training, dietary counseling, vascular risk management, and social engagement can all work together to protect cognitive function in older adults who are at risk (Ngandu et al., 2015; Rosenberg et al., 2020). Our study further emphasizes how crucial social engagement is to these kinds of multi-domain lifestyle interventions. The beneficial effects seen in the intervention group, especially in terms of improved cognitive performance, highlight the importance of encouraging social engagement as a fundamental element of programs aimed at improving older adults’ cognitive health.
Our findings on the potential benefits of combining daily brain exercise with social engagement have important implications for both practice and policy. In practice, this study encourages the use of social interaction in groups within cognitive interventions. Practitioners can encourage a more comprehensive approach to cognitive health in later life and improve the efficacy of interventions by encouraging social interactions as well as cognitive stimulation. Policymakers should emphasize legislation and financing that supports social integration initiatives within varied community settings to address the growing issues of social isolation as well as ADRD issues among older adults. More broadly, these initiatives can contribute to reducing healthcare costs by preventing or delaying the onset of ADRD and improving people’s overall well-being by increasing their opportunities to form meaningful social connections.
Conclusion
This study has some limitations that should be considered in future research. As an exploratory study, the sample size was small, which may restrict the generalizability of our findings and, as indicated by our post-hoc power analysis, provided limited statistical power to detect effects. Future research with larger, adequately powered samples is therefore crucial for validation. The relatively short 12-week intervention period inherently limits our ability to assess the long-term persistence of the observed benefits. Longer-term studies with extended follow-up periods are crucial to evaluate the sustained efficacy of this combined intervention and to understand the trajectory of cognitive and emotional well-being beyond the immediate post-intervention phase.
Furthermore, we observed that consistent and active social participation at our weekly meetings was likely a key factor in the overall effectiveness of the Stronger-Memory program. It would be interesting to investigate in future research how different levels of participation, both in terms of quantity and quality affect the success of the StrongerMemory program. This exploratory study did not include a formal quantitative assessment of intervention fidelity. future research should develop a more detailed protocol for the social engagement component and implement rigorous fidelity measures to support consistent delivery and improve the replicability of findings.
Additionally, the intentional recruitment of Korean participants suggests that StrongerMemory, which relies on everyday activities that are easily accessible across cultural boundaries, has the potential to overcome language barriers. While our study found similar program effects between Korean and American participants, future research should explore the impact of social engagement within the program on individuals from diverse racial and ethnic backgrounds.
Approximately 11.7% of older adults aged 65 and over reported some degree of cognitive impairment (CDC, 2019). A comprehensive understanding of cognitive impairment is essential for addressing the challenges associated with aging. Our efforts should focus on the development of a practice base that can serve as a guide to future research on interventions that can mitigate the effects of cognitive impairment among older adults. This study provides promising results about the potential benefits of incorporating social interaction into brain exercises, such as the StrongerMemory program, for older adults who are experiencing SCD.
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Virginia Center on Aging (2022-2023 Alzheimer’s & Related Diseases Research Award Fund).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
This study was not preregistered. Data and materials will be made available upon request.