Abstract
Background:
Women over the age of 50 often experience weight gain due to menopausal-related hormonal changes, increasing their risk for health complications. The Epitomee capsule, a nondrug medical device designed to enhance satiety, may offer significant benefits for weight loss in this demographic.
Methods:
This sub-analysis of the Randomized Evaluation of Efficacy and Safety of the Epitomee Capsule Trial (RESET) double-blind placebo-controlled study included 104 women aged ≥50, randomized to Epitomee capsule (n = 52) or placebo (n = 52) with lifestyle counseling. Outcomes included % weight loss (baseline to week 24), responder rates (≥5%, ≥10%, ≥15%), body mass index (BMI) change, early responder, waist circumference, and glycemic status.
Results:
Age and gender were not significant covariates influencing Epitomee capsule efficacy in the multivariate model of the RESET study. Among women aged ≥50 years, average weight reduction was 7.6% in the Epitomee group versus 4.4% in the placebo group (P = 0.0287). Significantly more women treated with Epitomee achieved ≥5% weight loss (63% vs 42%, P = 0.0463), and ≥10% weight loss (29% vs 9%, P = 0.0169). Among women aged older than 50 years, early response (ER) was defined as a weight loss of ≥2% at week 8. ER, average weight reduction at week 24 was 9.9% of baseline body weight in the Epitomee group versus 6.2% in the placebo group (P = 0.0074). The Epitomee group also showed greater reductions in BMI and waist circumference, along with more frequent shifts toward normoglycemia. Among women >50 years, a significantly greater proportion improved their BMI category compared with placebo (56% vs 21%, P = 0.0007). Adverse event rates were similar between groups, and no serious events occurred.
Conclusions:
The Epitomee capsule demonstrates significant efficacy in promoting weight loss, improving BMI, and a showing trend toward improved metabolic parameters in women over the age of 50, with a favorable safety profile, suggesting its potential as a valuable nonpharmaceutical intervention for the menopausal population.
Introduction
Approximately 50%–70% of women experience measurable weight gain during the menopausal transition.1–3 The SWAN study indicates an average annual weight gain of 1.5 kg during perimenopause, accumulating an increase of around 10 kg by the time menopause period reached. 3 Others report on weight gained of ≈2.0–2.3 kg over 3-year period around the menopause. 4 These changes are not solely attributable to aging but may be closely linked to the hormonal shifts of menopause, particularly the decline in estradiol, which contributes to increased fat mass, especially visceral adiposity. This increase in visceral adiposity is associated with cardiometabolic risk factors such as dyslipidemia, hypertension, vascular dysfunction, insulin resistance, type 2 diabetes, and coronary vascular disease.3–8
Beyond changes in body composition, menopause is increasingly recognized as a period of vulnerability for alterations in appetite regulation and satiety signaling. 9 Estradiol plays a central role in modulating energy homeostasis through its actions on hypothalamic pathways, influencing both anorexigenic and orexigenic signals.10,11 Observational and mechanistic clinical studies have shown that the menopausal transition is associated with increased hunger, greater desire to eat, and reduced fasting fullness.12,13 However, evidence that postmenopausal women experience increased hunger and reduced satiety has not yet been confirmed by large clinical trials. These changes may be driven by estrogen’s declining influence on key appetite-regulating hormones such as leptin, ghrelin, and GLP-1, as well as its effects on hypothalamic neurons involved in satiety.10,11,14 Recent systematic reviews have highlighted that disordered eating behaviors, including binge eating and emotional eating, may increase during perimenopause and postmenopause, potentially due to hormonal fluctuations, body dissatisfaction, and psychological stressors.15,16 Moreover, sleep disturbances common in midlife women, such as those caused by hot flashes, night sweats, frequent night time awakening, restorative sleep pattern, and nocturia, can further disrupt appetite regulation by altering leptin and ghrelin signaling.17,18 Midlife thus represents a critical window for intervention, as women face a convergence of physiological, psychological, and behavioral factors that increase the risk of weight gain and metabolic disease. 5 This demographic population is actively seeking effective, safe, and sustainable weight loss strategies. 19 While hormone therapy (HT) can partially mitigate changes in body composition and improve glucose metabolism,20–22 it is not suitable, accepted or preferred by all women.
Given that perimenopausal and postmenopausal women may have altered sensitivity to satiety cues, interventions that enhance satiety through nonhormonal mechanisms may be particularly well-suited for this population. The Epitomee capsule, a novel, orally twice daily administered, shape-shifting hydrogel device, is specifically designed to promote early satiety,23,24 through a purely mechanical mechanism of action. By expanding in the stomach and mimicking the presence of food, it resists the peristaltic waves of the stomach and activate sensory mechanoreceptors and the gut–brain axis signaling pathway to promote early signaling of satiety, potentially counteracting the blunted satiety cues observed in postmenopausal women.
The RESET study was published earlier.25–28 In this sub-analysis of the RESET study, we investigated the efficacy and safety of the Epitomee capsule in women aged ≥50 years, a population largely postmenopausal and not receiving HT. We hypothesized that the Epitomee capsule would be particularly effective in this group due to its satiety-enhancing mechanism, which may help overcome menopause-related changes in appetite regulation.
The Epitomee capsule is a Food and Drug Administration-approved, CE-cleared, novel, orally self-administered device, designed to induce weight reduction in participants with overweight and obesity (Fig. 1). The capsule is designed to expand in the stomach, mimicking the presence of food, which promotes the sensation of fullness with resultant clinical benefits of significantly reducing body weight, resulting in 5%–10% weight loss in a large percentage of patients, reduction in body mass index (BMI), improved metabolic parameters, and improved quality of life (QOL) coupled with a favorable tolerability and safety profile.23–29

Epitomee oral hydrogel capsule and after shape-shifting in the stomach.
Thus, we investigated the efficacy and safety of the Epitomee capsule compared with placebo, in conjunction with lifestyle intervention, in a subgroup of female participants from the RESET study aged over 50 years who were not receiving HT.
Methods
Study design
The Epitomee capsule investigated in this study, along with its underlying mechanism of action, has been previously described in detail in published work.23–29 These prior publications outline the clinical and mechanistic basis, predefined outcomes, and additional analyses that support the capsule’s evaluation in the current post-hoc study. The Randomized Evaluation of Efficacy and Safety of the Epitomee Capsule Trial (RESET) was a prospective, randomized, double-blind, placebo controlled, adaptive trial conducted in the United States. 25 The primary objective of the study was to compare the percentage change in baseline body weight at 24 weeks achieved with the Epitomee capsule versus a visually matching placebo capsule. Eligible participants were randomized in a 1:1 ratio using an interactive web response system to receive either the Epitomee capsule or placebo, both in combination with the same lifestyle intervention, for 24 weeks. The clinical trial is registered on ClinicalTrials.gov identifier (NCT04222322).
This post-hoc analysis evaluated the safety and efficacy of the Epitomee capsule, compared with placebo, in promoting weight loss and improving related metabolic indicators—including BMI classification, waist circumference, and glycemic status—among women aged ≥50 years of age.
Participants
The RESET study total population included adult participants aged ≥ 18 years with a BMI ranging from 27 to 40 kg/m2 who had normoglycemia or prediabetes. This post-hoc analysis included female participants ≥50 years who were enrolled in the RESET study. A total of 104 participants were included in this sub-analysis, with 52 participants in the Epitomee group and 52 in the placebo group (Supplementary Fig. S1).
Inclusion and exclusion criteria
Participants in the RESET study total population met detailed eligibility criteria, 25 including BMI 27–40 kg/m2, general good health, and willingness to consent and comply with study procedures. For this sub-analysis, an additional inclusion criterion was female participants aged ≥50 years. Key exclusion criteria were uncontrolled hypertension, severe cardiovascular disease, active gastrointestinal (GI) disorders, prior bariatric surgery, use of medications affecting weight or appetite, recent participation in other trials or weight loss programs, pregnancy, and known allergy to Epitomee components.
Intervention
The Epitomee capsule is a novel, minimally invasive, drug-free, oral medical device indicated for weight management in individuals with overweight or obesity (BMI = 25–40) (Fig. 1).23–29 It is composed of the following polymers: crosslinked polyacrylic acid sodium salt, hydroxy propyl methyl cellulose, cellulose acetate phthalate, hydroxypropyl cellulose, chitosan (vegan), diethyl phthalate, polyvinyl alcohol polyethylene copolymer (Kollicoat), triethyl citrate, cellulose acetate, acetyl tributyl citrate that self-expand in the stomach to create a semi-rigid, gel-based, space-occupying structure. In the stomach, it expands into a pH-sensitive, shape-shifting gel structure containing superabsorbent polymer microparticles that absorb up to 100 times their dry weight in water, decreasing in volume as ion concentration increases. The microparticles are contained within a pH-sensitive polymer envelope that remains stable at pH levels of up to 6.5. Upon ingestion and water absorption, the gel particles expand, forming an elastic triangular structure that resists stomach peristaltic waves, promoting early satiety signaling, constituting an entirely mechanical mechanism of action. Constituted of 97% water and 3% polymers, the Epitomee capsule maintains mechanical rigidity for several hours until envelope disintegration, with no caloric content. Within 30 min after reaching the intestine, the capsule disintegrates into small particles that are excreted through the GI tract.
Participants were randomized to receive the Epitomee capsule (Epitomee group) or a capsule identical in appearance but without the shape-shifting functionality (Placebo group). Both groups were instructed to take the capsule twice daily with two cups of water (16 oz or 480 mL) before lunch and dinner for 24 weeks.
All study participants received the same lifestyle intervention program, previously reported, 25 which included instructions for diet and physical activity modification, delivered over 24 weeks in 14 lifestyle counseling sessions of approximately 15 min each. Lifestyle counseling sessions were weekly in the first month and every other week thereafter and were provided by a registered dietitian, nurse practitioner, clinical or health psychologist, or research nurse. Participants were instructed to consume a diet of 1200–1800 calories per day based on their baseline body weight and to gradually increase their physical activity to 180 min or more per week by week 24.
Outcomes and data collection
The post-hoc primary end-point included the percentage change in body weight from baseline to week 24. Secondary end-point were responder rates for weight loss thresholds (≥5%, ≥10%, and ≥15%), changes in BMI, waist circumference, and glycemic status (normoglycemia, prediabetes, and diabetes). The safety end-points were the incidence of device-related serious adverse events (SAEs) from randomization through week 24 and incidents of all adverse events (AEs) coded according to the Medical Dictionary for Regulatory Activities (MedDRA).
Baseline characteristics such as gender, age, race, ethnicity, BMI, waist circumference, and menopausal status (if applicable) were collected at the screening visit. Weight was measured in duplicate at all study visits by trained assessors, while BMI was computed using the baseline height and the averaged weight measurements across study visits. Waist circumference was measured in duplicate at baseline, week 12, and week 24. Laboratory outcomes, including fasting plasma glucose and hemoglobin A1c levels, were collected at the screening visit, baseline, week 12, and week 24.
Statistical analysis
Data were analyzed using intention-to-treat principles. Continuous variables and categorical variables were analyzed using t-tests and chi-square tests, respectively. Age and gender were not significant covariates influencing Epitomee capsule efficacy based on the multivariate model of the RESET study. The subgroup threshold of 50 years was chosen based on both statistical and physiological considerations. This cutoff approximated the median age of the female RESET participants, 48 years in the Epitomee group and 49 years in the placebo group (Supplementary Fig. S2), thereby ensuring a balanced dichotomization of the cohort, with comparably sized groups.
Mixed-model repeated measures were used to estimate weight loss, adjusting for baseline weight. Responder analyses were conducted using logistic regression. Shifts in glycemic status were analyzed by comparing the proportion of participants who transitioned between normoglycemia and prediabetes categories from baseline to the end of treatment. Shifts in BMI categories were evaluated by comparing the proportion of participants in each treatment group who moved between BMI classes from baseline to end of treatment. All enrolled participants, including those with Class III obesity at baseline, were included in all BMI-related analyses. Safety end-points were analyzed in the safety dataset using the prespecified MedDRA system set and included the number of incidents of AEs and the number and percentage of participants who experienced AEs. Statistical significance was set at P < 0.05.
Ethical considerations
All participants provided written informed consent prior to performing any study related activities or evaluations. The study was conducted in accordance with International Council for Harmonisation E6, Guidelines for Good Clinical Practice, ISO 14155:2011, the U.S. Codes of Federal Regulations (21CFR parts 11, 50, 54, 56, 812, and 814), and the Declaration of Helsinki. Study protocols were approved by independent ethics committees or institutional review boards at each study site, and the study protocol is registered on clinicaltrials.gov (NCT04222322).
Results
Participant characteristics
Of the 279 individuals who enrolled in the RESET study, 223 (80%) were women. The age of female participants varied between 19 and 75 years old with an average ± standard deviation (SD) of 48 ± 12 years. The age distribution of the Epitomee and placebo treatment arms in the RESET study was comparable as shown in Supplementary Figure S2A and S2B.
A total of 104 women aged ≥50 years were included in this sub-analysis, with 52 participants in the Epitomee group and 52 in the placebo group. The baseline characteristics of the participants were comparable between the two groups (Table 1). The average age at screening was 59.2 years (SD = 6.4) for the Epitomee group and 57.6 years (SD = 6.1) for the placebo group. Of the participants aged ≥50 years, 90% were postmenopausal, based on self-reported data. Other baseline characteristics, including BMI, waist circumference, and glycemic status, showed no significant differences between the groups. This population of women was not treated with HT, except for three participants (one from the Epitomee group and two from the control group).
Demographic and Baseline Characteristics of Women Above 50 Years Old in the Randomized Evaluation of Efficacy and Safety of the Epitomee Capsule Trial Sub-Analysis
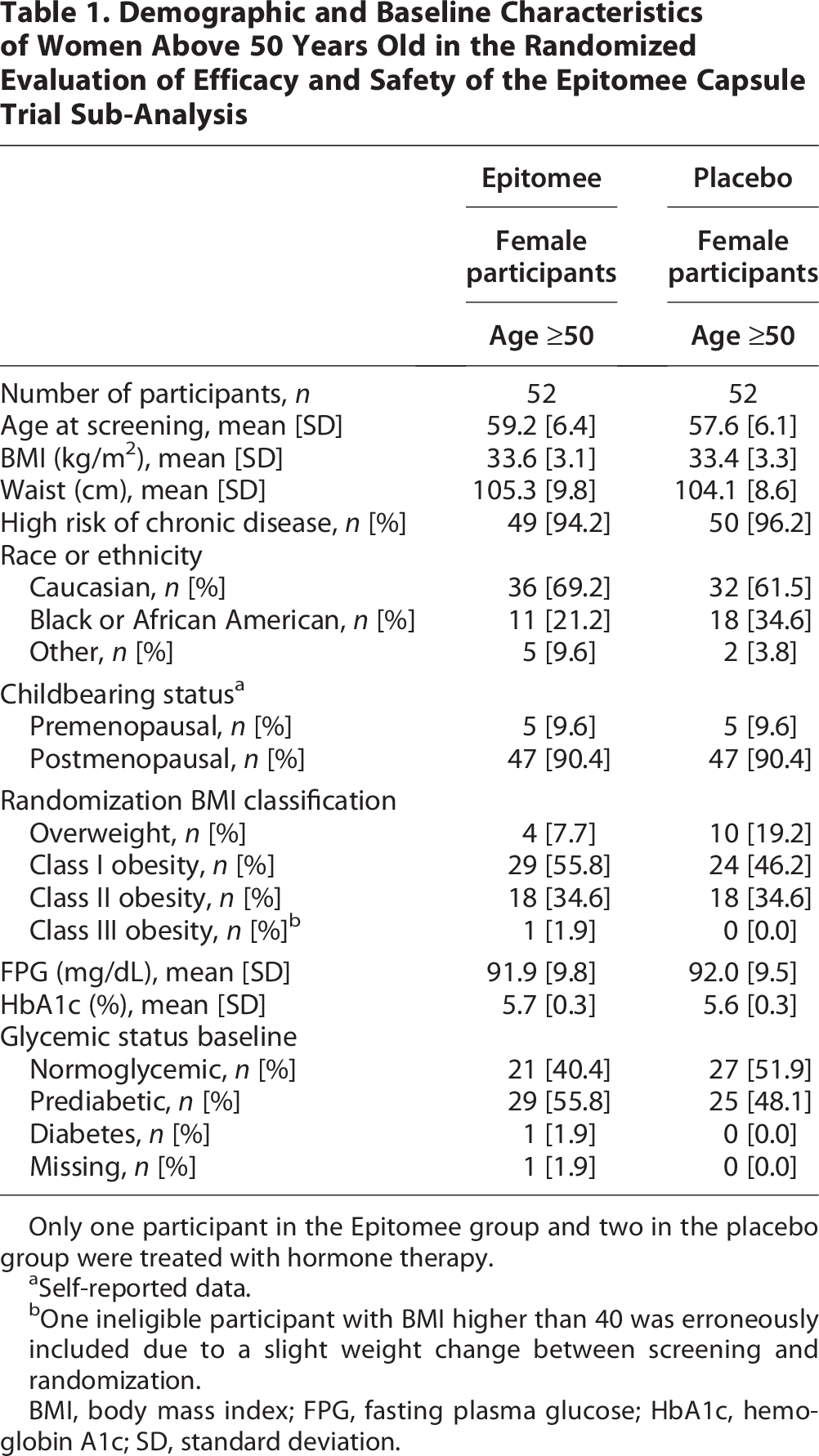
Only one participant in the Epitomee group and two in the placebo group were treated with hormone therapy.
Self-reported data.
One ineligible participant with BMI higher than 40 was erroneously included due to a slight weight change between screening and randomization.
BMI, body mass index; FPG, fasting plasma glucose; HbA1c, hemoglobin A1c; SD, standard deviation.
The dropout rate was numerically higher in the placebo group (17.3% [n = 9/52]) compared with the Epitomee group (7.7% [n = 4/52]), although this difference did not reach statistical significance. Only three participants were dropout due to AE (one from Epitomee and two from placebo). Reasons for dropout can be seen in Supplementary Table S1.
Weight loss outcomes
The main outcome of percentage change in body weight least squares mean from baseline to week 24 showed a significant difference with −7.6% in the Epitomee group compared with −4.4% in the placebo group (P = 0.0287) (Fig. 2).

Mean percent change in body weight from baseline to week 24 in female participants ≥50 years (n = 104).
Additionally, as shown in Figure 3, a significantly higher proportion of participants in the Epitomee group achieved clinically meaningful weight loss at clinically relevant weight loss thresholds. Significantly, more Epitomee-treated participants lost ≥5% of their baseline weight, compared with the placebo group (63% vs 42%, P = 0.0463). Furthermore, significantly more participants in the Epitomee group achieved ≥10% weight loss versus the placebo group (29% vs 9%, P = 0.0169). Although the percentage of participants achieving ≥15% weight loss was numerically higher in the Epitomee group (13% vs the placebo group 5%), this difference did not reach statistical significance.

Proportion of female participants ≥50 years with a reduction in body weight from baseline to week 24 of ≥5%, ≥10%, and ≥15%.
BMI improvements
The proportion of participants who demonstrated improved BMI categories was significantly higher in the Epitomee group compared with the placebo group (56% vs 21%, P = 0.0007). The descriptive distribution of BMI categories at baseline and week 24 in both groups is shown in Figure 4.
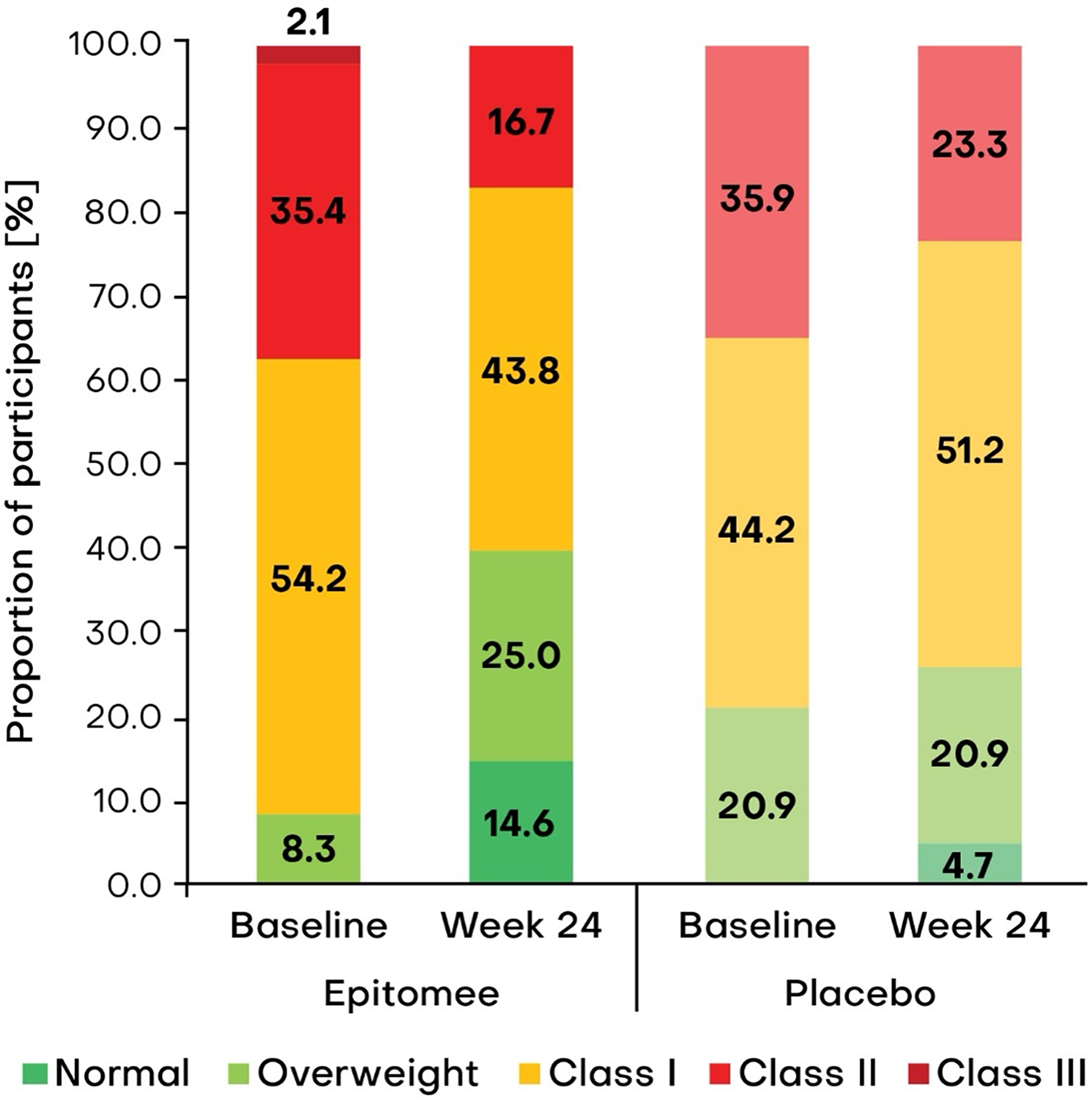
Change in body mass index (BMI) category among female participants aged ≥50 years. Distribution of BMI categories (normal, overweight, obesity class I, class II, and class III) at baseline and week 24 in female participants aged ≥50 years in Epitomee (left) and placebo (right) treatment arms. BMI category definitions: normal weight: BMI = 18.5–24.9 kg/m2; overweight: BMI = 25.0–29.9 kg/m2; obesity class I: BMI = 30.0–34.9 kg/m2; obesity class II: BMI = 35.0–39.9 kg/m2; obesity class III: BMI ≥40 kg/m2.
Among participants treated with Epitomee, the proportion classified as having normal weight increased to 14.6% at week 24 (from 0% at baseline), while the prevalence of Class III obesity was declined from 2.1% at baseline to 0%, and Class II obesity was reduced substantially (35.4% to 16.7%) observed following treatment with the Epitomee capsule. The proportion of participants in Class I obesity also declined (54.2% to 43.8%), accompanied by an increase in the overweight category (8.3%–25.0%). In contrast, the placebo group demonstrated only modest overall changes in BMI category distribution. Although the proportion of participants with Class II obesity decreased (35.9% to 23.3%), the proportion with Class I obesity increased (44.2%–51.2%), with no change in the overweight and fewer participants achieved normal weight (4.7% at week 24). Overall, Epitomee capsule treatment appeared to shift participants from higher obesity classes toward overweight and normal weight, whereas the placebo group showed limited overall change.
Waist circumference and glycemic status
At week 24, participants in the Epitomee group demonstrated a numerical greater waist circumference reduction compared with placebo (−6.2 cm ± 2.8 vs −4.8 cm ± 2.5). This numerical advantage was also evident at week 12 (−4.4 cm vs −2.3 cm) (Fig. 5A). In terms of glycemic status, 35% of participants treated with Epitomee shifted from prediabetes to normoglycemia versus 29% in the placebo group (Fig. 5B). Conversely, a lower proportion of Epitomee-treated participants transitioned from normoglycemia to prediabetes compared with placebo (22% vs 29%, P = NS), suggesting a trend toward improved metabolic parameters. This difference did not reach statistical significance.

Changes in waist circumference and glycemic status.
24-week weight loss based on early responder classification
Among women aged >50 years, early response (ER; ≥2% weight loss at week 8) was associated with greater weight reduction at week 24 in the Epitomee group compared with placebo (9.9% ± 5.7% vs 6.2% ± 4.4%, P = 0.0074). This finding is in line with observations reported for the entire RESET population in as reported by Jakicic et al. 27
Adverse events
AEs in the RESET study total population have been described in a recent publication and was found to be similar between Epitomee and placebo with no differences in the randomized and subpopulation Epitomee groups. 25 We conducted additional AE analysis among participants in this subpopulation.
There were no significant differences in AEs between the groups in the subpopulation, female participants >50 years, irrespective of treatment (Table 2 and Fig. 6). There was a similar overall AE incidence (86.5% in the Epitomee group and 90.4% in the placebo group, P = NS) with a numerically lower number of GI-related AEs in the Epitomee group versus the placebo group (38.5% vs 50.0%, P = NS) respectively. No device-related SAEs occurred (Table 2).

Adverse events in women 50 years old for Epitomee-treated subjects (n = 52) and Placebo-treated subjects (n = 52).
Total, Severe, Serious, and Gastrointestinal-Related Adverse Events in Women Above 50 Years Old

GI, gastrointestinal.
The AEs experienced by participants were also similar between the treatment groups (Fig. 6A). The most common AEs in the Epitomee group were constipation (14%), nasopharyngitis (14%), COVID-19 (12%), arthralgia (8%), abdominal pain (8%), upper respiratory tract infection (8%), headache (6%), and nausea (6%). In the placebo group, constipation (23%), arthralgia (15%), headache (10%), nausea (10%), diarrhea (10%), COVID-19 (8%), flatulent (8%), gastro reflux disease (6%), and vomiting (6%) were most common (Fig. 6A).
Figure 6B compares the proportion of participants experiencing total, GI, and GI-related AEs between Epitomee-treated and placebo-treated groups. Overall, the incidence and severity patterns are very similar between the two groups, with mild events being the most common and no SAEs reported. Across nearly all categories—especially total, GI, and GI-related AEs—the placebo group shows slightly higher proportions than the Epitomee group. The most common system organ classes (Fig. 6C) affected by AEs were GI disorders (Epitomee, 39% of participants; placebo, 50% of participants), followed by infections or infestations (Epitomee, 39% of participants; placebo, 39% of participants).
Discussion
In the RESET study, age and gender were not significant factors in the multivariate model, and the effect of Epitomee on weight loss was consistent across both sexes and all age groups. In this article, we specifically report on the subgroup of women ≥50 years, although their outcomes did not differ meaningfully from those of younger women or men. This focus is particularly relevant given the menopausal transition, a period marked by physiological changes in regulatory pathways. The results of this sub-analysis of the RESET study demonstrate the significant efficacy of the Epitomee capsule in promoting weight loss and suggest favorable trends in metabolic parameters in female participants over 50 years of age.
The Epitomee group achieved significant greater weight loss than placebo (7.6% vs 4.4%). Among women ≥50 years, those classified as early responders (≥2% weight loss at week 8) had significant greater weight reduction at week 24 with Epitomee compared with placebo (9.9% vs 6.2%). In addition, the responder analysis highlights the clinical relevance of the weight loss achieved with the Epitomee capsule. A significant higher proportion of participants in the entire population of the Epitomee group at the RESET achieved clinically significant weight loss thresholds (≥5% and ≥10%) compared with the placebo group.25,27 These findings underscore that treatment with Epitomee results in greater average weight loss compared with placebo and additionally increases the likelihood of patients reaching clinically meaningful weight-loss thresholds.
This finding is particularly noteworthy given the challenges associated with weight loss in postmenopausal women, who often experience increase in fat mass and weight gain due to hormonal changes and decreased metabolic rate.1–3 The significant improvements in BMI and trends of waist circumference reduction further support the efficacy of the Epitomee capsule in reducing central adiposity, which is a key risk factor for metabolic disease. 4
The numerical improvements in glycemic status observed in the Epitomee group could have clinical implications. More participants in the Epitomee group shifted from prediabetes to normoglycemia; and there was also less shift from normoglycemia to prediabetes, when compared with the placebo group. These findings may suggest that the Epitomee capsule not only promotes weight loss but also has, to some extent, a potential positive impact on metabolic health parameters such as waist circumference and change in glycemic status.
This is important because, as reported previously, moderate weight loss can lead to significant health benefits, including improved cardiovascular health, reduced risk of diabetes, and associated with overall improvement in QOL.30–32
The effect of Epitomee on weight loss was observed across all age groups and may be of particular importance for women undergoing the menopausal transition—a period characterized by multiple physiological changes in regulatory pathways. Of relevance, alterations in satiety, appetite, and eating behavior associated with hormonal changes during menopause are highly pertinent to various obesity treatments, including glucagon-like peptide (GLP)-1 receptor agonists (GLP-1 RAs), bupropion/naltrexone, bariatric surgery, and Epitomee capsule, all of which act on satiety pathways, through distinct mechanisms of action.24,33–35
Evidence from GLP-1 RAs studies supports the relevance of this approach in peri- and postmenopausal women: in a real-world cohort study, semaglutide produced greater total body weight loss in women receiving HT compared with those not receiving it (12-month: 16 ± 6% vs 12 ± 8%; 6-month: 13 ± 6% vs 9 ± 5%). 36 In the SURMOUNT-1, tirzepatide achieved substantial weight reductions across menopausal subgroups (26% in premenopausal, 23% in perimenopausal, and 23% in postmenopausal women at 72 weeks vs ∼2%–3% with placebo). 37 A study with low-dose semaglutide (1 mg) reported comparable weight loss between pre- and postmenopausal women after 4 months (5.8% vs 5.1%, P = NS). 38
Lifestyle interventions in the general population, comprising diet and physical activity, particularly resistance training, reliably improve body composition and cardiometabolic risk and are essential adjuncts. However, when applied alone, they generally yield smaller mean weight loss than modern pharmacologic agents in clinical trials. 39 Evidence specific to postmenopausal women with exercise as the sole intervention remains limited but is growing.40,41
Treatment with obesity pharmacotherapy should be assessed and tailored to individual postmenopausal women using HT, with key considerations including potential drug–drug interactions between concomitant medications and HT. 42 Incretin-based therapies are also associated with delayed gastric emptying, which may reduce the absorption of oral HT. 43 Although this effect is generally not considered clinically significant—even for drugs with a narrow therapeutic index or combined oral contraceptives—variability can occur depending on the agent. 44 For example, tirzepatide may present greater concern in single-dose settings. Therefore, caution is advised for patients with renal impairment and for medications with a narrow therapeutic index, where individualized assessment is recommended. Labeling warnings exist because GLP-1/GIP therapies can cause GI adverse effects (nausea, vomiting, diarrhea) and have been shown in clinical pharmacology studies to reduce exposure to oral hormonal contraceptives. From a broader clinical perspective, the use of GLP-1 receptor agonists in postmenopausal women requires consideration of their suitability for weight loss, strategies to minimize weight regain after discontinuation, 45 and tolerability and safety concerns. Postmenopausal women may be more vulnerable to GI AEs, 46 dehydration, and sarcopenia associated with rapid weight loss.
Encouragingly, the efficacy benefits demonstrated in this subgroup were achieved without compromising safety. The greater improvements observed in this Epitomee subpopulation, compared with the placebo subpopulation, were not accompanied by an increase in AEs. As Epitomee is not absorbed and has no systemic effects, its favorable benefit–risk profile reflects both the low rate of AEs versus placebo and the minimal risk associated with a nonsystemic drug–device combination, which may be particularly advantageous in the postmenopausal population. Of note, the vast majority of female participants in this sub-analysis were not taking HT, which is known to mitigate menopausal changes in body composition and metabolism, improve glucose regulation, and lower the risk of cardiovascular events.8,20–22
This sub-analysis is limited by its small sample size (n = 104) and short duration (24 weeks), restricting generalizability and long-term evaluation of the Epitomee capsule. Larger, longer studies are required to confirm efficacy, sustainability of weight loss, metabolic benefits, and underlying mechanisms.
Conclusion
The Epitomee capsule demonstrates significant efficacy in promoting weight loss and trends to improving several metabolic health parameters in women over 50 years of age. These findings support its potential as a valuable nonpharmaceutical intervention for weight management and potential metabolic health together with the favorable safety profile in this population of patients who suffer many challenges associated with weight loss.
Authors’ Contributions
G.S. and Y.K.: Conceptualization, methodology, formal analysis, investigation, writing—original draft, and writing—review and editing. M.K., A.G., and B.D.: Writing—review and editing. L.C.A.: Conceptualization, methodology, formal analysis, writing—original draft, and writing—review and editing. D.D.: Conceptualization, methodology, investigation, writing—original draft, and writing—review and editing. All authors have read and agreed to the published version of the article.
Footnotes
Acknowledgments
The authors would like to thank all the patient, medical sites (Department of Epidemiology and Prevention and Department of Medicine, Wake Forest University School of Medicine, Winston-Salem, North Carolina, USA; Weight Management Center, Department of Psychiatry and Behavioral Sciences, Department of Psychiatry, Perelman School of Medicine at the University of Pennsylvania, Philadelphia, Pennsylvania, USA; Medical University of South Carolina, Charleston, South Carolina, USA; Department of Medicine, Division of Endocrinology, Metabolism, and Molecular Medicine, Northwestern University Feinberg School of Medicine, Chicago, Illinois, USA; Department of Nutrition Sciences, The University of Alabama at Birmingham, Birmingham, Alabama, USA; Louisville Metabolic and Atherosclerosis Research Center, Louisville, Kentucky, USA; Division of Physical Activity and Weight Management, Department of Internal Medicine, University of Kansas Medical Center, Kansas City, Kansas, USA; and Pennington Biomedical Research Center, Louisiana State University, Baton Rouge, Louisiana, USA), and principal investigators of the RESET study (Jamy D. Ard, Patrick M. O’Neil, Robert F. Kushner, Holly R. Wyatt, Harold E. Bays, Frank L. Greenway, John M. Jakicic, and Thomas A. Wadden). In addition, the authors thank Donna H. Ryan from Pennington Biomedical Research Center for her involvement in this study. Finally, the authors wish to thank Dr. Ronell Klingman from Integremed for their assistance in the editing of this publication.
Author Disclosure Statement
G.S. reports advisory fees from Eli Lilly, Novo Nordisk, Rhythm, Quest Diagnostics, and Epitomee Medical; research grant support from Eli Lilly and Novo Nordisk; participation in the Speakers’ Bureau for Novo Nordisk; and serves as Editor-in-Chief for Diabetes Therapy and Obesity Management. Y.K. is an employee of Epitomee Medical and holds stock or stock options. M.K. reports consultancy fees from Exeltis, Uberlube, Reckitt Benckiser, and the California Medical Board. He serves as a consultant for Epitomee Medical but has not received consultancy fees to date. A.G. has received consultancy fees from Epitomee Medical, Eli Lilly, Novo Nordisk, Currax, and Boehringer Ingelheim. B.D. serves as a consultant for Epitomee Medical but has not received consultancy fees to date. L.C.A. has no conflict of interest and was past employee of Epitomee. D.D. has received speaker and advisory board fees from Boehringer Ingelheim, Eli Lilly, Novo Nordisk, AstraZeneca, and Roach; research grants from Eli Lilly, Novo Nordisk, and Boehringer Ingelheim; and support for attending meetings and/or travel from Novo Nordisk and Boehringer Ingelheim.
Funding Information
The study was funded by Epitomee Medical Ltd.
Supplemental Material
Abbreviations
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
