Abstract
Background:
The United States is grappling with an unprecedented overdose crisis, exacerbated by the proliferation of potent synthetic opioids like illicitly manufactured fentanyl. Despite the efficacy of methadone treatment in managing opioid use disorder, regulatory barriers hinder its widespread utilization. This article examines the complex landscape of methadone regulation across federal, state, and local levels, highlighting disparities and opportunities for reform.
Issue:
The COVID-19 public health emergency prompted temporary flexibility in methadone regulations, including expanded take-home doses and telehealth counseling, leading to improved treatment experiences and retention. Permanent revisions to federal guidelines have since been introduced by the Substance Abuse and Mental Health Services Administration, reflecting a progressive shift toward patient-centered care and streamlined access. State regulations, managed by Single State Agencies and State Opioid Treatment Authorities, vary widely, often imposing additional restrictions that impede access to methadone treatment. Local OTP clinics further exacerbate barriers through stringent policies, despite federal and state guidelines advocating for flexibility.
Recommendations:
Coordinated efforts among policymakers, healthcare providers, and communities are needed to promote the development of accountability measures, incentives, and community involvement to ensure equitable access and quality of care. To truly meet the demand needed to end the existing overdose crisis and enhance accessibility and comprehensive healthcare services, methadone treatment expansion beyond traditional OTP settings into primary care offices and community pharmacies should take place.
Highlights
Regulatory flexibility during COVID-19 improved methadone treatment experiences and retention, highlighting the potential benefits of permanent regulatory reforms.
Permanent revisions to updated federal guidelines are a welcome step toward improving methadone access, though establishing quality metrics and accountability measures, with input from patients with lived experience, will be crucial for meaningful reform.
The Association for Multidisciplinary Education, Research, Substance Use, and Addiction (AMERSA) advocates for expanded methadone treatment, including into primary care and community pharmacies, emphasizing the need for coordinated efforts to enhance accessibility and combat the overdose crisis.
Background
The overdose crisis in the United States has become one of the most pressing public health challenges of our time. National overdose deaths, often resulting from illicitly manufactured fentanyl and other potent synthetic opioids, exceeded 100 000 deaths in 2021 and 2022. 1 This crisis is further underlined by the fact that Black/African American individuals have 7 times higher overdose rates compared to their White peers. 2 Although various interventions have been deployed, the underutilization of methadone treatment remains a critical oversight in the country’s response. Methadone, a long-acting opioid agonist, has been a stalwart treatment in addiction medicine for decades. Its pharmacological properties, including its ability to occupy opioid receptors and suppress withdrawal symptoms, make it an invaluable tool in managing opioid use disorder (OUD), resulting in improved outcomes such as reduced overdose mortality, reduced opioid cravings and withdrawal, and overall improved quality of life. 3
Despite its proven efficacy, accessing methadone in the United States remains challenging due to regulatory barriers. While patients with chronic pain can access methadone from any pharmacy, methadone to treat OUD is only available through opioid treatment programs (OTPs), known colloquially as “methadone clinics.” Federally, the Substance Abuse and Mental Health Services Administration (SAMHSA) administers the regulatory framework for methadone treatment, while the Drug Enforcement Administration (DEA), within its mandate to enforce drug regulations, aims to prevent methadone diversion by requiring each clinic to submit a diversion safety plan. 4 In addition, individual states play a large role in overseeing OTPs, imposing their own regulations and licensing requirements which are often more restrictive than federal policies. Local county and city jurisdictions as well as OTPs’ own programmatic policies offer further restrictions, such as the hours of operation and location of methadone clinics, leading to a complex web of regulations that shape the accessibility and delivery of this essential treatment.
Patient advocate groups have critiqued the methadone treatment systems as “a culture of cruelty.” 5 Patients report having to comply with a series of onerous rules that are more aligned to carceral settings than to medical treatment centers and desire more tailored options to meet their specific needs. These rules include having to attend the OTP for daily methadone dosing under the watch of security guards and cameras, submitting frequent urine drug tests that are observed by 2-way mirrors or video cameras, being mandated to attend counseling, and other restrictive requirements that make long-term retention on methadone challenging.5,6
The complex interweaving of regulatory control at various levels was intended to create layers of protection from methadone diversion and overdose in the name of public safety. 4 However, many regulatory barriers were relaxed during the COVID-19 public health emergency. Early evidence suggests these feared consequences of loosened restrictions did not come to fruition, and both patients and clinicians experienced improved quality of care leading to increased treatment retention.7 -11 In light of the escalating overdose crisis, policymakers and researchers are increasingly interested in reexamining and reforming methadone treatment. This layered and shifting landscape of methadone regulation underscores the need for coordination among federal, state, and local entities as well as OTPs themselves to ensure the effectiveness and safety of methadone treatment while addressing the ongoing overdose crisis. A progressive shift toward modernizing methadone treatment is occurring, with the goal of increasing patient centeredness, reducing barriers to treatment, and further integrating it into evolving models of healthcare delivery outside of traditional OTPs. 12 In this perspective, we review the regulatory bodies at each level of governance, elucidating areas for future policy reform, and propose a vision for the future of methadone treatment (Table 1).
Policy Reforms and Levels of Action to Bridge Methadone Treatment Gaps.
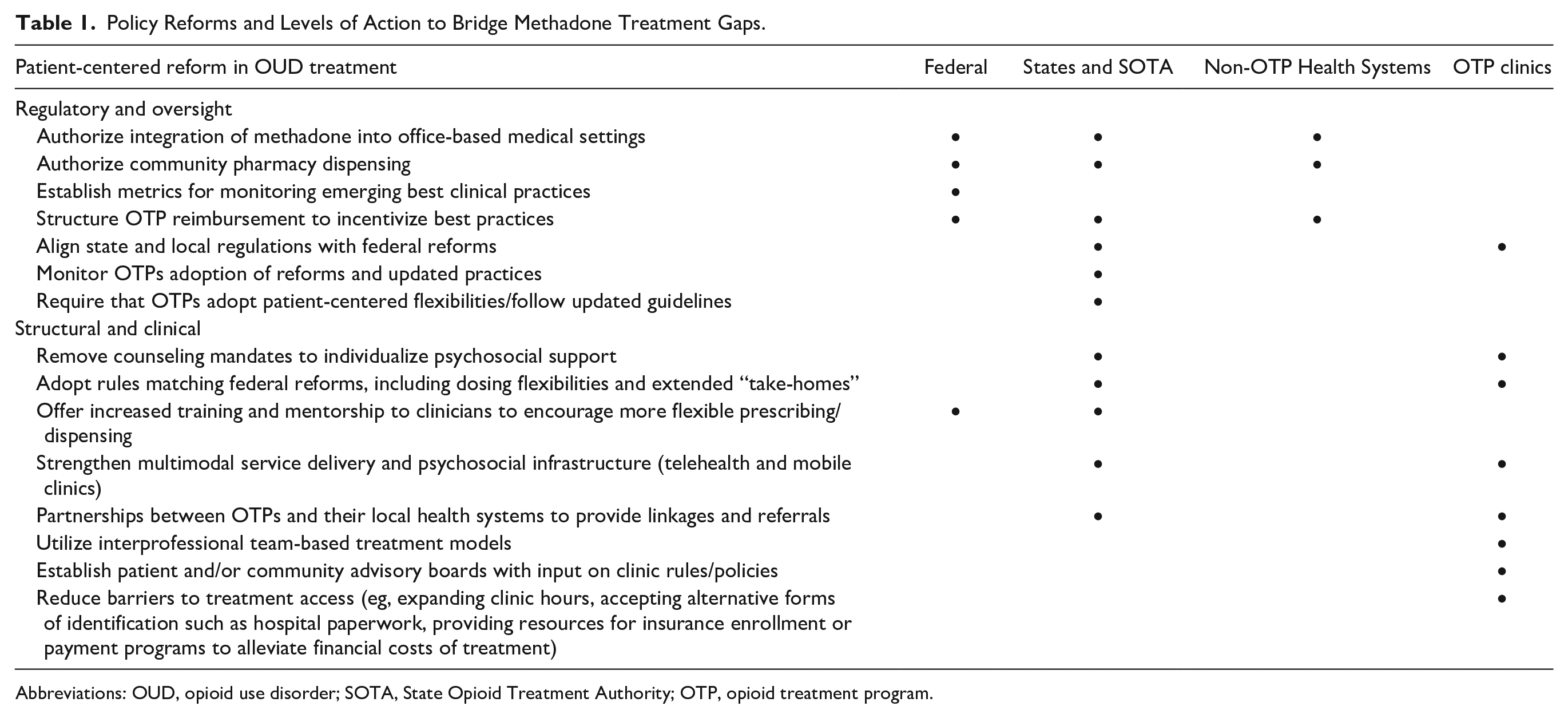
Abbreviations: OUD, opioid use disorder; SOTA, State Opioid Treatment Authority; OTP, opioid treatment program.
Methadone Federal Regulations and COVID-19
At the federal level, SAMHSA and the DEA are the agencies responsible for overseeing methadone treatment. SAMHSA provides the regulatory framework for methadone through the Code of Federal Regulations (CFR) in Title 42 CFR Part 8. 4 These regulations dictate the standards for methadone treatment, including rules on program accreditation, DEA registration, medication dispensing, and patient enrollment. OTPs must meet accreditation requirements and are subject to regular audits from the Joint Commission, state health agencies, and other regulatory bodies to ensure compliance with set statutes and guidelines.
Prior to COVD-19, the most widely known regulation from 42 CFR Part 8 was the requirement for most individuals, particularly those new to treatment, to present to the OTP daily to receive their methadone dosing. Only individuals with sufficient time in treatment (ie, at least 3 months) and meeting strict 8-point criteria were allowed unsupervised “take-home” doses. 4 This requirement often resulted in patients having to commute upward of an hour or more each way in the early morning, interfering with responsibilities such as employment, caregiving, and other necessary life activities.8,13
During the COVID-19 public health emergency, regulations underwent significant adjustments to ensure the continuity of methadone treatment while adhering to public health guidelines. SAMHSA introduced temporary flexibilities, including allowing for take-home doses and telehealth counseling appointments to reduce in-person interactions at clinics and minimize the risk of virus transmission. 14 Individuals, regardless of their time in treatment, were newly allowed by federal and state agencies to receive up to 14 to 28 take-home doses at a time at the discretion of the OTP medical director. In total, 43 states adopted methadone flexibilities during the COVID-19 pandemic.15,16 While the majority of states did apply for exemptions, not all clinics adopted them and not all patients experienced them. 17 Yet while most clinics did not take full advantage of the flexibilities, when adopted, the relaxations proved vital in maintaining access to methadone treatment amid the challenges posed by the pandemic. 18 Increasing research has found that loosening of regulations did not result in anticipated harms, such as increased overdose deaths. 19 Rather, patients and providers reported improved treatment experiences and retention in several early studies.7-11
Recognizing the potential benefits of these changes, SAMHSA announced the release of new revised guidelines of 42 CFR Part 8 in January 2024, representing the most significant updates to methadone regulations in over 30 years. 20 The revisions made the COVID-19 take-home exemptions permanent, allowing up to 7 days of take-homes in the first 2 weeks of treatment and up to 28 days after the first month. The new rules also notably removed the federal 8-point criteria for take-home dosing, no longer requiring negative urine drug testing to show abstinence, and instead implementing a 5-point criteria that largely relies on the judgment of OTP clinicians to determine risk and benefits of increased take-homes. 15 Other major changes included removing the requirement for patients to have a 1-year history of OUD to be eligible for methadone treatment, increasing allowances for methadone dosages on day 1 of treatment, allowances for telehealth, and updating terminology and definitions to reflect updated medical terminology, among several other changes. 20
In parallel, members of Congress are demonstrating growing interest in reforming the methadone treatment system. The Modernizing Opioid Treatment Access Act of 2023 (M-OTAA) (H.R.1359 and S.644 in the House and Senate, respectively) is a bipartisan bill that would allow physicians board certified in Addiction Medicine or Addiction Psychiatry to prescribe methadone outside of OTP settings and dispense it in community pharmacies, as is done in many other countries. The bill has received broad endorsement from national organizations, including the Association for Multidisciplinary Education and Research in Substance use and Addiction, the American Medical Association, the American Society of Addiction Medicine, and the American Pharmacists Association. The Senate version of the bill was approved by the Senate Health, Education, Labor, and Pensions Committee in December 2023 and currently awaits further consideration and voting by the full Senate and House before Presidential approval.
Methadone Regulation at the State Level
In addition to federal regulations, each US state has a designated Single State Agency (SSA), charged with managing federal funds dedicated to addressing substance use prevention, treatment, and recovery, with the vast majority of funding coming from SAMHSA. 21 Established through various laws in the 1970s (including the Comprehensive Alcohol Abuse and Alcoholism Prevention, Treatment and Rehabilitation Act of 1970, the reauthorization of the Hughes Act in 1974, and then strengthened through incentive grant programs established by the earlier Uniform Act of 1971), SSAs were designated as the exclusive bodies eligible for seeking and obtaining federal funding for tackling alcohol and drug use and addiction, as well as setting the state’s default policies and regulations regarding substance use services. 21
Each state’s SSA designates an individual who serves as the State Opioid Treatment Authority (SOTA). 22 The SOTA provides oversight and administrative guidance and support to OTPs; with primary duties including monitoring OTP operations and regulatory compliance; serving as a liaison between OTPs, SAMHSA, and the DEA; and managing patient or clinic complaints, requests, and exemptions. 23 Individual SOTAs have significant influence on SSA policies and practices regarding OTPs. There is wide variability in how SOTAs interpret federal guidelines and what practices they permit. Given the complex regulatory environment, SSAs and local OTPs must defer to SOTAs to interpret regulations and determine what is allowable. For the most part, very little is known about what, if any, oversight each SOTA receives from SAMHSA or SSAs, raising concerns about accountability for a position that yields so much power. Furthermore, questioning SOTAs may be challenging given they are also often involved in state funding allocations. Because of their significant role in state-level decision-making, the role of the SOTA presents a potential target that may warrant evaluation and reform for increased transparency.
States are bound by the requirement to be at least as strict as federal regulations demand. They will also often add restrictions to methadone treatment, impeding access to treatment and reducing quality of care, and states with the highest need for methadone treatment access tend to be those with the highest number of regulatory restrictions.24,25 For example, while federal regulations require counseling “as clinically necessary,” approximately half of the states mandate minimum monthly counseling requirements, including states like New York and California.24-26 Other examples of added state-level restrictions include increasing the annual number of required random drug tests, or increasing required time in treatment and/or imposing other criteria for take-home dose privileges. Some states also have restrictions limiting new OTPs from opening, including zoning restrictions on OTP locations, onerous approval processes, and bans on new OTPs. These restrictions have thus created “methadone deserts” in parts of the country, where as of 2019, 80% of US counties did not have an OTP. 27
Regulation at the Local City and Clinic Levels
Within particular OTP agencies and clinics, clinic administrators and medical directors have the authority to enact stricter policies than required by federal and state regulations. Individual OTP medical directors and clinical practitioners have significant discretion and influence on policy and practice, often choosing not to grant certain permissions to OTP clients, despite the ability to do so according to federal and state guidelines. For example, in its guidelines regarding the extension of take-home dosing flexibilities, SAMHSA writes, “OTP decisions regarding dispensing methadone for unsupervised use under this exemption shall be determined by an appropriately licensed OTP medical practitioner or the medical director.” 15 Studies finding that many OTPs did not enact federal flexibilities during the COVID-19 pandemic, underscoring how federal action alone was not enough to drive meaningful change without sufficient buy-in and subsequent implementation from local OTPs. 28
Opportunities for Reform at All Levels: The Need for Accountability and Incentives
While OTPs must comply with state and federal regulations, each clinic can set its own rules and can take steps to make care more accessible. 12 For example, OTPs typically only operate between the hours of 6:00 am and 2:00 pm, with some open for only 2 to 3 hours a day. They can expand hours of operation to increase accessibility for dosing and enrollment. They can waive requirements for identification cards, and ensure intake processes are not more burdensome than required by existing regulations. OTPs in states allowing take-home expansion can expand access to take-home doses, as well as incorporate varied avenues of treatment delivery including robust telehealth counseling and mobile clinic options. Many of these strategies have already demonstrated efficacy and safety during the COVID-19 public health emergency.7-11 OTPs can establish community advisory boards of patients with lived and living experience on methadone, including those with ongoing drug use to provide direct (and financially compensated) feedback to clinics on ways to improve patient experience.
To be sure, OTPs, particularly those receiving public funding, often already face challenges with staffing and resources. Ensuring OTP clinics implement new reforms will require development of new systems of accountability and incentives from state and federal governments. Currently, SAMHSA statutes and regulations explicitly “preserve states’ authority to regulate OTPs,” 29 and can only “describe a minimum acceptable standard for the operation of OTPs” while allowing states and individual OTPs to be the final decision-makers for clinical care delivered. 30 Overriding state authority to enact all federal OTP reforms would be unrealistic given the nature of state and clinical authority. However, SAMHSA can establish a new system of quality and accountability metrics that encourage adoption through partnerships with health systems and patient advocacy organizations. SAMHSA can link federal funds to newly created accountability metrics, pinpointing OTPs that adhere to outdated methods, and rewarding clinics who prioritize the integration of patient oriented-flexibilities.
To ensure all patients have equitable access and support to receive extended take-homes, relaxed entry criteria, and other already-established allowances, SAMHSA and SSAs should reexamine enforcement mechanisms and add metrics examining take-home receipt in their OTP monitoring and certification processes. For example, OTPs with significantly low offerings of take-home doses (eg, short durations to few clients) could be flagged for additional auditing measures to encourage OTPs to establish and maintain increased take-home access.
Notably, financial structures of methadone reimbursement services vary between clinics and states and must be examined to ensure alignment with improved outcomes and patient care goals. OTP clinics often under a fee-for-service model receive most of their funding through reimbursements for in-person services, such as dosing or counseling. Clinics may lose significant portions of their funding by offering more take-home doses, misaligning financial structures against treatment progress.31-33 States could remedy this problem through restructuring their Medicaid billing guidelines into bundled payment plans, as has been done in New York and Massachusetts, or alternative payment models that factor in quality of care metrics.31,34
Most important, the development of these new metrics, accountability measures, and incentives must involve the input of communities with lived and living experience of methadone treatment. Through the history of methadone treatment, those profoundly affected are often sidelined, resulting in regulations that fail to prioritize patient needs and realities. To avoid perpetuating our current system of high-barrier, nonpatient-centered methadone treatment, it is imperative to engage directly affected communities at all levels of regulatory reform to ensure meaningful and lasting transformation.
Moving Methadone Beyond the OTP
For the United States to truly advance more equitable access to evidence-based methadone treatment, we argue methadone must expand beyond the walls of traditional OTP service delivery models. 35 This must happen through concerted action by both OTP and non-OTP actors, enabled by policies built on patient priorities and modern research. 36 Methadone treatment must be integrated into office-based medical care, as it is in Australia, Canada, and throughout Western Europe.37,38 To be politically strategic and address initial concerns, this could begin in a stepwise fashion by first enabling board-certified addiction medicine specialists to prescribe methadone for dispensing at community pharmacies, with handoff to primary care clinicians after medication stabilization.37,39 Decades of experience in non-US settings support the efficacy and safety of such approaches, though further study should investigate implementation best practices and the impact of these changes in the US context on treatment access and efficacy, balancing risks such as adverse drug effects. 36 Once safety and effectiveness of such practice is established, all clinicians licensed by the DEA ultimately should be allowed to receive training and longitudinal mentorship in methadone prescribing and integrate it into their practice if desired. Because the supply of addiction-licensed physicians will never be enough to meet the demand for methadone treatment, expanding methadone into primary care is a necessary step that will require the coordination and partnership of federal, state, and local regulatory agencies. 40
Some argue that OTPs provision of psychosocial services and oversight alongside medication cannot be replicated successfully by clinicians prescribing outside of OTPs. While psychosocial services and formal treatment infrastructure are helpful for many individuals and should be made available to any individual seeking such services, pairing behavioral treatments alongside methadone treatment has not been found to be more effective than methadone medication treatment alone.41,42 Mandates to require counseling have also been seen as barriers for patients to engage in methadone treatment. 5
Providing multimodal treatment is not solely the purview of OTPs. Primary care is by far the largest provider of integrated healthcare and psychosocial services nationally, and many addiction specialists work within health systems that are equipped with behavioral health and case management support. Primary care clinics are also better equipped to screen, prevent, and manage many of the conditions related to OUD, including infectious diseases like hepatitis C and HIV. Bolstering treatment infrastructure in primary care and other community settings enables people with OUD to access whole-person healthcare through a team-based approach informed by robust experience with chronic disease management. This strategy recognizes there is no “one size fits all.” By offering patients the choice of selecting where they receive their methadone, they can be empowered to better meet their individual needs.
Critics also state that the relatively complex pharmacology of methadone renders it too dangerous for use outside of OTPs. In reality, any licensed healthcare provider can already prescribe methadone for chronic pain, along with several other widely used drugs with narrow therapeutic indices and significant potential adverse effects, like anticoagulants and insulin. While we agree training is necessary to safely prescribe and monitor methadone treatment, it is not beyond the capabilities of trained clinicians, especially in interprofessional practices. Such a model of primary care prescribing for methadone has also been effective in countries like Canada, the United Kingdom, and Australia. 37 Oversight by SAMHSA could help ensure that methadone prescribing is being safely integrated into community settings, and AMERSA would support SAMHSA championing the integration of methadone training into all healthcare professional programs. Moreover, it is imperative for leaders in health systems, clinics, and pharmacies to commit to establishing and maintaining the necessary framework to facilitate methadone treatment in nonspecialized settings.
Conclusion
Though federal policy change is necessary to unfetter methadone as a treatment modality, it is not sufficient by itself. National leaders in substance use policy, research, and clinical care must create training and oversight infrastructure for individuals who wish to integrate methadone into their practice. States and communities must ensure that their policies are not more restrictive than federal regulations and incentivize expansion of methadone treatment through investment in both OTP-based services and non-OTP treatment avenues. Locally, leadership within health systems, clinics, and pharmacies must be willing to develop and sustain infrastructure to support methadone treatment in office-based settings pursuant to SAMHSA oversight and regulations. To promote accountability, adoption of best practice, equity, and improvement, there also must be greater transparency in and evaluation of the policies that govern methadone across layered regulatory levels.
If we are to achieve progress in realizing methadone’s potential as treatment, it will take dedication, cooperation, and perseverance among a diverse set of stakeholders. The payoff will be a more accessible and equitable pathway to safety and quality of life for millions of individuals.
Footnotes
Acknowledgements
Position statements clarify key issues that are in alignment with the vision, mission, and values of AMERSA, Inc. This Position Statement, endorsed by their Board of Directors on March 27, 2024, amplifies the position of the organization, guides their activities, and informs the public and policy makers on the organization’s stance on this issue.
Author Contributions
All authors were involved in the conceptualization of the manuscript. LWS, MI, and RW wrote the original draft. All authors contributed substantially to its revision.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: LWS and CS are board members of the National Coalition to Liberate Methadone. JB is a consultant on a grant awarded to Brandeis University from the Pew Charitable Trusts to develop business models on pharmacy methadone dispensing.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Compliance,Ethical Standards,and Ethical Approval
Institutional Review Board approval was not required.
