Abstract
Dosing accuracy, activation force, maximum dose-setting torque, hold force, and injection time were determined for DuraTouch, a reusable insulin pen injector designed to optimize usability, across a range of insulin doses (1, 40, and 80 units) under various temperature and storage conditions. Additionally, a life-cycle assessment of DuraTouch was performed to determine its environmental impact. Dosing accuracy remained consistent across all conditions tested at each dose. Activation force, maximum dose-setting torque, hold force, and injection time remained comparable irrespective of test conditions. Per DuraTouch device per year, the total carbon footprint ranged from 482.2 g to 952.8 g of CO2 equivalents across geographical regions assessed, and the total plastic footprint was 0.47 kg including packaging, Penfill cartridge consumption, and use of NovoFine needles. These findings suggest that DuraTouch is suitable for use across a broad range of environments, and support its position as an alternative to disposable insulin pen injectors.
Introduction
Globally, 150–200 million people are reliant on insulin replacement to manage their diabetes, and this number continues to increase.1–4 There are several methods for insulin delivery, of which subcutaneous administration (vial and syringe, pen injectors) and continuous subcutaneous insulin infusion pumps are the most effective and widely used. 5 Although some individuals still inject insulin using a vial and syringe, in recent years, insulin pen injectors have become the predominant devices for insulin delivery in many countries. Compared with vial and syringe administration, pen injectors offer greater accuracy and precision of dosing; less pain; and improved ease of use, flexibility and convenience, thus enhancing user trust and adherence.6–9
Given the rising number of insulin users worldwide, the environmental impact of medical waste associated with insulin use, including pen injectors, is of increasing concern 10 ; indeed, in a study conducted in Bosnia and Herzegovina, it was estimated that 3.2 million insulin pen injectors were disposed of in 2020, resulting in over 600 tons of waste. 11 Some pen injectors are designed to be replaced once the insulin container is exhausted (disposable pen injectors), whereas others are designed with replaceable insulin cartridges (reusable pen injectors), which allows for refilling with insulin and reuse over several years. This latter approach may help to address environmental concerns while retaining functionality.
DuraTouch® (Novo Nordisk A/S, Bagsværd, Denmark) is a reusable insulin pen injector that incorporates several design enhancements to optimize key usability features of the reusable NovoPen® 4 (Novo Nordisk A/S) and the disposable FlexTouch® (Novo Nordisk A/S) pen injectors. DuraTouch can deliver the same maximum dose (80 units [U]) of U100 insulins as FlexTouch, in incremental steps of 1 U, which is greater than the maximum dose of NovoPen 4 (60 U).12,13 In addition, DuraTouch features an easy-to-read dose scale and a nonextending dose button, as used in FlexTouch.
Individuals who are reliant on insulin pen injectors need to be confident in the functionality, dosing accuracy, and ease of use of their device; however, many external factors may affect these parameters, such as fluctuations in temperature or storage conditions, which may occur when an individual is traveling in different climates. The aim of this study was to determine the dosing accuracy, activation force, dose-setting torque, hold force, and injection time of DuraTouch across a range of doses and under various temperature and storage conditions that reflect and exceed those at which the device is recommended for use. In addition, a life-cycle assessment of DuraTouch was performed to determine its environmental impact.
Methods
Dosing accuracy and user handling parameters
Materials and equipment
In total, 30 DuraTouch pen injectors from the same batch were evaluated using a test medium that was internally validated as a surrogate for insulin. The test medium had similar liquid properties to insulin formulations and was contained in 3 mL Penfill® cartridges (Novo Nordisk A/S). In accordance with ISO 11608-1:2022, 14 all test conditions were evaluated using the same set of DuraTouch pen injectors.
A climate chamber (WK3-340/40, Weiss Technik GmbH, Reiskirchen, Germany) with Simpati software (Weiss Technik GmbH) was used to establish three test temperature conditions: cool (5°C ± 3°C with no relative humidity [RH] requirement), standard (23°C ± 5°C with RH of 50% ± 25%), and warm (40°C ± 2°C with RH of 50% ± 10%). For the cool test condition, 32-gauge needles (NovoFine®, Novo Nordisk A/S; 6 mm) were used to provide a low flow rate. For the standard and warm test conditions, 30-gauge needles (NovoFine; 8 mm) were used to provide a high flow rate. Needles and DuraTouch pen injectors were placed inside the climate chamber (with the test medium cartridge inside the pen injector) and acclimatized to each temperature condition, for at least 4 h before dosing accuracy, activation force, dose-setting torque, hold force, and injection time were measured.
The climate chamber was also used to establish four test storage conditions: dry heat storage (70°C ± 2°C with RH of 50% ± 10% for ≥96 h), cold storage (−40°C ± 3°C for ≥96 h), damp heat storage (40°C ± 2°C with RH of 93% ± 5% noncondensing for ≥96 h), and cyclic storage (lower limit of 5°C ± 3°C, no humidity; upper limit of 55°C ± 2°C, with RH of 50% ± 25%; six cycles).
Dosing accuracy
In accordance with ISO 11608-1:2022, 14 the dosing accuracy of DuraTouch was evaluated at three insulin doses (minimum [1 U], mid-range [40 U], and maximum [80 U]) under the aforementioned test temperature and storage conditions. Each of the 30 DuraTouch pen injectors were used to deliver each dose twice. In total, 60 measurements were performed at each dose level. Air was expelled by performing air shots of two increments until liquid appeared at the tip of the needle. Each pen injector was then connected to a METDose2012 system (Novo Nordisk A/S) with a balance (XP205 Dual Range, Mettler-Toledo LLC, Ohio, USA) attached, and the appropriate dose was discharged and weighed. The acceptance criteria for weight measurements were corrected based on the density of the test medium (1.0041 g/mL at 5°C, 1.0022 g/mL at 20°C, and 0.9960 g/mL at 40°C).
Activation force, dose-setting torque, dosing flow rate, hold force, and injection time
To further assess user handling after the DuraTouch pen injector was exposed to various storage conditions, the activation force, maximum dose-setting torque (the effort needed to turn the dial and set the dose), hold force (the degree of force required to sustain the injection), and dosing flow rate or injection time (time taken for the full insulin dose to be administered) were measured.
The activation force for DuraTouch was measured in newtons, at the maximum injectable dose setting (80 U), using a LUNA pen tester (ZwickRoell Ltd., Worcester, United Kingdom). Activation force limits were defined as a minimum of 0.5 N and a maximum of 10 N. For dose-setting torque, acceptance limits were defined as a minimum of 10 milli-Newton meters (mNm) and a maximum of 75 mNm, in accordance with ISO 11608-1:2022. Minimum dose-setting torque was assessed only under standard conditions at each dose setting, with a rotational steady state speed of 25°/s ± 0.5°/s. For hold force, the acceptance limit was defined as a maximum force of less than 10 N, in accordance with ISO 11608-1:2022, and was assessed at the maximum dose under all test conditions. Injection time and dosing flow rate were assessed at the maximum dose under all test conditions, aiming for a maximum flow rate of 450 µL/s. Injection time was defined as the duration between the first and last clicks of the DuraTouch device during dosing, as indicated by audio captured with a microphone inside the LUNA pen tester. The maximum acceptance limit for injection time was 27.4 s.
Statistical analysis
All data were collected using testXpert software (ZwickRoell Ltd.) and analyzed using descriptive statistics. For dose-setting torque only, raw data were manually reviewed and corrected by trained personnel before analysis to ensure that peak torque was accurately identified by the software.
Life-cycle assessment
At the time of assessment, DuraTouch was not in full-scale production; therefore, data on energy consumption and product distribution collected by the manufacturer for FlexTouch during 2022 were used as a proxy to determine the estimated total carbon and plastic footprints of DuraTouch for the period 2024–2026. 15 Carbon and plastic footprints were calculated in accordance with sector guidance for pharmaceutical products and medical devices, namely, ISO 14040:2006 and ISO 14044:2006.16–18 Calculations included waste (e.g., plastic, cardboard) generated during production and packaging, and were based on production in China before distribution to pharmacies located in Europe (Munich, Germany), the USA (Denver, CO), and Japan (Kyoto). The use time of DuraTouch was assumed to be 2 years based on a defined daily dose of 40 U insulin per day. 19
Results
Dosing accuracy
The DuraTouch pen injector and its components are shown in Supplementary Fig. S1. Dosing accuracy results for each insulin dose (1, 40, and 80 U) under cool, standard, and warm temperature test conditions were consistent across conditions at each dose tested, and all remained within the ISO 11608-1:2022 acceptance limits (Fig. 1A–C). Similarly, when assessed under various storage conditions, the results for each dose under each test condition were comparable across test conditions and remained within the acceptance limits (Fig. 1D–F).
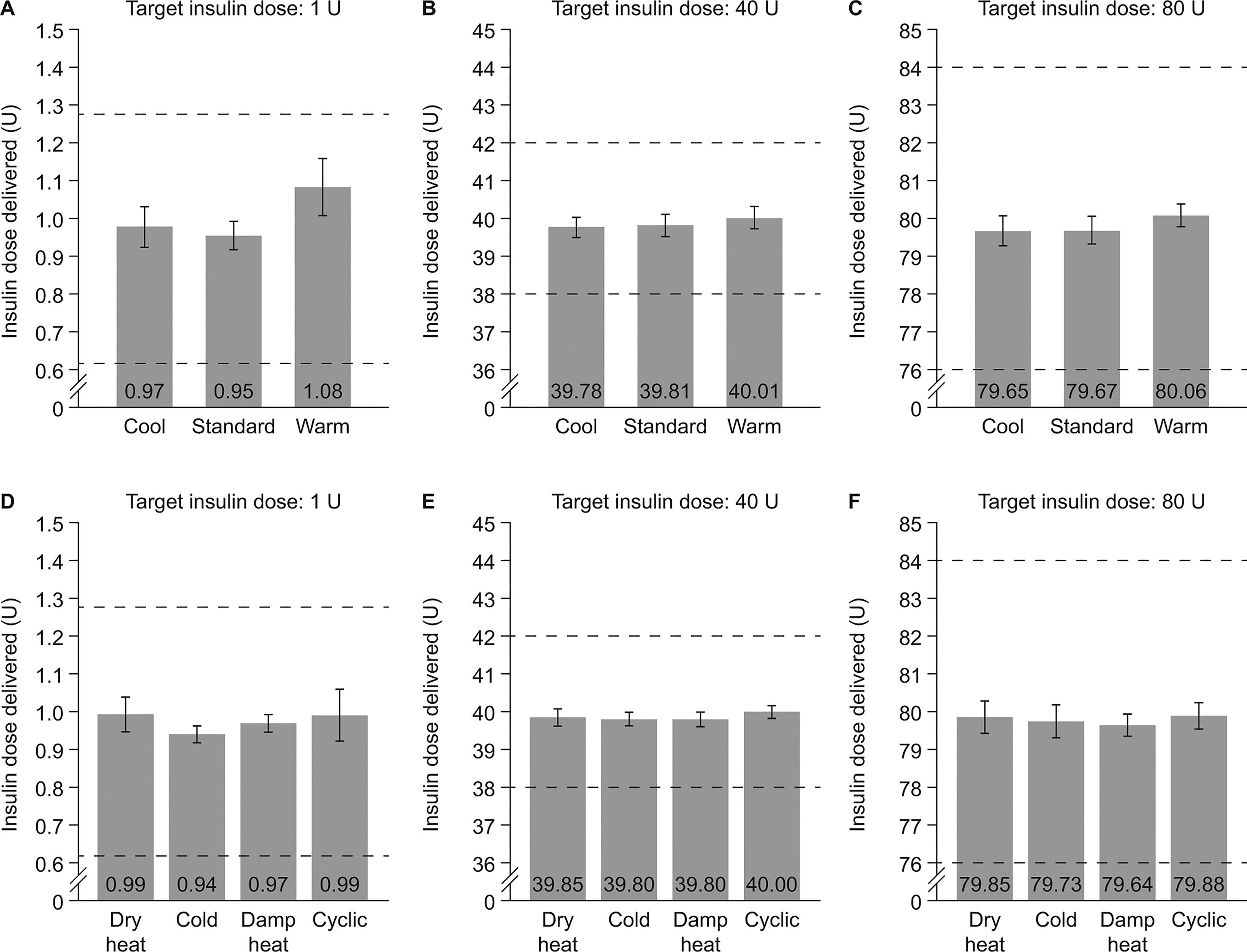
Dose accuracy results for low (1 U), medium (40 U), and high (80 U) doses of insulin under various temperature
User-handling parameters
When measured under test temperature or storage conditions, the mean (standard deviation [SD]) activation force, maximum dose-setting torque, hold force, dosing flow rate, and injection time for DuraTouch remained comparable irrespective of the test conditions and well within the acceptance limits specified for each parameter (Table 1). Similarly, when measured under exposure to various storage conditions, the mean (SD) activation force, maximum dose-setting torque, hold force, dosing flow rate, and injection time for DuraTouch remained comparable irrespective of the test conditions (Table 1). Under standard temperature conditions, the minimum dose-setting torque (SD) for DuraTouch was 26.5 (1.2) mNm. All measurements remained well within the specified acceptance limits.
Activation Force, Dose-Setting Torque, Hold Force, and Injection Time for DuraTouch Under Various Temperature and Storage Conditions
For all test conditions, at each dose (1 U, 40 U, and 80 U), the sample size was 30 DuraTouch pen injectors.
Dry heat storage was defined as 70°C ± 2°C with RH of 50% ± 10% for ≥96 h.
Cold storage was defined as −40°C ± 3°C for ≥96 h.
Damp heat storage was defined as 40°C ± 2°C with RH of 93% ± 5% noncondensing for ≥96 h.
Cyclic storage had a lower limit of 5°C ± 3°C with no humidity and an upper limit of 55°C ± 2°C with RH of 50% ± 25% for six cycles.
Minimum dose-setting torque is measured only under standard temperature conditions.
RH, relative humidity; U, unit(s).
Life-cycle assessment
Over a full life cycle, the total carbon footprint of DuraTouch was estimated to be 538.1 g of CO2 equivalents per device in Europe, 952.8 g of CO2 equivalents per device in the United States, and 482.2 g of CO2 equivalents per device in Japan. Raw materials and distribution were the primary contributors to the carbon footprint, accounting for over 80% of the total emissions for DuraTouch. The plastic footprint of DuraTouch, including Penfill cartridge consumption with NovoFine needles, was 0.12 kg per device per year excluding packaging, and 0.47 kg per device per year including packaging. 15 When Penfill consumption was excluded, the plastic footprint for DuraTouch was 0.012 kg per year. 15
Discussion
When assessed under a range of temperature and storage conditions, DuraTouch delivered consistent and accurate insulin doses, meeting industry standards, across its intended dose range (1–80 U). Based on these data, DuraTouch may be suitable for use by individuals in a broad range of environments.
The dosing accuracy findings were within the ISO 11608-1:2022 acceptance limits under all temperature conditions tested and were similar to those previously reported for the NovoPen 4 reusable pen injector 20 and the FlexTouch disposable pen injector,9,21,22 both of which are widely used in the current market and share some features with DuraTouch.
Similarly, when assessed under various storage conditions, the dosing accuracy of DuraTouch for each dose under each test condition was within the ISO 11608-1:2022 specified requirements for needle-based injection systems at all tested doses and under all conditions studied, and were similar to those previously reported for NovoPen 4.14,20 Minimizing variability in the delivered dose, that is, ensuring that an accurate dose is delivered consistently, thus enhancing or optimizing diabetes management, is an important factor in generating trust in a pen injector.
The nonextending dose button of DuraTouch is intended to reduce the required injection force, a design shared with the FlexTouch disposable pen injector, and was previously shown to be particularly beneficial for individuals who might struggle to apply the force needed to use traditional insulin pen injectors;9,23,24 indeed, across test conditions, the mean (SD) activation force reported for DuraTouch (3.9–4.3 N) was broadly similar to that previously reported for FlexTouch (5.33–5.94 N) and lower than that reported for NovoPen 4 (6.9 N).23,25 In addition, measurements for maximum dose-setting torque, hold force, injection time, and dosing flow rate were consistent for DuraTouch across the tested temperature and storage conditions, suggesting that the device remains stable and easy to use even following exposure to environmental stressors.
Multiple factors may affect the uptake of DuraTouch postlaunch, including ease of use and affordability. The present study did not assess patient-reported handling of DuraTouch; however, a recent handling study of DuraTouch in 100 adults with type 1 diabetes or type 2 diabetes with or without prior pen injector experience showed that ≥85% of participants found DuraTouch very easy or extremely easy to use and to learn how to use, irrespective of prior injector experience. 26 Furthermore, while variation in cost and access is expected on a local level, the affordability of reusable pens compared with disposable pens may also help facilitate the uptake of DuraTouch, as the former have been shown to be generally less expensive due to their replaceable cartridge-based design.27,28
Finally, data from the life-cycle assessment support the position of DuraTouch as an alternative to disposable insulin pen injectors, with a total plastic footprint of 0.47 kg per device per year including packaging, Penfill cartridge consumption, and use of NovoFine needles. 15 Although the estimated total carbon footprint of DuraTouch varied according to the geographical location, these differences are likely due to variations in distance from the production site in China and the transport method used for distribution. The lack of manufacturing data specific to DuraTouch was a key limitation of this environmental assessment (as it was not in full scale production at the time of analysis); however, best estimates were calculated based on proxy data from FlexTouch. Additional studies will be needed to update the environmental impact estimates provided herein.
Conclusions
Insulin pen injectors with good dosing accuracy help to facilitate optimal insulin delivery for people with diabetes. The results reported here demonstrate that the high dosing accuracy of DuraTouch was maintained across a range of temperatures and storage conditions. Similarly, activation force, maximum dose-setting torque, hold force, and injection time remained comparable over test conditions, and compliance with ISO 11608-1:2022 was confirmed for all parameters tested. These findings suggest that DuraTouch may be of benefit for those who self-administer insulin; moreover, the reusable design may also prove beneficial in reducing environmental waste.
Footnotes
Authors’ Contributions
All authors participated in developing the study concept and design, and in collecting the data. All authors were involved in the analysis and interpretation of data, participated in preparing the article, and approved the final article for submission.
Author Disclosure Statement
A.S.W., S.A., K.B.F., and T.S. are employees of Novo Nordisk and hold shares.
Funding Information
This study was funded by Novo Nordisk. Medical writing support was provided by Chloe Fletcher, MSc, of Oxford PharmaGenesis, Oxford, UK, funded by Novo Nordisk.
Abbreviations Used
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
