Abstract
Background:
Oxidative stress is crucial for the morbidity of diabetic complications. An abnormal metabolic milieu as a result of hyperglycemia leads to the onset of several complications, including diabetic retinopathy (DR). Treating eye diseases like DR is challenging due to the eye’s anatomy, physiology, and biochemistry. A biological antioxidant capable of inhibiting oxidative/nitrosative stress-mediated diabetic progression during hyperglycemia is still needed. We investigated the role of silica-coated gold nanoparticles (AuNP@SiO2) on high-glucose (HG)-induced oxidative stress, mitochondrial dysfunction, apoptosis, inflammation, and neurodegeneration in rat retinal ganglion cells (RGC-5).
Methodology:
Using advanced techniques, we investigated the profound inhibitory effect of AuNP@SiO2 in RGC-5 cells in the presence of a HG environment.
Results:
Overproduction of reactive oxygen species (ROS) and reactive nitrogen species (RNS) in the HG environment weakened the endogenous antioxidants, including catalase, superoxide dismutase, and glutathione peroxidase and AuNP@SiO2 treatment restored the HG effect. Furthermore, excess accumulation of ROS/RNS was allied with mitochondrial dysfunction, characterized by loss of mitochondrial membrane potential (ΔΨm) and increased mitochondrial mass in the HG environment, which Au@SiO2 nanoparticles (NPs) attenuated in RGC-5 cells. Moreover, the imbalance between the generation of ROS/RNS and their corresponding weakened endogenous antioxidant defense system leads to biomolecule damage, downregulation of 8-oxo guanine glycosylase, and accumulation of deoxyguanosine (8-OHdG) in DNA. However, treatment of AuNP@SiO2 attenuated the action. To broaden the knowledge, we further investigated the influence of AuNP@SiO2 on HG-induced increased expression of inflammatory mediators and neurodegenerative markers, leading to apoptosis, which was attenuated by Au@SiO2 NPs in RGC-5 cells.
Conclusion:
In summary, AuNP@SiO2 represents a potentially promising agent for facilitating defense against HG-induced oxidative/nitrosative stress, antioxidant depletion, mitochondrial dysfunction, apoptosis, and inflammation, thus generating a sustained control over ROS/RNS, which consequently evoke the potential of AuNP@SiO2 as an economic therapeutic remedy in diabetic treatments and its complications.

Introduction
Diabetes is a metabolic disorder of increasing occurrence and clinical relevance, contributing to high morbidity and mortality rates. 1 It has reached epidemic proportions among the challenging, unresolved health problems of the 21st century. Around 230 million people worldwide have been affected by diabetes, and around 367 million people are expected to be affected by 2030. Sustained hyperglycemia produces multiple biochemical sequelae, and diabetes-induced oxidative/nitrosative stress could play a role in the onset and progression of diabetes.2,3 Retinopathy is one of the most severe ocular complications of diabetes and is a leading cause of acquired blindness in young adults. Several pathogenic pathways are activated in diabetes, among which reactive oxygen species (ROS) and reactive nitrogen species (RNS) generated by high-glucose (HG) environments are responsible for metabolic abnormalities and chronic complications.4–6 Recently, studies have reported the biosynthesis and characterization of silica-coated gold nanoparticles (AuNP@SiO2) and their applications in cell culture models.2,7
The complex network of neuronal cells in the eye makes it a potential target of neuronal toxicity in diabetic conditions and a risk factor for visual loss. 8 Metal nanoparticles (NPs), such as gold and silver, have gained much attention in the ophthalmology community due to their potential to cross the barriers of the eye. 9 With the growing use of NPs in commercial and medical applications, including ophthalmology, there is a need for a reliable early prediction of the essential activity of AuNP@SiO2 on HG-induced ROS/RNS in rat retinal ganglion cells (RGC-5). 10 Direct glucose toxicity in the neurons usually occurs with increased free radicals or impaired antioxidant defense production.3,11 However, under diabetic conditions, glucose is prone to oxidation, resulting in the generation of hydrogen peroxide (H2O2), nitric oxide (NO), and reactive intermediates such as the hydroxyl radical (·OH), the most reactive and toxic of free radicals. 12 In addition, a simultaneous flux of NO and superoxide anion (O2•−) overproduction in each system with proximity to one another leads to the formation of peroxynitrite (ONOO−) in a diffusion-controlled fashion.13,14
The primary enzymatic defense mechanisms against the damage caused by free radicals are superoxide dismutase (SOD), glutathione peroxidase (GPx), and catalase (CAT). 6 Hyperglycemia, a hallmark of a diabetic condition, depletes natural antioxidants and facilitates the production of ROS/RNS. Excess production of ROS and RNS can react with all biological molecules like lipids, proteins, carbohydrates, and DNA and exert cytotoxic effects on cellular components in the retina.4,5,15 Furthermore, oxidative/nitrosative stress-induced cell death involves mitochondria in a primary role and is considered one of the major sources of cellular ROS. 16 Mitochondrial antioxidants MnSOD and CuZnSOD act as ROS detoxifiers. 17 However, an overproduction of ROS/RNS generation can overwhelm the defense ability, resulting in mitochondrial damage and generating oxidized proteins, leading to mitochondrial dysfunction by damaging its ΔΨm and mitochondrial mass. 18 Previously, our studies have revealed that reactive oxidant intermediates can trigger mitochondria to release cytochrome c, activating caspases and eventually leading to cell apoptosis.5,19 On the other hand, ROS/RNS damages the DNA. It initiates a vicious cycle leading to decreased transcription and protein synthesis, and this is further exacerbated due to a subsequent decrease in antioxidant defense. 20 Mitochondria utilize base excision repair (BER) as the primary DNA repair mechanism for genomic maintenance. Glycosylases, including 8-oxo guanine DNA glycosylase (OGG1), recognize and remove 8-hydroxy-2’-deoxyguanosine (8-OHdG), an indicator of oxidative damage to DNA. 21 Although it is reasonable to accept diabetes as a trigger for vascular inflammation, substantial evidence has shown that low-grade inflammation is an essential pathogenetic determinant of diabetes.3,22 Experimental observations have demonstrated the central mechanistic relevance of several cytokines and chemokine networks in diverse cell types, including HG-exposed RGC-5 cells. Treating eye diseases is challenging due to the complex anatomy, physiology, and biochemistry of the eye. A biological antioxidant capable of inhibiting oxidative/nitrosative stress-mediated diabetic progression during hyperglycemia is still needed. We investigated the role of AuNP@SiO2 on HG-induced oxidative stress, mitochondrial dysfunction, apoptosis, inflammation, and neurodegeneration in RGC-5.
Materials and Methods
Reagents and chemicals
Fetal bovine serum (FBS, Catalog Number: 10270106) and Dulbecco’s modified Eagle’s medium (DMEM, Catalog Number: 11885084) were purchased from GIBCO (Invitrogen, Thermo Fisher Scientific Inc., USA). Dihydroethidium (DHE, Catalog Number: D11347; Invitrogen, Thermo Fisher Scientific Inc., USA), dihydrorhodamine 123 (DHR, Catalog Number: D23806; Invitrogen, Thermo Fisher Scientific Inc., USA), JC-1 dye (Catalog Number: T3168; Invitrogen, Thermo Fisher Scientific Inc., USA), MitoSOX red (Catalog Number: M36008; Invitrogen, Thermo Fisher Scientific Inc., USA), and Mitotracker green FM (Catalog Number: M46750; Invitrogen, Thermo Fisher Scientific Inc., USA) were purchased from Molecular Probes (Eugene, USA). Propidium iodide (PI, Catalog Number: P1304MP; Invitrogen, Thermo Fisher Scientific Inc., USA), 4,6-diamidino-2-phenylindole (DAPI; Catalog Number: 62248; Invitrogen, Thermo Fisher Scientific Inc., USA), and 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazoliumbromide (MTT) were purchased from Sigma-Aldrich (Catalog Number: CT01-5; St Louis, USA). Neutral red (Catalog Number: SKU: TC448; Himedia, India), L-glutamine (Catalog Number: 25030081; Invitrogen, Thermo Fisher Scientific Inc., USA), phenol: chloroform: isoamyl alcohol solution (Catalog Number: MB078), and antibiotic were obtained from Himedia, Mumbai, India. Antibodies against OGG1 (Catalog Number: NB100-61664; Novus Biologicals, USA) and 8-OHdG (Catalog Number: Ab145594) were purchased from Abcam (UK). D-glucose (Catalog Number: G8769) and regular laboratory reagents were purchased from Sigma–Aldrich (USA).2–6
AuNP@SiO2 synthesis and characterization
As reported in our earlier work, the Au@SiO2 core-shell NPs were synthesized using the two-step approach. 7 Gold nanoparticles (AuNPs) were first prepared in the presence of cetyltrimethylammonium bromide (CTAB, Catalog Number: MB101), a cationic surfactant, followed by uniform silica coating on the AuNPs surface. In brief, 50 mL of 5 mM CTAB solution was adjusted to pH ∼10.5 with dilute NaOH solution and then maintained at 80°C using a water bath. The reaction mixture was equilibrated with constant magnetic stirring at that temperature for 15 min. Then, 2 mL of 3.7 wt.% formaldehyde solution was added to the above mixture. This was followed by adding 1.6 mL of 0.05 M HAuCl4 (chloroauric acid, Catalog Number: 16903-35-8, Merck, USA) solution under constant magnetic stirring. After some time, a wine-red colored solution appeared due to the formation of CTAB-capped AuNPs, and the reaction continued for 20 min. The resultant AuNPs were purified by centrifugation at 13,000 rpm and then redispersed in 50 mL Milli-Q water for further silica coating. To the above AuNPs dispersion, slowly added about 1.6 mL of 25% ethanolic solution of tetraethyl orthosilicate (TEOS, Catalog Number: 131903, Merck, USA) with gentle magnetic stirring for the whole night, which resulted in a milky appearance to the reaction mixture due to the formation of Au@SiO2 core-shell NPs. This was then purified by centrifugation, followed by three cycles of washing to remove unreacted products, and then redispersed in 50 mL Milli-Q water, the concentration of which was estimated based on the concentration of AuNPs before the silica coating. UV-vis absorption spectra of the synthesized AuNP@SiO2 were measured in an Agilent Cary 60 spectrophotometer (Agilent Cary 60 UV-Vis; USA). For this, a dilute solution of the NPs was taken in a quartz cuvette, and their absorption spectra were measured. The size analysis was carried out using transmission electron microscopy (TEM) images (Hitachi H-7650 TEM; Elk Grove Village, IL, USA) of the particles using ImageJ software. 2 For this, a dilute solution of the NPs was drop-cast on a carbon-coated copper grid and observed under TEM, which was followed by an analysis of the size and their polydispersity.
Cell culture
The RGC-5 cells were a kind gift from Dr. Jackson James, Rajiv Gandhi Center for Biotechnology, India, with prior consent from Dr. Neeraj Agarwal, National Eye Institute (NIH), USA. Cells were maintained in DMEM supplemented with 10% heat-inactivated FBS at 37°C in a humidified atmosphere with 5% CO2. For experiments, cells were incubated with 25 mM additional D-glucose followed by various concentrations of AuNP@SiO2 (0.005, 0.01, 0.025, 0.05, 0.1 nM) for 24 h, after getting 70–80% confluency in different plates, such as 96-well plates, 6-well plates, 60 mm plates, and 100 mm plates as per the experimental requirement.
Measurement of intracellular and mitochondrial ROS generation
The generation of intracellular ROS was detected using the nitro tetrazolium blue (NBT, Catalog Number: 11585029001, Merck, USA) reduction assay. 23 Briefly, cells were plated in tissue culture plates, treated with 0.025 nM of AuNP@SiO2 with/without HG (25 mM), and incubated for 24 h. After treatment, cells were harvested and suspended in NBT for 1 h. After centrifugation (1 min at 12,000 g), 50% (v/v) acetic acid was added to the cell pellet for NBT solubilization. Later, the absorbance of the supernatant was determined at 595 nm in a BioTek Epoch 96-well plate reader. The hydroxyl radical (·OH) level was determined as previously described by Kumar et al. 19 Briefly, treated cells were harvested, and 80 g protein from cell lysate was mixed with 700 μL of phosphate-buffered saline (PBS) solutions (2 mM sodium salicylate, 0.1 M EDTA, 40 μL 10 N HCl, and 0.25 g NaCl). An equal volume of ice-cold diethyl ether was added and incubated at 25°C for 30 min; absorbance was recorded at 510 nm. Furthermore, intracellular and mitochondrial superoxide production in RGC-5 cells was measured using DHE and MitoSOX dye, respectively.5,24 Briefly, seeded cells were exposed to 0.025 nM of AuNP@SiO2 with/without 25 mM of glucose for 24 h. Cells were first harvested and washed thrice with 1X PBS. Formerly, they were loaded with 5 μM DHE and MitoSOX solution at 37°C for 30 min. Then, 10,000 events per sample were examined by a fluorescence-activated cell sorting (FACS) analyzer (Becton Dickinson, San Jose, CA, USA).
Measurement of intracellular RNS
Nitric oxide production was determined by measuring the concentration of total nitrite in the sample. 25 Briefly, 100 μL of culture media was mixed with 100 μL Griess reagent (Catalog Number: G4410; Merck, USA), and after 15 min of incubation, absorbance was taken at 520 nm. Next, intracellular ONOO production was measured using the DHR dye. Cells treated with 0.025 nM of AuNP@SiO2 with/without 25 mM of glucose were harvested, washed in 1X PBS, and loaded with DHR (5 μM) solution at 37°C for half an hour. Cells were sorted using FACS, and 10,000 cells were examined for each sample.
Measurement of antioxidant enzymes
SOD, CAT, and GPx enzyme activity was determined in samples as described earlier.6,23 Briefly, cells were exposed to 0.025 nM of AuNP@SiO2 with/without 25 mM of HG after attaining 70–80% confluency. After 24 h, cells were harvested and centrifuged at 1,800 rpm. For the CAT assay, 80 μg of protein was added to a medium containing 50 mM phosphate buffer. This method is based on the rate of H2O2 degradation by the action of CAT. The kinetic analysis of CAT was measured spectrophotometrically at 240 nm after adding freshly prepared 30 mM H2O2. The results were expressed in M cat/mg. SOD activity in cell lysates was measured by spectrophotometer assay. Proteins were mixed with the reaction mixture (100 μM tris buffer, 20 μM L-methionine, 1% Triton X-100, 100 μM EDTA, 10 μM hydroxylamine hydrochloride) and 80 μL of riboflavin. Eventually, 1 mL of Griess’ reagent was added to the above mixture, and the absorbance was measured at 540 nm. GPx activity was measured using Subramanian method 26 with slight modification. Briefly, the sample protein was mixed with the reaction mixture containing 10 mM sodium azide, 0.8 mM EDTA, 2.5 mM H2O2, reduced glutathione, 0.4 M phosphate buffer (pH 7.0), followed by the addition of 0.5 mL of trichloroacetic acid (10%) to stop the reaction. The supernatant was taken out in a fresh tube, and 3 mL of disodium hydrogen phosphate (0.3 mM) and 0.04% 5,5′-dithiol-bis-(2-nitrobenzoic acid) were added, and immediately absorbance was recorded at 420 nm.
Measurement of mitochondrial membrane potential and mass
The mitochondrial membrane potential (ΔΨm) was measured by a flow cytometer using the lipophilic cationic fluorescent JC-1 dye as described previously. 20 The accumulation of JC-1 leads to the formation of J-aggregates (with a specific red fluorescence emission maximum at 590 nm), in addition to the typical green fluorescence of J-monomers (emission maximum of ∼525 nm). The lack of ΔΨm leads to the depression of JC-1 mitochondrial accumulation and a decrease in J-aggregate formation. 27 After three washes with 1X PBS, treated cells were harvested, washed with 1X PBS, diluted to 1 mL, then incubated with JC-1 dye (5 μg/ml) for 20 min at 37°C in the dark. Samples were washed with 1X PBS and analyzed immediately by FACS analyzer. Mitochondrial mass was monitored by using Mitotracker Green FM dye. 28 Briefly, treated cells were harvested and washed thrice with ice-cold 1X PBS, then incubated with Mitotracker Green FM (200 nM) for 20 min at 37°C in the dark. After incubation, cells were washed with 1X PBS and analyzed by the FACS analyzer.
Measurement of biomolecule damage
ROS/RNS are highly reactive molecules containing oxygen/nitrogen with unpaired electrons, which can lead to lipid peroxidation (LPO). The LPO level was measured as described by Sagar et al. 29 A reaction mixture was prepared with 100 μL of protein samples and incubated for 1 h at 37°C. Thiobarbituric acid [(0.375%, Catalog Number: 1.08180; Merck, USA) was then added and incubated in a boiling water bath for 15 min, followed by centrifugation at 800 rpm, and the absorbance was measured at 532 nm. For protein carbonylation (PC) measurement, 80 μg of protein samples were mixed with 450 μL of buffer, 100 μL of 10% streptomycin sulfate (Catalog Number: 1623003; Merck, USA), and centrifuged at 1,000 rpm for 10 min. Then, 400 μL of 10 mM DNPH (Catalog Number: 54278; Merck, USA) was mixed with the supernatant and kept in the dark for 30 min. After that, 20% ice-cold trichloroacetic acid was added and centrifuged at 1,000 rpm to stop the reaction. The pellet was washed carefully with absolute ethanol: Ethyl acetate solution (1:1). Finally, the pellet was dissolved in 6 M guanidine hydrochloride (Catalog Number: G3272; Merck, USA), and absorbance was measured at 366 nm. Comet assay in RGC-5 cells was performed as described earlier. 3 Cells were suspended in 0.5% agarose on comet slides. Next, slides were immersed in a lysis solution, followed by electrophoresis in the alkaline buffer. Then the slides were stained with PI and examined under a fluorescence microscope. The DNA ladder assay was carried out as described previously. 19
MTT assay and nuclear damage
MTT (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide) assay was used to detect the cell viability of RGC-5 cells. At first, ∼8,000–10,000 cells in triplicate were seeded into a 96-well plate and cultured overnight to attain 70–80% cell confluency. Furthermore, cells were exposed to various concentrations of AuNPs@SiO2 with/without HG for 24 h. Formerly, 100 μL MTT reagent (5 mg/mL) was added to each well, and the plate was incubated in the dark for 4 h. To dissolve the formazan crystal, MTT solution was added and incubated at 37°C in the dark conditions for 2 h. The optical density was recorded at 570 nm (BioTek Epoch Microplate Spectrophotometer, USA). For DAPI, cells were washed with 1XPBS after the desired treatment and fixed in 3.7% paraformaldehyde for 20 min. Then, cells were centrifuged at 2,000 rpm for 5 min before staining with 5 μg/mL DAPI solution, and apoptotic nuclei were observed under a fluorescence microscope.
Measurement of 8-OHdG
The expression of 8-OHdG was determined using ELISA (BioTek Epoch Microplate Spectrophotometer, USA). 23 In brief, cells were harvested after the desired treatment of 24 h, centrifuged, and the DNA was extracted. Then, the isolated DNA from cells was suspended in 20 mM sodium acetate and digested with nuclease P1. Next, 1 M Tris/HCl was added to each sample, followed by alkaline phosphatase treatment for 1 h at 37°C, and 8-OHdG in digested DNA was determined using ELISA.
Lysosomal staining
Lysosomal staining was carried out as described earlier by Mytych et al. 30 Briefly, after the desired treatment, neutral red (0.001%) was added and incubated for 3 h. The cells were then fixed in 0.5% formaldehyde in 1% CaCl2 solution for 20 min, washed with water, and taken to examination under a fluorescence microscope (FLoid Cell Imaging Station, Grand Island, USA).
RNA isolation and cDNA preparation
RGC-5 cells were harvested after the desired treatment and dissolved in TRIzol™ Reagent (Catalog Number:15596026; Invitrogen™, Thermo Fisher Scientific Inc., USA) for RNA extraction. Then 200 μL of chloroform was added per milliliter of TRIzol, the mixture was shaken vigorously for 10 s, incubated on ice for 10 min, and centrifuged at 10,000 g for 15 min at 4°C. Thereafter, samples were centrifuged, and the upper layer of supernatant was transferred to a fresh tube. The RNA was precipitated from the supernatant with isopropanol. The pellets were finally washed with 75% ethanol. The purity and concentration of RNA were evaluated using a NanoDrop ND-1000 Spectrophotometer (BioTek Epoch Microplate Spectrophotometer, USA). Reverse transcription was performed using the RevertAid First Strand cDNA Synthesis Kit (Catalog Number: K1622; Thermo Fisher Scientific Inc., USA), which, according to the manufacturer’s instructions, used oligo dT primers.
Quantitative real-time PCR
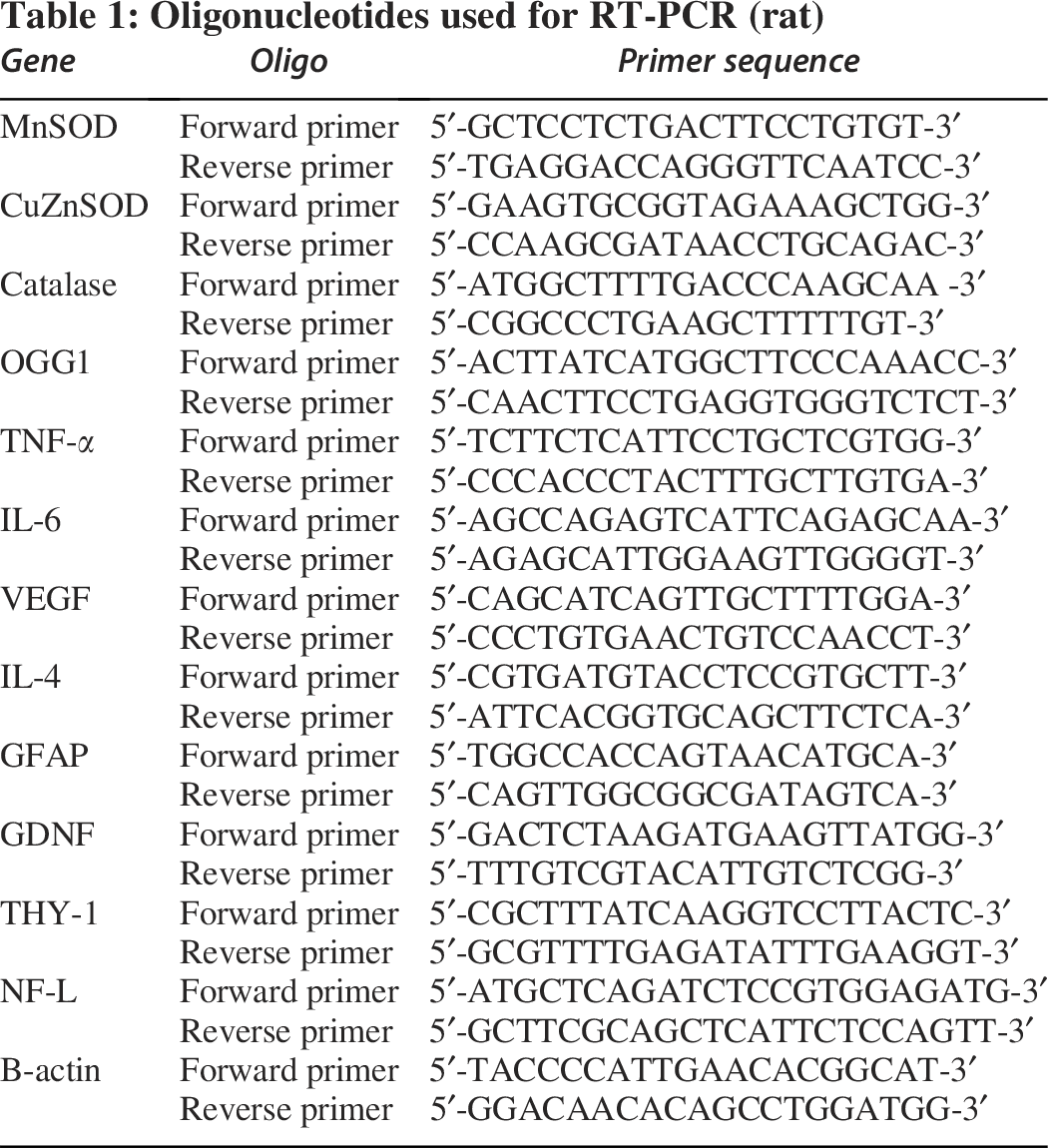
Total RNA from RGC-5 cells was isolated using the HiPurA® Total RNA Miniprep Purification Kit (SKU: MB602; Himedia, India) for mRNA quantification, and the RevertAid cDNA synthesis kit was used for cDNA preparation. Quantitative real-time PCR was performed with a CFX connect real-time system (BIO-RAD, USA) using the KAPA SYBR® FAST qPCR Kit (Catalog Number: KR0389-S – v3.20; KAPA Biosystems, Merck, USA). The 2−ΔΔCT method was used to quantify relative levels of target genes using oligonucleotides (Table 1).
Oligonucleotides used for RT-PCR (rat)
Data analysis and statistics
All the statistical analyses were performed using GraphPad Prism 8.0. A two-way analysis of variance (ANOVA) was used to evaluate the significance between the groups, followed by the students’ t-tests, as indicated in the figure legends. Two-way ANOVA was used to compare between control and treated group experiments. Values were calculated as mean ± SEM of at least three determinations, and p values ≤ 0.05 were considered significant.
Results
Synthesis and characterization of Au@SiO2 core-shell nanoparticles
The formation of AuNPs was demonstrated by the appearance of a ruby red color colloidal solution stabilized by CTAB micelles. A robust surface coating is necessary for biological studies to avoid the cytotoxicity of CTAB molecules and to provide better stability in a complex biological environment. Here we performed a thick silica coating on the CTAB-capped AuNPs, which also maintains the hydrophilicity of AuNPs, an utmost requirement for biological studies. Therefore, our approach is based on two consecutive steps: Colloidal synthesis of AuNPs and controlled silica coating using the modified Stӧber approach by hydrolyzing TEOS on CTAB-capped AuNPs. The resulting particles were first characterized by UV–vis absorption studies (Figure 1A). The spectra reveal a prominent surface plasmon band at 523 nm for CTAB-capped AuNPs and a red-shifted, broadened band at 527 nm after silica coating. The slight broadening and shifting of the plasmon band could be attributed to the change in the AuNPs surface environment after silica coating. A similar observation was made in our earlier work on AuNP@SiO2 and nanorods. The morphological evaluation of the resulting AuNPs was characterized by TEM, and a representative image is shown in Figure 1B, with the same scale bar for ease of comparison of AuNPs before and after silica coating. Size analysis of AuNPs and silica layer thickness was performed using ImageJ software, considering at least 50 particles. The average size of AuNPs was estimated to be 10 nm, and no change in size and morphology even after silica coating implies the practical significance of the coating process for a comparison study. Moreover, the uniform thickness of the silica layer on the AuNPs surface was estimated to be around 21 nm, accounting for the size of silica-coated AuNPs (Au NPs@SiO2) to be around 52 nm. The presence of such a thick silica layer provides hydrophilicity to CTAB-capped AuNPs and is sufficient to hide the cytotoxic CTAB molecules for potential use in a biological assay. The size evaluation by microscopic studies is in good accordance with their surface plasmon bands in UV–vis absorption studies. Furthermore, histogram analysis was performed to illustrate the AuNPs and Au NPs@SiO2 particle size distribution more comprehensively (Figure 1C). The size of AuNPs was calculated to be 10.5 ± 1.4 nm, and that of Au@SiO2 NPs was 52.4 ± 3.7 nm.
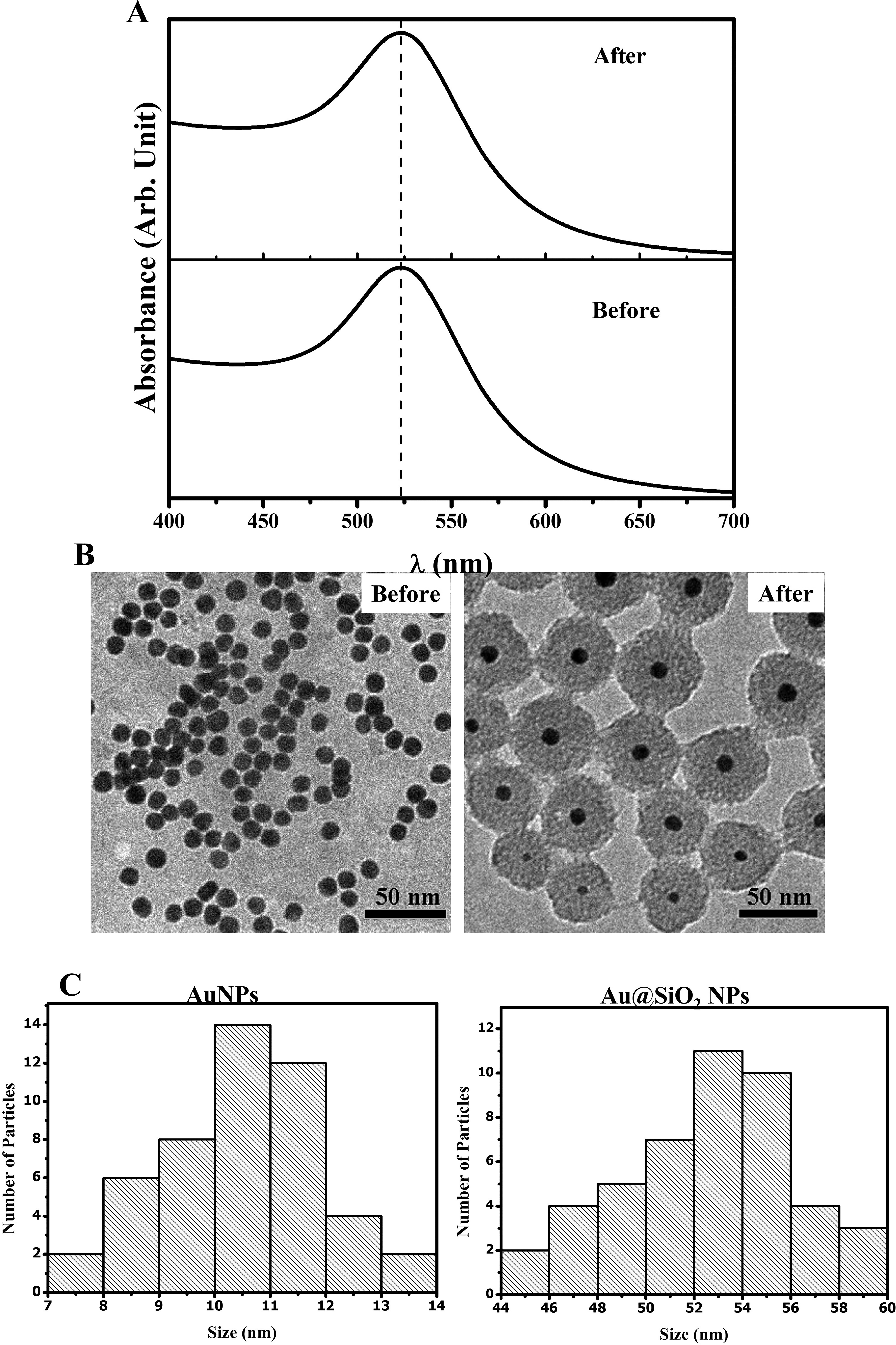
UV-vis absorption spectra and transmission electron microscopy (TEM) images of AuNPs before and after silica coating.
AuNP@SiO2 inhibits HG-induced ROS and RNS production in RGC-5 cells
ROS/RNS generated by the HG environment play a vital role in the development of diabetic complications. It results from the oxidative/nitrosative stress developed due to the release of free radicals. To check the intracellular ROS formation, cells were treated with 0.025 nM AuNP@SiO2 with/without HG (25 mM). Estimation of ROS was measured in terms of O2•− generation in RGC-5 cells. Figure 2A showed that HG significantly increased the level of O2•−, whereas AuNP@SiO2 attenuated the HG effect in RGC-5 cells. We measured the O2•− accumulation in cells by FACS analysis. The percent of fluorescent cells cultured in 25 mM HG increased from 3.8% to 24.7%, and exposure of cells pretreated with 0.025 nM AuNP@SiO2 led to a decrease in ROS-positive cells to 9.4% (Figure 2B). Next, we also measured •OH production in HG with/without AuNP@SiO2. An increased level of •OH in the HG environment was attenuated by 0.025 nM of AuNP@SiO2 in RGC-5 cells (Figure 2C, D). Byproducts of normal mitochondrial metabolism and homeostasis include the build-up of potentially damaging levels of ROS. So next, we determined mitochondria-derived ROS generation by using the mitochondrial-specific ROS indicator dye MitoSOX. The percentage of mitochondrial O2•− positive cells in HG was 7.5% and after AuNP@SiO2 treatment with HG, it reduced to 5.1%. The NO production significantly increased with 25 mM of HG, and an increase in the level of NO upon exposure to HG was attenuated by AuNP@SiO2 (Figure 2E). Furthermore, we followed the HG-induced ONOO− generation in RGC-5 cells cultured under 25 mM of HG, which show an increase in DHR fluorescence, and HG exposure with AuNP@SiO2 showed a decrease in expression (Figure 2F).
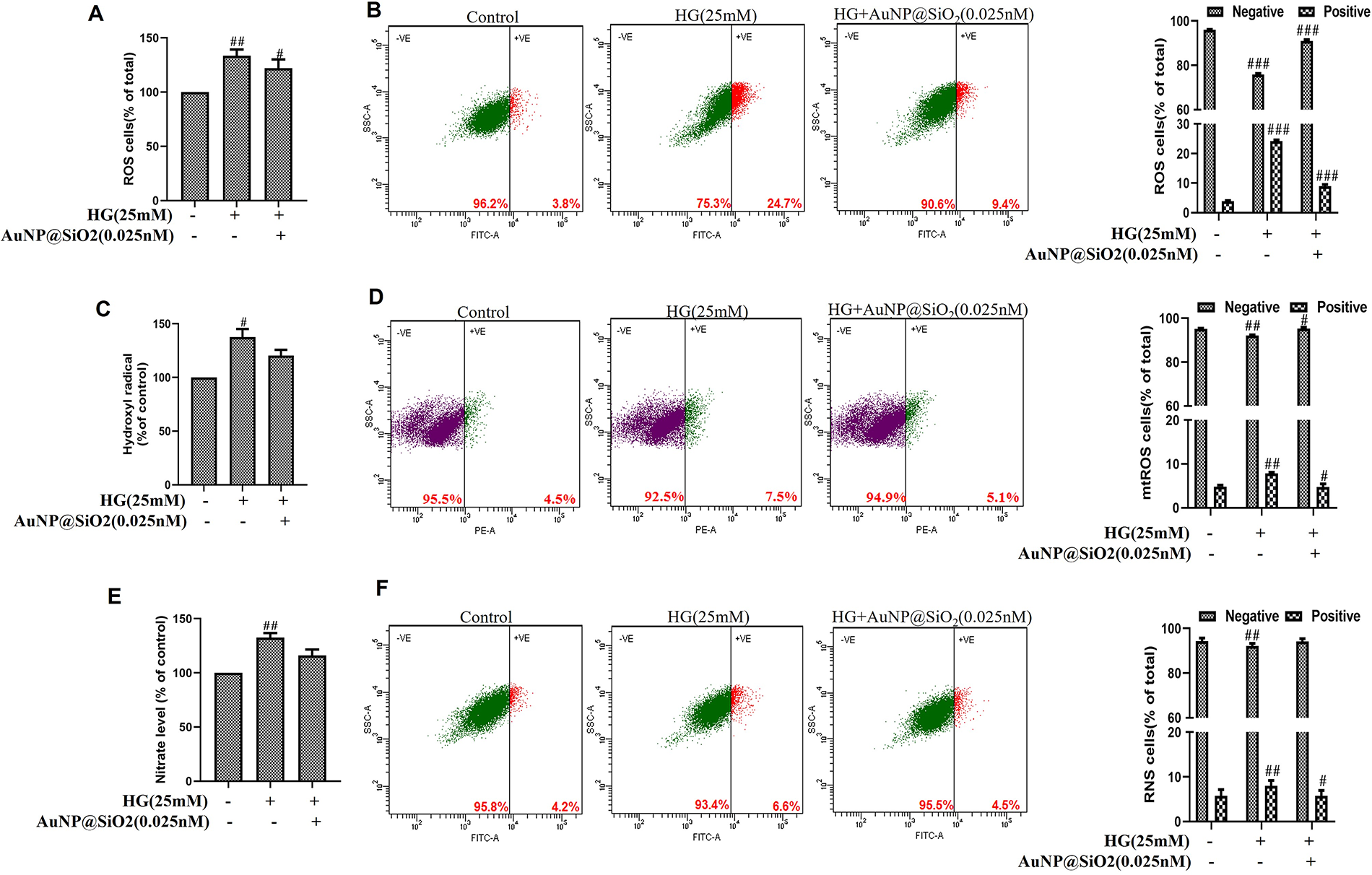
Silica-coated gold nanoparticles (AuNP@SiO2) inhibit high-glucose (HG)-induced reactive oxygen species (ROS) and reactive nitrogen species (RNS) production in RGC-5 cells.
AuNP@SiO2 restores HG-induced antioxidant depletion in RGC-5 cells
The effects of antioxidants on ROS/RNS are measured through specific observable biomarkers. These markers include the enzymatic activities of CAT, SOD, and GPx. Cells were cultured in 0.025 nM AuNP@SiO2 with/without HG (25 mM) to measure antioxidant levels. In initial experiments, the cellular levels of the SOD enzyme were significantly downregulated by 25 mM of HG (Figure 3A), and pretreatment of AuNP@SiO2 restored the enzyme activity. Furthermore, mitochondrial SOD (MnSOD) and cytosolic SOD (Cu-ZnSOD) antioxidant enzymes were evaluated to understand AuNP@SiO2 action on antioxidant levels. Figure 3B showed that 0.025 nM of AuNP@SiO2 restored the HG-induced increased fold of MnSOD and Cu-ZnSOD in RGC-5 cells. In HG-induced RGC-5 cells, it was observed that the level of antioxidant enzyme CAT was reduced, and a statistically significant (p < 0.05) increase in CAT level was seen with AuNP@SiO2 exposure (Figure 3C). In addition, we checked the mRNA expression of the CAT enzyme in HG-induced RGC-5 cells. It was observed that CAT expression significantly decreased (p < 0.05). However, pretreatment with 0.025 nM AuNP@SiO2 restored the antioxidant levels (Figure 3D). Similarly, the reduced GPx level in HG-exposed cells was restored with AuNP@SiO2 treatment in RGC-5 cells (Figure 3E).
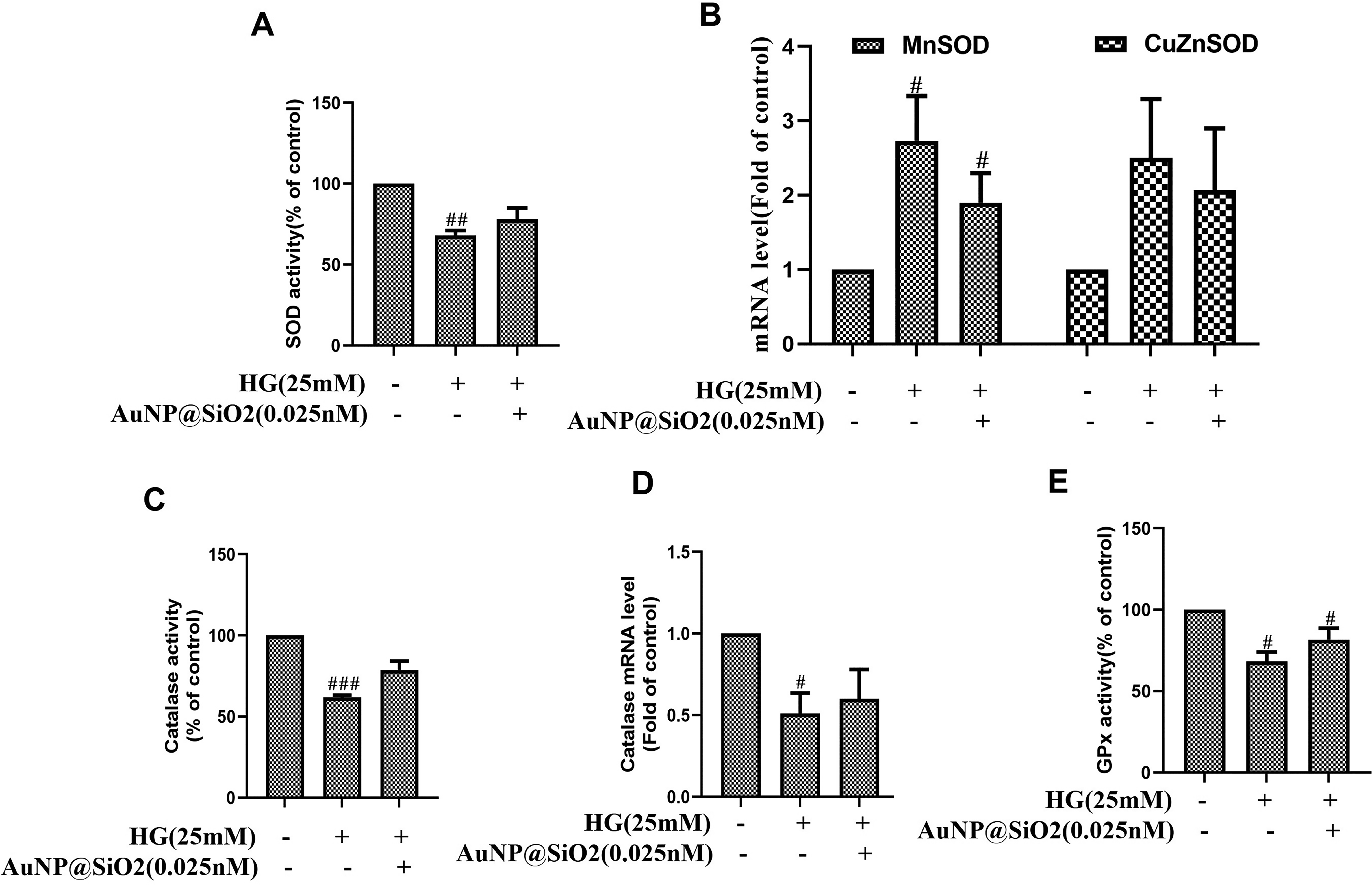
Silica-coated gold nanoparticles (AuNP@SiO2) restore high glucose (HG)-induced antioxidant depletion in RGC-5 cells.
AuNP@SiO2 protects HG-induced mitochondrial damage in RGC-5 cells
Mitochondrial dysfunction is defined as an alteration in membrane potential and mitochondrial number due to the accumulation of ROS/RNS in cells. JC-1, a cationic dye, emits red fluorescent J-aggregates at higher mitochondrial concentrations, reflecting higher mitochondrial potential, and green fluorescent J-monomers at lower mitochondrial concentrations, indicating lost membrane potential. The red/green fluorescence ratio is independent of the mitochondrial shape, density, or size, and it depends only on the membrane potential. To check the effect of AuNP@SiO2 on HG-induced alteration of mitochondrial ΔΨm, we used JC-1 dye in RGC-5 cells. As shown in Figure 4A, a decrease in ΔΨm was observed after treatment with 25 mM HG for 24 h, which was further attenuated by pretreatment of AuNP@SiO2 in RGC-5 cells. Next, we assessed the ability of Mito-Tracker Green FM Dye to measure mitochondrial mass under 0.025 nM AuNP@SiO2 with/without HG exposure. Notably, there was an increase in mitochondrial mass with the treatment of 25 mM HG (Figure 4B). More importantly, AuNP@SiO2 exposure caused a statistically significant decrease in mitochondrial mass within 24 h (p < 0.01).

Silica-coated gold nanoparticles (AuNP@SiO2) protect against high glucose (HG)-induced mitochondrial damage in RGC-5 cells.
AuNP@SiO2 inhibits HG-mediated apoptosis in RGC-5 cells
To investigate the proapoptotic role of AuNP@SiO2 on HG-induced RGC-5 cells, cells were cultured for the indicated durations in media containing 0.025 nM AuNP@SiO2 with/without an HG environment. Incubation in 25 mM of additional HG for 24 h exhibited a significant (p < 0.05) reduction in cell viability. Almost all cytotoxic effects of HG were abrogated by increasing the concentration of 0.025 nM of AuNP@SiO2 (Figure 5A). Nuclear staining of cells exposed to HG by DAPI displayed apoptotic nuclei. The number of apoptotic nuclei decreases with AuNP@SiO2 treatment (Figure 5B). Accumulating excessive ROS levels can lead to direct oxidative modification of proteins, lipids, carbohydrates, and DNA, resulting in tissue damage. An increased LPO level in HG treatment was significantly decreased after exposure to 0.025 nM of AuNP@SiO2 in RGC-5 cells (Figure 5C). After evaluating the effect of AuNP@SiO2 on RGC-5 cell viability, we also analyzed the changes in lysosome content by neutral red staining in HG-treated RGC-5 cells with AuNP@SiO2 exposure. As shown in Figure 5D, cells with HG show extra lysosomal staining compared with untreated cells and HG with AuNP@SiO2, inhibiting the effect in RGC-5 cells. Subsequently, we examined the PC level by 0.025 nM AuNP@SiO2 with/without HG treatment in RGC-5 cells. As shown in Figure 5E, increased PC was observed in HG-treated RGC-5 cells, and pretreatment of AuNP@SiO2 for 24 h attenuated the effect of HG. Comet assay showed that oxidative/nitrosative DNA damage, which is an inevitable consequence of cellular metabolism with a propensity for increased levels following toxic insult, was higher in HG-induced cells (Figure 5F). Pretreatment of RGC-5 cells with AuNP@SiO2 attenuated HG-induced DNA damage.
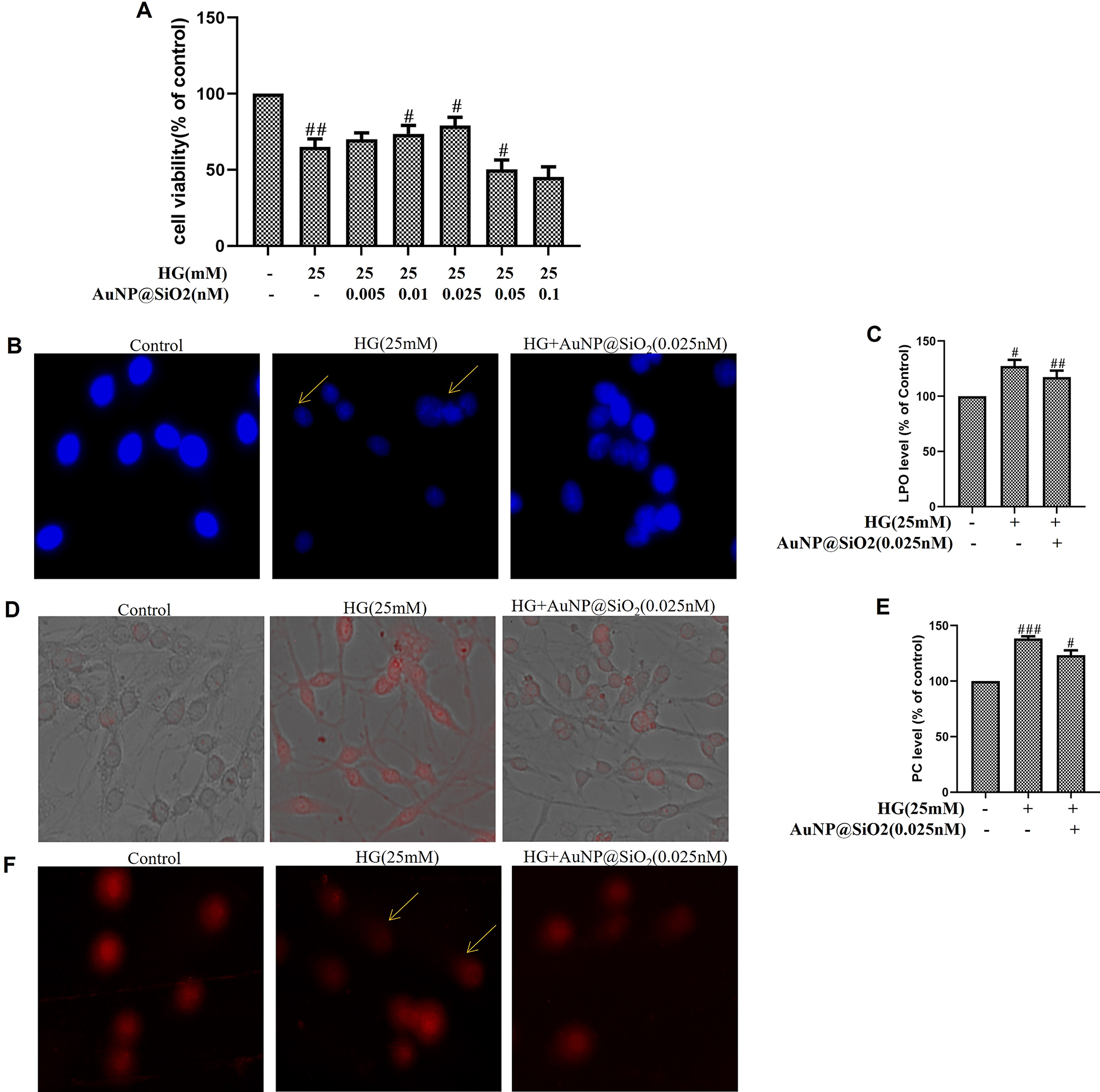
Silica-coated gold nanoparticles (AuNP@SiO2) inhibit high glucose (HG)-mediated biomolecule damage in RGC-5 cells.
AuNP@SiO2 inhibits HG-induced OGG1 downregulation and 8-OHdG accumulation in RGC-5 cells
OGG1 is the major DNA BER enzyme that recognizes and excises 8-oxodG. Therefore, we determined whether the change in OGG1 abundance influenced the accumulation of 8-oxodG in vitro. Figure 6A demonstrates a rapid increase in the downregulation of OGG1 level in HG-induced RGC-5 cells. Furthermore, we incubated cells with AuNP@SiO2 in RGC-5 cells before HG exposure and observed that AuNP@SiO2 exposure attenuated the effect of HG. qRT-PCR was conducted to confirm whether a drop in OGG1 could be attributed to lower transcription of OGG1 mRNA. Figure 6B shows that HG inhibited OGG1 expression and the damage was recovered by AuNP@SiO2 treatment in RGC-5 cells. Free radicals can attack guanosine residues of DNA and form 8-OHdG, which may adversely affect transcription of many genes. Hence, we investigated the 8-OHdG accumulations in cells with 0.025 nM AuNP@SiO2 with/without HG treatment. 8-OHdG accumulation increased with 25 mM of additional HG for 24 h, and treatment of cells with AuNP@SiO2 attenuated the effect of HG (Figure 6C). To confirm the impact of AuNP@SiO2 on 8-OHdG in the presence or absence of HG, we also performed an ELISA. The findings demonstrate the inhibitory effect of AuNP@SiO2 on HG in RGC-5 cells (Figure 6D).
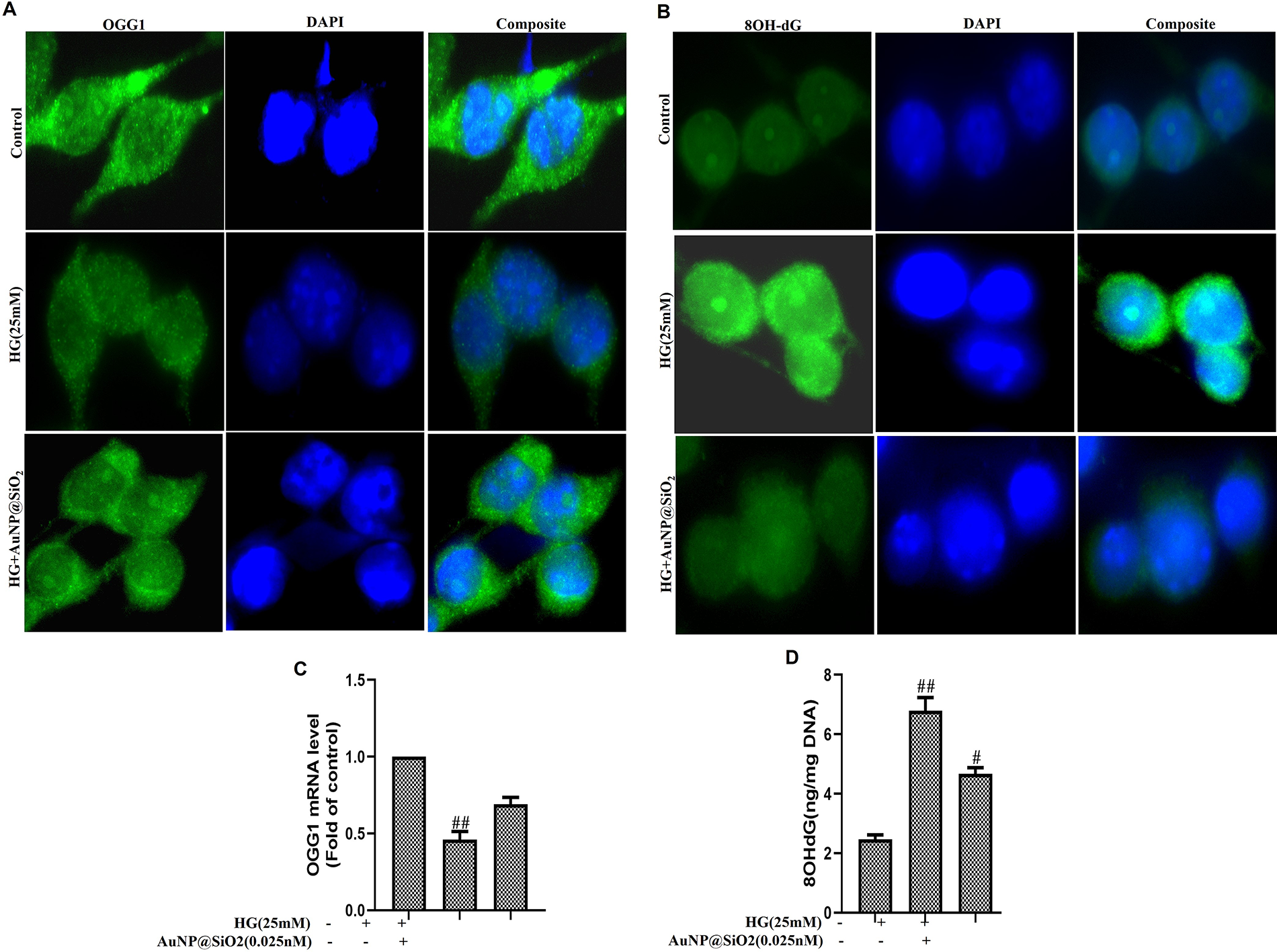
Silica-coated gold nanoparticles (AuNP@SiO2) inhibit high glucose (HG)-induced OGG1 downregulation and 8-OHdG accumulation in RGC-5 cells.
AuNP@SiO2 inhibits HG-induced inflammation and neurodegeneration in RGC-5 cells
Many studies have reported in different cell types that HG-induced inflammation involves oxidative/nitrosative stress. Therefore, we have evaluated the AuNP@SiO2 inhibition of inflammation in RGC-5 cells exposed to HG. In the HG environment, inflammatory marker generation was increased compared with control cells. The presence of AuNP@SiO2 further decreases HG-induced inflammation (Figure 7A-D). Hyperglycemia in diabetic brain and HG-treated neuronal cells significantly altered the mRNA expression of several neuromarkers, including GFAP, GDNF, Thy-1, and NF-L, compared with control (Figure 7E-H). Pretreatment of RGC-5 cells with AuNP@SiO2 reversed the HG-induced alteration in neuromarkers.

Silica-coated gold nanoparticles (AuNP@SiO2) inhibit high glucose (HG)-induced inflammation and neurodegeneration in RGC-5 cells.
Discussion
Chronic or intermittent hyperglycemia is associated with the development of diabetic complications. 31 The complications of diabetes predominantly relate to vascular damage of the microvasculature in the retina, resulting in diabetic retinopathy (DR). 32 Oxidative/nitrosative stress is a general term that mainly describes the steady state level of oxidative damage in a cell, tissue, or organ caused by ROS/RNS.19,33 Evidence has been reported that ROS/RNS toxicity remains an undisputed cause and link between diabetes and DR.5,34 Although, in normal conditions, ROS/RNS act as cellular messengers in redox signaling, increased ROS/RNS disrupt standard cellular mechanisms via antioxidant depletion, DNA damage, activation of the signaling pathway, and apoptosis.6,35 Thus, an effective and economical therapeutic molecule capable of delivering the treatments for diabetes by controlling the oxidative/nitrosative stress induced by hyperglycemia and thereby preventing the onset of complications is still needed. Gold NPs, an emerging nanomedicine, are renowned for their promising therapeutic possibilities due to their significant properties against hyperglycemic and inflammatory responses.9,36 The preliminary objective of this study was to demonstrate the nontoxic properties of AuNP@SiO2. We have demonstrated that the treatment of AuNP@SiO2 conquered HG-induced increased oxidative/nitrosative stress in RGC-5 cells. We have also presented evidence that pretreatment of AuNP@SiO2 causes a dramatic decrease in the release of inflammatory mediators.
Many studies have shown that Au@SiO2 systems are appropriate intrants as surface-enhanced resonance Raman scattering encoded particles for their use in biolabeling and biomedical imaging.2,37 In our study, the typical yield of AuNP@SiO2 was assumed to be 100%, based on the initial precursor concentration and the final recovered product. Reaction efficiency was evaluated by monitoring the complete silica coating on gold NPs, which was confirmed via UV–Vis spectroscopy and TEM analysis. Slight variations in particle size and uniformity across different batches are common in NP synthesis, which may arise due to fluctuations in reaction parameters, precursor concentration, mixing efficiency, etc. To minimize these variations, utmost percussions were taken care, like precise control of reaction parameters, use of freshly prepared reagents and real-time spectroscopic monitoring of reaction progress, etc. Therefore, optimizing reaction parameters were critical to enhance reproducibility.
Furthermore, in our study, after confirmation of the nontoxic nature of the AuNP@SiO2, the effect of AuNP@SiO2 over the oxidative/nitrosative stress induced at hyperglycemic conditions was investigated. Auspiciously, our data showed a significant decrease in ROS and RNS levels by a 0.025 nM dose of AuNP@SiO2 in RGC-5 cells. The ONOO− is a potent oxidant and nitrating agent generated by NO and O2• reaction in stress conditions. In this study, we demonstrate that a hyperglycemia-induced increase in NO and ONOO− in RGC-5 cells was attenuated by AuNP@SiO2 treatment. Furthermore, cells are equipped with various antioxidants that counterbalance the effect of oxidants and provide a first line of defense, preventing free radical-mediated cell damage. 23 The antioxidants CAT, SOD, and GPx are detoxifying enzymes that counteract oxidative/nitrosative stress. Since superoxide is the primary ROS produced from a variety of sources, its dismutation by SOD is of primary importance for each cell. SOD isoforms MnSOD and CuZnSOD are enzymes that neutralize toxic O2•− to water and less poisonous H2O2 and prevent ONOO− formation from O2•− and NO. CAT hydrolyzes H2O2, and GPx reduces organic hydroperoxides in different types of cells.38–40 However, the levels of CAT, SOD, and GPx decreased with 25 mM of glucose, and the levels of free radicals increased in the presence of AuNP@SiO2.
The mitochondrial respiratory chain is the primary site of ROS/RNS production because of unavoidable oxidative phosphorylation. Formation of mitochondrial ROS due to hyperglycemia should be either scavenged or reduced into metabolically less harmful molecules to protect cells. Here, we have demonstrated that increased mitochondrial ROS was attenuated by pretreatment with AuNP@SiO2. It was also observed that expression of MnSOD and CuZnSOD increased in RGC-5 cells after HG exposure and gradually decreased with pretreatment of AuNP@SiO2. It is not surprising that RGCs are particularly vulnerable to mitochondrial dysfunction. Oxidative/nitrosative stress led to mitochondrial dysfunction by a lack of balance in ΔΨm and an increase in the mass of mitochondria.17,18 Our previous studies demonstrated that HG induces the loss of mitochondrial membrane potential and increases the mitochondrial mass. This study found that AuNP@SiO2 restored HG-induced damage to mitochondria by restoring ΔΨm and mass in RGC-5 cells.
Several lines of evidence presented in the study also support the notion that ROS/RNS overproduction leads to biomolecule damage, which is considered an essential factor in the pathogenesis of macro/microvascular complications of diabetes. Our results show that HG increases the LPO and PC in RGC-5 cells, while AuNP@SiO2 has a protective effect. Despite the mechanisms’ complexity, mitochondria appear to release apoptotic factors that may trigger DNA fragmentation in nuclei.19,23 Consistent with this report, a morphological study of nuclei confirmed the protective side of AuNP@SiO2 in RGC-5 cells. We also determined the lysosomal content in RGC-5 cells in HG with/without AuNP@SiO2. Furthermore, our outcomes demonstrated that RGC-5 cell viability was decreased significantly when cells were grown in an HG environment, but when cells were exposed to the different doses of AuNP@SiO2, cell viability was increased, and the 0.025 nM dose was highly effective in restoring the number of live cells. Oxidative/nitrosative stress-related DNA damage has been proposed as an important factor in the pathogenesis of diabetes. Based on our previous finding that OGG1 was downregulated in HG-induced RGC-5 cells, which is consistent with a recent study, the downregulation of OGG1 via HG-induced oxidative/nitrosative stress is restored by AuNP@SiO2. Next, the 8-OHdG accumulation due to oxidative/nitrosative stress is conquered by the presence of AuNP@SiO2.
Elevated levels of circulating inflammatory mediators, including cytokines, are hallmarks of chronic inflammation and are now found to promote the progression of diabetes.5,41 Several studies have demonstrated that inflammation with increased cytokine levels in the retina plays an essential role in the pathogenic development of DR. Our data indicate that HG-induced oxidative/nitrosative stress triggers the activation of inflammatory cytokines, and AuNP@SiO2 restores that. In the present study, we also found that HG-induced ROS/RNS production leads to alteration in neuronal markers, further restored by AuNP@SiO2 treatment, confirming the protective effect of AuNP@SiO2 upon neurodegeneration. AuNP@SiO2 has antioxidative, anti-inflammatory, antiangiogenic, and antiproliferative effects, and because of these properties, it may find beneficial applications as therapeutic agents in angiogenesis-dependent disorders, such as DR. As AuNP@SiO2 inhibits the proliferation of VEGF, it can effectively inhibit the formation of new blood vessels in the retina, a key feature of proliferative DR. The key findings of the study are very limited, with common markers, and different animal models are required to verify the current findings.
Conclusions
In conclusion, the antihyperglycemic effect of AuNP@SiO2 and their protective effect on the retina during hyperglycemia-induced oxidative/nitrosative stress may be attained by inhibiting ROS/RNS production. Our study suggests that treatment with AuNP@SiO2 in RGC-5 cells decreases ROS/RNS production caused by HG, improves the depletion of antioxidant levels, and triggers the normalization of mitochondrial dysfunction, biomolecule damage, apoptosis, inflammation, and neurodegeneration in RGC-5 cells. Here, we have demonstrated the promising protective effect of AuNP@SiO2 for preventing DR associated with oxidative/nitrosative stress, which could be used for the treatment of retinal complications. Taken together, AuNP@SiO2 has antioxidative, anti-inflammatory, antiangiogenic, and antiproliferative effects, and because of these properties, it may find beneficial applications as therapeutic agents in angiogenesis-dependent disorders, such as DR. As AuNP@SiO2 is inhibiting the proliferation of VEGF, it can effectively inhibit the formation of new blood vessels in the retina, a key feature of proliferative DR.
Authors’ Contributions
S.P., S.S., B.K.B., and G.N.R. performed the experiments and draft preparation. A.P. conceived and designed the study, supervised the experiments, and wrote the manuscript. All authors reviewed and approved the final version of the manuscript.
Footnotes
Acknowledgments
Author Disclosure Statement
All the authors declare that they have no conflict of interest.
Funding Information
This work was supported by the Department of Biotechnology, Government of India (Grant no: BT/PR14241/MED/30/423) to A.P. The authors also thank the Department of Science and Technology, Government of India, for the Ramanujan Fellowship to S.S.
