Abstract
Objectives
Recent data demonstrating superior outcomes and enhanced life expectancy with the Ross procedure (RP) have sparked renewed interest and increased utilization. However, the RP remains technically challenging compared to conventional aortic valve replacement (AVR), necessitating specialized expertise and careful patient selection. This study provides global insights into current RP practices to establish best practices and inform new or evolving programs.
Methods
A web-based survey (> 60 questions) was distributed globally to cardiac surgeons known to perform the RP from May 1 to June 30, 2023. The survey queried surgeon experience, RP volumes, patient selection criteria, preoperative imaging, intraoperative techniques, and postoperative care practices. Responses were analyzed to identify global trends and optimal approaches for RP management.
Results
Of the 167 respondents, 123 (74%) performed the RP, with 75% performing 5 to 30 procedures annually. Approximately half involved a second attending surgeon. Most treated younger patients and preferred homografts for right ventricular outflow tract reconstruction. Variations existed in the ventriculoaortic and sinotubular junction support (30% vs 45% always support). Most surgeons (89%) enforce strict blood pressure control postoperatively, with 65% prescribing beta-blockers. About 54% use postoperative inflammatory prophylaxis, and 90% obtain imaging prior to discharge, with 78% performing annual echocardiograms.
Conclusions
This international survey highlights wide variability in RP practices, underscoring the need for standardized training and procedural protocols. These insights may guide new or evolving RP programs, improve access, and ensure durable outcomes by aligning global practices with contemporary evidence.
This is a visual representation of the abstract.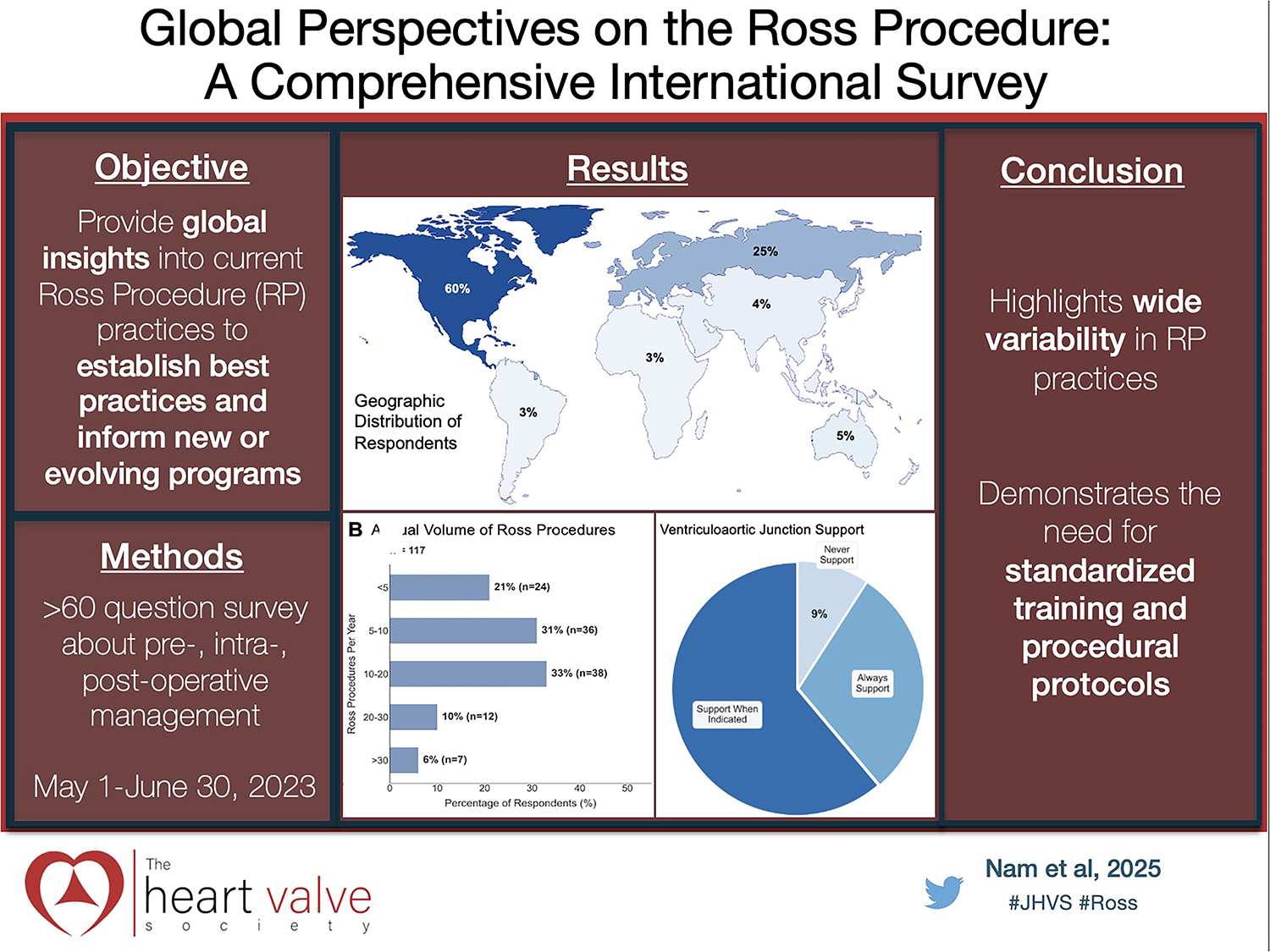
Keywords
Key Points
Significant variability exists among surgeons in technical approaches, patient selection, and postoperative management, highlighting the need for standardized guidelines and comprehensive training programs.
The Ross procedure (RP) represents a nuanced surgical approach where patient selection succeeds traditional contraindication checklists, requiring individualized assessment based on patient characteristics and surgeon expertise.
High-volume surgeons demonstrate more consistent approaches to surgical technique and patient management compared to low-volume surgeons, suggesting that experience plays a crucial role in procedural refinement.
Introduction
Prosthetic aortic valve replacement (AVR) is associated with loss of life expectancy in young adults,1–4 while data suggests the Ross procedure (RP) offers superior outcomes and restores life expectancy compared to the matched general population.5–8 This has sparked a “Ross Renaissance,” with increasing numbers of centers adopting this approach for appropriate candidates. The optimization of RP practices is crucial for patient outcomes.
Despite its proven benefits, significant variation exists in surgeon training pathways, patient selection criteria, operative techniques, and postoperative management of RP patients worldwide.9,10 The absence of standardized approaches to this technically demanding procedure could potentially limit its wider adoption and impact on long-term outcomes. These variations represent a critical knowledge gap in current practice, particularly for surgeons who are learning the procedure or performing it infrequently.
To address these gaps, we conducted the first comprehensive international survey of RP practitioners, gathering detailed data on training pathways, patient selection considerations, technical variations, and postoperative care protocols from 167 surgeons across 6 continents. These global perspectives allow us to identify both consensus practices and areas of significant variation that require standardization to facilitate broader adoption of the procedure. While not intended as a procedural manual, this survey offers granular insights into the pre-, intra-, and postoperative management of the Ross patient, thus providing guidance for surgeons with limited RP experience or those looking to establish new RP programs.
Methods
This survey study received exemption from the Mass General Brigham Institutional Review Board (protocol #2023P000424, approved 3/24/2023), with consent implied by voluntary participation.
The authors collaboratively created a comprehensive questionnaire addressing more than 60 detailed considerations for the RP. Survey sections included surgeon demographics, RP volumes and experience, patient selection criteria, preoperative testing protocols, intraoperative and technical details, postoperative management strategies, and case-based assessment of patient candidacy using 10 scenarios rated from 0 (not a candidate) to 10 (ideal candidate).
The questionnaire, included as Supplemental Material 1, was used to generate a web-based survey, which was maintained using Research Electronic Database Capture (REDCap, Vanderbilt University). The survey link was disseminated through social media platforms and via email to cardiac surgeons known to perform the RP. To increase participation and ensure international representation, a PubMed search for “Ross procedure” yielded over 300 publications within the last 5 years. The authors’ names were compiled, and CTSNet's Surgeon Profile search feature was used to obtain email addresses for those attending/consultant surgeons who have a CTSNet account. Surgeons who received the survey but did not perform the RP were asked about their prior experience or training with the RP and whether they would be interested in learning how to perform it. They were also encouraged to forward the survey to colleagues who do perform the RP. The survey was available for completion between May 1, 2023, and June 30, 2023.
Descriptive statistics were used to analyze response patterns, with categorical variables presented as frequencies and percentages, and continuous variables presented as medians with interquartile ranges (IQR). No formal statistical testing was performed, given the descriptive nature of this survey study.
Results
Survey Respondents
Of the 167 respondents, 123 (74%) perform the RP and 44 (26%) do not (Figure 1A). Most respondents practice in North America, followed by Europe (Figure 1B). Respondents represent various career stages, with 44% in late career (> 15 years), 26.7% in early career (first 5 years), and 29.0% in mid-career (5-15 years) (Figure 1C).
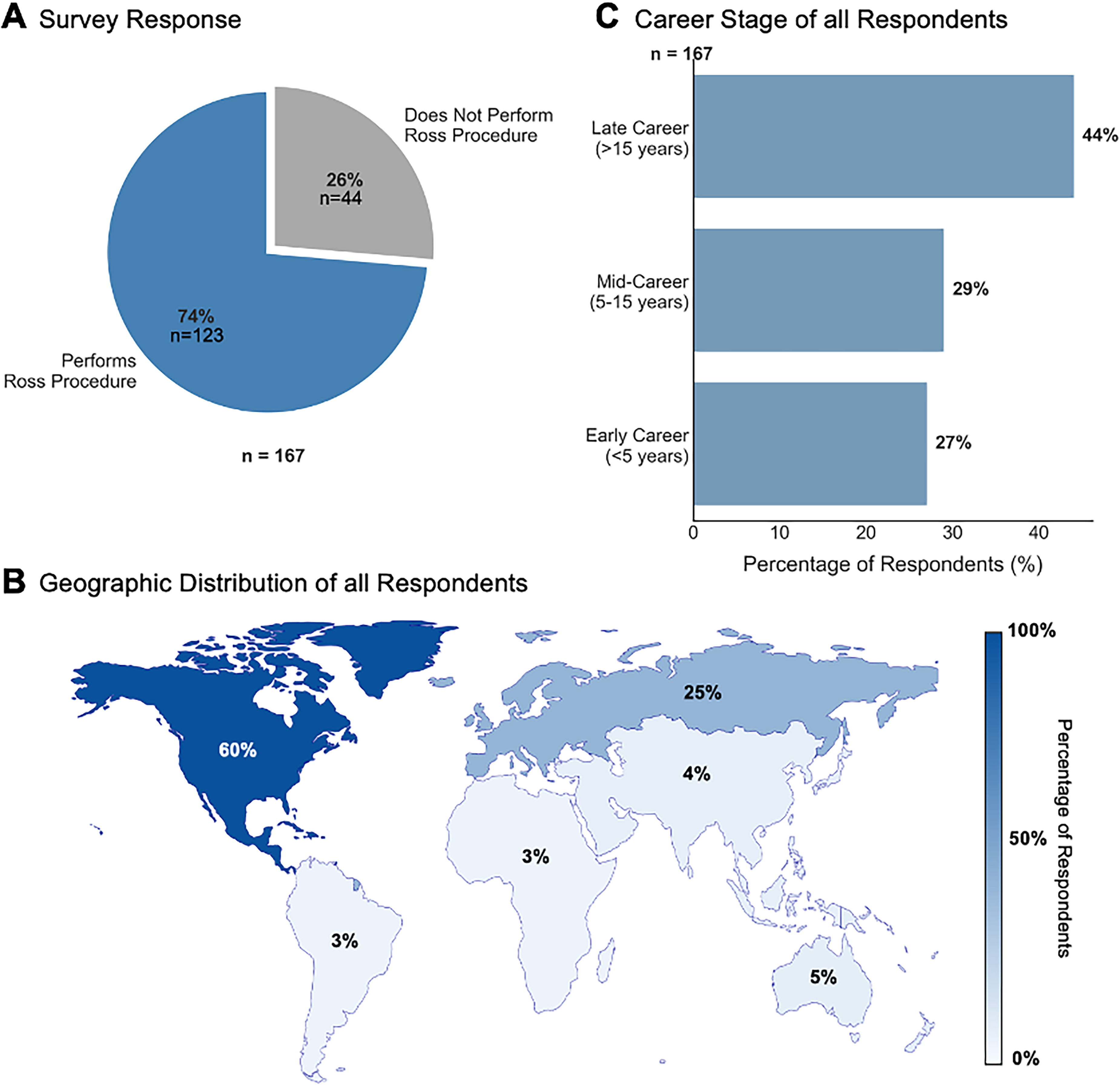
Survey Respondent Demographics. (A) Distribution of Ross procedure (RP) practitioners (74%) versus nonpractitioners (26%). (B) The geographic distribution of respondents with North America (60%) and Europe (25%), predominating. (C) Career stage distribution showing 44% late-career surgeons (> 15 years), with balanced representation of mid-career (29%) and early-career (27%) surgeons.
Non-RP Surgeon Respondents
Among the 44 respondents who do not perform the RP, 27 (68%) cited lack of experience or training as the primary reason, while 13 (33%) do not favor the procedure. Notably, 25 (57%) of these surgeons expressed interest in learning to perform the RP, highlighting an unmet need for training opportunities.
RP Surgeon Respondents
Among RP surgeons, the majority (60%) learned the procedure as attending surgeons, while 29% learned during cardiothoracic surgery residency and 22% during an advanced fellowship (Figure 2A). Most identify as adult cardiac surgeons (61%), followed by congenital (45%) and aortic surgeons (22%). Our survey includes surgeons who have been performing the RP for 1 to 42 years, with a median of 6 years.
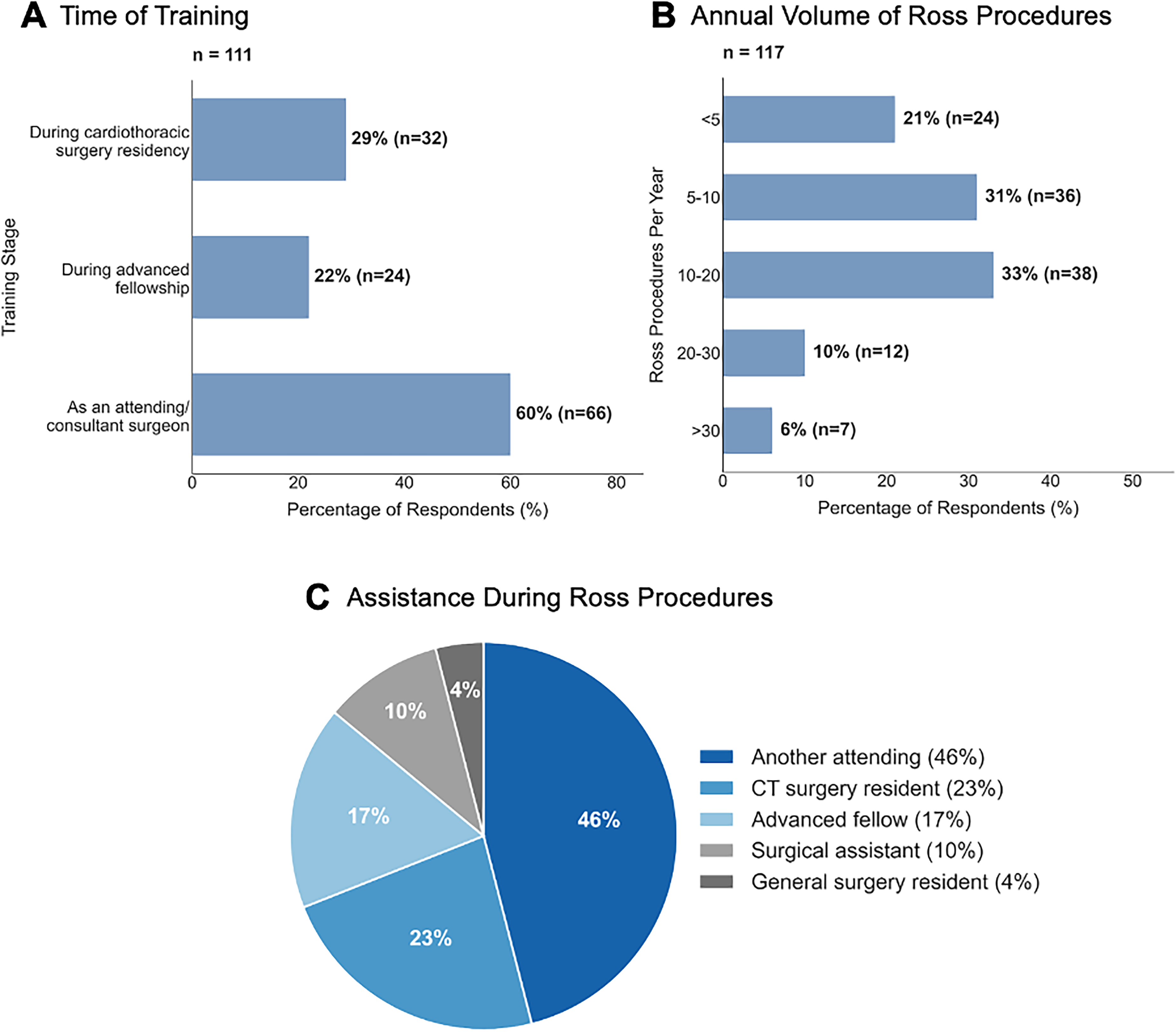
Ross Procedure (RP) Training and Current Practice Patterns Amongst RP Respondents. (A) Most surgeons learned the RP as an attending/consultant (60%), with fewer trained during residency (29%) or fellowship (22%). (B) Annual RP volume distribution, with most surgeons performing 5 to 20 procedures annually (64%). (C) Surgical assistance during RP is primarily provided by another attending surgeon (46%) or cardiothoracic surgery resident (23%).
Regarding annual volume, 33.3% perform 10 to 20 RP per year, 31% perform 5 to 10 procedures, and 6% perform more than 30 procedures annually (Figure 2B). Nearly half (46%) are assisted by another attending surgeon during the procedure, while others are assisted by cardiothoracic surgery residents, advanced fellows, surgical assistants, or general surgery residents (Figure 2C).
Patient Selection
Most RP surgeons (77%) report having an upper age limit for offering the procedure, with a median age of 60 years (IQR 55-65, range 45-80 years).
Preoperative Testing
RP surgeons consistently obtain preoperative testing, with 96% performing preoperative echocardiography, and around 70% obtaining either axial or coronary imaging. Among those obtaining echocardiography, 90% use transthoracic echocardiography (TTE) and 31% transesophageal echocardiography (TEE). For axial imaging, 95% obtain computed tomography (CT) scans and 20% cardiac magnetic resonance imaging (MRI). For coronary assessment, 77% obtain CT angiography (CTA) and 57% perform left heart catheterization (Figure 3).
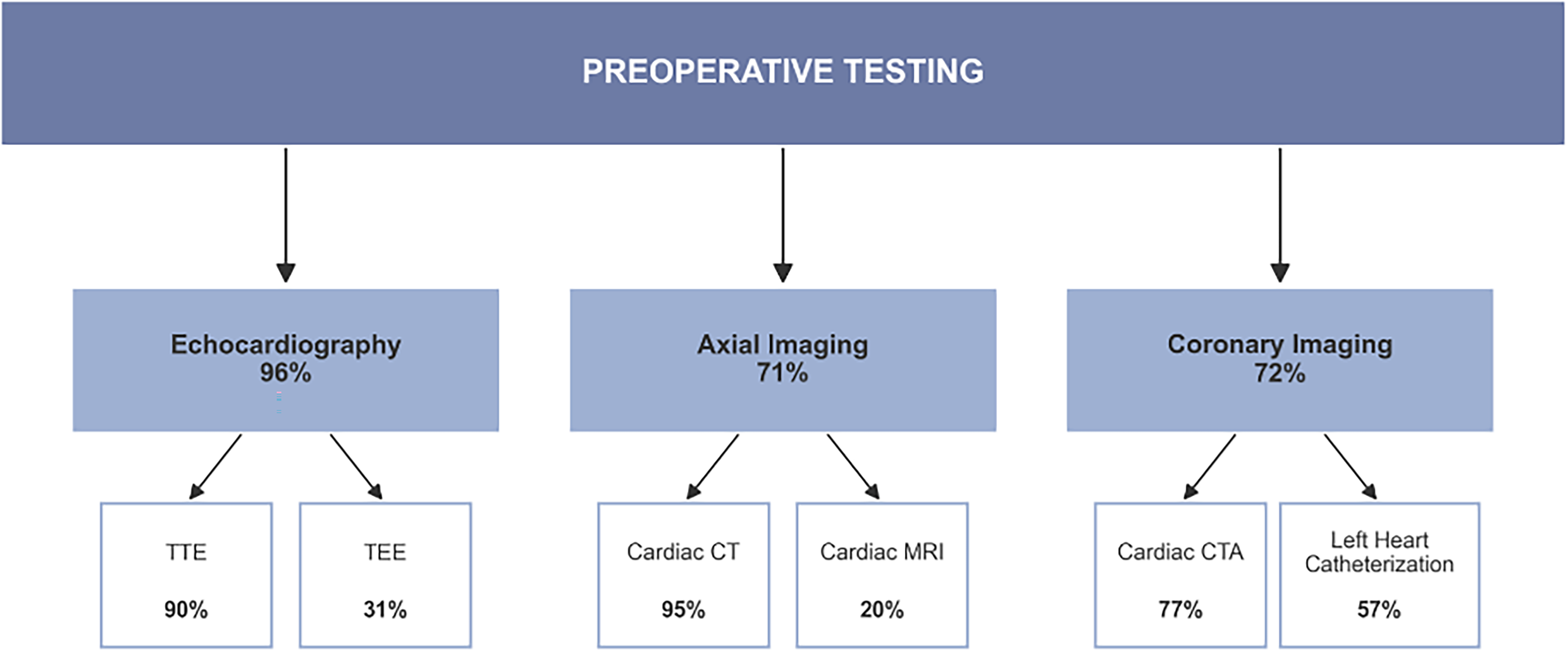
Preferences for Preoperative Testing. Percentages Represent the Proportion of Ross Procedure (RP) Surgeons Obtaining Each Type of Imaging.
Operative Indications
The most common pathologies for which surgeons consider the RP include aortic stenosis (94%), bicuspid/unicuspid aortic valve (86%), aortic regurgitation (78%), endocarditis (57%), and rheumatic heart disease (44%). Figure 4 demonstrates the percentage of surgeons who considered each pathology as an absolute or relative contraindication.
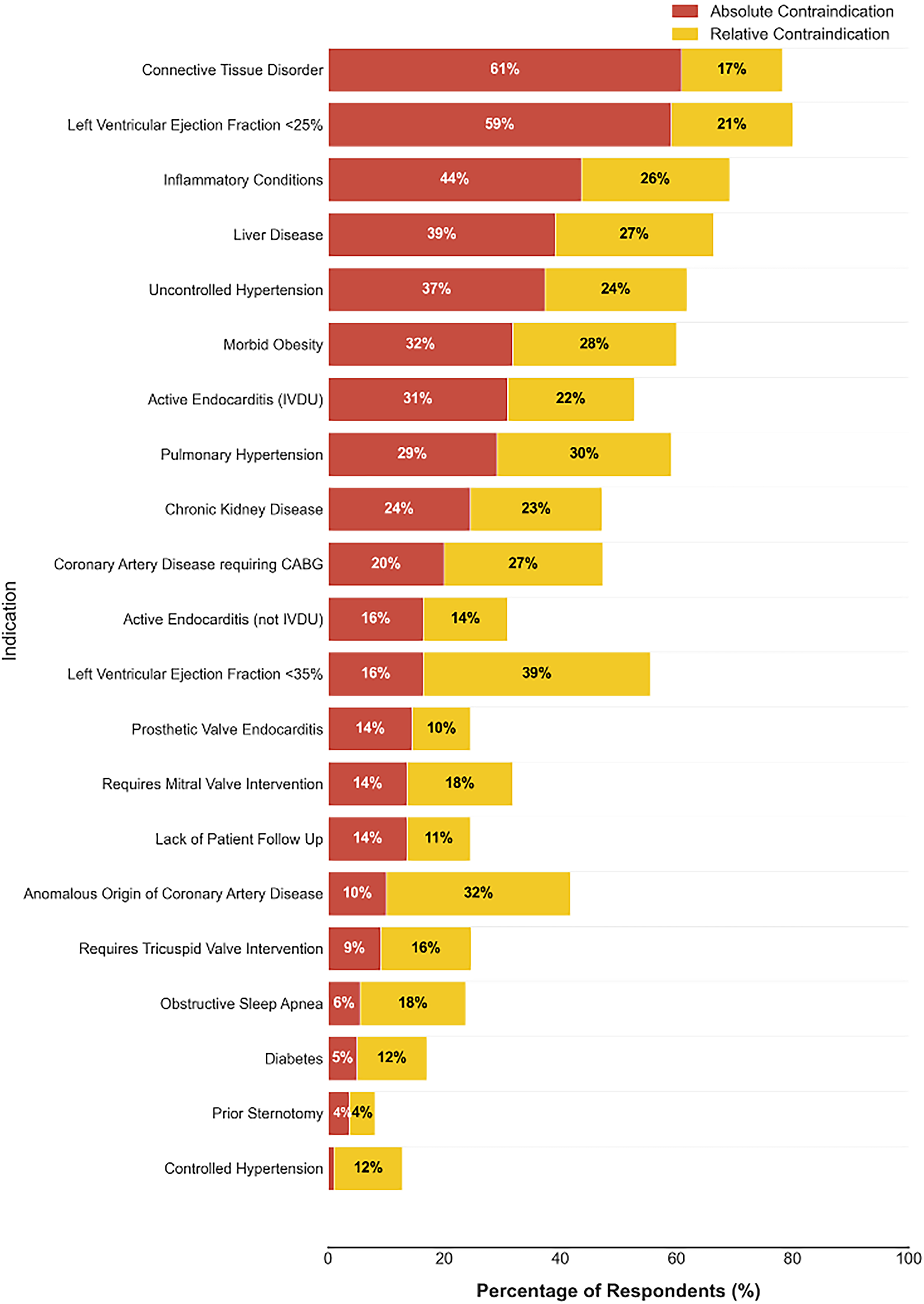
Operative Contraindications. The figure shows the relative proportions of respondents who answered “Yes” for absolute and relative contraindications. The list is organized by the percentage of individuals who answered “Yes” for absolute contraindications, with the fewest percentages at the bottom and the largest percentages at the top. 1% of respondents said controlled hypertension was an absolute contraindication.
The survey revealed substantial variation in how respondents categorized potential contraindications. While certain conditions were more frequently identified as absolute contraindications, the data demonstrated a lack of clear consensus across most clinical scenarios. This heterogeneity suggests that patient selection for the RP frequently involves multidisciplinary assessment, balancing individual patient characteristics alongside surgeon experience rather than adherence to rigid contraindication criteria. The most cited absolute contraindications were connective tissue disorders (60%) and left ventricular ejection fraction (LVEF) < 25% (59%), while controlled hypertension (1%) and prior sternotomy (3%) were rarely considered absolute barriers to the procedure. Among the 60 low-volume surgeons (those performing 10 or fewer RP annually), LVEF < 25% emerged as the most common absolute contraindication, with an LVEF < 35% serving as their primary relative contraindication.
When rating specific patient scenarios (Figure 5), respondents showed strongest consensus for a 59-year-old healthy male with a bicuspid valve and severe aortic stenosis as an ideal candidate. However, the wide range of ratings across all scenarios demonstrates significant variability in patient selection approaches among experts.
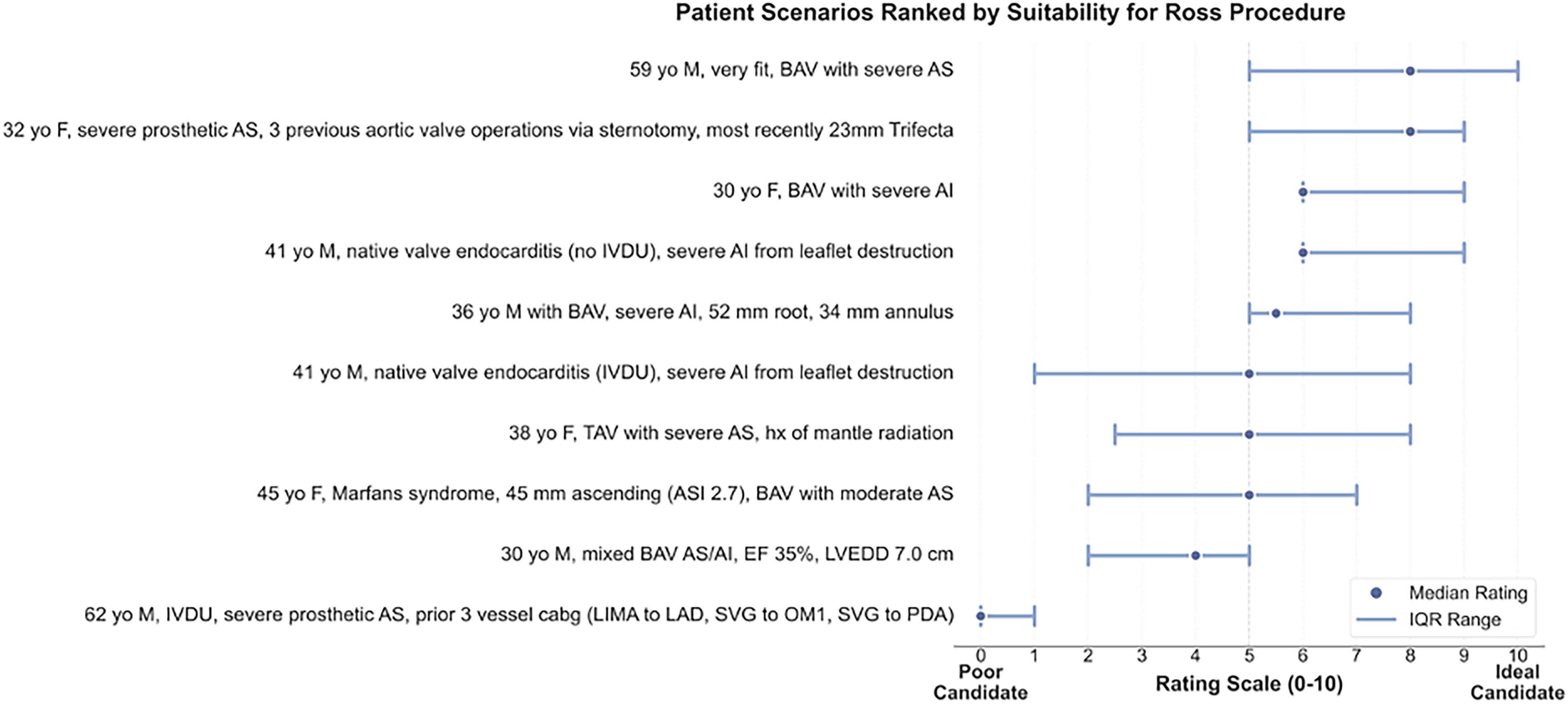
Assessment of Patient Suitability for the Ross Procedure (RP). Patient scenarios are ranked most suitable (top) to least suitable (bottom) based on expert ratings (0-10 scale). Blue dots represent median ratings, and horizontal lines show the interquartile range. The dashed vertical line at 5 marks the midpoint between “Poor Candidate” and “Ideal Candidate.” Higher ratings and narrower ranges indicate stronger consensus on patient suitability for the RP.
Operative Details
Cardioplegia and Bypass
Most respondents (78%) perform bicaval cannulation. The majority (70%) cool patients while on bypass, typically to 32° (63%). An equal number of respondents use standard 4:1 cardioplegia (46%) versus del Nido cardioplegia (46%), with the remainder (8%) using other solutions. For del Nido cardioplegia, the median redosing interval is 60 min (IQR 45-60, range 10-100 min). Most surgeons (58%) deliver antegrade-only cardioplegia, followed by antegrade and retrograde (36%) and retrograde-only (6%).
Autograft Harvest Technique
Only 10% of respondents perform beating heart autograft harvest. Approximately half (52%) scallop the autograft by cutting up into the intercommissural triangles. The median length of the muscular cuff retained is 3 mm (IQR 3-5, range 0-10 mm).
Autograft Implantation
The predominant technique for autograft implantation is full root replacement (87%) (Figure 6A). Most surgeons (59%) use an interrupted suture technique, while 36% use running sutures (Figure 6B). Prolene is the most common suture material (74%), followed by Ethibond (21%), silk (4%), and other materials (2%). Most commonly, surgeons use 4-0 suture size (70%), with fewer using 5-0 (15%), 3-0 (14%), or 6-0 (1%). Only 37% of respondents routinely use material to support the suture line, with Dacron (37%), felt (34%), and pericardium (21%) being the most common materials.
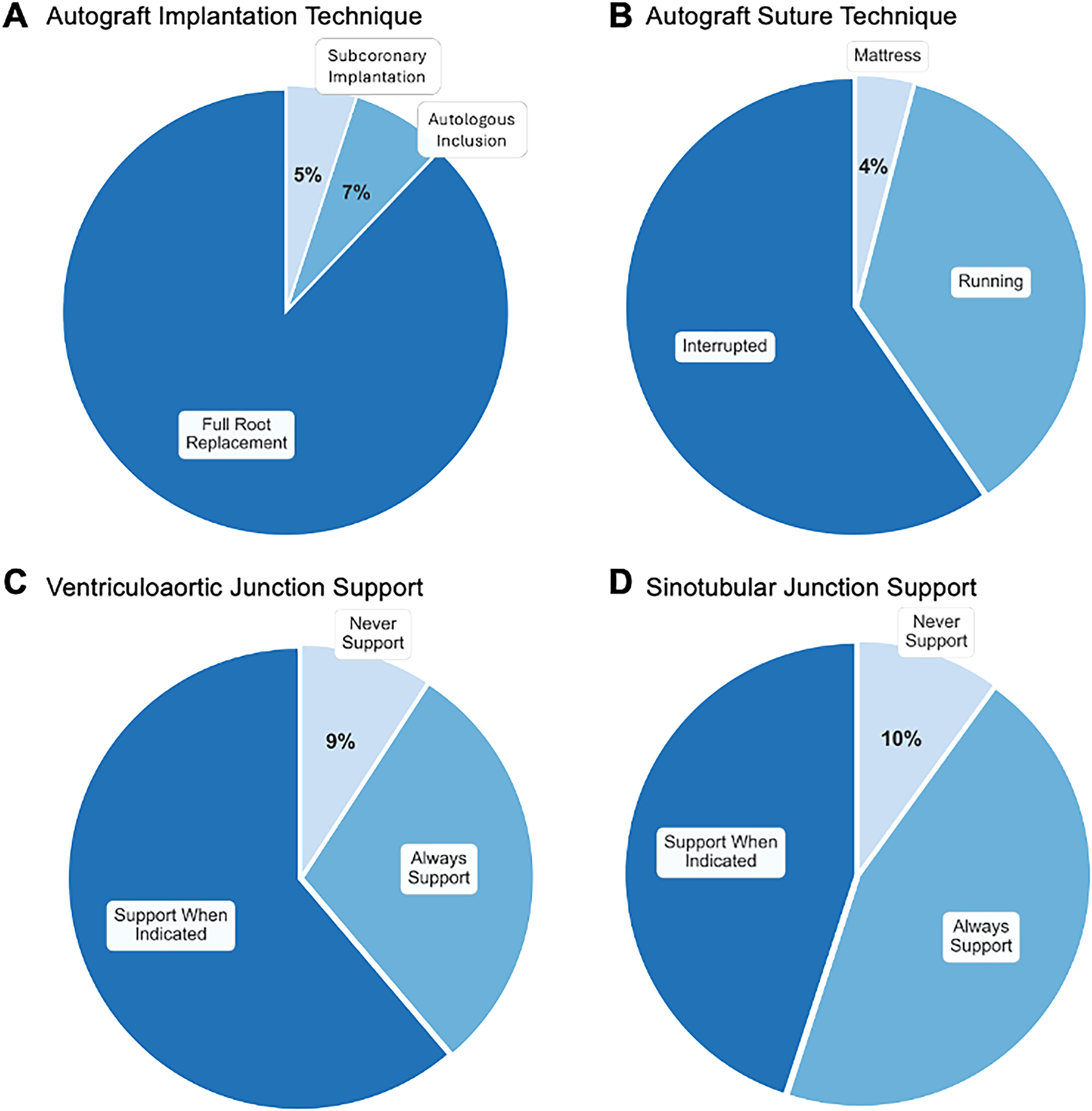
Operative Techniques. (A) The full root replacement technique predominates over inclusion and subcoronary approaches. (B) Interrupted suture technique is slightly favored over running sutures, with minimal mattress suture usage. (C) Most surgeons support the ventriculoaortic junction selectively when indicated rather than routinely. (D) For the sinotubular junction, surgeons either provide selective or routine support, with few opting for no support.
Extra-Arterial Support: Ventriculoaortic Junction (VAJ) and Sinotubular Junction (STJ)
Significant variation exists in approaches to autograft support. Most respondents (61%) support the VAJ when indicated, while 29% always support it and 10% never support it (Figure 6C). Among those who support the VAJ when indicated, all would do so for a dilated aortic annulus, 41% for primary aortic insufficiency, 21% for dilated ascending aorta (median size threshold 35 mm, IQR 25-40), and 11% for bicuspid aortic valve. The most common aortic annulus threshold for reinforcement is > 27 mm (54%), followed by > 25 mm (32%). Among those who always support the VAJ, 43% use a full Dacron inclusion, 17% use a Dacron ring, 10% use an autologous pericardial strip, 10% use a bovine pericardial strip, and 20% use other methods (felt strip, PEARS, or others).
For the STJ, equal proportions (45% each) of respondents always support it or support it when indicated, while 10% never provide support (Figure 6D). Among those who always support the STJ, 37% use a full Dacron inclusion, 33% use a Dacron interposition graft, 13% use an external Dacron ring, and 17% use other methods (felt strip, pericardial strip, PEARS, or others). The most common indication for STJ reinforcement is dilated ascending aorta (89%, median size threshold 35 mm, IQR 30-39), followed by mismatch between ascending aorta and pulmonary STJ (72%), primary aortic insufficiency (20%), dilated aortic annulus (17%), and bicuspid aortic valve (15%).
Right Ventricular Outflow Tract Reconstruction
A decellularized cryopreserved pulmonary homograft (Artivion Synergraft) is the preferred conduit for RVOT reconstruction for 43.6% of respondents, followed by fresh cryopreserved homograft (19%), local tissue bank homograft (16%), Lifenet pulmonary homograft (10%), and LeMaitre RestoreFlow homograft (8%). The median ideal minimum homograft size is 26 mm (IQR 25-28) for males and 25 mm (IQR 23-26) for females. Only 18% of respondents routinely gender-type or blood-type match homografts, but 55% of those who do not currently match would be interested in doing so if feasible at their institution.
Most respondents (83%) trim the homograft muscle prior to implantation, and 19% perform beating heart implantation. If a pulmonary homograft were unavailable, 55% would still perform the RP, using alternatives such as freestyle graft (45%), bovine pericardial valve (32%), porcine valve (21%), or aortic homograft (18%).
Postoperative Management
Three major considerations following the RP are critical for improving durability and quality monitoring, including blood pressure management, postoperative inflammation prophylaxis, and postoperative imaging (Figure 7).
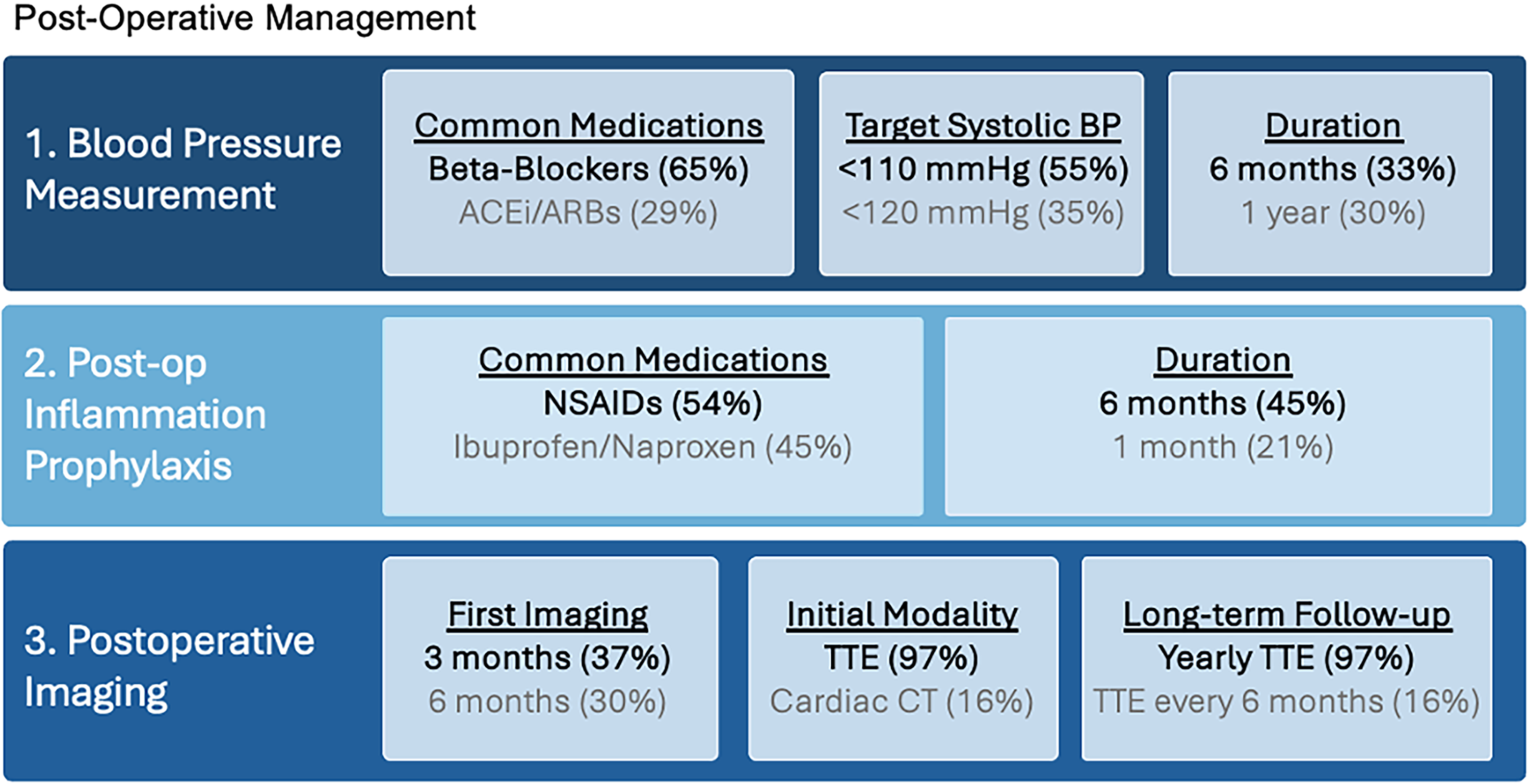
Postoperative Management Following Ross Procedure (RP). This figure presents the 2 most common approaches for 3 major postoperative consideration based on the survey: (1) blood pressure management, showing preferred medications, target systolic values, and restriction durations; (2) postoperative inflammation prophylaxis, indicating most common anti-inflammatory agents and treatment durations; and (3) postoperative imaging, displaying typical timing of first imaging, preferred modalities, and long-term follow-up strategies.
Blood Pressure Management
Most respondents (89%) monitor strict blood pressure control after the RP. The majority target a systolic blood pressure < 110 mm Hg (55%), while 35% target < 120 mm Hg. Most maintain these restrictions for either 6 months (33%) or 1 year (30%), with others restricting for 3 months (19%), 1 month (5%), or 1 week (6%). Beta-blockers are the most common first-line antihypertensive (65%), followed by angiotensin-converting enzyme inhibitors (ACEi)/angiotensin receptor blockers (ARBs) (28%) and calcium channel blockers (6%).
Postoperative Inflammation Prophylaxis
Around half the respondents (54%) use postoperative inflammatory prophylaxis to mitigate inflammation of the homograft, with around the other half using nonsteroidal anti-inflammatory drugs (NSAIDs) (ibuprofen or naproxen) and the rest with anti-inflammatories or corticosteroids. Most maintain anti-inflammatory therapy for 6 months (45%), followed by 1 month (21%), 3 months (17%), indefinitely (8%), or 1 week (6%). The majority (69%) also prescribe a proton pump inhibitor or H2 blocker for gastrointestinal prophylaxis.
Postoperative Imaging
Most respondents (90%) reimagine patients prior to discharge. The first postdischarge imaging is typically performed at 3 months (37%) or 6 months (30%). For initial postdischarge imaging, 97% obtain a TTE, 16% obtain a CT, and 5% obtain a TEE. For long-term follow-up, 78% obtain yearly echocardiograms, 14% obtain echocardiograms every 6 months, 6% every 2 years, and 1% at other intervals. Practices for follow-up cross-sectional imaging vary considerably: 33% obtain it every 2 years, 29% never obtain it after discharge, 26% obtain it yearly, 10% at other intervals, and 3% twice per year.
Discussion
Donald Ross first described the RP in his seminal manuscript published in the Lancet in 1967. 10 The RP is a complex operation for adult or pediatric patients in need of AVR that involves transplanting the native pulmonary valve into the aortic position and reconstruction of the right ventricular outflow tract. 5 Interest and utilization in the RP have fluctuated greatly since its inception, mostly driven by mixed results and technical complexity. More recently, numerous international studies have demonstrated superior outcomes when compared to prosthetic AVR and a repeatedly normal life expectancy after the RP.8,11–20 This has ignited a Ross revival, with the RP now accounting for nearly 10% of AVR in patients under the age of 60 according to data from the STS database. The RP is a technically demanding operation which is highly nuanced, and for it to be successful, it must be done with great care and in centers of excellence with adequate volume. 21 Due to its complexity, understanding global practice patterns is particularly valuable for surgeons who are learning the procedure or performing it infrequently. For new or evolving programs, access to the practices of established centers serves as an essential resource.
Ross Training and Learning Curve
Our survey highlights significant heterogeneity in training pathways for RP surgeons. Notably, 60% of respondents reported learning the RP as attending surgeons, underscoring a lack of formalized training opportunities, though this might also reflect the fact that renewed interest in the RP has gained momentum in the last 5 to 10 years only. Nevertheless, this gap reinforces the need for structured educational programs to broaden access and ensure competency. Our data also reveal a volume–outcome relationship worth emphasizing: while approximately one-third of respondents perform 10 to 20 RPs annually, 20% perform fewer than 5, raising critical concerns about the minimum procedural volume required to maintain proficiency. Perhaps most striking is that 46% of respondents are assisted by a second attending surgeon during the RP—underscoring the technical complexity of the procedure and the essential role of having an established surgeon assisting, especially during the early learning curve. The assisting surgeon may not necessarily serve as a formal mentor, but their presence and expertise provide valuable support during this challenging procedure.
The RP is a highly nuanced operation which has a high degree of difficulty and requires a unique skillset.22–24 The ideal RP surgeon is one with advanced expertise in aortic root surgery and comfort in the right ventricular outflow tract (RVOT). This dual skillset is rarely consolidated in either adult or congenital training alone—adult cardiac surgeons tend to have more experience with aortic root surgery, while congenital surgeons are more comfortable with the RVOT. 25 Our survey demonstrates that most surgeons performing the RP are not congenitally trained surgeons, suggesting that the necessary comfort on the right side of the heart can be learned.
The procedure requires mastery of numerous technical aspects, including autograft harvest, preparation, anastomotic technique, coronary reimplantation, VAJ and STJ support, and homograft selection, preparation, and implantation. The variations in techniques observed in our survey highlight the need for standardized approaches based on the best available evidence.
Patient Selection for the RP
Patient selection is important for optimal outcomes when performing the RP. Classically, the ideal patient is a young or middle-aged person who is otherwise healthy, with aortic stenosis and a small or normal-sized aortic annulus. 5 However, our survey reveals a far more nuanced approach to patient selection that extends beyond these classic parameters.
For patients 50 to 60 years of age, the RP should be reserved for those most likely to derive a benefit from the operation. Specifically, these are patients with a projected life expectancy of ≥ 15 years, an active lifestyle, favorable anatomy, no major concomitant cardiac disease, and few comorbidities.5,26 The RP may also be ideal for older patients who have normal life expectancy, given the fact that the need for reintervention may be less likely.12,27–29 A tailored RP can be perfect for the patient with nonrepairable aortic insufficiency, and there are data to confirm excellent long-term durability for this indication.30–32
Our study uncovered a critical insight: the disconnect between reported contraindications and actual surgical decision-making. While 61% of respondents cited connective tissue disorders as an absolute contraindication, the patient scenario ratings revealed a far more personalized approach—no surgeons indicated that they would deny a 45-year-old Marfan syndrome patient from the RP. This finding underscores the importance in patient selection for the RP: individual patient characteristics, surgeon experience, and comprehensive clinical assessment invariably succeed checklists, as none of the surgeons would consider any of the patient scenarios (aside from the patient with IVDU, AS, and prior 3 vessel CABG) (Figure 5), as an absolute contraindication, in contrast to the pathologies that many would consider as absolute contraindications (Figure 4).
The ultimate decision for when to perform the RP depends on a delicate interplay between patient-specific factors and surgeon expertise. Our data demonstrates remarkable variability in approach, with no clear consensus on absolute contraindications. Our findings emphasize the need for a flexible, nuanced approach to patient selection that goes beyond but builds upon traditional contraindication frameworks.
Care for Patients After the RP
There are several postoperative considerations for patients undergoing the RP. The first and probably most important is strict blood pressure monitoring. 33 While the pulmonary root is adapting to the new systemic environment, aggressive blood pressure management may result in increased durability. 34 A total of 89% of the respondents state they regulate blood pressure after an RP, with the majority (55%) keeping it under 110 mm Hg systolic. The majority keep this goal for 6 months and use beta-blockers as their first line, followed by ACEi/ARBs. This area of relative consensus suggests that blood pressure management should be considered a standard component of post-Ross care.
Half the respondents (54%) prescribe NSAIDs or steroids to mitigate inflammation of the homograft, with maintenance typically for 6 months (45%). The widespread practice of reimaging before discharge (90%) and annual follow-up imaging (78%) suggests consensus on surveillance. Furthermore, our analysis revealed that these standard practices vary between low-volume and high-volume surgeons.
High Versus Low Volume Surgeons: Practice Variations
With the resurgence of the RP and the technical complexity of the procedure, it should still be carefully performed by expert centers, with clear guidelines for those seeking to establish an RP program. 58% of low-volume surgeons (who perform 10 or fewer RP procedures annually) operate with a second surgeon, compared to 30% of surgeons who perform more than 10 RP procedures annually.
Regarding technical and postoperative management, low-volume surgeons showed greater variability. For example, while 75% to 80% of low-volume surgeons would either always support or support when indicated the VAJ and STJ, all high-volume surgeons who perform > 30 RP procedures annually support the VAJ and STJ when indicated. There was also uniformity among high-volume surgeons in postoperative management. They all consistently had strict systolic blood pressure goals (<110 or 120 mm Hg), and all would reimage after 6 or 12 months.
The variations between high and low volume surgeons further elucidate the need for standardized guidelines. The divergence between low and high-volume surgeons emphasizes the critical need for structured training programs, mentorship, and guidelines that can bridge the experience gap.
Limitations
Our study has several limitations that warrant consideration. As with all survey-based research, response bias may influence findings, potentially overrepresenting high-volume or academic surgeons. Our survey shows that nearly half of the respondents perform > 10 RP per year, which is a significant contrast from national data from the Society of Thoracic Surgeons, which has shown that the median RP performed per center is around 2 in the United States. 35 This discrepancy highlights potential sampling bias in the survey.
Furthermore, the sample size, while robust for a specialized procedure, may not capture the full spectrum of practice variations globally. Additionally, the cross-sectional nature of our survey does not allow for correlation of practice patterns with long-term outcomes.
Conclusion
This comprehensive international survey outlines current RP practices and highlights the need for global alignment around training, patient selection, and technical execution. We hope these findings will guide evolving programs, enhance reproducibility, and further support the growing momentum behind the RP.
Supplemental Material
sj-docx-1-hvs-10.1177_30494826251359904 - Supplemental material for Global Perspectives on the Ross Procedure: A Comprehensive International Survey
Supplemental material, sj-docx-1-hvs-10.1177_30494826251359904 for Global Perspectives on the Ross Procedure: A Comprehensive International Survey by Lucy Nam, Dane C Paneitz, Michael E. Ibrahim, Christopher R Burke, Maral Ouzounian, Michael WA Chu, Darryl F Shore, Ismail El-Hamamsy and Jordan P Bloom in Journal of the Heart Valve Society
Footnotes
Abbreviations
Ethical Considerations
Exempt from review by the Mass General Brigham IRB (protocol #2023P000424, approved 3/24/2023).
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
