Abstract
Gastroparesis, a form of autonomic neuropathy, causes delayed gastric emptying and unpredictable postprandial glycemic fluctuations, increasing hypoglycemia risk due to mismatched insulin timing. We demonstrate the effective application of sensor-augmented pump (SAP) therapy using an advanced hybrid closed-loop (AHCL) system (MiniMed 780G) in an individual with type 2 diabetes complicated by gastroparesis. To assess the adaptability of the AHCL algorithm, we conducted meal tests using two distinct macronutrient compositions—high carbohydrate (bread) and high fat (curry with rice)—and demonstrated that the AHCL system appropriately responded to complex postprandial glycemic fluctuations. The system autonomously adjusted basal rates and delivered correction boluses in response to delayed glucose rises, markedly reducing hypoglycemia. The patient achieved excellent glycemic management, with time in range (70–180 mg/dL) of 88% and time below range (<70 mg/dL) of <1%. AHCL-based SAP therapy may be especially beneficial in gastroparesis-associated glycemic variability.
Introduction
Gastroparesis is a form of autonomic neuropathy with delayed gastric emptying unrelated to obstruction. It is commonly observed in people with diabetes who have other microangiopathy and experience difficulties in glycemic management. 1 In a subset of the Diabetes Control and Complications Trial, gastroparesis was found to be associated with elevated HbA1c levels, suggesting that chronic difficulties in glycemic management are implicated in gastroparesis. 2 However, despite elevated HbA1c levels in individuals with gastroparesis, the mismatch between insulin administration timing and nutrient absorption in gastroparesis increases the risk of severe hypoglycemia. Previous reports have also indicated that individuals with gastroparesis experience a higher frequency of severe hypoglycemia compared with those without gastroparesis. 3 Therefore, these factors contribute to the difficulty in achieving target blood glucose levels.
To address the challenges of glycemic management in individuals with gastroparesis, continuous subcutaneous insulin infusion (CSII) therapy has been explored as a treatment option. A pilot study (GLUMIT-DG) conducted by the National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK) Gastroparesis Clinical Research Consortium (GpCRC) evaluated the safety, feasibility, and potential efficacy of continuous glucose monitoring (CGM) and CSII therapy in diabetic gastroparesis. The study demonstrated the safety of CSII therapy, along with reductions in HbA1c, improvements in blood glucose profiles as measured by CGM, and alleviation of gastroparesis symptoms. 4 In a case series using sensor-augmented pump (SAP) therapy with a hybrid closed-loop (HCL) system, which automatically adjusts the basal rate based on blood glucose levels, a significant improvement in time in range (TIR, 70–180 mg/dL) has been reported. 5 Advanced hybrid closed-loop (AHCL) insulin delivery systems represent a significant evolution in diabetes technology, offering algorithm-driven insulin adjustments based on real-time CGM sensor data. While their use has expanded in type 1 diabetes, 6 AHCL systems are also considered promising in other settings where glycemic management is particularly challenging—such as in individuals with high glycemic variability or those with diabetes in pregnancy requiring tight glucose targets. However, clinical evidence supporting their efficacy in these populations remains limited. Among such complex scenarios, gastroparesis—a form of autonomic neuropathy—leads to delayed gastric emptying and unpredictable postprandial glycemic variability, thereby increasing the risk of hypoglycemia due to mismatched insulin timing. Several reports have demonstrated that, regardless of CGM use, CSII therapy improves glycemic trends and reduces glucose variability compared with multiple daily injection (MDI) regimens in individuals with gastroparesis.4,5,7 However, there have been very few reports objectively evaluating the clinical impact of AHCL system in real-world settings. Here, we describe a clinically instructive case in which an AHCL system (MiniMed 780G; hereafter referred to as 780G AHCL system) was successfully deployed to address the complex glycemic patterns associated with gastroparesis, providing important practical insights for real-world application.
Case Report
A 52-year-old man was referred to our hospital for glycemic management. At the age of 14, he began consuming large amounts of sugary beverages, which triggered a rapid weight loss from 104 kg to 80 kg within a year. Later, he was found unconscious at home and was transported to the emergency department, where he was diagnosed with diabetes due to hyperglycemic coma, leading to the initiation of insulin therapy. Further evaluation revealed that islet-related autoantibodies were negative, and he was in a noninsulin-dependent state, leading to a diagnosis of type 2 diabetes. Although insulin therapy was initially continued, his blood glucose levels stabilized without medication by the age of 16, leading him to discontinue follow-up visits on his own. At the age of 24, hyperglycemia was incidentally detected during a pre-employment medical checkup, prompting the reinitiation of insulin therapy. However, due to a demanding work schedule, adherence to insulin injections was poor, and his HbA1c remained above 10% (87.79 mmol/mol). Approximately 2 years before presenting to our hospital, he developed bilateral plantar numbness and impaired temperature and pain sensation. About 1 year prior to his visit, he also noticed bilateral pitting edema. At presentation to our clinic, fasting plasma glucose was elevated at 264 mg/dL (14.6 mmol/L), with a concurrently low C-peptide level of 1.26 ng/mL (0.42 nmol/L), suggesting impaired endogenous insulin secretion. He was managed with insulin alone throughout the clinical course, without the use of any other antidiabetic medications.
Regarding complications, he exhibited peripheral neuropathy, including impaired temperature and pain sensation. His retinopathy had required vitrectomy, and his nephropathy was classified as stage 3, with a daily proteinuria of 8 g/day (8000 mg/day). Autonomic dysfunction was confirmed by orthostatic hypotension, fecal incontinence, anhidrosis, and a low coefficient of variation in R-R intervals.
Gastroparesis was strongly suspected based on clinical symptoms and CGM data showing a biphasic postprandial glucose pattern. This suspicion was further supported by upper gastrointestinal endoscopy, which revealed significant food retention in the stomach. This was confirmed by a 13C-acetate breath test, which is commonly used for the evaluation of gastroparesis.8,9 We conducted the 13C-acetate breath test in five healthy individuals and the patient to compare the gastric emptying rates, following a previously reported protocol, as outlined below. 10 End-tidal breath samples were collected using small exhalation bags (PYLORI-BAG20, Otsuka Electronics Company, Osaka, Japan) at −15 min, 0 min, every 5 min until 120 min, and every 30 min until 240 min, in accordance with the recommendations of the Japan Society of Smooth Muscle Research. In addition, end-tidal breath samples were collected into three large exhalation bags (PYLORI-BAG1.3L, Otsuka Electronics Company) at −15 min for use as reference samples in exhalation gas analysis. The test meal was prepared by steaming rice with 13C-labeled sodium acetate (catalog number CLM-156-1, Cambridge Isotope Inc., MA, USA), using a ratio of 150 g of steamed rice to 200 mg of 13C-labeled sodium acetate. The prepared meal was stored at −20°C until use. Prior to administration, both the steamed rice and accompanying side dishes were reheated in a microwave. In the test, the time required to reach 50% of the breath CO2 concentration, with the 240-min value set as 100%, was compared. While healthy individuals reached 50% in approximately 30 min, the patient required around 90 min, strongly suggesting the presence of gastroparesis (Fig. 1).

Results of 13C-acetate breath test using 150 g steamed rice prepared with 13C-sodium acetate. Black circle represents the average of 5 healthy individuals, and white circle represents the patient in this case. The time required to reach 50% of the breath CO2 concentration as 50% gastric emptying, with the 240-min value set as 100%. Time to 50% gastric emptying: healthy individuals (mean around 30 min), and the patient (around 90 min).
Switching from rapid-acting insulin analog to regular human insulin, which has a slower onset and longer duration of action, modestly improved the timing mismatch between postprandial glycemic fluctuation and insulin action. However, TIR remained around 50%, and time above range (TAR, >180 mg/dL) exceeded 40%, primarily due to late postprandial hyperglycemia from the wanning effect of bolus insulin.
Given the persistent biphasic glycemic pattern associated with delayed gastric emptying, we hypothesized that setting higher carbohydrate-to-insulin ratios (CIRs) could help prevent early postprandial hypoglycemia by reducing prandial insulin dosing, while relying on the AHCL system’s automated correction boluses to address subsequent hyperglycemia. Based on this rationale, we initiated SAP therapy with an 780G AHCL system, using insulin aspart. To mitigate early hypoglycemia and enable effective automated correction of delayed glucose excursions, the CIRs were substantially increased—to 27 g/U before breakfast, 28 g/U before lunch, and 28 g/U before dinner—compared with 10 g/U, 12 g/U, and 15 g/U respectively, during MDI. In addition, the target glucose level was raised from 100 to 120 mg/dL to help prevent hypoglycemia due to excessive automated correction boluses. To assess the ability of the system to handle postprandial variabilities, we conducted meal tests using two distinct macronutrient compositions: high-carbohydrate (bread) and high-fat (curry with rice). The system responded appropriately in both cases: basal insulin was suspended during glycemic decline and automated correction boluses were delivered in response to late glycemic rises. Notably, the number and timing of correction boluses varied by meal type, demonstrating adaptive algorithmic responses to differing postprandial glycemic patterns (Fig. 2).
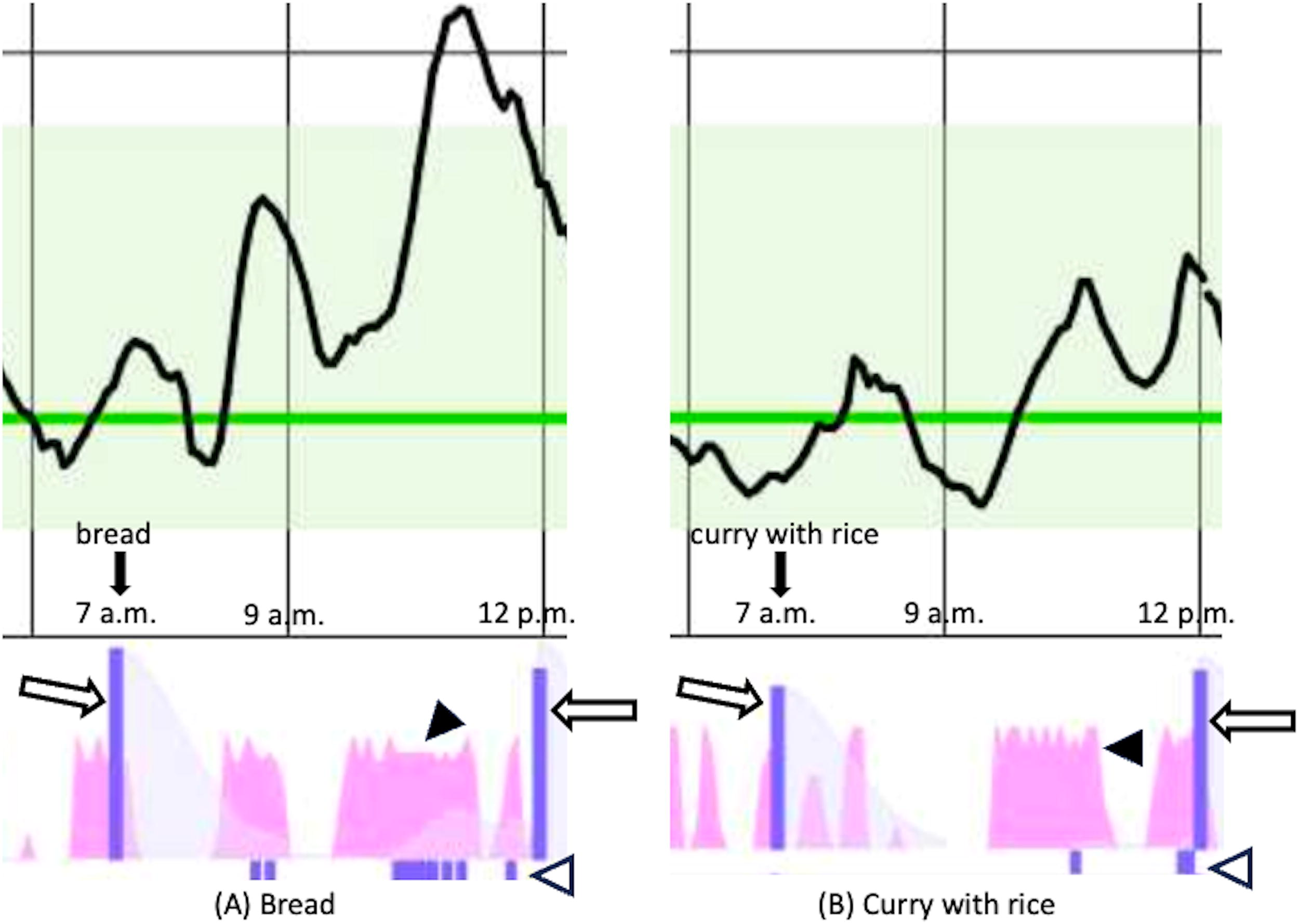
Postprandial glucose fluctuations and the response of basal rate and automatic correction boluses. Target sensor glucose level sets to a default of 100 mg/dL in this test (green line), and the light green shaded area represents the TIR range (sensor glucose levels between 70 and 180 mg/dL). White arrow indicates preprandial bolus administration; white arrowhead indicates automated correction bolus; black arrowhead indicates basal insulin infusion rate.
The 780G AHCL system was able to accommodate complex postprandial glycemic fluctuations, particularly by effectively mitigating postprandial hyperglycemia. Notably, it facilitated an appropriate decline in glucose levels after dinner through automated correction, thereby preventing both nocturnal hypoglycemia and the dawn phenomenon, and achieving a stable, flat overnight glycemic profile. Glycemic management improved substantially. TIR increased from 50% to 88%, and time below range (TBR, <70 mg/dL) declined from 3% to <1%. This case highlights the potential of 780G AHCL systems to manage the complex and unpredictable glycemic dynamics characteristic of gastroparesis.
Discussion
In individuals with gastroparesis, the mismatch between the timing of nutrient absorption and insulin action makes glycemic management challenging, often leading to early postprandial hypoglycemia and late postprandial hyperglycemia. 4 To improve postprandial glycemic excursions, administering bolus insulin 15–20 min before meals is one possible approach. 11 However, in individuals with gastroparesis, the variability in gastric emptying time increases the risk of early postprandial hypoglycemia, making this approach often ineffective. A common approach for glycemic management in individuals with gastroparesis involves the use of a regular insulin with a relatively slow onset of action, along with frequent small meals. However, this method can be quite cumbersome and burdensome for the individuals involved. 12 The use of CGM and CSII has enabled more detailed monitoring of glucose trends, making it easier to administer insulin in accordance with glucose absorption. A study conducted on individuals with gastroparesis, regardless of the use of CGM, demonstrated that CSII therapy improved glycemic trends and reduced glucose variability compared with MDI regimens.4,7
In addition, a case series using SAP therapy with HCL systems in individuals with gastroparesis showed that the TIR increased from 26.0% ± 15.7% to 58.4% ± 8.6%, without increasing hypoglycemic events, demonstrating the efficacy of HCL. 5 AHCL insulin pumps represent the latest innovation in CSII technology, incorporating artificial intelligence and machine learning algorithms to predict individual glycemic trends and automatically deliver correction boluses in response to dynamic glucose changes. These systems are expected to offer superior adaptability to complex glycemic patterns, such as those observed in individuals with gastroparesis. While AHCL systems are theoretically well-suited to manage complex postprandial glycemic fluctuations through automated correction boluses, no previous reports have demonstrated their effectiveness in this context, particularly in real-world cases such as gastroparesis. With the introduction of SAP therapy, we achieved a significant improvement in the glycemic profile, with a TIR of 88% and a TBR of <1%, without causing hypoglycemia. Notably, here we demonstrated that the 780G AHCL system can effectively adapt to complex postprandial glycemic fluctuations in gastroparesis. By using two meals with distinctly different macronutrient compositions, we showed that the system appropriately suspended basal insulin and delivered automated correction boluses in a timely manner, thereby minimizing hypoglycemia and promoting glucose levels toward the target range. From a practical standpoint, two key strategies emerged: setting relatively high CIRs to avoid early hypoglycemia and slightly increasing the target glucose level to reduce the risk of excessive correction boluses. These adjustments allowed the algorithm to function more efficiently in the setting of delayed and variable gastric emptying. Importantly, these improvements were achieved without placing additional burden on the individual, as most corrective actions were carried out by the algorithm in the background. This level of automation may be especially beneficial for individuals with complex glycemic profiles, including those with gastroparesis, who often struggle to predict glycemic responses to meals.
The present case underscores that SAP therapy with the 780G AHCL system can be a highly effective approach for individuals with diabetes complicated by gastroparesis, where glycemic management is challenging. While this is a single case, the findings suggest that AHCL technology may offer a significant advancement in the management of diabetes with gastroparesis, warranting further investigation.
Footnotes
Acknowledgments
The authors are grateful to the members of the Center for Diabetes, Endocrinology and Metabolism, who engaged in fruitful discussion. The authors also thank the patient who participated in this study.
Authors’ Contributions
All authors made individual contributions to authorship. All authors were involved in the diagnosis and management of this patient. M.M. and Y.Y. were involved in writing the first draft and submission. M.M., K.K., M.K., M.I., H.K., and Y.Y. contributed to the design and analysis of the 13C-acetate breath test. K.K., M.K., M.I., H.K., Y.Y., Y.H., and Y.S. critically revised the article with important intellectual content. All authors reviewed and approved the final draft.
Author Disclosure Statement
There are no conflicts of interest to declare.
Funding Information
No public or commercial funding was provided for this study.
Informed Patient Consent for Publication
Signed informed consent was obtained directly from the patient.
Ethics
This study received approval from the Ethics Committee of Kansai Electric Power Hospital (25-011) and was conducted in accordance with the Declaration of Helsinki and the Ethical Guidelines for Medical and Health Research Involving Human Subjects issued by the Japan Ministry of Education, Culture, Sports, Science and Technology, and the Ministry of Health, Labour and Welfare.
Data Availability Statement
Some or all datasets generated or analyzed during the current study are not publicly available but are available from the corresponding author on reasonable request. The resources are available on request.
