Abstract
Objective:
To evaluate the feasibility of using continuous glucose monitors (CGM) and provide preliminary estimates of interstitial glucose patterns among individuals with polycystic ovary syndrome (PCOS).
Methods:
We performed a feasibility study of individuals with PCOS aged 18–45 years without a diabetes diagnosis. Participants wore a FreeStyle Libre 2TM CGM for 42 days. Feasibility outcomes were rate of recruitment, participant attrition, CGM glucose data completeness, and participant satisfaction with CGM use. Secondary exploratory analyses examined glucose parameters: mean glucose, overnight glucose (23:00–07:00), daytime glucose (07:01–22:59), and percent time in range (70–180 mg/dL), below range (
Results:
We enrolled 35 participants in the study. Four (11%) participants withdrew. The mean recruitment rate was 5.8 participants/month. Median CGM data completeness was 91.8% (interquartile range 71.9%–97.9%). Most participants (n = 21, 60%) indicated they would use a CGM again if provided. Stratification by androgen status, BMI, and phenotype revealed no significant differences in glucose parameters.
Conclusion:
We demonstrated the feasibility of CGM in assessing dysglycemia among individuals with PCOS. No significant glucose differences were observed across androgen status, BMI, or phenotype groups. Further research is needed to determine how CGM could be incorporated into PCOS management.
Introduction
Polycystic ovary syndrome (PCOS) is a common endocrine disorder affecting 10%–13% of people with ovaries, with reproductive, metabolic, and psychological implications across the lifespan. 1 PCOS is diagnosed based on the presence of at least two of the following: hyperandrogenism, irregular menstrual cycles, and polycystic ovarian morphology. 1 Individuals with PCOS have an increased risk of metabolic complications, including obesity, insulin resistance, metabolic syndrome, and prediabetes, all of which contribute to a significantly higher likelihood of developing type 2 diabetes. 2 Females with PCOS are two to five times more likely to develop type 2 diabetes compared with those without PCOS, with this risk further amplified in individuals with excess weight.3–5 As a result, international guidelines recommend preventive strategies and routine screening for diabetes, including oral glucose tolerance testing (OGTT) at diagnosis and every 3 years thereafter, or more frequently in the presence of additional risk factors. 1
Continuous glucose monitors (CGM) have become a valuable tool in diabetes management. 6 CGM offer real-time tracking of glucose levels in interstitial fluid and provide a comprehensive view of daily glucose fluctuations. 6 CGM can detect early abnormalities in glucose regulation, which may be missed by standard screening methods like hemoglobin A1c (HbA1c). 6 Although CGM have been widely applied in diabetes care, their use in PCOS remains underexplored.7–9 These studies had small sample sizes and short monitoring periods, and most were published over a decade ago using older CGM technology. Earlier CGM technologies were limited by short sensor lifespans, frequent calibration requirements, and reduced accuracy over time. 10
This pilot study aimed to evaluate the feasibility of using CGM to evaluate glucose patterns in individuals with PCOS to inform the design of future, larger-scale trials investigating glucose dysregulation in this population. Specifically, this study sought to assess recruitment rates, participant retention, the quality and completeness of collected CGM data, and participant satisfaction with CGM use. To inform planning of future trials, our secondary objective was to provide preliminary estimates of blood glucose patterns among people living with PCOS.
Methods
Study design
We conducted a cross-sectional study at the University of Calgary, Canada, between April and September 2024. The protocol was registered prospectively on Open Science Framework (osf.io/f8txd) and approved by the University of Calgary Conjoint Health Research Ethics Board (REB23-1576). All participants provided written informed consent.
Participants
Females aged 18–45 years with self-reported PCOS were included, with a diagnosis subsequently confirmed by a physician using the Rotterdam criteria. 1 When ultrasound data were unavailable, anti-Müllerin hormone (AMH) was used as a proxy for polycystic ovarian morphology, in accordance with the 2023 International PCOS Guidelines. 1 A cutoff of 22.8 pmol/L was used based on findings from previous literature. 11
Participants were recruited using social media, posters, word of mouth, and health care provider referrals. Participants were excluded if they had any known medical condition that could affect glucose metabolism (e.g., diabetes, history of bariatric surgery) or had previously taken medications or supplements that could impact reproductive hormone levels (e.g., oral contraceptive pills, spironolactone) or glucose metabolism (e.g., weight loss or diabetes medications) within the last 3 months. Participants taking medications that could interfere with the accuracy of the Freestyle Libre 2TM CGM 12 (e.g., aspirin or vitamin C ≥500 mg/day) were also excluded. Individuals were not eligible to participate if pregnant, undergoing treatment for an eating disorder, had an implantable device, or planning to undergo diagnostic imaging procedures (e.g., X-ray) during the study period. Potential participants were screened for inclusion and exclusion criteria by email or telephone. The study was incentivized, and participants received compensation for their involvement.
Outcomes
The primary outcome of this pilot study was to evaluate the feasibility of CGM use in individuals with PCOS. Four feasibility metrics were defined a priori: (1) recruitment of ≥30 participants; (2) participant attrition of <25%, consistent with other PCOS studies;13,14 (3) having ≥50% of the expected glucose readings obtained over the 42-day study period for participants, based on expert opinion; and (4) participant satisfaction with CGM use. The secondary outcome was to provide preliminary estimates of interstitial glucose patterns among people with PCOS.
Study measurements
Baseline data were collected at study enrollment, including demographics, anthropometric measurements, self-reported medical history, and laboratory assessments. Participants recorded the dates of their menstrual periods during the study and reported them upon study completion. Height and weight were collected using a standard stadiometer (Pelstar 402KL scale, McCook, IL, USA) and a calibrated weighing scale (Pelstar 402KL scale, McCook, IL, USA), respectively. Waist circumference was taken at the midpoint between the lowest rib and the iliac crest with a nonelastic flexible tape measure and recorded to the nearest 0.5 cm. Hip circumference was measured using a flexible tape measure at the widest point of the buttocks, with the participant standing upright and feet together and recorded to the nearest 0.5 cm. Blood pressure was measured in accordance with Hypertension Canada guidelines 15 using a sphygmomanometer (7670–01S, Welch Allyn Inc., NY, USA). Grip strength was measured using a standard dynamometer. All measurements were conducted by an exercise physiologist or research staff trained by an exercise physiologist to ensure consistency and reliability.
Bioelectrical impedance analysis (Quantum IV, RJL Systems Inc., MI, USA) was used to assess body composition, including fat mass (kg), fat-free mass (kg), and percentage of fat in total weight. Exercise testing included assessments of peak oxygen consumption (VO2 peak) with a graded treadmill test adapted from the Bruce protocol. 16
Blood samples were drawn in the morning after fasting for at least 8 h. Total cholesterol, high-density lipoprotein cholesterol, triglycerides, alanine aminotransferase, and creatinine were measured on the Cobas c701 (Roche Diagnostics). HbA1c was measured on the Cobas c523 analyzer (Roche Diagnostics). Total testosterone, estradiol, progesterone, sex hormone binding globulin (SHBG), AMH, luteinizing hormone, and follicle-stimulating hormone were measured on the Cobas e801 analyzer (Roche Diagnostics). Free androgen index (FAI) was calculated as the ratio of total testosterone to SHBG. Homeostatic model assessment of insulin resistance 17 and low-density lipoprotein cholesterol 18 were calculated. Insulin was measured using a sandwich assay (Cobas e801 analyzer, Roche Diagnostics). A 75-g OGTT was also performed, with glucose measured at fasting and 2 hours post-load.
Participant satisfaction with CGM was evaluated at the end of the study using a survey adapted from previous literature. 19 The survey assessed acceptability of CGM use through overall satisfaction with CGM use, the likelihood of future use, and experiences related to sensor application, comfort, discretion, and integration into daily life. 19
CGM measurements
Interstitial glucose levels were collected for 42 consecutive days using the Freestyle Libre 2TM CGM. The CGM captured readings every 15 min and reported them to a smartphone app on the participants’ phones or to a reader device. 20 Participants received three sensors at the beginning of the study, with the first sensor applied by the study team. The CGM was worn on the back of the arm. Additional adhesives were applied directly to the skin to further secure the CGM. Participants were asked to scan the sensor to the smartphone app or reader device at least once every eight hours to ensure CGM connectivity and prevent any loss of data. All data from the CGM were automatically uploaded and stored on the LibreView system 12 for later analysis. Each sensor had a measuring period of 14 days, after which participants needed to remove the old sensor and apply a new one. Participants were given the option to apply the subsequent sensors themselves or with the assistance of the research team. Replacement sensors were provided in cases of sensor malfunction or if sensors detached before the full 14-day wear period. Adverse events were documented on a standard form throughout the study.
Sample size
This pilot study aimed to assess the pre-specified feasibility outcomes, including participant recruitment, protocol adherence, and participant satisfaction. Therefore, a sample of 35 participants was recruited to allow for this preliminary evaluation.
Statistical analysis
Feasibility outcomes were summarized using descriptive statistics. Recruitment was assessed based on whether the target sample size of participants was achieved, followed by calculating the recruitment rate, which was defined as the total number of participants divided by the 6-month recruitment period. Attrition was calculated as the number of participants who dropped out of the study relative to the total number of enrolled individuals and presented as a percentage. The completeness of interstitial glucose data was assessed by calculating the percentage of the expected CGM readings obtained during the wear period. The CGM recorded interstitial glucose every 15 min, resulting in 96 readings per day and an expected total of 4032 readings over the 42-day study period. CGM data completeness was reported as the median and interquartile range (IQR) across participants. Participant satisfaction responses were grouped into categories and summarized.
Data retrieved from the CGM were downloaded and systematically processed via LibreView. 12 We extracted glucose readings along with their corresponding timestamps and subsequently separated the date and time to accurately correlate each reading with its specific occurrence.
Exploratory analysis of glucose patterns was conducted among participants for whom ≥50% of glucose data was available over the 42-day study period. Participants were excluded if diagnosed with type 2 diabetes. Among eligible participants, we used all available interstitial glucose values for each participant to calculate mean interstitial glucose, mean overnight glucose (23:00–07:00), mean daytime glucose (07:01–22:59), percentage of time spent within the target range (70–180 mg/dL), percentage of time below range (
Statistical analyses were conducted using GraphPad Prism software (version 8.4.2, GraphPad Software Inc., San Diego, CA, USA) and Microsoft Excel 365 (Microsoft Corporation, Redmond, WA, USA). To evaluate the normality of the data, the Shapiro–Wilk test was utilized, with statistical significance assigned as P < 0.05.
Results
Participant characteristics
A total of 35 participants consented to participate in the study. Participant characteristics are presented in Table 1. The mean age was 32 years (±4.9). All participants identified as cisgender women. Most participants self-identified as being of European/Mediterranean descent (19, 54%). Educational attainment was high, with over 85% of participants reporting education beyond high school, including 20 (57%) with an undergraduate degree. Thirteen (37%) participants identified as former smokers.
Participant Characteristics
n = 34.
Ethnic categories adapted from Statistics Canada classification (https://www.statcan.gc.ca/en/start).
Phenotype: (A) clinical or biochemical hyperandrogenism, ovulatory dysfunction, polycystic ovaries; (B) clinical or biochemical hyperandrogenism, ovulatory dysfunction; (C) clinical or biochemical hyperandrogenism, polycystic ovaries; (D) ovulatory dysfunction, polycystic ovaries.
Participant Anthropometric Measures
The mean BMI was 30.4 kg/m2 (±8.4), with 13 (38%) participants classified as having obesity (BMI ≥30 kg/m2) (Table 2). The mean VO2 peak was 35.3 (±9.7) mL/kg/min. On average, participants had a fasting glucose of 92.8 mg/dL (±14.3), a fasting insulin level of 10.5 mIU/L (±7.6), and mean HOMA-IR of 2.9 (±2.3) (Table 3).
Participant Laboratory Work
Twenty-two participants (63%) reported experiencing ≥
Feasibility outcomes
A total of 284 individuals contacted our research team to express interest in participating in the study. Of these, 183 (64%) learned about the study through social media and online PCOS support groups, 55 (19%) through posters, 23 (8%) were recruited through the University of Calgary’s research participation website, 15 (5%) were referred by word of mouth, and 5 (2%) were referred by physicians.
Of the 284 individuals who contacted us—either by email or completing the online study interest form—90 (33%) did not complete eligibility screening. Of the remaining 194 individuals, 54 (19%) were eligible for the study, while 140 (49%) were not. The primary reason for ineligibility was the use of medications (e.g., metformin, semaglutide, combined oral contraceptive pill) or supplements (e.g., inositol, berberine) that could affect blood glucose and/or hormone levels (Fig. 1). Among the 54 (19%) eligible individuals, 35 (12%) consented to participate in the study.
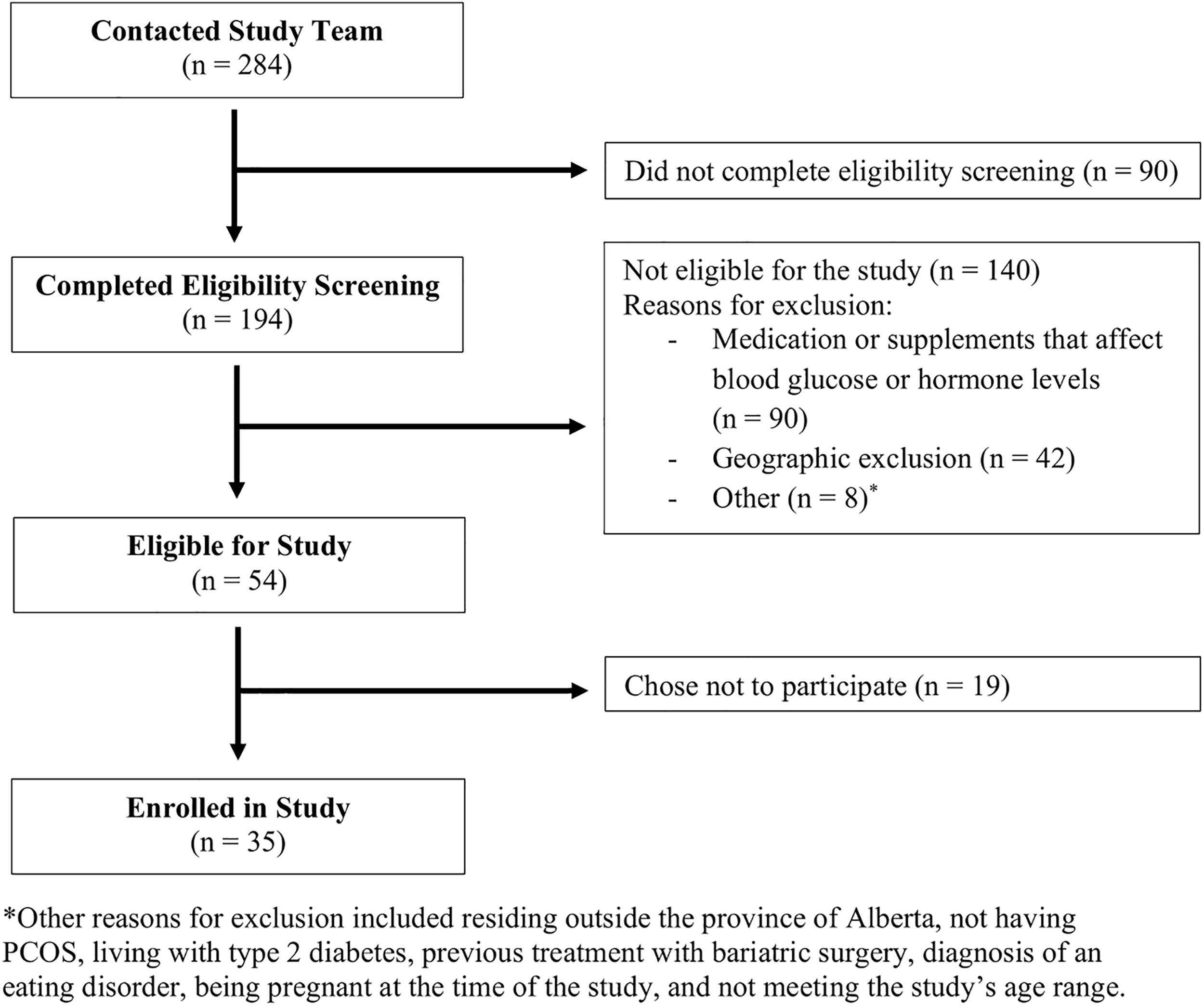
Participant flow diagram.
As recruitment occurred over 6 months from April to September 2024, the recruitment rate was 5.8 participants per month. Four of the 35 (11%) participants dropped out, which was less than the a priori feasibility threshold of 25%. Two of those who withdrew chose to stop participating for personal reasons, while the other two withdrew due to issues with the CGM. The CGM repeatedly fell off for the individuals who had issues with the CGM, with both participants reporting use of all three provided sensors in 1 week rather than over 42 days. Consequently, they decided not to continue with the study.
CGM data completeness was high, with a median of 91.8% (IQR: 71.9%–97.9%) of the expected glucose readings successfully recorded across participants. Thirty-one (88.6%) participants had ≥50% of the glucose readings over the 42-day study period. One common reason for the loss of CGM data was connectivity issues between the CGM and the smartphone device. The CGM frequently connected and disconnected from the device, even when in proximity, resulting in the loss of interstitial glucose data. Additional missing data arose from time gaps between removing one sensor and placing the next after the 14-day wear period.
As shown in Figure 2, participants rated their experience with the CGM through a series of statements regarding sensor wear, comfort, and satisfaction. Overall, satisfaction with CGM use was high, with 25 (81%) participants agreeing or strongly agreeing that the sensor was comfortable to wear and 21 (66%) reporting no discomfort under the skin. Most participants (25, 78%) found the sensor easy to wear due to its small size, while 21 (66%) indicated it did not interfere with their daily activities. The sensor was perceived to integrate well into participants’ everyday activities, with 20 (69%) participants agreeing it aligned with their lifestyle. Additionally, 23 (74%) participants felt comfortable applying the sensor. Notably, only 9 (29%) participants felt the CGM was not noticeable, meaning over two thirds of participants felt it was noticeable, highlighting concerns about device visibility.
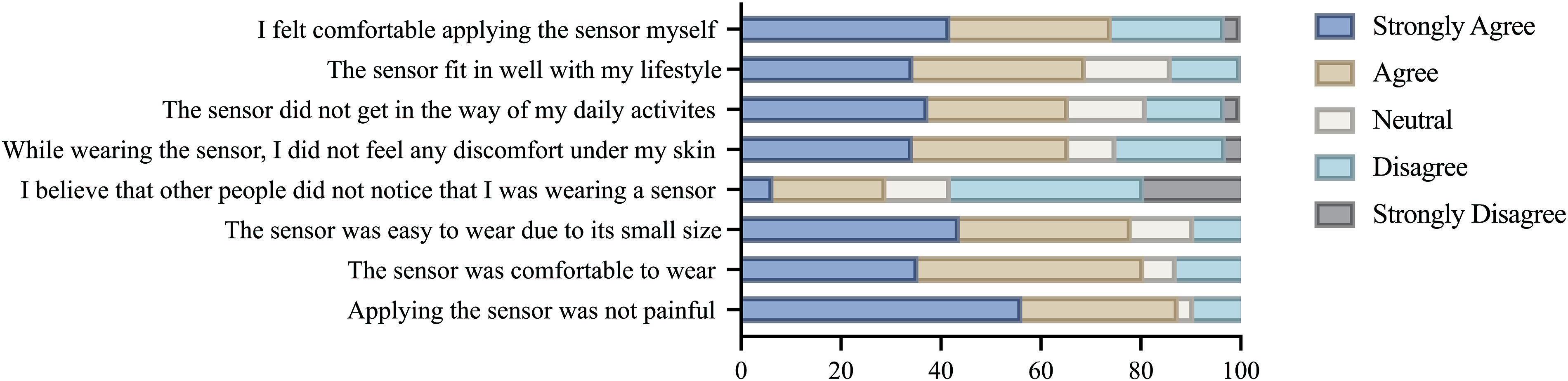
Participation satisfaction with CGM use. CGM, continuous glucose monitor.
A total of 105 sensors were distributed among the 35 participants, of which 10 (10%) required replacement due to sensor detachment or malfunction.
Glucose parameters
Two participants were diagnosed with type 2 diabetes, and six were identified as having impaired glucose tolerance (IGT) as a result of participating in this study. The two participants diagnosed with type 2 diabetes were excluded from the exploratory glucose analysis. The analysis included data from 29 participants with ≥50% of the CGM data over the 42-day study period. The mean glucose level over the 42 days was 105 ± 9.5 mg/dL, with a daytime mean of 107.1 ± 10.1 mg/dL and a nighttime mean of 101 ± 9.2 mg/dL. The mean percent time in range was 97.6% ± 3.0%, above range was 0.5% ± 1.5%, and below range was 1.96% ± 3.1%.
In the subgroup analysis, when stratified by androgen status, BMI, and phenotype, we observed no statistically significant differences in the mean 42-day glucose, mean day and overnight glucose, and percent time in range, above range, and below range (Supplementary Fig. S1, Supplementary Table S1). The androgen group tended to have higher glucose levels than the nonandrogen group, with this pattern observed across mean 42-day, daytime, and nighttime measures. The same trend was observed in the phenotype analysis, where phenotypes A/B/C showed lower glucose levels compared with phenotype D. In addition, BMI subgroup analysis indicated that individuals in the higher BMI subgroup tended to show higher glucose levels on mean 42-day and daytime measures. One participant did not complete blood work and was therefore excluded from the androgen status subgroup analysis.
Discussion
In this cross-sectional study of 35 individuals living with PCOS, we demonstrated the feasibility of using CGM among those with PCOS. Recruitment goals were met with a recruitment rate of approximately six participants per month, combined with low attrition, as only four participants (11%) withdrew from the study. Most participants (31, 89%) wore the CGM for the entire 42-day study period. CGM data capture was high, with 91.8% (IQR: 71.9%–97.9%) of possible interstitial glucose readings recorded. A positive response was noted by individuals regarding CGM use, with 81% of individuals reporting that the device was comfortable to wear, 78% reporting that the sensor was easy to wear, and 74% feeling comfortable applying the sensor themselves. Although nonsignificant, subgroup analyses consistently showed higher glucose levels in participants with hyperandrogenism, PCOS phenotype D, and a higher BMI, highlighting patterns that should be explored in larger studies.
Our findings align with those reported in recent studies on CGM feasibility in individuals with diabetes regarding CGM data completeness and satisfaction.23,24 Johansson et al. 23 conducted a 28-day study where participants with type 2 diabetes in a Swedish primary care setting wore CGM (FreeStyle Libre 1TM CGM). The availability of CGM data within their study exceeded 70%, and assessment of the acceptability of the CGM through participant feedback and focus group interviews with health care professionals revealed a positive user experience. 23 Similarly, Richardson et al. 24 incorporated a brief CGM intervention into a diabetes prevention program for adults with prediabetes (Dexcom G6TM). In this study, participants wore the CGM for 10 days and attended one educational session focused on CGM use. 24 The acceptability of CGM was evaluated through postintervention surveys and focus groups, showing high ratings and support for the use of CGM as part of the program. 24 A median of 100% data sufficiency was reported, with 88% of participants stating they would wear the CGM again. 24 This favorable rating is higher than the rating provided by participants in our study, where only 65% of participants indicated that they would wear the CGM again. A possible reason for this could be that the study was conducted during the spring-summer seasons when CGM are more visible to the public due to clothing choices for warmer weather (i.e., short sleeves). Such perceptions may influence CGM acceptability with a previous study reporting that some participants were reluctant to wear CGM on visible areas, like their upper arm, because they did not want others to associate them with diabetes or ask intrusive questions due to the stigma surrounding the condition. 25 Some individuals may be hesitant to wear a CGM again due to health anxiety from real-time glucose data, misconceptions about its use being limited to diabetes, or financial barriers when coverage is unavailable.26,27 Beyond immediate acceptability, these concerns may also influence participant retention and long-term adherence in research settings. Individuals who experience stigma or discomfort may be less likely to continue wearing a CGM in longitudinal studies, which has implications for study design, data completeness, and generalizability. Future research should therefore examine how device visibility and psychosocial factors impact CGM adherence over time and incorporate strategies to support CGM usage in both clinical practice and research contexts.
Despite increasing use, there is a lack of guidelines and standardized interpretation frameworks for how CGM can be used in individuals without diabetes.28–32 This concern was underscored by Spartano et al., 31 who found that CGM experts were inconsistent in interpreting CGM reports from people living without diabetes—more than half recommended follow-up when the time above 180 mg/dL exceeded 2%. While CGM provide real-time data and trends throughout the day and night, the physiological relevance of these data remains unclear. 28 In our exploratory analysis, we found that people with PCOS without a diagnosis of diabetes had high percent time in range as reported by CGM. This is consistent with another recent study, which also used the Freestyle Libre 2 CGM. 9 While the overall time in range is high, small observational studies have reported that people with PCOS may have prolonged glucose responses and that those with insulin resistance may have lower glycemic variability.7–9 Spartano et al. reported that normoglycemic adults spend the vast majority of their CGM time within the broad range of 70–180 mg/dL. 32 These observations provide preliminary reference points for physiological glucose ranges but were not intended as clinical targets. More research is needed to define practices and interpretative thresholds outside of diabetes.
PCOS is strongly associated with an increased risk of developing type 2 diabetes and IGT.2–5 Individuals with PCOS have a two to five times higher likelihood of developing type 2 diabetes compared with those without PCOS, even after adjusting for BMI and other factors.3–5 PCOS and diabetes are primarily connected through insulin resistance, obesity, genetic factors, and hormonal imbalances, which further elevate the risk.33,34 CGM technology has evolved substantially in recent years, enhancing diabetes care, management and diagnosis. 6 With these advancements, CGMs now offer personalized feedback on the effects of diet, activity, and stress. 35 CGMs support proactive lifestyle adjustments, which are a vital part of precision care, presenting as a promising, underexplored opportunity to advance precision care in this underrepresented population. Demonstrating feasibility in this population is a critical first step toward unlocking that potential.
One area where the use of CGM raises important questions is how we traditionally assess glucose tolerance. The OGTT has long been the standard for detecting dysglycemia, but its practical limitations create barriers for patients. 36 OGTTs are time-consuming and require scheduled lab visits, making them difficult to complete. 36 The need for fasting, the unpleasant glucose drink, and potential side effects such as nausea or dizziness further add to the burden. 36 These challenges often lead to missed testing and delayed recognition of glucose abnormalities. 36 In contrast, CGM provide a more convenient, patient-centered option offering continuous data in a real-world setting that can complement and, in some cases, replace OGTTs. 37
This feasibility study had many strengths. We had a diverse range of participants regarding age, and ethnicity, enhancing our findings’ relevance and applicability to different groups. All data collection was conducted during the same season (spring-summer). We also adhered to a rigorous protocol to ensure consistency across all participants. However, there are important limitations to consider. As a pilot study, we had a small sample size, which reduced the power to detect differences between groups. We also excluded people with PCOS who were taking medications and supplements used for the treatment of PCOS, including combined oral contraceptive pills, metformin and inositol, which limits the external validity of our findings to the broader PCOS population. Future studies should consider stratified inclusion or subgroup analyses comparing individuals taking medications and supplements such as metformin, oral contraceptives, and inositol to those not taking them, to better reflect real world PCOS management and assess potential differences in glucose patterns. In addition, this was a single-site study in Calgary, limiting generalizability to other settings. It is also possible that our sample reflected individuals with relatively high socioeconomic status and health literacy, which could further impact generalizability; however, this was not formally assessed.
In summary, our pilot study demonstrated that CGM are feasible for people with PCOS. Traditional glucose tests, such as OGTT, may overlook early indicators of IGT, as they provide only a single snapshot in time. 38 Additionally, these tests require scheduled laboratory visits, which are frequently missed or deferred, limiting their utility in routine screening. The advantage of CGM in providing real-time data and day/night trends is that it offers a promising alternative to OGTT for identifying IGT. Nevertheless, a significant challenge remains due to the lack of standardized guidelines for CGM interpretation beyond diabetes management.28,30,31 Further research is necessary to understand how CGM can be integrated into PCOS care. Larger studies are required to explore the connections between CGM data and potential metabolic risks associated with PCOS and to evaluate how these sensors can guide early intervention strategies.
Footnotes
Acknowledgments
The authors would like to thank the participants of this study for their time.
Authors’ Contributions
I.S.: Data curation; formal analysis; investigation; visualization; writing—original draft. R.V.: Conceptualization; methodology; writing—review and editing. H.A.H.: Investigation; validation; writing—review and editing. H.V.B.: Conceptualization; methodology; writing—review and editing. E.A.B.: Conceptualization; methodology; writing—review and editing. S.M.D.: Conceptualization; methodology; writing—review and editing. J.M.Y.: Conceptualization; methodology; writing—review and editing. J.L.B.: Conceptualization; funding acquisition; investigation; methodology; project administration; resources; supervision; validation; writing—review and editing.
Author Disclosure Statement
I.S., R.V., H.A.H., E.A.B., H.V.B., S.M.D., and J.L.B. have no disclosures to report. J.M.Y. reports nonfinancial support (study devices) from Abbott Devices for a clinical study not reported in this article.
Funding Information
This study was funded by the Alberta Health Services/University of Calgary Clinical Research Fund Grant.
Abbreviations Used
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
