Abstract
Objectives:
The aims of the study were to investigate the proportion of frailty among inpatients having Acute Coronary Syndrome (ACS) and to compare the prognostic value of three frailty screening methods in predicting in-hospital adverse events among the study population.
Methods:
This prospective, observational study design was conducted on older patients with ACS. Data was collected using a structured questionnaire on general characteristics (age, gender, comorbidities, body mass index) and medical records (admission diagnosis, ACS type, angiography results, treatment therapy, left ventrical ejection fraction, and length of hospital stay). Frailty was assessed using three frailty screening scales: Reported Edmonton Frail Scale (REFS), Clinical Frailty Scale (CFS), and Frail Scale (FS).
Results:
A total of 116 older patients, the mean age was 72.9 (SD: 6.2) years. Prevalence of frailty in older inpatients with ACS was 44.8%, 35.3%, and 32.7% according to REFS; CFS, and FS, respectively. In addition, 75.9% were treated with percutaneous coronary intervention and the length of hospitalization was 6.3 (SD: 3.8) days. The AUC in the prognosis of net adverse clinical events (NACE) for patients with ACS was 0.74 (with REFS ≥ 7 points), 0.76 (with CFS ≥ 5 points), and 0.80 (with FS ≥ 2 points). Kappa values of 0.49, 0.57, and 0.47 were observed for REFS, CFS, and FS.
Conclusion:
This study compared three frailty screening tools in predicting in-hospital adverse events among Acute Coronary Syndrome. The Frail Scale showed the highest value to predict NACE and demonstrated its superiority over other frailty scales.
Introduction
According to the World Health Organization (WHO), ischemic heart disease is the leading cause of mortality globally, responsible for 16% of the world’s total death (WHO, 2020). With the fast population aging, the incidence and the health-related burden of Acute Coronary Syndrome (ACS) is steadily increasing. Advanced age, along with geriatric syndromes, is also an important risk factor for complications and adverse outcomes of ACS.
Frailty is a common geriatric syndrome defined as a state of decreased vulnerability and decreased physiological reserve following stressor events (A. T. Nguyen et al., 2019). In Vietnam, a typical developing country in South East Asia with a rapidly aging population, nearly half of hospitalized older patients with ACS were diagnosed as frailty (T. V. Nguyen et al., 2019). The presence of frailty in older people can cause a concerned burden for patients with ACS. The previous structured review highlighted that frailty was independently associated with mortality in patients having ACS (Adjusted all-cause mortality HR for patients with frailty: ranged from 1.5 to 5.4; Bebb et al., 2018). Today, frailty is well-known as a chronic condition in older patients, however, has not yet become a part of routine examination and management of ACS in clinical practice.
The geriatric condition of frailty was widespread among elderly patients in Vietnam (Vu et al., 2017). It is a biological vulnerability to adverse effects brought on by aging-related alterations in several physiological systems. Patients who are frail are more prone to experience poorer outcomes, a slower rate of recovery, more iatrogenic complications, and a greater death rate (Murad & Kitzman, 2012; Singh et al., 2014). Frailty was also connected to a higher chance of dying, being hospitalized, falling, and losing function (Dupre et al., 2009; Gharacholou et al., 2012; McNallan et al., 2013). It is well known that fragility is associated with a worse prognosis in ACS patients (Ekerstad et al., 2011; Graham et al., 2013; Matsuzawa et al., 2013; Wallace et al., 2014). However, the majority of tools focused on the basic phenotypic categories of frailty as assessed by physical performance tests, and frailty was quantified in the early stages of ACS utilizing specific difficult-to-measure items (walk speed, handgrip strength; Dodson et al., 2016; Graham et al., 2013; Matsuzawa et al., 2013; White et al., 2016). In this situation, simple scales such as REFS, CFS, and FS are appealing tools for assessing frailty since they are concise, interview-based, and straightforward to administer, score and interpret in clinical practice. It is important to have a valuable and feasible scale to screen for frailty in ACS patients. Early diagnosis of frailty helps medical staff to develop a comprehensive healthcare strategy, to prevent complications and reduce mortality for ACS patients. That makes more sense when it helps to reduce direct and indirect treatment costs and improve the quality of life for patients. The aims of the study were to investigate the proportion of frailty among inpatients having ACS and to compare the prognostic value of three frailty screening methods in predicting adverse events in patients with ACS.
Methods
Study Design and Participants
This prospective, observational study design was conducted in older patients with ACS admitted to Vietnam National Heart Institute, Bach Mai Hospital, Hanoi, Vietnam from February 2021 to August 2021.
Participants were recruited if they: (1) were ≥60 years old; (2) were diagnosed with ACS at admission according to 2016 American Heart Association (AHA) and American College of Cardiology (ACC) guidelines (WHO, 2020). Participants were excluded if they: (1) were unable to understand and/or answer the questions, (2) suffered from other acute or severe illness or received intensive care, (3) had cognitive impairment or dementia or delirium, or (4) did not agree to participate in the study.
The study protocol was approved by the institutional review board of Hanoi Medical University (Reference number: 3916/QD-DHYHN). All participants or their representatives/guardians were asked to give their written informed consent and they could withdraw anytime. Their information was kept confidential and used only for research purposes.
Sample Size and Sampling
The sample size was calculated using a formula, sample size: n = Z2 1 − α/2 × (p × (1 − p)/d2), with n = the required sample size, Z1 − α/2 = 1.96 (with α = .05 and 95% confidence interval), p = .113 (the prevalence of major adverse events in the frail group in the study by Alonso Salinas, Sanmartín Fernández et al. (2016)), and d = precision (assumed to be 0.05). Therefore, our study’s sample size was calculated to be at least 79 participants.
All patients meeting inclusion and exclusion criteria were recruited for a period of 6 months of the study. A total of 116 older patients with ACS were recruited in the study.
Data Collection
Data was collected using a structured questionnaire on general characteristics and from medical records. The face-to-face interviews were conducted on the day of admission by cardiologists working at the Vietnam National Heart Institute, Bach Mai Hospital.
Frailty Definition
Frailty was assessed using three frailty screening scales: Reported Edmonton Frail Scale (REFS), Clinical Frailty Scale (CFS), and Frail Scale (FS). The REFS which was developed by Hilmer et al in 2009, includes nine frailty domains: cognition, general health status, functional independence, social support, medication use, nutrition, mood, continence, and functional performance. The Reported Edmonton Frail Scale (REFS) has a score range from 0 to 18. The Clinical Frailty Scale (CFS) was created by Rockwood et al. in 2005. The score evaluates specific domains including comorbidity, loss of physical and cognitive function. The score ranges from very fit (CFS = 1) to very severely frail (CFS = 8) and terminally ill (CFS = 9). The Frail Scale (FS) was developed by the International Association of Nutrition and Aging to provide a simple approach with good predictive value. The score evaluates 5 specific domains including fatigue, resistance, ambulation, illnesses, and loss of weight. The score ranges from very fit (FS = 0) to frail (FS = 5).
Outcome Variables
In-hospital adverse outcomes included arrhythmia (defined as any of the following arrhythmias: atrial fibrillation, atrioventricular block, ventricular tachycardia, ventricular flutter), acquired pneumonia, stroke, major bleeding (bleeding that provoked a decrease of over 5 g/dl hemoglobin or 15% hematocrit or required blood transfusions) and all-cause mortality. Net adverse clinical events (NACE) was defined as a composite of all-cause mortality, stroke, and major bleeding).
Other Measures
Socio-demographic variables comprised sex (male and female), and age (years). Comorbidities included hypertension, diabetes, dyslipidemia, previous stroke, chronic obstructive pulmonary disease (COPD), and heart failure. Comorbidities were obtained from medical records based on doctors’ diagnoses.
Weight and height were measured and used to calculate the body mass index (BMI) using the formula weight/height2 (kg/m2). BMI was categorized into three groups: underweight (<18.5), normal (18.5–22.9), overweight and obesity (≥23).
Information obtained from medical records included: ACS type (including ST-elevation myocardial Infarction; non-ST elevation myocardial infarction and unstable angina), coronary angiography assessed by Quantitative Coronary Angiography (one-vessel coronary artery disease (CAD) was defined as over 70% stenosis in one major coronary vessel, two-vessel CAD was defined as over 70% stenosis in two major coronary vessels or in left main, three-vessel CAD was defined as over 70% stenosis in three major coronary vessels or in left main and right coronary artery), treatment therapy (percutaneous coronary intervention—PCI during hospitalization or medication treatment). Left ventricle ejection fraction (LVEF) in echocardiography was divided into two groups: heart failure with reduced ejection fraction LVEF < 53% and normal ejection fraction with LVEF ≥ 53% (according to the American Society of Echocardiography). Length of hospital stay (days) was also collected from medical records.
Statistical Analysis
All data analysis used the Stata/SE 16.0. Characteristic variables were expressed in numbers, percentages, and mean (and standard deviations, SD). The qualitative variables are presented with percentages. T-test and Chi-squared test were used to measure relationships between frail and non-frail groups and other characteristics. ROC curve statistical analysis was done to determine the cut-off and evaluate the prognostic efficacy of three frailty scales. A p-value < .05 was considered a statistical significance.
Results
A total of 116 older patients were recruited in the study. The mean age was 72.9 (SD: 6.2) years. Women accounted for 40%. The most popular type of ACS was unstable angina (51.7%). In the study population, 75.9% were treated with PCI and the length of hospitalization was 6.3 (SD: 3.8) days. Prevalence of frailty in older inpatients with ACS was 44.8%, 35.3%, and 32.7% according to REFS; CFS, and FS, respectively (Table 1).
Patient Demographic and Clinical Characteristics.

Note. COPD = chronic obstructive pulmonary disease; BMI = body mass index; ACS = acute coronary syndrome; STEMI = ST elevation myocardial infarction; NSTEMI = non-ST elevation myocardial infarction; CAD = coronary artery disease; PCI = percutaneous coronary intervention; LVEF = left ventricle ejection fraction; NACE = net adverse clinical outcome.
The AUC in the prognosis of NACE for patients with ACS was 0.74 (with REFS ≥ 7 points), 0.76 (with CFS ≥ 5 points), and 0.80 (with FS ≥ 2 points; Table 2).
Prognostic Characteristic of Net Adverse Clinical Outcome (NACE) for Patients With ACS.
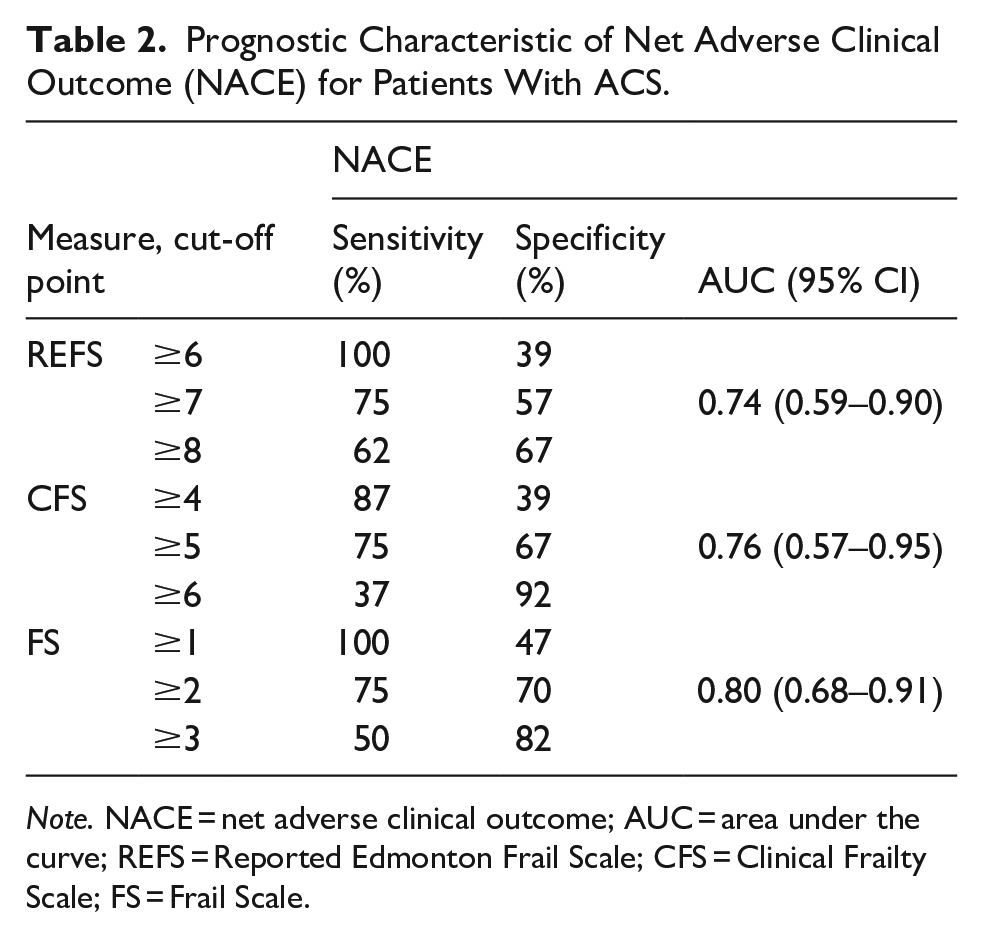
Note. NACE = net adverse clinical outcome; AUC = area under the curve; REFS = Reported Edmonton Frail Scale; CFS = Clinical Frailty Scale; FS = Frail Scale.
The Kappa coefficient between three frailty scales was analyzed in our sample. Kappa values of 0.49, 0.57, and 0.47 were observed for REFS, CFS, and FS respectively, indicating a moderate agreement between the three frailty scales.
The ROC curve analysis showed that the Frail Scale had the largest area under the curve (AUC = 0.80) among the three scales, linking frailty to adverse outcomes. The AUC of the REFS, CFS, and Frail scales are shown in Figure 1.

ROC curves of the different frailty scales to analyze the strength of association with NACE.
We conducted multivariate regression analysis, evaluating the impact of factors such as frailty scale, age, gender, and treatment to NACE.
Discussion
The main result of this study was that the proportion of frailty in elderly patients with ACS was relatively high, accounting for 32% to 45% of patients, and frailty tools showed valuable in predicting NACE risk in older patients with ACS. This finding is in line with some prior studies showing that the high prevalence of frailty in older patients with ACS ranges from 27% to 49% (Alegre et al., 2018; Ekerstad et al., 2014; Tonet et al., 2019) depending on the tools used in the study. Some previous studies also have demonstrated the prognostic value of these tools in predicting adverse outcomes in patients with ACS (Alegre et al., 2018; Dodson et al., 2016; Ekerstad et al., 2011, 2014; Graham et al., 2013; Matsuzawa et al., 2013; T. V. Nguyen et al., 2019; Núñez et al., 2017; White et al., 2016). Some studies use tools with fairly objective assessments, based on measurements of physical performance such as the gait speed in older patients with myocardial infarction (Matsuzawa et al., 2013) or the Short Physical Performance Battery (SPPB; Campo et al., 2017) in patients receiving cardiac interventional procedures. Some other studies used frailty assessment tools mixed interview and measurements of physical performance such as Fried score (White et al., 2016) or SHARE-FI (Alonso Salinas, Sanmartín Fernández, & Pascual Izco, 2016) which is a mix of questions about exhaustion, appetite, physical activity, ambulation, resistance and measurement of grip strength, or The Edmonton Frail Scale (EFS) (Graham et al., 2013) is a scale including questions and tasks about nutrition, symptoms, mood and physical performance. However, these tools do not demonstrate differences with scales that use only interviews. In fact, in the context of people with ACS, they need maximum quietness and limited physical activities, so that interview-based tools such as REFS, FS, CFS. . .seem more suitable for ACS. Besides, several studies have used practical scales applicable to ACS patients such as CFS (Canadian Clinical Frailty Scale; Ekerstad et al., 2011, 2014) or REFS (Reported Edmonton Frail Scale; T. V. Nguyen et al., 2019) or FRAIL scale (Alegre et al., 2018) to evaluate the value of frailty in predicting events in hospital stay and follow-up. However, these researches were all used a single scale, without head-to-head comparison among these tools, therefore no scale has demonstrated its superiority over the others to discriminate frailty in patients with ACS.
This study presented a concurrent comparison of three frailty instruments, including REFS, CFS, and FS in the same elderly ACS patients. As far as we are aware, this is the initial simultaneous evaluation of these three frailty measures in elderly individuals with ACS in Vietnam. Given that the statistical difference among the AUC of the three scales was small, the results were significant in daily clinical practice. The interesting result is that although two scales, The Clinical Frailty Scale and the FRAIL scale, were able to predict the risk of NACE in ACS. The results of multivariate regression analysis also supplemented the independent prognostic value of FS and CFS scores with NACE events during hospitalization. The FRAIL scale showed the best and acceptable balance between sensitivity and specificity at cut-off ≥ 2. These findings may be explained by different factors. First, the FRAIL scale was also the test obtaining the lower prevalence of frailty (32.7%), which probably increased the specificity as a predictor of adverse events (Leeflang et al., 2013). Second, the FRAIL Scale uses questionnaires related to multidomain evaluation including fatigue, resistance, ambulation, weight loss, and comorbidities, while the CFS seems to lack multidomain evaluation, and some questions lack specific criteria and defined parameters, thus, the assessment might be biased by subjective considerations (Ekerstad et al., 2014).
This study suggested the superiority of the Frail scale compared with the CFS and REFS, as a short frailty screening instrument for use in coronary settings. This data also directly supports the Frail Scale as a new risk stratification in older ACS persons, in addition to currently recommended risk scores. Refining risk evaluation of elderly patients with ACS could add to improving clinical results, accordingly, possibly prompting significant clinical, social, and financial outcomes. This is in accordance with recent guidelines (Lawton et al., 2022) where the recommendation of cautious consideration of the risks and benefits accounting for frailty is vital in decisions about suitable invasive strategy, dual antiplatelet therapy, and secondary prevention programs. In fact, on the first days of hospitalization with ACS, a highly practical, interview-based, non-physical activity assessment frailty tool such as the Frail Scale is feasible and applicable. In contrast, the REFS scale, although it is assessed in many aspects, does not show prognostic value for adverse outcomes, and requires more time to evaluate, so it does not show superior compared to the Frail Scale in urgent ACS. Furthermore, in a previous study, the Frail Scale has also been shown to be effective in assessing frailty and predicting adverse outcomes in patients with NSTEMI (Alegre et al., 2018).
Due to a lack of resource, our study has some limitations, such as the modest sample size and a single-center study design which may limit the generalizability of the findings to all older inpatients with ACS in Vietnam and its observational nature. Consequently, we could not preclude the presence of selection bias and unmeasured confounding. However, despite these limitations, several clinical and subclinical characteristics were comprehensively assessed by face-to-face interviews and physical examinations. This study retrieves interesting data about the superior role of the simple FRAIL scale in the risk stratification of older patients with ACS. Besides, this is only a preliminary study, other longitudinal research with larger sample size is needed to further confirm its comprehensive values in clinical practice.
Conclusion
In conclusion, this study revealed the high proportion of frailty in elderly patients with ACS and compared three frailty screening tools in predicting in-hospital adverse events among ACS. We found that the Frail Scale showed the highest sensitivity–specificity pair to predict NACE and demonstrated its superiority over other frailty scales.
Footnotes
Acknowledgements
We would like to thank all patients participating in this study. We are deeply grateful to all participants who gave their time to participate in this study and thank to all staffs from Vietnam National Heart Institute, Bach Mai Hospital for support me to complete this study.
ORCID iDs
Ethical Considerations
The study protocol was approved by the institutional review board of Hanoi Medical University (Reference number: 3916/QD-DHYHN). All participants or their representatives/guardians were asked to give their written informed consent and they could withdraw anytime. Their information was kept confidential and used only for research purposes.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
