Abstract
Background & Aims:
Hepatitis C virus (HCV) infection is a prevalent disease at drug treatment centers (DTC). Alcohol abuse may also contribute to liver fibrosis at DTC, which may affect the prescription of HCV therapy. This study evaluated the risk of alcohol intake and other predictive factors for fibrosis progression.
Methods:
An on-site dried-blood-spot (DBS) testing program for hepatitis C care at DTC from 2017 to 2019 was used to identify patients with previous HCV-antibody testing and fibrosis score data. Patients were grouped based on RNA status, alcohol intake and advanced liver disease (ALD ⩾ F3). Fibrosis progression was assessed using APRI and FIB-4. Kaplan-Meier and Cox-regression analyses were performed.
Results:
Positive HCV RNA patients (n = 138) exhibited a higher rate of ALD (11% vs 2.2%, P < .001) and a higher risk of fibrosis progression during follow-up (HR = 3.14, 95% CI: 1.2-8.2) than patients who did not have an infection (n = 230). Overall, 25% (n = 84) reported high-risk alcohol consumption, which was associated with an increased risk of ALD in patients with RNA positivity (HR = 3.2; 95% CI: 1.1-9.1). Alcohol consumption at any dose regardless of HCV infection status was not associated with ALD. Age, high-risk alcohol consumption, and RNA positivity were independent factors for progression to ALD.
Conclusions:
Patients with active HCV infections at DTC have an increased risk for ALD compared to patients without HCV. High-risk alcohol consumption is present in a minority of patients aggravating fibrosis. These results suggest that HCV treatment should not be delayed at DTC regardless of alcohol consumption.
Introduction
The World Health Organization (WHO) has set a goal to eliminate hepatitis C virus (HCV) infections by 2030 through increased diagnosis and treatment rates, aiming for an 80% reduction in new HCV infections and a 65% decrease in HCV-related deaths. 1 The implementation of micro-elimination strategies and changes in policies, practices, and procedures are necessary to achieve this goal, especially in patient populations with a high prevalence of HCV. 2
People who injected drugs (PWID) have the highest HCV seroprevalence of any population. 3 Despite the recent availability of effective treatments with minimal adverse effects, this high-risk population has remained largely undiagnosed and untreated. 4 Recent European data highlight both progress and ongoing gaps in HCV testing and treatment, especially in substance use treatment settings. Chronic HCV prevalence in the European Union (EU) remains at 0.50%, with over one-third of cases attributed to injecting drug use. In 2017, only 23 of 30 EU regions provided HCV testing at harm reduction sites, and fewer than half had referral systems for treatment. 5
Advances in HCV treatment have enabled new care diagnostic models, such as dried-blood-spot (DBS) testing at drug treatment centers, improving access for high-risk populations. 6 DBS testing for HCV, as part of linkage-to-care programs in drug treatment centers, has shown to be effective in narrowing treatment gaps in the HCV care cascade, as it simplifies sample collection and transport, reduces associated logistical costs, and maintains high diagnostic accuracy, making it particularly suitable for PWID.7,8
Despite the availability of therapies, 9 treatment uptake remains low among patients at drug treatment centers. Concerns persist regarding the appropriateness of treating individuals with ongoing drug or alcohol use, which may contribute to the progression of liver fibrosis after a virological response is achieved, which remains controversial due to stigma and limits the number of patients being treated. 10 Few studies have specifically evaluated the impact of alcohol consumption on liver fibrosis progression in HCV-infected patients attending drug treatment centers. Therefore, this study aimed to assess liver fibrosis severity and associated risks factors, including alcohol consumption, for the progression to advanced liver fibrosis (ALF) among individuals screened for HCV using DBS testing at drug treatment centers.
Methods
Patients
In January 2017, an HCV screening program was launched across 6 drug treatment centers that provide care for potentially 480 000 patients in the northern area of Tenerife (Canary Islands, Spain). The program included psychological counseling, opioid substitution therapy, and on-siteDBS testing for HCV, regardless of previous HCV testing and status. 6 The program was conducted in 2 phases: the first phase (January 2017-July 2018) involved anti-HCV antibody testing, while the second phase (August 2018-September 2019) implemented DBS testing for RNA detection and re-engaged patients lost to follow-up. DBS testing for RNA was conducted as previously described. 11 The analysis was based on all available patients who underwent HCV screening via dried blood spot testing in the defined study period (January 2017-September 2019) across 6 drug treatment centers. This pragmatic approach reflects the total eligible population for analysis within the program framework.Therefore, a retrospective cohort study was designed for evaluating the progression of liver fibrosis and its predictors among this population screened for HCV.
Patients were eligible if they: (1) were ⩾18 years old, (2) attended 1 of the 6 participating drug treatment centers, (3) underwent dried blood spot (DBS) testing for HCV antibodies and/or RNA during the study period, and (4) had available clinical and laboratory data to allow assessment of liver fibrosis.
The DBS method involves collecting a drop of capillary blood from a finger prick onto a filter paper card (Whatman 903™ Protein Saver Card, 10 534 612, Germany), which is then air-dried and stored for subsequent analysis. The dried blood samples were then incubated in PBS elution buffer with 0.5% Tween 20 with agitation at 22°C for 16 hours before being centrifuged (10 000g for 5 minutes). The supernatant was used to detect the presence of anti-HCV antibodies via indirect enzyme-linked immunosorbent assays using the TRITURUS® platform with a fourth-generation Dia. Pro kit (Diagnostic Bioprobes, Milan, Italy).
Patients who tested positive for HCV were evaluated for HCV infection and grade of liver disease by a hepatologist at the university hospital based on current HCV management guidelines. 9
All patients were interviewed using a standardized data collection form to record demographic and socioeconomic status, current drug use, and alcohol intake; not only in the first visit but also during follow-up, so changes in drugs habits could be tracked. Alcohol intake was categorized as non-consumption or consumption, and consumption was graded as low-moderate (<20 g/day in women and <40 g/day in men) and high-risk (⩾20 g/day in women and ⩾40 g/day in men). 12
Data regarding human immunodeficiency virus (HIV), hepatitis B virus (HBV), obesity (defined as body mass index >30 kg/m2), diabetes, arterial hypertension, statin use, previous HCV testing (antibodies and RNA), previous HCV treatment and achievement of sustained virological response, alanine aminotransferase (ALT), aspartate aminotransferase (AST; abnormal value defined by >40 U/L), platelet count, gamma-glutamyl transpeptidase (GGT), mean corpuscular volume (MCV), and cholesterol were collected from the medical records.
Patients were categorized according to HCV test results as having an active HCV infection (HCV RNA positive) or a non-active HCV infection (HCV antibody negative and HCV antibody positive with undetectable HCV RNA).
Baseline fibrosis was assessed by calculating the APRI (AST to Platelet Ratio Index) and FIB-4 (Fibrosis-4) scores using laboratory results obtained at the time of or within 6 months after HCV antibody detection. To estimate the risk of liver fibrosis progression, the APRI score was calculated at the time of DBS testing. Advanced liver disease was defined as an APRI score ⩾F3. 13 The cut-off values for APRI (⩾F3 > 1.5) and FIB-4 (⩾F3 > 3.25) scores used in this study were previously validated. 14
Patients who did not undergo HCV antibody testing prior to the DBS testing, those who did not have RNA test results after testing positive for HCV antibodies, and those with missing data required for the calculation of liver fibrosis scores within 6 months of HCV testing were excluded from this study.
Statistical Analysis
Qualitative variables are expressed as absolute frequency and percentage and quantitative variables are expressed as mean and standard deviation or median and range. The chi-squared test was used to compare proportions and the Student’s t-test or Mann-Whitney U test were used to compare continuous variables. Logistic regression analyses were used to identify predictors of significant fibrosis and cirrhosis. The Kaplan-Meier method and log-rank test were used to evaluate the progression of liver fibrosis. Statistical significance was set at P < .05. All analyses were conducted using SPSS version 25.0 (IBM Corporation, Armonk, NY).
Ethical Considerations
This study was conducted according to the principles of the Declaration of Helsinki. The Clinical Research Ethics Committee of the Hospital Universitario de Canarias approved this study (approval number: 2016_57 (Hepc-cribado2016)). All patients provided written informed consent for their participation in this study.
Results
Patient Characteristics
A total of 907 patients (mean age: 46.3 ± 10.3 years, 82.2% men) were offered on-site DBS testing between January 2017 and September 2019. Between January 2017 and July 2018, 484 patients underwent DBS testing for HCV antibodies; and between August 2018 and September 2019, 423 patients underwent DBS testing for HCV RNA. Approximately half of the patients reported alcohol consumption, and 24.9% (n = 226) reported high-risk alcohol consumption (Supplemental Table 1) which did not change during follow-up. A total of 551 (66.7%) patients had previously been tested for HCV antibodies before the on-site DBS test (Figure 1). Of those with a positive HCV antibody test (n = 252), previous HCV-RNA data was available for 246 patients, of which 178 (72.4%) had detectable RNA. A non-active HCV infection was diagnosed in 367 patients, including 68 patients with HCV antibodies but undetectable RNA levels and 299 patients with negative HCV antibodies.

Patient flow chart. From 907 subjects attending DBS testing program, 551 had a previous HCV-antibody test, of those with a positive HCV antibody test (n = 252), previous HCV-RNA data was available for 246 patients, of which 178 (72.4%) had detectable RNA. A non-active HCV infection was diagnosed in 367 patients, including 68 patients with HCV antibodies but undetectable RNA levels and 299 patients with negative HCV antibodies. A total of 368 (66.8%) patients had sufficient data with which to calculate the liver fibrosis scores at baseline and at the time of DBS testing. These patients were categorized as those with active HCV infections (n = 138, 37.5%) and those with non-active HCV infections (162 with negative HCV antibodies and 68 with HCV antibodies but undetectable RNA; 62.5%).
Liver fibrosis scores at baseline and at the time of DBS testing could be calculated for 368 (66.8%) patients with complete data. These patients were categorized as those with active HCV infections (n = 138, 37.5%) and those with non-active HCV infections (162 with negative HCV antibodies and 68 with HCV antibodies but undetectable RNA; 62.5%).Patients with an active HCV infection were more likely to be male than those with a non-active HCV infection (90.8% vs 80.3%, P = .006). The active HCV infection group also had greater percentages of HIV-infected patients (6.5% vs 0.9%, P < .001) and patients receiving opioid substitution therapy (96.4% vs 68.3%, P < .001). The active HCV infection group had higher levels of AST, ALT, and GGT and a lower platelet count (all P < .001). The mean age was not significantly different between the groups, and no differences in other variables that may influence fibrosis progression, including alcohol consumption (53.5% vs 61.8%, P = .132) and high-risk alcohol consumption (24.6% vs 25.2%, P = .136) were found (Table 1). Supplemental Table 2 shows characteristics of patients with non-active infection based on if they tested positive (with undetectable RNA) or negative for HCV antibody.
Patient characteristics based on HCV infection status (n = 368).

Data are presented as median (interquartile range) or number (percentage).
Abbreviations: ALT, alanine aminotransferase; AST, aspartate transaminase; GGT, gamma-glutamyl transpeptidase; HBV, hepatitis B virus; HCV, hepatitis C virus; HIV, human immunodeficiency virus; INR, international normalized ratio; MCV, mean corpuscular volume.
HIV was tested in 112 and 106 patients in each group, respectively.
HBV was tested 5in 113 and 108 patients in each group, respectively.
Baseline Liver Fibrosis Scores
The APRI liver fibrosis scores were higher in patients with active HCV infections (median score: 0.49; range: 0.1-3.1) than in patients with non-active HCV infections (median score: 0.23; range: 0.1-4.2) at baseline (P < .001). Analyses using the FIB-4 score had similar results: active HCV infections (median score 1.2; range 0.3-6.4) compared to non-active infections (median score 0.97; range 0.3-12.9) demonstrate higher fibrosis scores (P = .042). The active HCV infection group had a greater percentage of patients progress to ALD (11% vs 2.2%, P < .001) based on the APRI score.
Liver Fibrosis Progression
The median time from previous HCV testing to the DBS testing conducted during this study was 85.9 months (interquartile range: 35.3-125.8 months). During this time, patients with active HCV infection were more likely to progress to ALD than those with non-active HCV infections (log-rank: 6.05; P = .014; hazard ratio (HR) = 3.14; 95% confidence interval (CI): 1.2-8.2; Figure 2).

Progression to advanced liver disease. Patients with active HCV infection were more likely to progress to advanced liver disease (ALD) than those with non-active HCV infections (log-rank: 6.05; P = .014; hazard ratio (HR) = 3.14; 95% confidence interval (CI): 1.2-8.2).
Predictive Factors of Liver Fibrosis Progression
Among patients with active HCV infection, high-risk alcohol consumption was identified as a predictive factor of liver fibrosis progression (log-rank: 4.95; P = .026; HR = 3.2, 95% CI: 1.1-9.1), though alcohol consumption at any dose was not associated with an increased risk of liver fibrosis progression to ALD in this population (log-rank: 0.615; P = .433; HR = 1.6, 95% CI: 0.5-5.2; Figure 3A and B). In the non-active infection group, high-risk alcohol consumption (log-rank: 2.1; P = .147; HR = 3.1; 95% CI: 0.6-15.4) and alcohol consumption at any dose (log-rank: 3.04, P = .081; HR = 38.8, 95% CI: 0.03-46 592) did not increase the risk of liver fibrosis progression to ALD (Figure 3C and D).

Probability of advanced liver disease based on level of alcohol consumption. In the group of patients with active HCV infection, only patients with high-risk alcohol consumption exhibited a higher probability of developing ALD compared to non-high-risk alcohol consumers (log-rank 4.95, P = .026; HR = 3.2, CI 95% [1.1-9.1]). Alcohol consumption (at any dose; yes/no) did not interfere increasing the probability of ALD (log-rank 0.615, P = .433; HR = 1.6, CI 95% [0.5-5.2]) (A and B). In the non-active infection group, neither high-risk alcohol consumption (log-rank 2.1, P = .147; HR = 3.1, CI 95% [0.6-15.4]) or at any dose (log-rank 3.04, P = .081; HR = 38.8, CI 95% [0.03-46592]) increased probability of ALD (C and D).
Liver Fibrosis Progression and Alcohol Consumption
The patients were divided according to their alcohol consumption (Supplemental Table 3). Both groups were similar in terms of age, sex, injected drug use, and presence of an active HCV infection. The AST (26 vs 25 U/L, P = .012), GGT (37 vs 27 U/L, P < .001), and mean corpuscular volume (93.2 vs 91.3 fl, P = .027) were higher in patients who reported alcohol consumption compared to those without alcohol consumption. Patients who reported alcohol consumption also had higher APRI and FIB-4 scores.
Patients who reported high-risk alcohol consumption were more likely to have liver fibrosis progression if they also had an active HCV infection (log-rank: 4.1; P = .043; HR = 3.7; 95% CI: 1-14.6; Figure 4A). Among patients who reported any level of alcohol consumption, there was no significant difference in liver fibrosis progression between patients with and without an active HCV infection (log-rank: 2.7; P = .098; HR = 2.3; 95% CI: 0.8-6.5; Figure 4B).
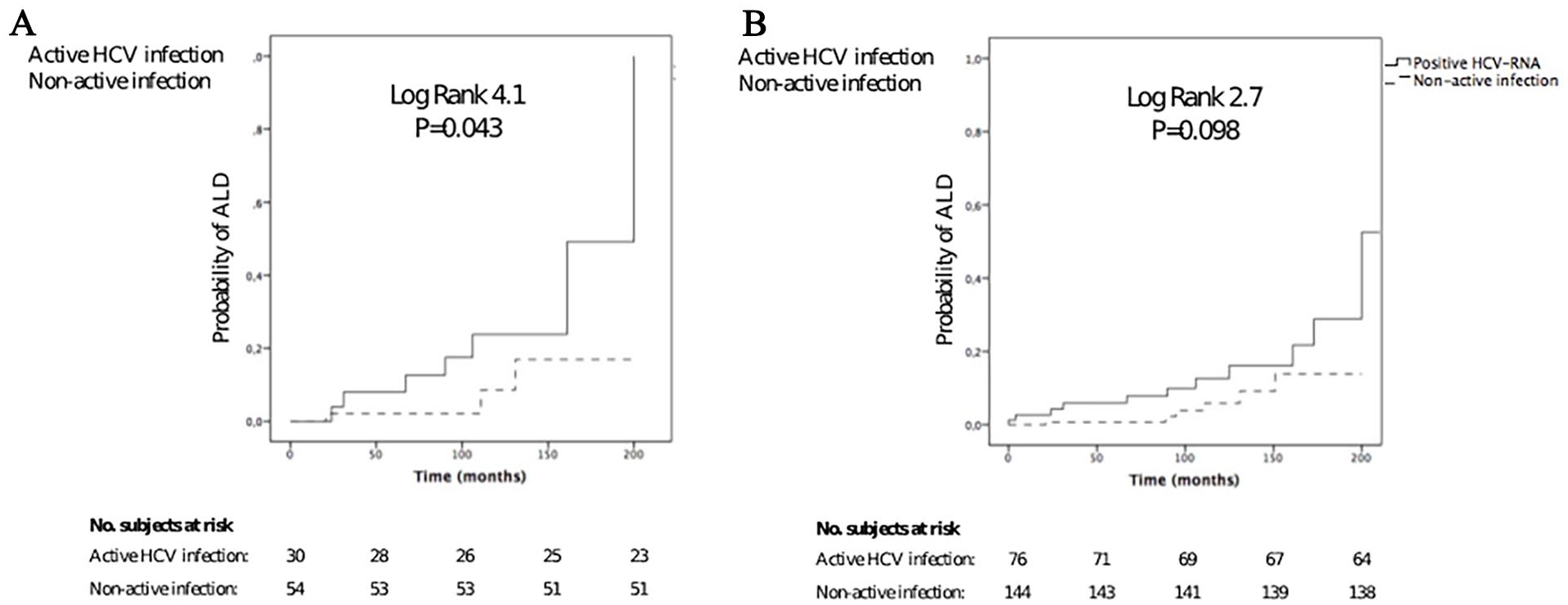
Probability of advanced liver disease based on HCV-RNA status. Patients with high-risk alcohol consumption presented more probability of developing ALD if coexisted detectable HCV-RNA (log-rank 4.1, P = 0.043); HR = 3.7, CI 95% [1-14.6]) (A). When this was evaluated in alcohol consumers (at any dose) no differences were seen between having or not detectable HCV-RNA (log-rank 2.7, P = .098; HR = 2.3, CI 95% [0.8-6.5]) (B).
Independent Predictors of Liver Fibrosis Progression
Age > 49 years (HR: 4.3; 95% CI: 1.36-13.8; P = .01), an active HCV infection (HR = 4.3; 95% CI: 1.5-12.1; P = .006), and high-risk alcohol consumption (HR = 3.8; 95% CI: 1.4-10.1; P = .007) were identified as independent predictors of liver fibrosis progression to ALD (Table 2). Other analysis were perfomed, adding alcohol consumption at any dose, with similar results (Supplemental Table 4).
Predictive factors for liver fibrosis progression to advanced liver disease.

Abbreviations: HCV, hepatitis C virus; RNA, ribonucleic acid.
Discussion
This study identified age >49 years, active HCV infection, and high-risk alcohol consumption as independent predictors of liver fibrosis progression to ALD in patients attending drug treatment centers. Importantly, and contrary to extended general opinion, non-high risk alcohol consumption by this group of patients does not impact as much as having an active HCV infection. Our findings suggest that patients at drug treatment centers are at high risk of liver fibrosis progression to ALD despite available antiviral treatments and alcohol consumption should not be an evidence-based reason for antiviral treatment delay. 15
Active HCV infection is associated with ALD and a high risk of liver fibrosis progression among patients of the general population who are infected with HCV.14,15 Our study extends these findings to PWID receiving care at drug treatment centers. In this study, approximately 20% of patients had detectable HCV RNA, suggesting a high prevalence of active hepatitis C infections in this patient population.16,17 Most patients participating in the on-site DBS testing program had a previous HCV diagnosis (median time between diagnosis and participation: 7 years). The implementation of on-site HCV testing at drug treatment centers confirmed that providing HCV treatment to this population remains a logistical and clinical challenge, highlighting the importance of on-site evaluations and treatment. 18 Recent studies report that linking the diagnosis and treatment of HCV infections is the most effective method to prevent ALD and eliminate HCV infections among PWID.19 -21
Late presentation of ALD is common among people with chronic viral hepatitis.22,23 Age, years since HCV diagnosis, and HCV transmission route are risk factors for late presentation. 24 These risk factors are present in the majority of patients in this study; therefore, policies and plans to reduce late presentation should be prioritized at drug treatment centers. Additional risk factors associated with ALD in this patient population are likely related to social factors. These factors should be identified to reduce ALD. Arterial hypertension, obesity, diabetes, and the use of statins have not been associated with fibrosis in this population. However, high-risk alcohol consumption has been reported to be independently associated with the risk of developing ALD over time in this population, as alcohol abuse increases the risk of liver disease progression in patients with HCV infections.25,26 In this study, nearly half of the patients reported alcohol intake and approximately 25% reported high-risk consumption.
These results are consistent with a previous population-based cohort study. 27 The real-life clinical data used in this study revealed that patients with high-risk consumption have a higher risk of fibrosis progression, but not patients with any level of alcohol consumption. As HCV treatment in this patient population is often controversial due to the theoretical interactions of risk factors of liver disease (including alcohol), these results may significantly affect the HCV treatment received by patients in drug treatment centers. 28 Patients who abstain from alcohol, including those with a history of an alcohol use disorder, have similar behavioral treatment-related risks as patients without a history of alcohol use, supporting the theory that these patients should be considered as candidates for HCV treatment. 29 Moreover, a recent study found that HCV treatment does not increase injection drug use or risk behaviors among PWID and that drug or alcohol use remains stable or decreases slightly during the 2-year follow-up period after treatment. 10
However, patients with high-risk alcohol consumption have a high risk of liver fibrosis progression. Therefore, prompt treatment is recommended to reduce virus transmission and avoid late presentation. 13
The study suggests that non high-risk alcohol consumption does not contribute to liver fibrosis progression; however, alcohol cannot be considered a non-risk factor in other scenarios. The amount of alcohol intake necessary to increase the risk of liver fibrosis in patients with HCV infection is controversial; it may be assumed that no level of alcohol consumption is safe for patients with chronic hepatitis C. 30 Patients with non-active HCV infections had a lower risk of ALD regardless of alcohol intake. This may be related to the relatively short follow-up period compared to the natural history of alcohol liver disease. In any case, patients at drug treatment centers should be advised to stop drinking alcohol as part of the standard care for alcohol abuse and use disorder treatments.31,32
Genes related to liver fibrosis progression were not investigated in this study. 33 Future studies should explore genetic polymorphisms that may protect or worsen alcohol effects or aggressive fibrosis progression. The underrepresentation of women limits the ability to explore potential gender differences in fibrosis progression and response to interventions. Future studies should aim to include a more balanced gender distribution to assess sex-specific outcomes and improve generalizability. 34 In addition, the social stigma associated with HCV and alcohol-related liver disease remains a significant barrier to testing and treatment, particularly among PWID. 35 More research is needed to understand the roots of this stigma in the local context and to evaluate the impact of community-based interventions aimed at destigmatization. Finally, the findings of this study may be influenced by local determinants—such as access to healthcare, cultural attitudes toward drug use, and the structure of drug treatment centers in the Canary Islands—which may limit external validity. 36 Comparative studies across different regions could help identify contextual factors and expand generalizability.
This study has limitations. First, the alcohol consumption may have been underreported by the participants. However, a low cut-off was used to define alcohol abuse (⩾20 g/day for women and ⩾40 g/day for men), which may have compensated for the underreporting. Second, although questionnaires were made during the follow-up, alcohol consumption may have varied between appointments. Third, there is absence of a prior sample size or power calculation. Given the retrospective nature of the analysis, all eligible patients from the existing database were included, but the study may be underpowered to detect certain subgroup differences or small effect sizes. Lastly, the fibrotic scores may have overestimated the fibrosis stage as alcohol consumption can elevate GGT, AST, and cholesterol levels (32). However, fibrosis scores originally studied for HIV and HCV fibrotic staging have been applied for different liver diseases, including alcohol abuse, and their use in this study is acceptable.37,38 In addition, a high cut-off for advanced liver disease was used in this study.
This study provides relevant insights for a range of stakeholders involved in HCV care. Primarily, the findings are useful for drug treatment centers professionals and addiction services aiming to optimize screening and treatment pathways. Policy makers and public health authorities could also use these results to support the implementation of decentralized and stigma-sensitive testing strategies. Given the identified risk factors, including age, active HCV infection, and high-risk alcohol use, targeted interventions for these populations should be prioritized to prevent late-stage liver disease. Therefore, this study reinforces the value of on-site testing programs and highlights the urgent need for policies promoting early diagnosis and treatment, regardless of alcohol consumption status.
In conclusion, patients with active HCV infections receiving care at drug treatment centers who are >49 years old have an increased risk of liver fibrosis progression to ALD, especially those who are high-risk alcohol consumers. These findings support immediate initiation of HCV therapy regardless of alcohol consumption, and highlight the value of integrating decentralized testing and care models into addiction services to accelerate HCV elimination.
Supplemental Material
sj-docx-1-sat-10.1177_29768357251347821 – Supplemental material for Alcohol Consumption Impacts Liver Fibrosis Progression in Minority of Patients Screened for Hepatitis C at Drug Treatment Centers
Supplemental material, sj-docx-1-sat-10.1177_29768357251347821 for Alcohol Consumption Impacts Liver Fibrosis Progression in Minority of Patients Screened for Hepatitis C at Drug Treatment Centers by Alberto Hernández-Bustabad, Dalia Morales-Arraez, Ruth Ramos, Luz Goretti Santiago Gutiérrez, María Jesús Medina Alonso, Zaida Ramos-Marrero, Victor Pérez Pérez, Eva Molina Carballo, Felicitas Diaz-Flores, Julia Nazco, Fernando Gutiérrez and Manuel Hernández-Guerra in Substance Use: Research and Treatment
Footnotes
Acknowledgements
We thank BIOAVANCE and CIBICAN for editorial support.
Abbreviations
HCV: hepatitis C virus; DTC: drug treatment centers; DBS: on-site dried-blood-spot; ALD: advanced liver disease; PWID: people who injected drugs; HIV: human immunodeficiency virus (HIV); HBV: hepatitis B virus; ALT: alanine aminotransferase; AST: aspartate aminotransferase; GGT: gamma-glutamyl transpeptidase; VCM: mean corpuscular volume; INR: international normalized ratio.
Author Contribution
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by Alberto Hernández-Bustabad and Manuel Hernández-Guerra. The first draft of the manuscript was written by Alberto Hernández-Bustabad and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported in part by grants from Fondo Europeo de Desarrollo Regional (FEDER). Dr. M. Hernandez-Guerra is the recipient of a grant from Instituto de Salud Carlos III (PI19/01756).
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. M. Hernandez-Guerra has received research grants from Abbvie and Gilead and has participated in consultant advisories for Bayer and Intercept. All other authors report no conflicts of interest.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
