Abstract
With the rising adoption of virtual reality (VR) in health care, research publications on the topic are increasing rapidly. Guidance on reporting VR interventions is limited, which risks misinterpretation of findings, inappropriate data synthesis in reviews and meta-analyses, and reduced reproducibility. This could also result in possible misuse of VR interventions and might hinder further implementation in clinical practice. The aim of this study was to develop a reporting guideline for therapeutic, immersive VR interventions using head-mounted displays in scientific publications. To reach this aim, we intended to achieve expert consensus on key items and categories for reporting these interventions. We conducted a three-round, modified, online Delphi study with a validation meeting to develop a VR intervention reporting guideline. Participants were international experts in therapeutic VR research, working as academic or industrial researchers. Convenience, purposive, and snowball sampling were used to recruit participants. Consensus was defined a priori as ≥75% agreement on each reporting item and category. Of the 280 invited experts, 61 participants (22%) completed all three rounds of this Delphi study. Participants (32 females [52%]) had an average of 6 years’ experience with VR research and diverse backgrounds in VR research settings and disciplines. The final consensus-based VR intervention reporting guideline consists of 16 reporting items divided into six categories (i.e., theory, content, deployment, development, safety, and context). The INVIRTUE intervention reporting guideline provides a framework for describing therapeutic VR interventions. Using this tool may enhance the uniformity and completeness of VR intervention reporting and support study replicability and scientific progress. The reporting guideline will also facilitate efficient empirical comparison across existing VR interventions and ultimately contribute to the appropriate use and effective implementation of therapeutic VR in clinical practice.
Introduction
Extended reality (XR) is increasingly used in health care as an (adjunct) intervention. 1 XR is an umbrella term that encompasses virtual reality (VR), mixed reality, and augmented reality. VR can be defined as a “computer-generated digital environment that can be experienced and interacted with as if that environment was real” 2 and is usually provided through a head-mounted display (HMD). By using an HMD, the user generally is sensory immersed in the virtual environment. 3 Therapeutic VR is used for a variety of conditions, including (chronic) pain, 4 phobias, 5 and stress. 6
Recently, a cross-sectional study reviewed the quality of reporting XR interventions in health care publications and showed that current intervention reporting is very poor. 7 This corresponds with a recent review on XR interventions in pain management research, which showed that only 23% of the examined scientific papers included a substantial content description of the VR intervention, and only 46% included a substantial hardware description. 8 This lack of detail and completeness in VR intervention reporting can lead to, for example, inappropriate interpretation, as well as comparisons and pooling of heterogeneous interventions in reviews and meta-analyses.9,10 This is a potential factor that hinders the adoption of therapeutic VR in health care settings and the replication of VR effectiveness studies. 11 Recent literature on the current state of clinical VR research described it as “the Wild West,” with a lack of clear guidelines, standards, or substantial progress.12,13 Multiple sources suggest that the current state of VR scientific inquiry is underpinned by the absence of consensus on an intervention reporting guideline. This was mentioned as a limitation of recent VR research in different health care settings, including psychology, 14 chronic pain, 15 and physical rehabilitation research. 16
Several intervention reporting guidelines of therapeutic interventions exist; the Template for Intervention Description and Replication (TIDieR) checklist is one of the most commonly used in health care. 17 This checklist includes items delineating why, how, and where the intervention was delivered. More specific intervention reporting guidelines were developed for exercise interventions such as the Consensus on Exercise Reporting Template, 18 but not yet for therapeutic VR. Given the multiple specific technical, content, and user-interactive elements involved in VR applications, existing therapeutic reporting guidelines are limited in their ability to capture the essential components of therapeutic VR interventions. 7
The aim of this study was to develop an internationally accepted reporting guideline on how to describe therapeutic VR interventions in scientific papers. In this study, therapeutic VR is considered to be sensory immersive and provided through an HMD. We chose to focus specifically on VR rather than other forms of XR because it is the most extensively researched area within the XR domain.8,19 To reach this objective, we aimed to achieve consensus on key items and categories for reporting therapeutic VR interventions.
Methods
Design
The three-round, modified, electronic Delphi (eDelphi) survey and additional validation meeting was conducted among international VR research experts. Delphi techniques are often used to identify consensus or develop standards by collecting knowledge from domain-specific experts.20,21 Traditional Delphi studies use open-ended questions to identify responses for future Delphi rounds, while modified Delphi studies provide a set of initial statements that are developed a priori. 22 Consequently, in modified Delphi studies, the coordinating researchers have an active role in generating consensus and generally two to three Delphi rounds suffice, rather than at least four rounds required in traditional Delphi studies. 21 We chose a modified Delphi design to facilitate timely study completion, diminish burden on study participants, and permit tailoring of the process to study needs. In addition, existing intervention reporting guidelines served as a useful foundation for determining key items and categories. This Delphi study included three rounds: round 1 (demographic information, rating, and proposing [new] items and categories), round 2 (rating and testing stability of items and categories), and round 3 (rating and testing stability of items and categories). The reporting guideline was finalized during a validation meeting.
The study was based on the Enhancing the QUAlity and Transparency Of health Research (EQUATOR) network recommendations for developing reporting guidelines 23 and reported according to the guidance on Conducting and REporting DElphi Studies guidelines. 24
Approval to conduct the study was granted by the ethics committee of the HAN University of Applied Sciences (HAN ECO: 564.06/24). All participants provided electronic informed consent before participating in the study. The study was registered prospectively on the Open Science Framework (OSF) (doi: 10.17605/osf.io/hfp4m) and with the EQUATOR network. The Delphi rounds were conducted between June 2024 and March 2025, and the validation meeting was conducted in June 2025.
Contributors
The study was primarily conducted by a day-to-day committee, supported by an international steering committee. Members of both committees were all researchers studying the application of therapeutic VR in health care interventions.
Day-to-day committee
The day-to-day committee of the Delphi study consisted of S.S., M.v.d.H., H.v.G., R.O., J.B.S., and J.K. The committee conceptualized, designed, coordinated, and coauthored this scientific paper on the Delphi study.
Steering committee
The international steering committee of this Delphi study consisted of D.H., L.B., B.D., T.D.G., P.A.v.d.H., M.K., J.B.S., Z.T., and D.U. The committee supported the day-to-day committee in providing feedback and coauthored this scientific paper.
INVIRTUE expert panel
Potential candidates for the expert panel were identified by the day-to-day committee. Expert panel members studied VR interventions in different health care areas and were included if they (1) (co)authored a scientific paper on therapeutic VR in a peer-reviewed journal and (2) were employed as researchers in an academic institution or industry. Participants were recruited through convenience sampling (e.g., by inviting colleagues of committee members), purposive sampling (e.g., by searching scientific databases for specific researchers), and snowball sampling (e.g., by asking participants to propose other participants in the first Delphi round). We strived to include at least 40 experts in the final round of the Delphi study, which is considered to be sufficient. 25 Participants in the expert panel who completed all three Delphi rounds were mentioned as final authors of this article as part of the “VR intervention reporting international consensus (INVIRTUE) group” (see section “Acknowledgments”).
Procedure and analysis
Preparation
Before the first round, a list of reporting items and categories was developed. The list was based on thematic analysis of >100 scientific papers that reported on the effectiveness of a therapeutic VR intervention (derived from Trost et al. [2021] 26 ), other intervention reporting guidelines (i.e., TIDieR 17 and RATE-XR 27 checklist), and group discussions with the day-to-day and steering committees. Relevant features describing the therapeutic VR intervention of the aforementioned sources were systematically reviewed and inductively coded by one researcher (M.v.d.H.) and subsequently discussed with two additional researchers (S.S. and J.K.). Next, these three researchers categorized the codes into themes through iterative comparison and discussion. 28 The a priori constructed list consisted of 26 intervention reporting items divided into five categories (i.e., theory, dose, content, preparation, and additional).
Before conducting the Delphi rounds, pilot tests were conducted to ensure study procedure feasibility. Results of the pilot tests were included in the final analysis, as no major issues were identified. Participation in the study was confidential, but not anonymous, because the coordinating researcher could identify participants based on their email address. Participant identification was necessary to be able to invite participants who completed a Delphi round for the next round. Personal data were stored on a secure server.
Round 1
All potential candidates of the expert panel were invited by the day-to-day committee through email to participate in round 1 of the study. The email contained general information about the Delphi study and a link to the survey on the web platform Qualtrics (Qualtrics, Provo, USA).
This first round included three parts. The first part contained the information letter and accompanied informed consent (Appendix A). After agreeing to participate in the study, participants continued to the actual survey. The second part requested the following (demographic) information about the participants: email address, age, sex, country of residence, VR research context, primary VR research area, and number of years’ experience in VR research. The third part comprised the a priori constructed list of 26 intervention reporting items with explanation per item and five categories (Appendix B), which were rated on a 9-point Likert scale per item (1 = of no importance to 9 = extremely important). The rating was based on the following question: “How important do you think this topic is for a therapeutic VR intervention reporting guideline?” Besides rating, there were two types of free-text boxes, one to comment on individual reporting items and suggest new items, and one to comment on the categories suggested in the a priori constructed list.
Participants received an email reminder to participate in the study after 7 and 14 days. Three weeks after the study started, this first round was closed. Items were proposed in the next round for inclusion in the reporting guideline when 75% of the participants rated an item 7 or higher, and less than 15% of the participants rated the item 3 or lower. Items were proposed in the next round to be excluded from the reporting guideline when 75% of the participants rated an item 3 or lower, and less than 15% of the participants rated the item 7 or higher. 29 Item and category clarifications or modifications and new items proposed by participants were screened and analyzed inductively by the day-to-day committee and, if deemed of added value, included in the second round. This ensured that expert input, not just quantitative consensus, played a central role in shaping the subsequent rounds.
Round 2
The second round provided controlled feedback in the form of an anonymized summary of results from round 1. This round had three parts. First, to study item stability, items from round 1 that scored as sufficient to be included or excluded were presented with the question of whether the intervention reporting item should indeed be included or excluded in the reporting guideline. 21 An item was deemed stable if 85% of the participants agreed that an item should be included or excluded. 30 Second, items and categories without consensus were presented again with the median and interquartile range scores that the item scored in round 1. 31 Third, new items proposed during round 1 were presented. Both items without consensus and new items were again rated based on the procedure presented for round 1. There was no option to suggest new items in this or subsequent rounds, but participants had the option to suggest minor revisions for both the items and their descriptions.
Round 3
The procedure of round 3 was similar to the survey procedure of round 2 and was based on the results from round 2. In addition, participants were asked to classify all proposed intervention reporting items in the following categories: “essential in reporting guideline,” “nice to have in reporting guideline,” or “not necessary in reporting guideline,” based on Duncan et al. (2020). 32 Intervention reporting items were considered as included in the final reporting guideline if they scored a median rating of 7–9 33 and/or if >50% of participants categorized an item in the “essential in reporting guideline” category. Similarly, intervention reporting items were considered excluded from the final reporting guideline if they scored a median rating of 1–3 and/or if >50% categorized an item in the “not necessary in reporting guideline” category.
Validation meeting
Results of round 3 were discussed with members of the day-to-day and steering committees during an online consensus meeting. In this meeting, each individual reporting item and associated category was discussed, and the members were asked to improve, as necessary, the information regarding the suggested reporting items (i.e., reporting item title, category, description, and example). The results of the consensus meeting formed the validated version of the VR intervention reporting guideline.
Results
Of the 280 international VR research experts were invited for the first Delphi round, 61 (22%) completed all three Delphi rounds (see Table 1).
Flow of Participants
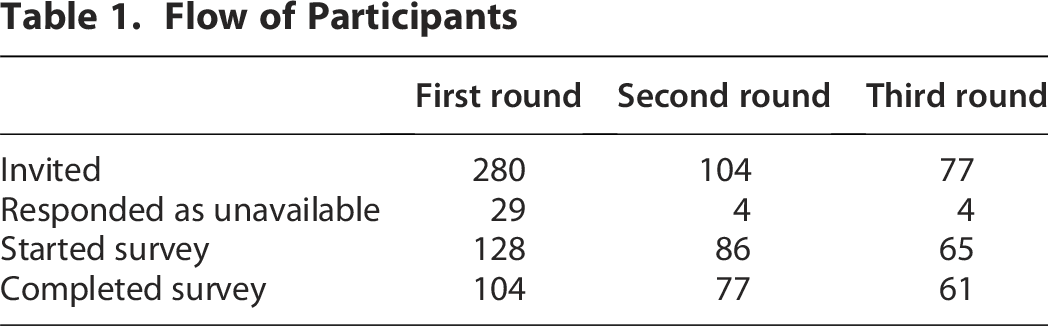
The 61 participants who completed the third round had a mean age (standard deviation) of 39 (9.7) and an average of 6 years’ experience with VR research. They were located in 13 countries from 4 continents and studied VR in a variety of disciplines and settings. For more information on participant demographics information, see Table 2. Comparison between the participants who completed all three rounds and non-completers showed no large differences in demographic information (see Appendix C).
Participant Characteristics

Multiple options possible.
For example, cognitive rehabilitation.
SD, standard deviation; VR, virtual reality.
The flow of the included, excluded, new, incorporated, and split reporting guideline items throughout the three Delphi rounds and validation meeting is shown in Figure 1.

Flowchart of reporting items.
In the first round, the 26 items (divided into five categories) from the a priori constructed list of intervention reporting items were rated. After this round, seven items (i.e., clinical aim, time per session, intervention duration, frequency, intervention setup, application/game, and interactivity) were rated sufficiently important to include in the reporting guideline, six new items were proposed (i.e., purpose of VR intervention, scientific evidence, participant involvement, availability/accessibility, professional training, and data security), and two items were excluded (i.e., intensity and software for development). Intervention procedure was incorporated into the existing reporting item application/game, and four items were split (i.e., proposed working mechanism was split into clinical aim and proposed working mechanism; hardware was split into technical requirements and hardware; intervention instruction was split into preparation time and intervention instruction; and intervention time session was split into time per session and VR time).
In the second round, 33 items (divided into six categories) were rated based on the results of the first round. Three new categories (i.e., usability, population, and safety) were added, and two categories (i.e., preparation and additional) were removed. The 33 items of the second round included the 7 items that were included after the first round to test their stability. After this round, five items were rated sufficiently important to include in the reporting guideline (i.e., clinical aim, application/game, interactivity and feedback, dosage, and setting and supervision), and three items were excluded (i.e., costs, presence, and embodiment). Fourteen items were incorporated into existing reporting items (i.e., purpose of VR intervention was incorporated into clinical aim; scientific evidence was incorporated into proposed working mechanism; feedback mechanism was incorporated into interactivity and feedback; intervention modification was incorporated into modification, personalization, and tailoring; motion tracking, audio, and technical requirements were incorporated into hardware; time per session, intervention duration, and frequency were incorporated into dosage; intervention setup, body position, and safety measures were incorporated into setting and supervision; and provision/delivery was incorporated into instruction).
In the third round, 16 items (divided into six categories) were rated based on the results of the prior rounds and included in the reporting guideline. One category was added (i.e., deployment), and one category was removed (i.e., dose).
In the validation meeting, these 16 items were discussed, and their descriptions were improved if necessary (e.g., including more extensive examples per reporting item).
The final consensus INVIRTUE reporting guideline on how to describe therapeutic VR interventions contains 16 items divided among six categories: theory, content, deployment, development, safety, and context. The full version of the reporting guideline is shown in Table 3, and Appendix D provides a customizable version of the guideline. Appendix B gives a full overview of the reporting items and categories per round.
Final INVIRTUE Reporting Guideline

HMD, head-mounted display.
Discussion
This Delphi study reached agreement on reporting items and associated categories that resulted in the consensus INVIRTUE intervention reporting guideline, which could improve uniformity and completeness of therapeutic VR intervention reporting in scientific papers. The consensus reporting guideline consists of 16 items divided into six categories.
Discussion of intervention reporting guideline content
The Delphi rounds facilitated important discussions and revealed challenges in defining the final reporting guideline. One unexpected challenge was orienting all participants to the discussion of intervention reporting rather than traditional article reporting. This is reflected, for example, in participants suggesting including an item on adverse events, even though this would be more suitable for article results instead of the intervention description. The final reporting guideline includes an item on anticipated adverse events and safety considerations to prevent these (item 15). Another topic where consensus proved difficult was how detailed the reporting guideline should be. Some participants suggested including detailed technical reporting (e.g., utilized HMD refresh rate). This level of technical detail, while essential for hardware and software design, was generally considered less relevant for reporting of applied therapeutic VR research. After the three rounds, this item was excluded from the reporting guideline, but we urge authors to include this if they think it is appropriate. Further, the a priori list of reporting items included 26 reporting items, and after the first Delphi round, only six new items were proposed. This suggests that the initial list was already quite comprehensive and captured substantial relevant content. There was likewise some debate on using the term tailoring or personalization to describe reporting for item 6. In line with the TIDieR checklist and a recent review on the concepts of personalization and tailoring in eHealth interventions, we chose the term tailoring as we think this best reflects the description of the reporting item. 34 Finally, although participants struggled to reach consensus on certain reporting items in the first and second rounds, consensus was reached on all items after the third Delphi round. This could be due to the novelty of the field and the broad range of therapeutic VR applications, which is reflected in other current debates in VR research, including the struggle to define (immersive) VR in general.35–37
Comparison with other reporting guidelines
This is the first guideline specifically for VR intervention reporting, whereas more general article and intervention reporting guidelines already exist. Comparing our reporting guideline to the established TIDieR reporting checklist for interventions in general, it seems our reporting guideline is comprehensive. 17 Most items of the TIDieR checklist are reflected in our reporting guideline, that is brief name (item 4 of our reporting guideline), why (items 1 and 2), what (items 4–8 and 11), who provided (items 10 and 13), how (item 10), where (item 10), when and how much (item 9), tailoring (item 6), and modifications (item 6). The only item not reflected in our reporting guideline is the final item of the TIDieR checklist (i.e., how well), which describes the planned and actual intervention adherence and fidelity. This is possibly due to the fact that this item is not strictly about describing the intervention itself but rather about the correct deployment of the intervention. Our reporting guideline is specifically designed for therapeutic VR interventions, providing more specific guidance. This is, for example, illustrated in the why reporting item of the TIDieR checklist (i.e., describe any rationale, theory, or goal of the elements essential to the intervention). Our reporting guideline divides this item into multiple items that ask for a therapeutic VR-specific clinical aim (item 1) and proposed working mechanism (item 2). Similarly, the intervention items of the RATE-XR checklist 27 are reflected in our reporting guideline, that is, 4—setting and location (item 10), 5b—providers and training (items 10 and 13), 6—application description (items 4–12), 7—intervention development (item 14), and 8—participant timeline (item 16). The RATE-XR article reporting guideline aims to improve the reporting of XR evaluation studies and includes items from the title to statements. However, the RATE-XR focus was not specifically on the in-depth description of therapeutic VR interventions.
Intervention reporting guideline use
Sole use of this reporting guideline may not suffice for uniform and complete VR intervention reporting and possible study replication. It should be noted that this reporting guideline is not exhaustive, and additional details are warranted to ensure an in-depth and comprehensive intervention description for individual VR interventions. However, not all suggested reporting items are always necessary to report in a scientific paper, for example, when a VR intervention is described in-depth in a separate paper. In such cases, it is crucial that authors explain why they omitted this reportable information or where more information can be found. Unfortunately, it is not possible to establish a single, definitive reporting guideline that fits all VR interventions, given the considerable breadth of the therapeutic VR research field, which spans among others, physical, psychological, and cognitive rehabilitation applications. Moreover, it should be stressed that this reporting guideline focuses specifically on VR intervention description in a scientific paper. Other guidelines were developed to provide general guidance across a variety of scientific reporting, for example, the Consolidated Standards of Reporting Trials reporting guideline for randomized trials 38 and Preferred Reporting Items for Systematic Reviews and Meta-Analyses reporting guideline for systematic reviews and meta-analyses. 39 Also, the RATE-XR article reporting guideline was recently published to report early-phase clinical evaluation of XR applications.27,40 Therefore, we suggest combining the INVIRTUE intervention reporting guideline with a fitting article reporting guideline.
Strengths and limitations
This study has several strengths. First, the study employed a comprehensive methodological approach including a literature review, multiple Delphi rounds, and a validation meeting, resulting in rich qualitative and quantitative data. Also, the international EQUATOR network recommendations to develop intervention reporting guidelines were adopted, which aim to advance high-quality reporting of health research studies. 41 Furthermore, the study was registered a priori on OSF and the EQUATOR network. Second, the included expert sample was larger than anticipated and sufficient for a Delphi study. 25 Also, no considerable differences in characteristics were observed between responders and nonresponders, suggesting decent external validity. The sample further included experienced VR experts with a large heterogeneity in locations, expertise, and backgrounds, which ensured comprehensive and trustworthy study results.
This study has the following limitations. First, participants were mostly from Western countries and especially from the Netherlands, since most members of the day-to-day and steering committee were from the Netherlands. In addition, the study lacked input from lower- and middle-income countries even though experts from these countries were invited. Consequently, the guideline may not reflect a global perspective on a VR intervention reporting guideline and, for example, may miss important topics like the availability of therapeutic VR interventions in different languages. Moreover, differences across countries in, for example, health care infrastructure and access to VR may limit the generalizability and global applicability of the reporting guideline. Second, since participants were invited through convenience, purposive, and snowball sampling, selection bias might have influenced the results. Also, using the a priori constructed list of reporting items may have introduced confirmation bias, which we mitigated by incorporating open-ended feedback and revising items across rounds. Third, we limited participants to those who (co)authored a scientific paper on therapeutic VR in a peer-reviewed journal, which may have overlooked variability in research expertise. Accordingly, the consensus achieved might not represent all expert perspectives, and certain guideline details could differ among different participant groups. Fourth, the reporting guideline was developed for scientists, and therefore, only researchers who published VR-related scientific papers were invited. Although several participating researchers also work as clinicians, the lack of variety in stakeholders may have affected the generalizability of the results.
Future research
Future research could address several topics based on the results of this study. First, studies could increase the scope of this research and, for instance, extend these results to XR or digital health in general, although the current version of the reporting guideline is likely already well suited for describing XR interventions. Also, while our results are limited to therapeutic VR, VR is increasingly being used in diagnostics, 42 and it might be worthwhile to study this in detail. Second, this study is a small step in advancing research on therapeutic VR, but more steps are needed to mature this relatively new area of research. One of these steps could be the development of a research agenda with the most urgent topics to study in the field of therapeutic VR, to which the current guideline can be applied. The fast-paced evolution of VR warrants continuous updating of this study’s outcomes, including input from lower- and middle-income countries and different VR stakeholders (e.g., clinicians, users, and developers). In addition, new technological advancements, including the use of haptics and eye tracking, are likely to improve and be integrated into therapeutic VR interventions of the future, necessitating further expansion or modification for optimal VR intervention reporting. To ensure continuous relevance, the INVIRTUE group will update the reporting guideline every 5 years, taking into account significant developments within the field.
Impact
Implementation of this reporting guideline could be achieved in multiple ways. We will notify academic journals and researchers (including Delphi participants) about the completion of the guideline. Furthermore, this reporting guideline is freely available, and we will continue cooperating with EQUATOR to disseminate this reporting guideline. Implementation of this guideline is expected to have an impact on different stakeholders in research on therapeutic VR. Researchers could use the reporting guidelines for transparent and comprehensive intervention reporting. This way, the reporting guideline would support appropriate interpretation, comparison, and pooling of VR interventions. Clinicians could use the reporting guideline to extract essential VR intervention elements (e.g., dosage) required for accurate intervention application, which would ultimately support intervention adoption and effectiveness in clinical practice. Finally, the reporting guideline may also inform other stakeholders, including funding bodies and industry, by improving the clarity and consistency of intervention reporting and facilitating the translation of research findings into real-world applications.
Conclusion
To conclude, this study reached consensus on key items and categories to report therapeutic VR interventions in scientific papers. These results culminated in the new consensus INVIRTUE VR intervention reporting guideline, which provides the scientific community with a meaningful tool for uniform, detailed, and comprehensive VR intervention description. Utilizing this tool could support researchers in study interpretation, comparison, and replicability, ultimately building a stronger empirical foundation for VR utility and supporting clinical practice in the appropriate application and effective implementation of therapeutic VR in health care.
Authors’ Contributions
S.S. was the principal investigator of this study, part of the day-to-day committee, and drafted the first version of the article. H.v.G., R.O., B.S., and J.K. were part of the day-to-day committee, provided feedback on the article, and supervised by S.S. M.v.d.H. participated in this project as part of his thesis and was supervised by S.S. and J.K. D.H., L.B., B.D., T.D.G., P.A.v.d.H., M.K., B.S., Z.T., and D.U. were part of the steering committee and provided feedback on the article.
Footnotes
Acknowledgments
The members of the INVIRTUE Expert Panel are Reinoud Achterkamp, Corinne Ammann, Sara Arlati, Jane Aspell, Nancy Baker, Lina Bareišytė, Aislinn Bergin, Sylvie Bernaerts, Paul Best, Chris Bevan, Inge Blauw, Iris Brunner, L.B., Błażej Cieślik, Davide Clemente, Nigel Cowan, Tom van Daele, B.D., Denzel Drop, Alexander Elser, Maximilian Friehs, Linda Garms, Andrew Goodsell, T.D.G., Ineke van der Ham, D.H., Lieke Heesink, Renée van den Heuvel, Sander van de Hoef, Alexandre Hudon, Marian Hurmuz, Stephanie Jansen, Hendrik Knoche, J.K., Marileen Kouijzer, Janienke Lier, Lukas Lorentz, Alexandre Luc, Erin Macintyre, Todd Maddox, Thomas Matheve, Ryan McConnell, Abele Michela, Lynn Nakad, Daniel Perez, Susan Persky, Ivan Phelan, Jennefer Pouwels, Federica Ramírez, Aurora Ruiz-Rodriguez, Aniek Siezenga, Nathan Skidmore, Karin Slegers, Ivan Steenstra, Vincent Stirler, Jordan Tsigarides, D.U., Ilse Verveer, Harald Vonkeman, Marjan de Vries, and Steven Watson.
Author Disclosure Statement
The authors declare they have no competing interests.
Funding Information
This study was funded by ZonMw (case number: 10270032021502). The funder had no role in the design, organization, and execution of the study.
Availability of Data and Materials
The data generated during these studies will not be publicly available but will be available upon reasonable request to the corresponding author.
Ethics Approval and Consent to Participate
Ethical approval of this study was obtained from the ethics committee of the HAN University of Applied Sciences (case number: 564.06/24). Participants provided informed consent before study commencement.
