Abstract
Background:
Addictive disorders are significant global public health burdens. Treatment uptake with these disorders is low and outcomes can be mixed. Electronic screening, brief intervention, and referral to treatment (e-SBIRT) programs have potential to improve uptake and treatment outcomes. To date, however, no prior review of the literature has been conducted to gauge the effectiveness of e-SBIRT for addictive disorders.
Methods:
We conducted a systematic review and meta-analysis of the literature concerning e-SBIRT for addictive disorders by surveying the MEDLINE, PubMed, Web of Science, Scopus, Embase, and PsycInfo databases on January 17, 2023.
Results:
Ten articles were included at analysis reporting evaluation of e-SBIRT interventions for substance use disorders including alcohol use in a variety of settings. No articles were identified regarding treatment for behavioral addictions such as disordered/harmful gambling. Meta-analysis found e-SBIRT to be effective at reducing drinking frequency in the short term only. e-SBIRT was not found to be advantageous over control conditions for abstinence or other treatment outcomes. We identified and described common components of e-SBIRT programs and assessed the quality of available evidence, which was generally poor.
Conclusion:
The present findings suggest that research regarding e-SBIRT is concentrated exclusively on higher-risk substance use. There is a lack of consensus regarding the effectiveness of e-SBIRT for addictive disorders. Although common features exist, e-SBIRT designs are variable, which complicates identification of the most effective components. Overall, the quality of outcome evidence is low, and furthermore, high-quality experimental treatment evaluation research is needed.
Keywords
Highlights
Electronic Screening, Brief Intervention and Referral to Treatment (e-SBIRT) programs have potential in identifying persons at risk of addiction related harms, providing limited help and support, and facilitating entry to treatment services.
e-SBIRT vary in composition, but are unified by the inclusion of Motivational Interviewing (MI) techniques.
The current review finds that the evidence base in support of e-SBIRT is limited and of low quality, and so the effectiveness of e-SBIRT for people at risk of addictive disorders remains in equipoise.
Introduction
Substance use disorders (SUDs) and disordered gambling present considerable public health burdens1,2 and commonly co-occur. 3 Although they are not both listed as addictive disorders in fifth edition of the Diagnostic and Statistical Manual of Mental Disorders (DSM V), 4 they share an urge to engage in a behavior (eg, gambling or substance use) despite harmful negative consequences or a desire to stop. 5 A range of mental and physical health problems are associated with harmful (eg, causing significant problems or distress to the individual and others close to them 6 ) use of substances like alcohol, 7 cannabis, 8 opioids, 9 and cocaine. 10 Similarly, disordered or harmful gambling is associated with physical and psychological ill-health and an increased risk of suicide compared to the general population.11,12 Despite being at increased risk of health problems, persons at higher risk of substance use-related harms infrequently use specialist treatment services.13,14 This lack of service access can be attributed to many factors including transport, insurance cover where applicable, and stigma. 15 Similarly, only a quarter of those at risk of harmful gambling, and one-fifth of those actively experiencing harmful gambling, seek help for gambling-related harms. 16
Screening, brief intervention, and referral to treatment (SBIRT) is an approach that can be used to identify persons at risk of drug or gambling-related harm, provide intervention to reduce risky behaviors, and/or facilitate entry to treatment services. 17 Primarily, SBIRT is considered a routine preventative service delivered in settings to people who might not otherwise access help and who might not meet diagnostic criteria but are still engaging in harmful addictive behaviors. The aim of SBIRT programs is to rapidly assess the severity of problematic substance use (or other addictive behavior, such as gambling), and then provide either feedback and education regarding risk, brief intervention, and/or referral to formal treatment depending on the severity of the problem. 17 Frameworks such as those by the Substance Abuse and Mental Health Services Administration provide an outline for content, yet program delivery is variable with the individual practitioner highly autonomous. 18 Typically, SBIRT will be delivered in primary care settings by a qualified healthcare provider (eg, nursing, social work, or medical staff) beginning with the application of a rapid self-report screening tool. If the client is assessed as low risk for drug or gambling-related harm, then the healthcare provider will provide positive feedback about the client’s current usage and education regarding the dangers of increased usage. If the client is found to be at moderate risk of harm, then the healthcare provider will deliver a brief intervention designed to motivate changes which might include reduced usage, help-seeking, or both. The brief intervention will usually include elements of education about the harms of continued usage at the reported levels and Motivational Interviewing (MI) exercises. If the client is deemed to be at high risk of harm, the healthcare provider will offer or advise a referral to treatment.
These programs have been found to be clinically and economically effective in reducing risks related to harmful substance use19,20 but there is a comparative paucity of evidence in relation to disordered/harmful gambling compared to substance use. This is likely related at least in part to the origins of SBIRT as a means of managing hazardous drinking 21 and the absence of clinical and empirical extension to disordered gambling.
Generally, SBIRTs vary in their structure, delivery, and timescales. 22 Practitioner-delivered SBIRT is rarely applied in emergency settings (other than for problem drinking) where staff are limited by time pressures and unable to deliver interventions for a wider range of problems. 23 The use of SBIRT programs to address harmful substance use is also limited in primary care, despite calls for their increased use in these settings. 24
Client-accessed electronic or e-SBIRT programs via software applications or websites have gained provenance in recent years. 25 However, there is a paucity of research evidence regarding the effectiveness of e-SBIRT compared to traditional SBIRT or lack of intervention.26,27 A systematic review of the literature concerning e-SBIRT for addictive disorders would therefore be of value to researchers and service providers.
Current Study
We sought to conduct the first systematic review and meta-analysis to identify the most effective models of e-SBIRT design and delivery for the treatment of addictive behaviors. As a secondary aim, we sought to identify knowledge gaps and make recommendations for future research.
Methods
We carried out a systematic review and meta-analysis in accordance with PRISMA (Preferred Reporting Items for Systematic reviews and Meta-Analyses) 28 and Cochrane 29 guidelines.
Inclusion and Exclusion Criteria
Our population of interest were adults at risk of experiencing or currently experiencing any addictive disorder with or without formal diagnosis. We defined e-SBIRT as any SBIRT program which is delivered electronically and entirely or partially self-accessed by a member of the target population. Peer-reviewed articles reporting experimental or quasi-experimental studies of e-SBIRT making between or within-group comparisons were included. Our outcomes of interest were broad encompassing any reported changes in help-seeking behavior, addictive behavior, or related symptomatology such as financial worry or depression. We decided on these outcomes of interest as encompassing the aims of SBIRT as a means of reducing the prevalence of adverse consequences of substance misuse. 21 Studies conducted in any clinical or research setting were included. Table 1 shows the inclusion and exclusion criteria summarized using a Population, Exposure (or intervention), Comparator, Outcomes, Timings and Settings (PECOTS) format.
PECOTS Inclusion and Exclusion Criteria.
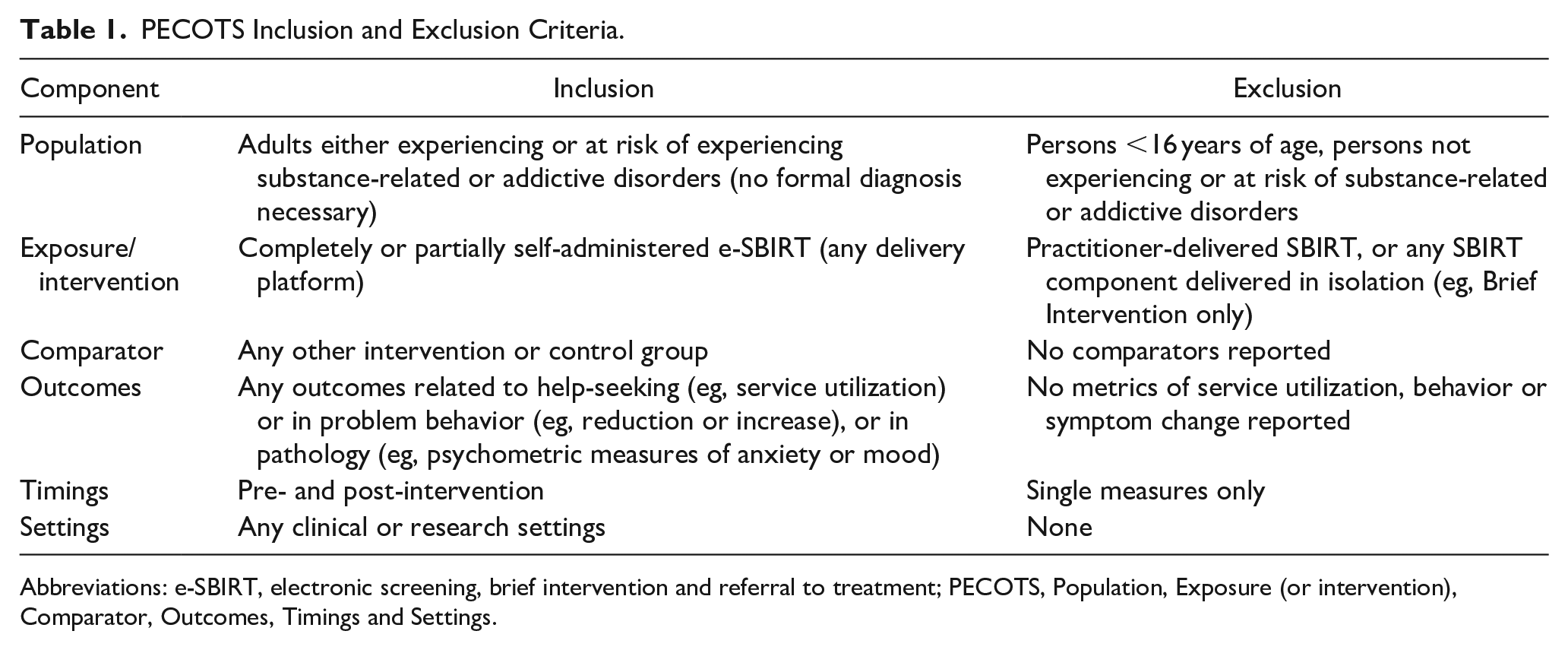
Abbreviations: e-SBIRT, electronic screening, brief intervention and referral to treatment; PECOTS, Population, Exposure (or intervention), Comparator, Outcomes, Timings and Settings.
Data Sources and Searches
We selected databases of peer-reviewed research articles pertinent to the fields of medicine including psychiatry (MEDLINE, PubMed), general healthcare (Web of Science, Scopus, Embase), and clinical or health psychology (PsycInfo). Our search strategy was developed to identify keywords related to e-SBIRT platforms and experimental or quasi-experimental research designs. Controlled vocabulary such as MeSH (Medical Subject Headings) terms were used where appropriate.
Our search strategy consisted of 4 text queries combined using parentheses and Boolean operators. The first 3 queries described the intervention of interest and read as: ((e-SBIRT OR “e SBIRT” OR eSBIRT) OR (digital OR e-health OR computer OR computerised OR computerized OR online OR internet OR electronic OR web OR web-based OR “web based” OR app OR app-based OR “app based”) AND (SBIRT OR “Screening, Brief Intervention and Referral to Treatment” OR “Screening, Brief Intervention, and Referral to Treatment” OR “Screening Brief Intervention and Referral to Treatment”)). The fourth text query described the study methodologies of interest and read as: (“randomized controlled trial” OR “randomised controlled trial” OR “non-randomized controlled trial” OR “non-randomised controlled trial” OR “natural experiment” OR trial OR “clinical trial” OR “nonequivalent group design” OR “non-equivalent group design” OR “nonequivalence group design” OR “non-equivalence group design” OR “pre and post test design” OR “pre-test post-test design”).
The search strategy was applied to 6 large databases and the results from each, along with the search field option used, are as follows: PsycInfo (All text) 78 records; Web of Science (All fields) 97 records; Scopus (Title, Abstract, Keywords) 85 records; MEDLINE (All text) 176 records; Pubmed (All fields) 99 records; and Embase 171 records. In total, our search returned 706 records, which was reduced to 311 following removals of duplicates (Figure 1).
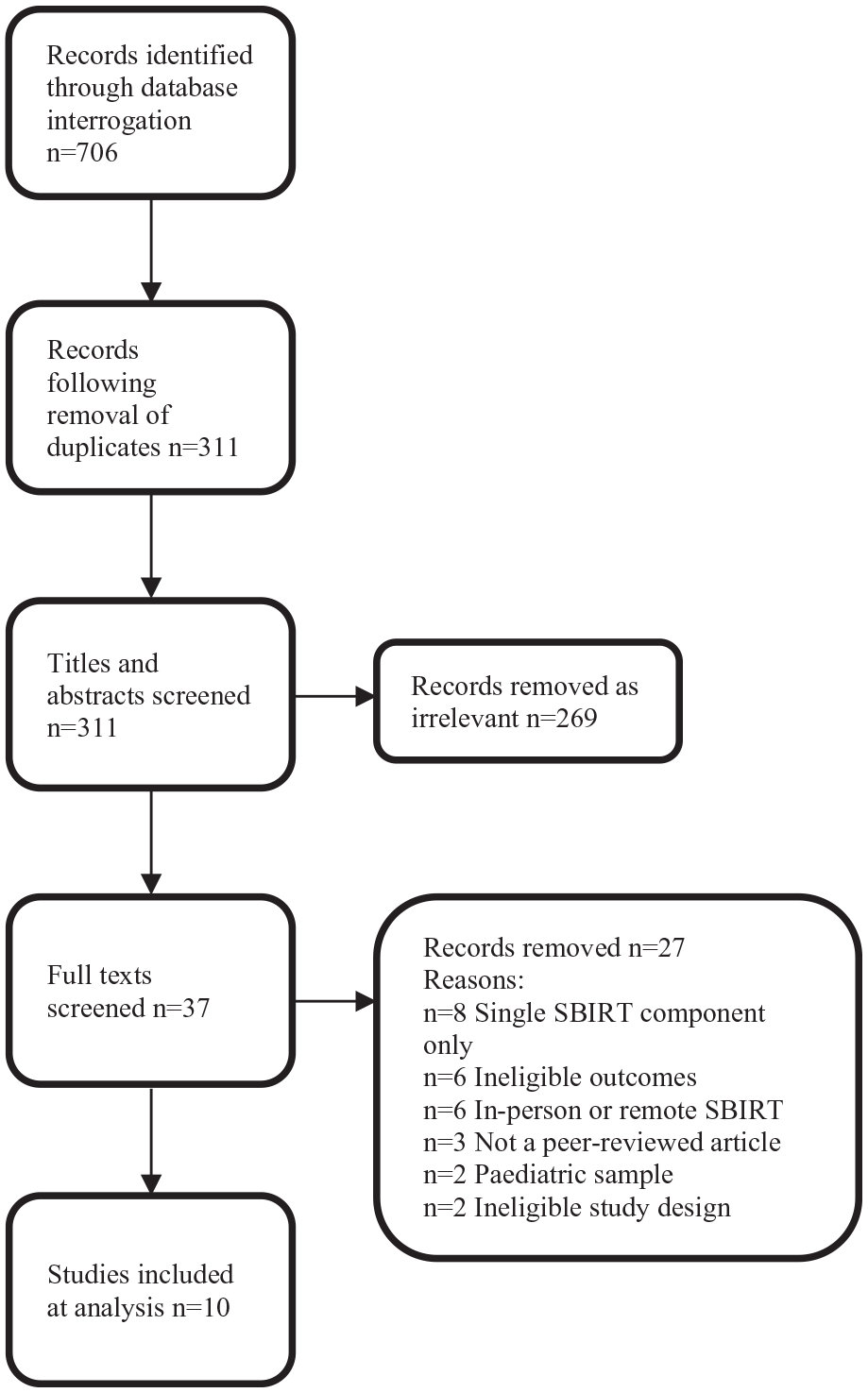
PRISMA flowchart.
Study Selection
We surveyed the databases using our search strategy on January 17, 2023, and all records were imported into Endnote 20. 30 Using the Covidence web-based application for undertaking systematic reviews, 31 reviewer 1 (MJ) surveyed the titles and abstracts of the imported records and excluded or included each record for further review based on the inclusion and exclusion criteria as summarized in Table 1. Reviewer 2 (CJS) also surveyed titles and abstracts against the inclusion and exclusion criteria. After agreement had been reached on the suitability for inclusion based on titles and abstracts, the reviewers surveyed remaining full-text records based on the inclusion and exclusion criteria (Figure 1). At all stages of screening and extraction, both MJ and JCS were blind to one another’s decisions. At the end of each stage of screening and extraction, conflicts were flagged by the Covidence system and discussed on a case-by-case basis by MJ and CJS with reference to the agreed inclusion criteria.
Data Extraction
We extracted study author names, publication title, year of publication, nation where the study was carried out, study design, sample size, comparators, screening method, brief intervention method, referral method, outcomes, and any conflict of interests.
Quality and Risk of Bias
The GRADE (Grading of Recommendation, Assessment, Development and Evaluation 32 ) checklist was employed to assess quality of outcomes over 5 domains of within-study risk of bias, imprecision, inconsistency, indirectness, and publication bias.
Synthesis and Analysis
Extracted study data were summarized and presented as a narrative synthesis of the findings. Quality and risk of bias was reported and described, as were gaps in the available evidence. Statistical meta-analysis of outcomes data was undertaken using RevMan analysis software. 33 Where authors reported continuous outcome data, calculated effect using mean differences with 95% confidence intervals (CIs) were reported. If different methods of measurement were used for comparable outcomes, we reported standard mean difference (SMD) with 95% CIs. Where discrete outcomes were reported, we calculated risk ratios (RRs) with 95% CIs. We contacted the authors of all the included articles and requested missing data where necessary.
Results
We included 10 studies at analysis evaluating 9 different e-SBIRT programs.
Demographics
The combined sample size of the 10 included studies was 4048 (SD 412.94). Weighted average attrition of participants from baseline data capture to final follow-up was 11.5% (SD 0.11). Total weighted average sex ratio was 56% female (SD 0.37) with 4 studies including only female participants.
Overview of Included Articles
A summary of the 10 included articles can be found in Table 2. All the included articles concerned use of addictive substances. The included articles reported trials carried out in a variety of settings. Four were conducted in perinatal and/or women’s health settings, 3 studies in emergency departments (EDs), 1 in probation services, 1 in a HIV clinic, and 1 among a subset of the general population who were registered with a private research company. Eight of the studies were carried out in the United States, 1 in France, and 1 in South Korea.
Study Characteristics.
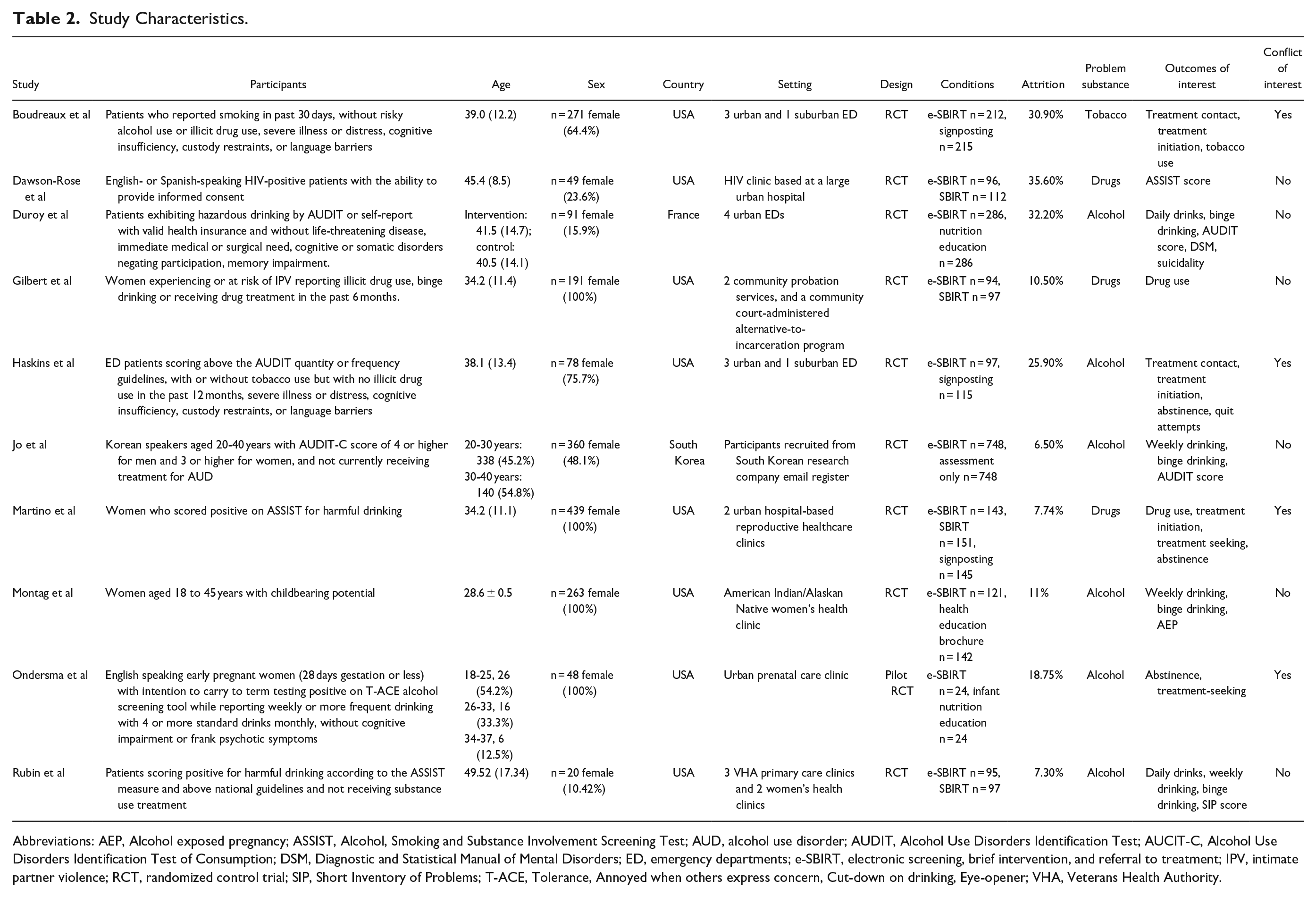
Abbreviations: AEP, Alcohol exposed pregnancy; ASSIST, Alcohol, Smoking and Substance Involvement Screening Test; AUD, alcohol use disorder; AUDIT, Alcohol Use Disorders Identification Test; AUCIT-C, Alcohol Use Disorders Identification Test of Consumption; DSM, Diagnostic and Statistical Manual of Mental Disorders; ED, emergency departments; e-SBIRT, electronic screening, brief intervention, and referral to treatment; IPV, intimate partner violence; RCT, randomized control trial; SIP, Short Inventory of Problems; T-ACE, Tolerance, Annoyed when others express concern, Cut-down on drinking, Eye-opener; VHA, Veterans Health Authority.
Eight studies reported on e-SBIRT in relation to alcohol-related harms, 2 were focussed on illicit substance use, and 1 study was concerned with tobacco use. No studies were concerned with the application of e-SBIRT to address behavioral addiction such as gambling.
All included articles reported randomized control trials (RCTs); however, one described a pilot study rather than a fully powered RCT. A variety of controls and comparators were used in the reported studies, with 3 studies comparing e-SBIRT to standard in-person SBIRT, 3 comparing e-SBIRT to signposting, 3 studies compared e-SBIRT to nutrition or health education, and 1 study compared e-SBIRT plus treatment as usual (TAU) with TAU alone.
e-SBIRT for Populations at Risk of Harmful Alcohol Use
Of the 4 articles reporting e-SBIRT and alcohol-related harms, 2 studies were carried out in ED settings (Duroy et al 34 and Haskins et al 35 ), 1 was carried out in 2 Veterans Health Authority (VHA) primary care centers and a VHA women’s health clinic (Rubin et al 36 ); and 1 study included a subset of the general population registered with a private research company (Jo et al 37 ).
e-SBIRT for Harmful Alcohol Use in ED Settings
In the first of the 2 studies from ED settings (Duroy et al 34 ), outcomes of interest pertinent to this review were daily number of drinks, episodes of intoxication and alcohol-related problem behaviors, Alcohol Use Disorders Identification Test (AUDIT) score, DSM V criteria of alcohol abuse, acceptance of long-term follow-up for alcohol use disorder, and suicidality at 3, 6, and 12 months. One group received e-SBIRT followed by 2 MI telephone appointments at 1- and 3-month post intervention, while a control group received nutritional education and advice during 2 phone appointments at the same time points. The authors controlled for sex only. In the second study (Haskins et al 35 ), outcomes of interest were treatment contact, treatment initiation, abstinence, and the number of attempts to reduce or quit using alcohol at 1 and 3 months. Baseline AUDIT scores and readiness to quit were controlled for as covariates. The control group received signposting by way of a printed list of alcohol support services.
Duroy et al 34 reported no statistical between groups regarding any outcomes of interest, whereas Haskins et al 35 reported differences in efforts to reduce or quit alcohol use at 3 months only, which were more common among control participants (odds ration [OR] 0.44, 95% CI [0.26-0.77], P = .004; OR 0.66, 95% CI [0.51-0.87], P = .01, respectively).
e-SBIRT for Harmful Alcohol Use in Outpatient Settings
Three articles reported trials of e-SBIRT in outpatient settings. Rubin et al’s 36 study took place at multiple VHA run primary care and women’s health clinic locations and involved e-SBIRT and TAU versus TAU alone. Here, TAU alone included an undescribed brief intervention carried out in person using a prepared checklist of items followed by referral to specialty treatment depending on problem severity. The authors controlled for gender and alcohol use severity at baseline. The studies by Montag et al 38 and Ondersma et al 39 trialed e-SBIRT against brochures displaying general health advice and an interactive nutrition education program, respectively, and both took place at perinatal healthcare clinics. Montag et al 38 stratified some of their analysis on baseline depression score, but these analyses were not pertinent to our review. Ondersma et al 39 controlled for baseline alcohol use.
Outcomes of interest reported by Rubin et al 40 were drinks per day, drinking days per week and proportion of heavy drinking days (not defined in standard drinks), and Short Inventory of Problems (SIP) scores. The authors applied a generalized linear model to their outcome data and found significant decrease in average drinks per day (b = −0.39, z = −5.79, P ≤ .001) and drinking days per week (b = −0.21, z = −3.84, P ≤ .001), but with no statistical difference between groups. According to a linear mixed model proportion of heavy drinking days decreased in both arms (b = −0.07, z = −4.81, P ≤ .001) with no statistical difference between e-SBIRT and TAU and TAU in isolation. e-SBIRT and TAU receiving participants exhibited greater rates of change in SIP score compared to those receiving TAU alone (b = −0.02, t = −2.25, P = .02).
Alcohol exposed pregnancies’ was the primary focus of Montag et al 38 ; the authors also reported drinks per week and binge episodes per week. Participants in the e-SBIRT and control groups reported improvement in all these outcome variables, but there was no statistically significant difference between groups and nor was there a significant treatment effect overall.
Ondersma et al 39 carried out a pilot trial of the acceptability rather than effectiveness of e-SBIRT in an outpatient setting. Nevertheless, the authors found a statistically nonsignificant difference in abstinence at 90 days post-intervention in favor of the e-SBIRT arm.
e-SBIRT for Harmful Alcohol Use in Nonclinical Settings
A single article by Jo et al 37 described a study involving participants recruited from the general population. The authors trialed e-SBIRT against a control condition of generalized feedback on alcohol intake. Outcomes of interest were weekly alcohol consumption, binge drinking (defined as ≥7 standard drinks for men or ≥5 for women in 1 drinking session), and AUDIT score at 1-month follow-up. Age, sex, and baseline AUDIT-K (Alcohol Use Disorders Identification Test, Korean language) were controlled for at follow-up.
The intervention was found to be effective for all primary outcomes of drinks per week (RR 0.13, 95% CI [0.018-0.906], P = .012), binge episodes (RR 0.69, 95% CI [0.562-0.848], P ≤ .001), and AUDIT score (RR 0.59, 95% CI [0.361-0.956], P = .009) at 1 month.
e-SBIRT for Populations at Risk of Harmful Illicit Drug Use
e-SBIRT for Harmful Illicit Drug Use in Probation Service Settings
Gilbert et al 41 reported a study trialing e-SBIRT known as Women Initiating New Goals of Safety (WINGS) among women from 2 probation sites and 1 community court-administered alternative-to-incarceration program. WINGS which were designed to identify and improve intimate partner violence (IPV) among women under community supervision compared to an in-person SBIRT designed for the same purposes. The authors assessed the feasibility, safety, and efficacy of the e-SBIRT. Although the primary focus was concerned with IPV, the program did integrate drug use components due to the high rates of drug use among the sample population and the overlap between SUDs and IPV, 42 and thus was included in the present review. The relevant outcome of interest was frequency of illicit drug use. The authors found that days using drugs at 1-month post intervention was significantly reduced in both e-SBIRT (OR 1.19, 95% CI [1.05-1.35], P ≤ .01) and SBIRT arms (OR 1.30, 95% CI [1.17-1.45], P ≤ .01) compared with control conditions. There was no significant difference between the 2 conditions, and the authors controlled for a variety of background characteristics including age and baseline drug and alcohol use.
e-SBIRT for Harmful Illicit Drug Use in Outpatient Settings
Two articles reported trials of e-SBIRT in outpatient settings. Martino et al 43 described a trial including women attending 2 hospital-based reproductive healthcare clinics, and Dawson-Rose et al 44 reported a trial of e-SBIRT in an in-hospital-based HIV clinic. Martino et al 43 trialed e-SBIRT against in-person SBIRT as a comparator and enhanced usual care (EUC) which was a control condition of a brief assessment of drug use followed by signposting. Reported outcomes of interest included substance use and treatment initiation at 1, 3, and 6 months. The authors provided additional findings on abstinence at the same time points. Dawson-Rose et al 44 compared e-SBIRT with traditional in-person SBIRT. Outcomes of interest were specific Substance Involvement Scores using the Alcohol, Smoking and Substance Involvement Screening Test (ASSIST) measure. The authors made use of bootstrap-based bias correction prior to regression analyses due to right-skewed baseline substance use scores.
Martino et al 43 described a generalized estimating equations (GEE) analysis controlling for baseline substance use and pregnancy status. GEE is a method of regression estimate ideal for use with repeated measures and nonnormal response variables. The authors found that independent of pregnancy status, days using substances were reduced at 1 and 3 months but not at 6 months compared with EUC. There was no significant difference between e-SBIRT and SBIRT, or between either intervention or EUC with regard to treatment initiation. The authors did not report differences in abstinence, but these data were requested from the authors, received, and are included in meta-analysis.
Dawson-Rose et al 44 reported no difference between e-SBIRT and SBIRT conditions on any outcomes of interest. The authors reported change in scores between participants screened as low risk and those found to be medium to high risk at 6 months. A statistically significant increase in scores across all domains was found for those at lower risk excepting clients reporting harmful amphetamine and sedative use. Correspondingly, the authors also found a significant decrease for those reporting medium to high-risk scores regarding—cocaine (OR 0.82, 95% CI [−1.39 to −0.25]) and amphetamines (OR 0.69, 95% CI [−1.32 to −0.10]), but increases for those using sedatives (OR 1.58, 95% CI [−2.21 to −0.92] and opioids (OR 1.31, 95% CI [−2.13 to −0.36]). No significant changes were observed for participants reporting harmful use of alcohol, tobacco, or cannabis.
e-SBIRT for Populations at Risk of Harmful Tobacco Use
e-SBIRT for Harmful Tobacco Use in ED Settings
One article by Boudreaux et al 45 reported a multicenter trial e-SBIRT in EDs in the context of harmful tobacco use. The authors trialed e-SBIRT against signposting to tobacco cessation treatment providers. Outcomes of interest were tobacco treatment provider contact, treatment initiation, and tobacco use at 1 and 3 months, controlling for baseline Heavy Smoking Index (HSI) scores and readiness to change. The authors found via GEE analysis that e-SBIRT participants were significantly more likely to contact tobacco treatment provider at both follow-up points (OR 2.69, 95% CI [1.65-4.39], P < .001), and no significant differences were found in relation to other outcomes of interest.
Meta-Analysis
Multiple outcomes were reported, with authors utilizing a range of measures of frequency of problem behavior (eg, drinks per week, daily drinks, days consuming any alcohol). Due to this variation in outcome reporting, we were only able to pool data from a minimum of 2 and a maximum of 4 studies for each reported outcome.
SMDs and RRs were calculated for frequency of standard drinks in the past week at 1 month post intervention. A significant effect size was found to explain reduced frequency of past week drinks in the experimental arm (n = 850) compared to the control arm (n = 853; d = −0.23, 95% CI [−0.33 to −0.14], P ≤ .0001; Figure 2). Episodes of binge drinking (or intoxication) at 6 months were not found to be significantly different between experimental (n = 397) and control arms (n = 399).
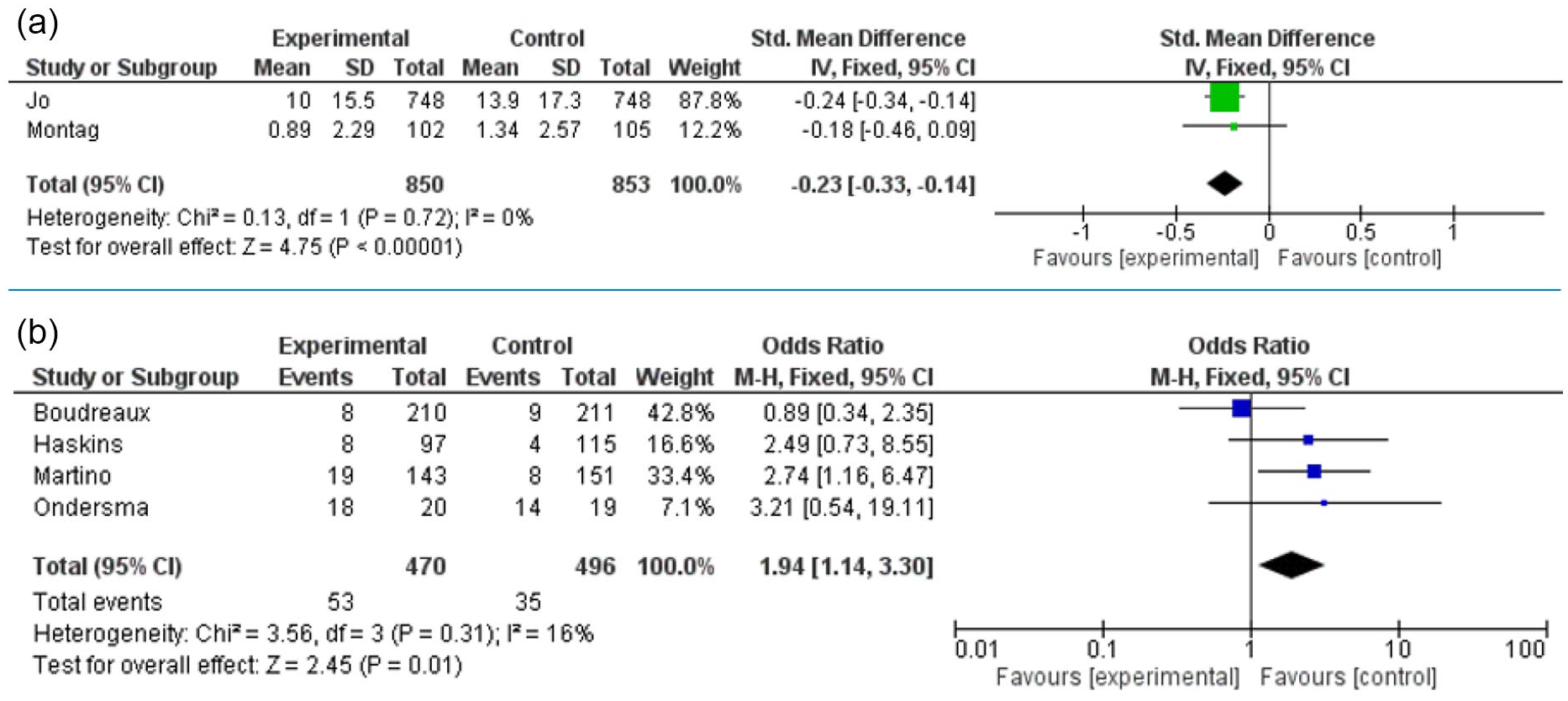
Abstinence from problem substance at 1 month (A) and 3 months post intervention (B).
There was no significant difference between experimental (n = 307) and control arms (n = 326) in terms of abstinence at 1 month. At 3 months post intervention, control participants were more likely to be abstinent, with those receiving e-SBIRT at greater risk of relapse (RR 1.94, 95% CI [1.14-3.30], P = .01; Figure 3).
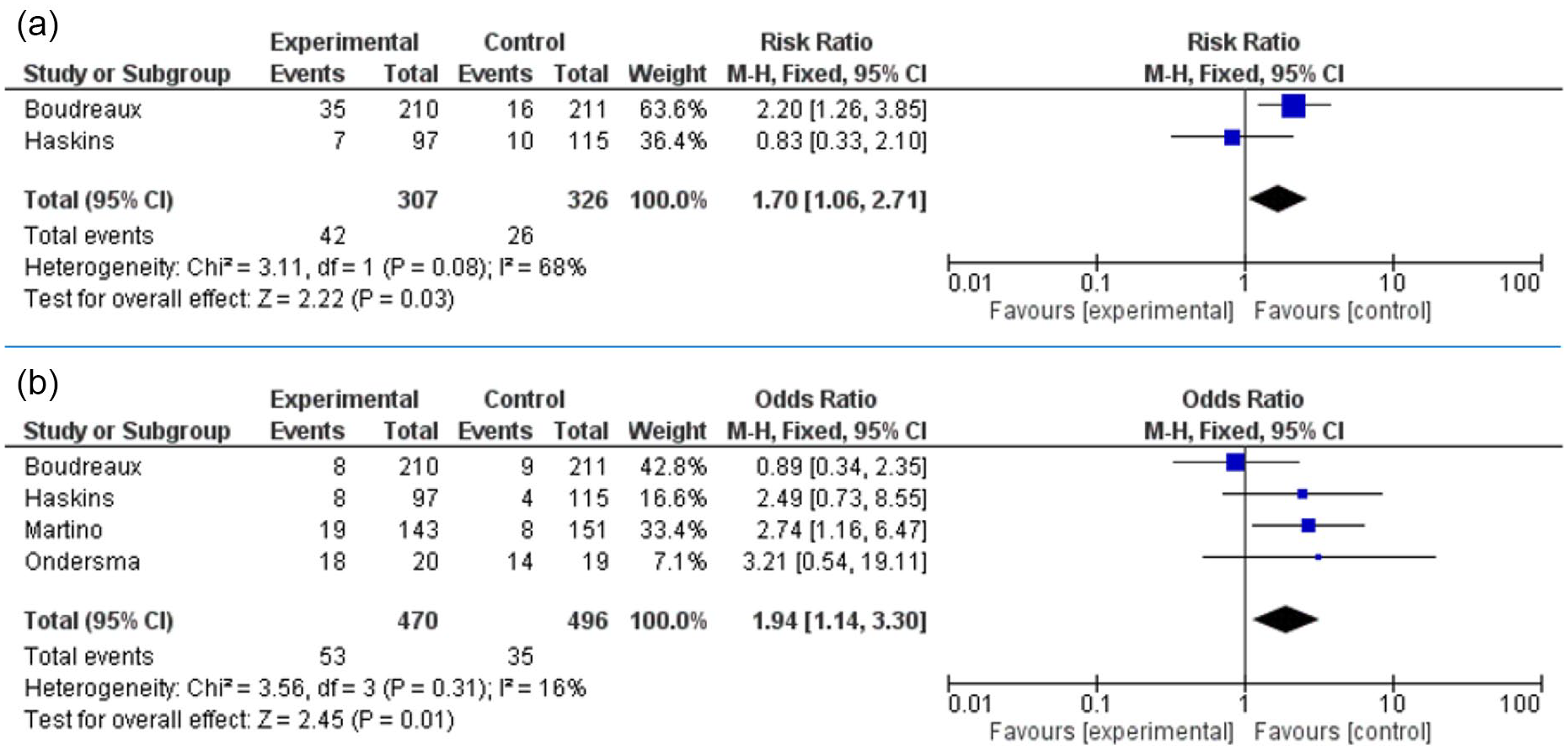
Treatment contact at 1 month (A) and 3 months (B) post intervention.
Control participants (n = 326) were more likely to make treatment contact at 1 month with participants receiving e-SBIRT at greater risk of absence (n = 307; RR 1.70, 95% CI [1.06-2.71], P = .03) and this elevated risk of absence from follow-up was maintained at 3 months (RR 1.6, 95% CI [1.11-2.32], P = .01; Figures 4).
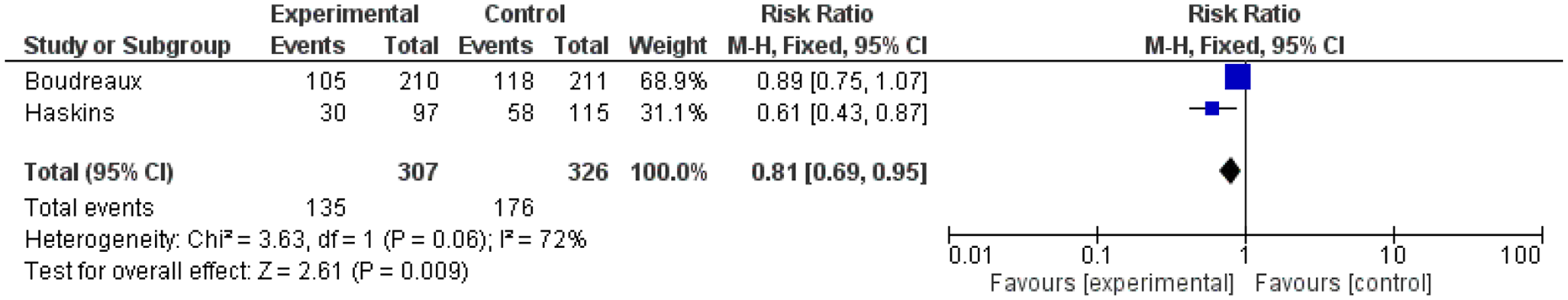
Quit attempt at 3 months post intervention.
There was no significant difference in treatment initiation at 1 and 3 months between experimental (n = 307) or control participants (n = 326). Likewise, there were no significant differences in treatment seeking between the experimental (n = 163) and control (n = 170) arms at 5 to 6 months. No significant difference was found for presence of a quit attempt at 1 month, but e-SBIRT participants (n = 135) were more likely to have attempted to quit at 3 months compared to controls (n = 176; Figure 4). There was no difference in attempts to reduce problem substance use behavior between e-SBIRT (n = 307) and control (n = 326) arms at 1 and 3 months.
Key e-SBIRT Components
Nine e-SBIRT programs were described in the included studies. Designs varied, but inclusion criteria dictated that each study reported the trialing of an e-SBIRT which included at least 2 of the 3 components of SBIRT. The key components of the included articles are summarized in Table 3.
e-SBIRT Components.
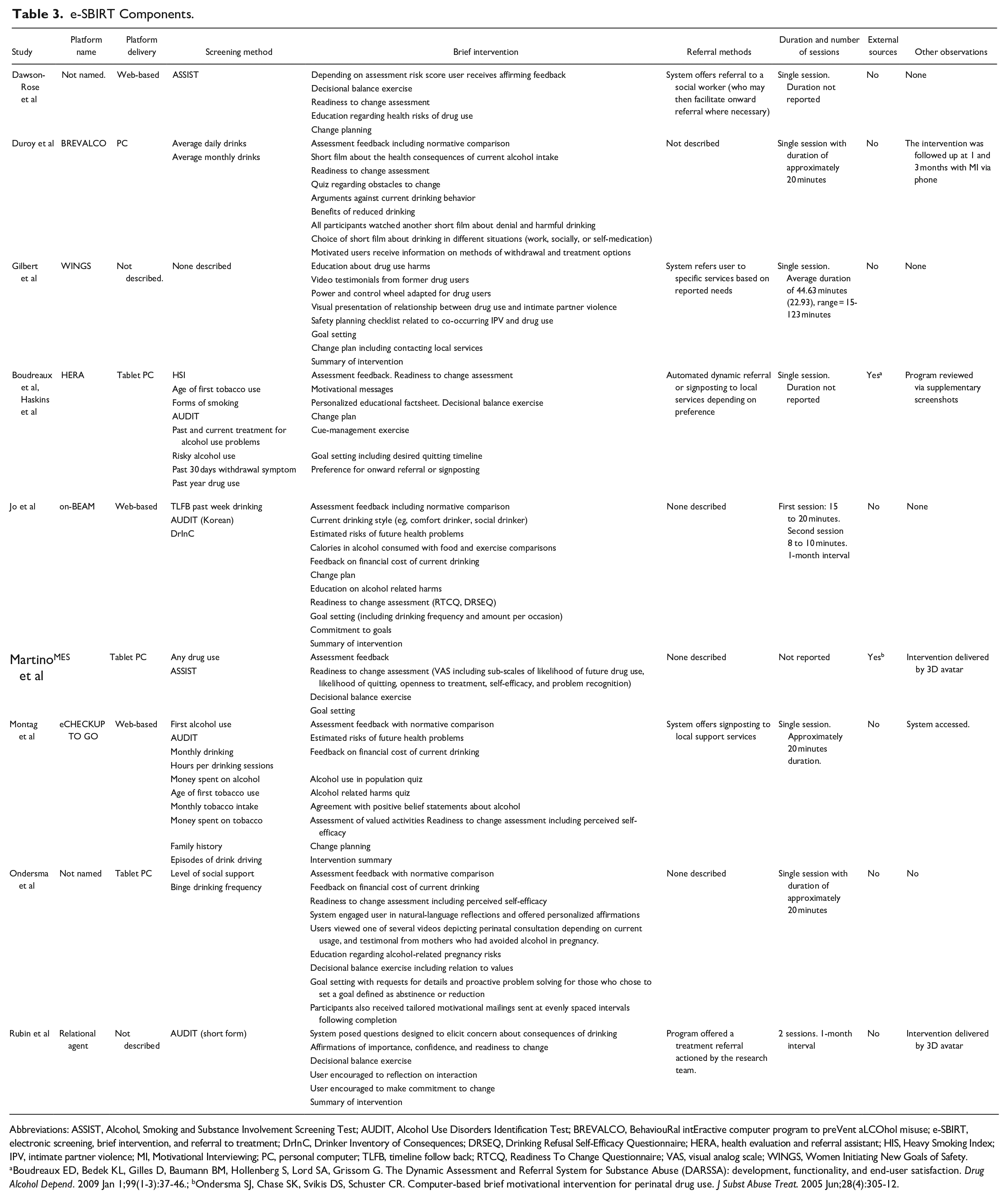
Abbreviations: ASSIST, Alcohol, Smoking and Substance Involvement Screening Test; AUDIT, Alcohol Use Disorders Identification Test; BREVALCO, BehaviouRal intEractive computer program to preVent aLCOhol misuse; e-SBIRT, electronic screening, brief intervention, and referral to treatment; DrInC, Drinker Inventory of Consequences; DRSEQ, Drinking Refusal Self-Efficacy Questionnaire; HERA, health evaluation and referral assistant; HIS, Heavy Smoking Index; IPV, intimate partner violence; MI, Motivational Interviewing; PC, personal computer; TLFB, timeline follow back; RTCQ, Readiness To Change Questionnaire; VAS, visual analog scale; WINGS, Women Initiating New Goals of Safety.
Boudreaux ED, Bedek KL, Gilles D, Baumann BM, Hollenberg S, Lord SA, Grissom G. The Dynamic Assessment and Referral System for Substance Abuse (DARSSA): development, functionality, and end-user satisfaction. Drug Alcohol Depend. 2009 Jan 1;99(1-3):37-46.;bOndersma SJ, Chase SK, Svikis DS, Schuster CR. Computer-based brief motivational intervention for perinatal drug use. J Subst Abuse Treat. 2005 Jun;28(4):305-12.
Screening
Six of the 9 e-SBIRT programs described in this review used a previously validated assessment measure of problem behavior to screen participants as no/low risk, moderate or high risk regarding alcohol, drug, or tobacco use. Four programs used the AUDIT, which is a 10-item screening tool developed by the World Health Organization (WHO) to assess alcohol consumption, drinking-related behaviors, and alcohol-related problems. A risk score is calculated representing low risk, increasing risk, high risk, or possible dependence. 46 Two programs used the WHO’s ASSIST measure which consists of 8 questions covering tobacco, alcohol, cannabis, cocaine, amphetamine-type stimulants (including ecstasy) inhalants, sedatives, hallucinogens, opioids, and “other drugs” which do not fit easily in to existing pharmacological classes such as new psychoactive substances. A risk score is provided for each substance, and scores are grouped into “low risk,” “moderate risk,” or “high risk.” 47 One program used several questions from the 50-item Inventory of Drug Use Consequences to measure the adverse physical, social, intrapersonal, and interpersonal consequences of alcohol and substance use, 48 while another program used the 15-item SIP. 40 One e-SBIRT program used the 2-item HIS measuring time to first cigarette and number of cigarettes smoked per day, 49 and 1 e-SBIRT (WINGS) did not include a screening component.
Brief Intervention
Not all the content of the brief interventions included in these programs were fully applicable to the remit of this review. For example, the WINGS e-SBIRT program described by Gilbert et al 41 included a power and control wheel adapted for drug users, a visual presentation of relationship between drug use and IPV, and a safety planning checklist related to IPV. The programs described by Ondersma et al and Martino et al were designed for perinatal populations, and so included some components which were specifically related to the needs of these populations. Articles also varied in the amount of information provided to describe the brief interventions included in the various e-SBIRTs. In some cases, a paucity of description in the text was offset with the inclusion of supplementary materials (eg, screenshots), and in 1 case, we were able to access the system in question (eCHECKUP TO GO) and review the components directly.
All 9 programs offered some form of brief intervention based at least in part on MI where in users were prompted to engage in some level of “change talk” by reflecting on their desire to change, their current ability to change, their readiness to change, and their personal need to change.
Seven of the 9 e-SBIRT programs offered personalized assessment feedback with reference to population norms. Depending on risk at screening, the user would then proceed on to other components of a brief intervention, or following the standard structure of SBIRT programs, receive positive feedback to motivate the user to maintain current usage. One program was described as communicating to the user their current “drinking style” (eg, social, comfort, impulsive, etc).
Eight e-SBIRT programs included a readiness to change assessment as part of the brief intervention component, 3 of which included measures of self-efficacy (ie, perceived ability to resist engaging in a problem behavior in difficult situations such as those which include social pressure or experiential avoidance of difficult emotions). Only one e-SBIRT program used evaluated measures of readiness to change as part of the brief intervention component, including both the Readiness to Change Questionnaire (a 12-item self-report questionnaire which assesses readiness to change along 3 stages of change) 50 ; and the Drinking Refusal Self-Efficacy Questionnaire (a 31-item measure of drinking self-efficacy covering situations including those characterized by social pressure, opportunistic drinking, and emotional relief). 51 One program used a visual analog scale including subscales of likelihood of future drug use, likelihood of quitting, openness to treatment, self-efficacy, and problem recognition to measure readiness to change.
All programs offered education related to drug, alcohol, or tobacco use (eg, prevalence or economic costs), with 1 program was described as using a quiz format for this purpose. Four specifically focussed on health risks (2 of which offered personalized risk estimation based on current habits). Five programs included a form of decisional change exercise wherein the user is instructed on listing the costs and benefits of changing their alcohol, drug, or tobacco usage. One program was described as presenting the user with set arguments for change, and a list of likely benefits. Two programs included values assessment with 1 program described as incorporating “deeply held” values data into the decisional balance exercise. Another assessed values along set domains similar to the Valued Living Questionnaire. 52 Three studies calculated the financial cost of current drinking and communicated this to the user, with one relating feedback to user and incorporating previously input information regarding their personal values by communicating to the user how much money they would have to spend on valued activities. One program calculated calorific content of the user’s alcohol intake and communicated the equivalent food intake and time and intensity of exercise needed to burn off this excess calorific intake. Five studies described engaging the user in devising a change plan, and 5 programs were described as including goal setting. One program described the goal setting process as including “proactive problem solving,” and 1 was reported as utilizing an “interactive quitting timeline.” Two programs proactively assessed obstacles to change, with 1 delivering this exercise in the style of a quiz, and 2 invited users to commit to making a change. Three programs used 1 or more educational films. One used video testimonial from people who have experienced substance use problems, and 1 used videos to educate the user about alcohol-related health problems and to encourage users to think about their alcohol use in different situations including work, social events, and when alone. Four programs described providing tailored motivational messages or affirmations to the user, with 1 offering the user the option of receiving regular motivational messages via email for a set duration after use. One program included a cue-management component (eg, strategic avoidance of behavioral cues which may precipitate substance use), while another offered education on treatment options available and methods of withdrawal from alcohol or drugs.
Referral to Treatment
Five of the 9 e-SBIRT programs were described as involving a referral to treatment component. Two e-SBIRT programs were reported as automating onward referral. Of these, 1 described incorporating a system wherein the user could opt for signposting or “dynamic” automated referral. If the user chose the dynamic option, then a referral would be automatically faxed to a local treatment service based on the individual’s address, health insurance status, and preference for telephone or in-person treatment. Detailed information was lacking regarding the other program’s automated referral system. One program offered higher-risk users a referral to a social worker who in turn would be able to offer support to the client or make onward referrals. It was not clear in the reporting if the initial referral was automated or carried out by an e-SBIRT system administrator. One program described offering the user a referral to a local treatment service which was completed manually by a system administrator (in the included article this person was the lead author). One system only offered signposting to local services and 1 program gave the user a preference for onward referral or signposting.
Components and Outcomes
Four of the 9 e-SBIRT programs were trialed against control conditions and found to be superior in at least 1 outcome of interest. These were the HERA (Health Evaluation and Referral Assistant), MES (Motivational Enhancement System), Relational Agent, eCHECKUP TO GO, and on-BEAM (online-based Brief Empowerment Program for Alcohol-Use Monitor) programs. The HERA program was found to be superior to a comparator of in-person SBIRT in 1 outcome of interest. At meta-analysis, the on-BEAM and the eCHECKUP TO GO programs were associated with reduced alcohol intake compared to controls, and HERA was associated with greater likelihood of quit attempts at 3 months post intervention. The components of the programs reported as part of this review are summarized in Table 3.
In terms of commonality, these 5 programs were reported as employing 1 or more validated screening measures (HSI, AUDIT, ASSIST, Drinker Inventory of Consequences). Also, all included a readiness to change assessment. Four of the 5 were described as including a personalized assessment feedback, and 3 of the 5 were described as including a decisional balance exercise, a goal setting exercise, a change plan, and of providing a summary of the intervention to the user. In addition, 3 of the 5 included a referral to treatment component, which was either a dynamic automatic referral, signposting, or manual referral by a system administrator.
Evidence Quality
Based on the GRADE system to help inform clinical practice recommendations, 32 outcomes were considered along 4 levels of quality of evidence: very low, low, moderate, and high (Table 4). Outcomes from RCTs were automatically considered high quality but downgraded if the outcome suffered from risk of bias, imprecision, inconsistency, indirectness, or publication bias. If risk of bias was deemed likely by lack of allocation concealment, lack of blinding, high attrition, or funding or sponsorship bias as evidenced by declared conflict of interest, the outcome was downgraded.
GRADE Outcome Quality.
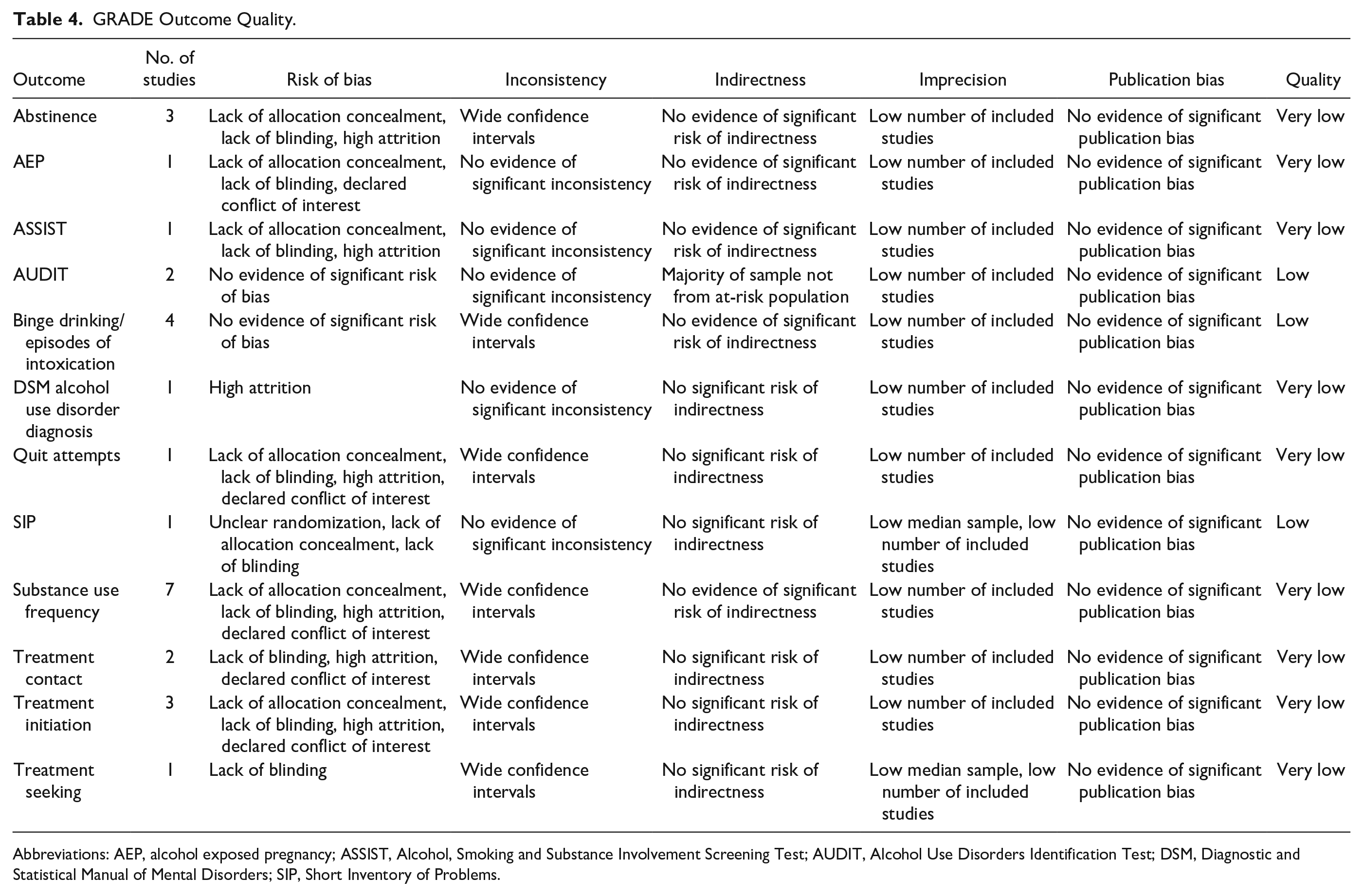
Abbreviations: AEP, alcohol exposed pregnancy; ASSIST, Alcohol, Smoking and Substance Involvement Screening Test; AUDIT, Alcohol Use Disorders Identification Test; DSM, Diagnostic and Statistical Manual of Mental Disorders; SIP, Short Inventory of Problems.
In terms of imprecision, if most of the sample was found to be from a population not directly relevant to the aims of the review (eg, not at risk of addictive disorders), the outcome was measured indirectly using a surrogate marker, or where measurements were made over too brief or prolonged a time frame, the outcomes were downgraded. Applicability of the intervention was not relevant here as all studies reported on e-SBIRT findings in comparison with traditional SBIRT or a control condition.
In terms of inconsistency, where excessively wide CIs were reported for most of the sample, median sample size was under 100, or included studies were under 10, the outcome was downgraded. Multiple outcomes were supported in this review including multiple time points and measures of frequency of behavior (eg, drinks per week, daily drinks, days consuming any alcohol). In the limited meta-analysis, we pooled alcohol, drug and tobacco use as substance use, and calculated SMDs and RRs where this were possible. Study authors were often found to control for age, sex, and baseline usage in their analyses which strengthens the precision of the data; however, we see wide CIs making it reasonable to assert that overall, the data lacked homogeneity.
As we carried out a comprehensive search, did not exclude by language, experienced no industry influence on our work, and found no evidence of underreporting of null results, there was no need to assume significant publication bias. After application of the GRADE approach to assessing the quality of evidence to outcomes of interest pertinent to this review, no outcomes were found to be of high quality, with most being of very low quality and 3 being of low quality (Table 4).
Discussion
In carrying out this review and meta-analysis, we sought primarily to identify effective models of e-SBIRT design and delivery for the treatment of addictive behavior and identify knowledge gaps. The quality of the available literature made satisfying our primary objective difficult; however, we were able to describe the kinds of problem substances and settings represented in e-SBIRT trials and report the common components of e-SBIRT.
We did not find any articles describing e-SBIRT is relation to harmful gambling. Considering the impact that harmful gambling has as a public health concern, this absence may be of concern to the clinical research community.
A review of the literature surmised that although people at risk of drug-related harms often present at the ED, time pressures and competing priorities, lack of knowledge and skill deficits, as well as stigma, hamper the practical application of SBIRT in these settings. 53 Based on the findings of this review, we cannot conclude that people at risk of harmful alcohol use cannot be said to benefit from e-SBIRT as alcohol-related harm constitutes a significant proportion of all ED presentations.54,55
Our review supports continued clinical trialing of e-SBIRT for populations known to be at a significantly increased risk of drug-related harms (or at an increased likelihood of experiencing the more severe end of drug-related harms) such as women on probation, 56 women receiving perinatal care, 57 and people with HIV. 58
Traditional SBIRT has been implemented in primary care settings successfully in that programs have been found to be acceptable and well received by staff and frequently utilized where available. 59 We conclude that although the current evidence is lacking, e-SBIRT has the potential to offer much of the same benefits, while being less resource hungry, and routine data research confirms that primary care settings are disproportionately utilized by those at risk of the most serious drug-related harms. 60
More evidence is needed regarding not just efficacy and effectiveness of e-SBIRT, but also adverse consequences. Among the included studies an increase in drug use was reported among those screened as being at lower risk, and so there might be a deleterious effect for this subsample. More research is needed to understand this effect.
To compare pooled outcomes, we calculated RRs. In doing so, we are simply calculating the number of persons experiencing the outcome of interest (eg, abstinence) in the exposed and unexposed groups post intervention, and then dividing by the total sample in each arm. RR is a simple and intuitive measure of association, which has greater precision when events are relatively rare among the groups under observation compared with the alternative of calculating ORs. In addition, RR has the benefit of being collapsible meaning that unadjusted RRs will not change when analysts adjust for other variables excluding confounders. For this reason, the difference in population and setting may not harm the validity of our results (as far as the purposes of the review are concerned). However, treatment trajectories associated with different problem substances prior to the intervention may have a more significant effect on the validity of the analysis as it was undertaken. This is especially pertinent as substance differences were reported by the authors of an included study. 44
The present review has some limitations. It is limited by the search terms used to include all 3 SBIRT components together (screening, brief intervention, and referral to treatment). Therefore, it is possible that programs that have some but not all the components of SBIRT, such as screening and brief intervention (SBI) or screening and referral to treatment, may have been missed. Included articles were illustrative of heterogeneity in methodology and reported outcomes. However, our findings do support the assertion that e-SBIRT may be useful in reducing problem substance use in the short term, but not in the longer term. In addition, we found that attempts to quit were more commonly reported at 3 months post-intervention slightly but significantly favored e-SBIRT. This suggests that e-SBIRT may be associated with maintained motivation to change over the longer term, even if a significant difference in outcomes is not apparent in the data at this point. Poor-quality outcome evidence curtails the extent to which the findings of this review can be applied to changes or developments within practice. Finally, much of the outcome data reported in this review come from self-report measures, and although many of the articles reported the use of empirically validated measures, these outcomes remain subjective and open to self-report bias.
Conclusion
The design of e-SBIRT programs vary in their composition, though the foundation in MI principles remains a unifying factor. The components most often associated with effectiveness across the outcomes of interest in this review were validated screening measures, personalized assessment feedback, readiness to change assessment, decisional balance exercises, goal setting, and change plans. There is currently no clarity of consensus on which referral methods may have an advantage over others.
The evidence to support the use of e-SBIRT to screen for harmful substance use behaviors, deliver brief intervention, and referral for formalized treatment is limited and of a low quality. The evidence to support e-SBIRT in relation to harmful gambling, or any other behavioral addiction, is nonexistence. High-quality experimental research is needed to evaluate the effectiveness of e-SBIRT for people at risk of addictive problems. This should include not only substance-related problems but also behavioral problems such as harmful gambling. The development and trialing of e-SBIRT programs which utilize evidence-based psychotherapeutic models other than only MI is encouraged. The current review finds that the effectiveness of e-SBIRT for people at risk of addictive disorders remains in equipoise. Therefore, we recommend the development of a longitudinal double blinded multi-site RCT to evaluate the effectiveness of e-SBIRT against TAU in NHS primary care settings with onward referrals to NHS SUD and NHS behavioral addictions settings (eg, the National Problem Gambling Clinic) as the primary outcome variable.
Footnotes
Acknowledgements
Funding for this review was provided by Greo Evidence Insights. The authors would like to thank Martino and colleagues for providing additional data for meta-analysis.
Author Contributions
All authors contributed to the review conception and design, collection of data, analysis, interpretation of the results, writing, and revision of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by Gambling Research Exchange Ontario (GREO). The funding organization had no role in the design and conduct of the study; collection, management, analysis, and interpretation of the data; preparation, review, or approval of the manuscript; and decision to submit the manuscript for publication.
Compliance,Ethical Standards,and Ethical Approval
Institutional Review Board approval was not required.
