Abstract
Background:
Healthcare providers may be utilizing central nervous system (CNS) depressants to reduce opioid use due to recent changes in public policy. Combination use of these agents with opioids increases the risk of respiratory depression and death. Healthcare expenditures by individuals using these drug combinations have not been previously quantified. We sought to characterize healthcare costs and expenditures associated with a population reporting concurrent CNS depressants and opioid use compared with nonopioid analgesics in the United States from 2009 to 2019.
Methods:
A serial cross-sectional design was used to compare the healthcare expenditures of adult Medical Expenditure Panel Survey respondents who were prescribed nonopioid analgesics, opioids only, opioids/benzodiazepines (BZD), opioids/BZD/skeletal muscle relaxants (SMR), or opioids/gabapentin (gaba) using pooled data from 2009 to 2019. Expenditure (cost and resource utilization) categories included inpatient, outpatient, office-based, and prescription medicine. Average marginal effects were used to compare survey-weighted annual costs and resource utilizations across the groups as compared to nonopioid analgesic respondents, adjusted for covariates.
Results:
A weighted total of 34 241 838 individuals were identified. Most were opioid-only respondents (46.5%), followed by nonopioid analgesic (43.4%), opioid/BZD (5.3%), opioid-gaba (3.5%), and opioid/BZD/SMR respondents (1.3%). In comparison to the study groups with nonopioid analgesics, opioid-gaba users had the highest significant incremental cost difference among the different pairings (+$11 684, P < .001). Opioid-gaba, opioid/BZD, and opioid/BZD/SMR respondents had significantly higher inpatient, emergency department, and prescription drug costs and use compared to nonopioid analgesic respondents. Opioid-only respondents had higher outpatient and office-based costs and visits compared to nonopioid analgesic respondents.
Conclusions:
As healthcare providers seek to utilize fewer opioids for pain management, attention must be paid to ensuring safe and effective use of concurrent CNS depressants to mitigate high healthcare costs and burden.
Highlights
Of all concurrent opioid drug categories, opioid-gabapentin respondents had the highest total annual medical costs compared to nonopioid analgesic respondents.
Opioid-only respondents had a significant increase in outpatient use and a nonsignificant increase in other healthcare services over time.
Opioid/benzodiazepines (BZD) and opioid/BZD/skeletal muscle relaxants respondents had the highest emergency department use of the study groups compared to nonopioid analgesics, likely driven by complex patient presentations.
Introduction
In 2019, an estimated 50.2 million US adults reported suffering from chronic pain. 1 The use of prescription opioids as treatment for chronic pain has resulted in significant morbidity and mortality. 1 Recently, the coronavirus pandemic exacerbated the occurrence of opioid use disorder and associated mortality rates by aggravating social and economic stressors. 2 In addition, individuals with opioid use disorder faced increased challenges in accessing addiction treatment during this time.2,3 In 2021, the number of drug overdose deaths involving a prescription opioid was almost 5 times as high as those reported in 1999. 4 Between 2008 and 2017, it was estimated that the total annual health-related economic burden of patients using a prescription opioid was $524 billion. 5 Although the release of the Centers for Disease Control and Prevention’s (CDC’s) opioid prescribing guidelines in 2016 has been associated with a reduction in opioid prescription rates over time, the deleterious effects of the opioid epidemic continue to persist.6,7 While focus has been centered on the abuse of opioids alone, less has been devoted to understanding the potential harmful effects of opioids with co-occurring use of other central nervous system depressants, including benzodiazepines, gabapentinoids, and skeletal muscle relaxants (referred to as “concurrent opioid use”).
Use of these medications in combination with each other increases risk of overdose from opioid-induced respiratory depression, sedation, and impaired sensory-perceptual processing. 8 Concurrent use of opioids with benzodiazepines, skeletal muscle relaxants, and/or gabapentinoids results in significantly higher magnitudes of mortality, drug-related hospitalization, and emergency department (ED) use for respiratory depression compared to use of these drug classes alone.9-13 These risks are further underscored in the case of triple combinations, where patients concurrently utilize an opioid, a benzodiazepine, and a skeletal muscle relaxant, colloquially referred to as the “Holy Trinity.” 14 In 2017, the CDC estimated that of all deaths related to prescription opioids, 5.7% had evidence of the triple combination in the 6-month period preceding death. 15 Triple combination use has also been estimated to increase the risk of ED visits by almost 9 times the rate of nonopioid users, surpassing comparable ED rates for patients using concurrent opioids and benzodiazepines. 16
It has been hypothesized that utilization of concurrent opioid and triple combination use has resulted from mounting regulatory pressure to reduce use of opioids as treatment for pain.17-19 As clinical guidelines continue to advocate for a multimodal analgesic approach to pain, clinicians may find themselves gravitating toward the use of nonopioid analgesics, skeletal muscle relaxants, gabapentinoids, and/or benzodiazepines to mitigate the risk of death imparted by opioids. 18 Moreover, as individuals with chronic pain often present with comorbid anxiety, use of benzodiazepines or gabapentinoids to treat both conditions may be highly desirable.20-23 Despite the high risk of death demonstrated by the triple combination or concurrent opioid combinations, the CDC’s updated 2022 Clinical Guidelines for Pain Management have only warned against concurrent opioid use with either benzodiazepines or gabapentinoids, based on limited evidence. 18 We sought to expand on this evidence base by investigating the differences in healthcare costs and resource utilizations among nonopioid analgesic, opioid-only, opioid and concurrent benzodiazepine, opioid and concurrent gabapentin (gaba), and opioid concurrent skeletal muscle relaxant and benzodiazepine adults using pooled data from 2009 to 2019 in a nationally representative cohort. We also sought to evaluate changes in these outcomes over time among the 5 groups.
Materials and Methods
Study Design
A serial cross-sectional analysis design using pooled data from the Medical Expenditure Panel Survey (MEPS) between 2009 and 2019 was used to evaluate differences in economic burden and resource utilization between 5 groups: nonopioid analgesic respondents, opioid-only respondents, concurrent opioid/benzodiazepine respondents (“double threat”), concurrent opioid/gabapentinoid respondents (opioid-gaba), and triple combination respondents, consisting of concurrent opioid, benzodiazepine, and skeletal muscle relaxant respondents (“triple threat”). MEPS is a nationally representative survey of the US civilian, noninstitutionalized population that utilizes complex survey design methods to yield nationally weighted estimates using clustering, stratification, and multistage and disproportional sampling curated by the Agency for Healthcare Research and Quality (AHRQ).24,25 MEPS collects data for up to 5 rounds within a 24-month period, including prescription fills.23,24 This study followed the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines for cross-sectional study designs. 26
Study Sample
We pooled data from adult MEPS participants (18 years or older) between 2009 and 2019. The full-year consolidated MEPS Household files, Prescription Medicines files, and Medical Conditions files were used to identify and assess participants.24,27,28 The Full-Year Consolidated files contain data on demographics, socioeconomic status, insurance status, and health-related data. 24 The Medical Conditions file contains data on self-reported diagnoses from MEPS participants. 28 The Prescription Medicines file contains data on self-reported prescription drug fills from participants in outpatient pharmacies. 27 Drugs administered in an inpatient setting or over-the-counter drugs are not captured in the Prescription Medicines file. Healthcare resource use and related costs reported by MEPS respondents are validated with each respondent’s medical provider and pharmacy. 26
Characterization of Study Groups
Respondents in the “non-opioid analgesic” group were defined as those who reported purchasing a prescription for a nonopioid analgesic (eTable1, Supplementary Materials) at any round in that specific year. Respondents in the “non-opioid analgesic” group did not report any opioids, benzodiazepines, skeletal muscle relaxants, or gabapentinoids throughout the study period. We excluded selective serotonin reuptake inhibitors (SSRIs), serotonin-norepinephrine reuptake inhibitors (SNRIs), tricyclic antidepressants, antipsychotics, antiepileptics, and hydroxyzine as “non-opioid analgesics” because we could not confirm whether these medications were reported for pain control or for another indication (eg, major depressive disorder, general anxiety disorder, etc). “Opioid-only” respondents were defined as those who only reported purchase of one or more opioids at any round in that specific year. Opioid and benzodiazepine respondents (“double threat”) were characterized as those reporting prescription purchases of both an opioid and a benzodiazepine in the same round for that year. Opioid and benzodiazepine and skeletal muscle relaxant (“triple threat”) respondents were defined as those reporting prescription purchases of an opioid, a benzodiazepine, and a skeletal muscle relaxant in the same round for that year. Opioid-gaba respondents were defined as those reporting purchase of an opioid and a gabapentinoid (pregabalin or gaba) prescription in the same round for that year. These categories were mutually exclusive (eg, double-threat respondents did not report use of a gabapentinoid or a skeletal muscle relaxant during any other round for that year). Medications were flagged using Multum Lexicon Variables from Cerner Multum, Inc (Supplementary Materials) and were manually reviewed for appropriate inclusion into the study. We did not restrict our analyses to a single type of drug formulation (eg, oral, topical, injectable, etc) to best evaluate respondents who may have used analgesics for various types of pain (eg, neuropathic, visceral, somatic, etc). Methadone was excluded as an opioid from this analysis as we could not confirm whether reported methadone was for pain control or for opioid use disorder.29 -31
Study Outcomes
The primary outcomes of interest were total yearly healthcare costs and yearly resource utilizations. Secondary outcomes of interest included prescription costs, outpatient costs, ED costs, inpatient costs, number of prescriptions filled, number of office-based visits, number of ED visits, number of outpatient visits, and number of inpatient hospitalizations. Costs are the sum of direct payments provided, including out-of-pocket payments and payments by private insurance, Medicare, Medicaid, or other sources and were adjusted for inflation using the Personal Consumption Expenditure Health price index as recommended by AHRQ to reflect 2021 USD.24,32 Data on prescriptions filled included information on medication name, number of fills and refills for that medication, and round reported. Medical provider visits were captured using the number of office-based visits and number of outpatient visits to a medical provider. Data on ED visits included the count of all ED visits reported for that year. Inpatient hospitalizations were captured as the total number of hospital discharges.
Demographic and Other Variables
Participant demographic data used in our analysis included age category (18-24 years, 25-44 years, 45-64 years, and 65 years or older), sex (female/male), race (White/Black or African American/American Indian or Alaska Native/Asian or Native Hawaiian or Pacific Islander/Multiple), ethnicity (Hispanic/Non-Hispanic), marital status (married/widowed/divorced/separated/never), education (no diploma/General Education Development or high school/associate, other, <4 years college/BS degree/Master’s, doctorate, professional degree), geographic region (Northeast/Midwest/South/West), poverty status (poor or negative/near poor/low income/middle income/high income), insurance coverage (any private/public/uninsured), and Charlson Comorbidity Index (CCI) score. CCI was calculated using a previously published algorithm using International Classification of Diseases 10 (ICD-10) diagnosis codes to identify comorbidities from each year’s Medical Conditions file to account for disease burden. 33 For years 2009 to 2015, ICD-9 diagnosis codes were used to identify CCI conditions (Supplementary Materials). Poverty status category was determined by each participant’s total annual income of his or her family for each year of data. 34 Categories were defined according to federal poverty level (FPL): poor or negative (<100% of FPL), near poor (100% to <120% of FPL), low income (125% to <200% of FPL), middle income (200% to <400% of FPL), and high income (greater than or equal to 400% of FPL). 34
Statistical Analysis
Demographic data were descriptively analyzed between the 5 study groups using Analysis of Variance (ANOVA) and chi-squared tests for continuous and categorical variables, respectively. Means and standard deviations were derived for continuous data, and frequencies with proportions were summarized for categorical variables. Survey weights were applied to estimate the population standard errors and 95% confidence intervals (CIs) using STATA’s svy command. 35
For our primary aim, we individually compared opioid-only, double-threat, triple-threat, and opioid-gaba group healthcare costs and resource utilizations to the nonopioid analgesic group. We further compared the groups on total prescription drug costs, total outpatient costs, total ED costs, total inpatient hospitalization costs, total office-based costs, total number of prescriptions filled, total number of office-based visits, total number of ED visits, and total number of inpatient hospitalizations. Survey-weighted two-part models were used to evaluate cost-related outcomes due to a large number of respondents with $0 expenditure with STATA’s twopm package.36 -38
We used logistic regression for the model’s first part to estimate the likelihood of a respondent having nonzero healthcare costs. Generalized linear models with gamma distribution were used for the second part of the models to estimate the relationship between healthcare cost and drug group contingent on whether the participant had nonzero healthcare costs in the first part, adjusting for covariates.36 -38 All models were adjusted for covariates including age, year, sex, race/ethnicity, marital status, education attained, geographic region, income category, insurance status, and CCI score. All cost model results were reported using average marginal effects in USD with 95% CIs. Differences in resource utilization categories (eg, number of prescription medications, etc) between the study drug groups were evaluated using survey-weighted negative binomial regression models to account for overdispersion, adjusting for covariates listed previously. Results were reported as incidence rate ratios (IRRs) with 95% CIs and robust standard errors. All models utilized the nonopioid analgesic group as a reference group.
For our secondary aim, we evaluated whether the study outcomes (costs, resource utilizations) significantly changed over time for each study group compared to the nonopioid analgesic group. This was performed using a series of linear regression models with an interaction term between year and study group, adjusting for covariates. Findings were presented as average annual differences using regression model coefficients with standard errors and 95% CIs. Statistical significance was defined using a 2-tailed alpha value of <0.05. All analyses were performed using STATA version 17 (StataCorp, College Station, TX).
Results
In total, 370 738 unique unweighted MEPS respondents were pooled between 2009 and 2019 (Figure 1). Among these, 18 214 (4.9%) purchased only a nonopioid analgesic at least once, 16 796 (4.5%) purchased only an opioid at least once, 1742 (0.47%) purchased an opioid and a benzodiazepine in the same round, 439 (0.12%) purchased an opioid, a benzodiazepine, and a skeletal muscle relaxant in the same round, and 1230 (0.33%) purchased an opioid and a gabapentinoid in the same round (Table 1). A total of 370 738 unweighted MEPS respondents represented an annual weighted population of 34.2 million lives. Opioid respondents accounted for 172.1 billion dollars spent in total medical expenditure, followed by nonnarcotic opioid respondents ($103.2 billion), double-threat respondents ($29.4 billion), opioid-gaba respondents ($26.3 billion), and triple-threat respondents ($7.5 billion). Demographic characteristics varied across the study groups. Nonopioid analgesic respondents were predominately middle-aged (37.4%), female (56.9 %), white (75.2%), not Hispanic (81.4%), and married (53.0%) with higher education and income levels (high income: 37.8%). Opioid-only respondents were younger with a higher proportion of white individuals (82.0%), slightly higher income (high income: 40.1%), and a high rate of private insurance coverage (7.2%). Double-threat respondents were mostly male (62.6%), white (88.5%), and non-Hispanic (93.7%) with lower income (poor/negative: 18.4%) and more public insurance coverage (37.1%). Triple-threat respondents had similar characteristics to nonopioid analgesic respondents. Opioid-gaba respondents were predominantly over age 65 (46.6%) with a higher comorbidity burden (mean CCI: 3.2).
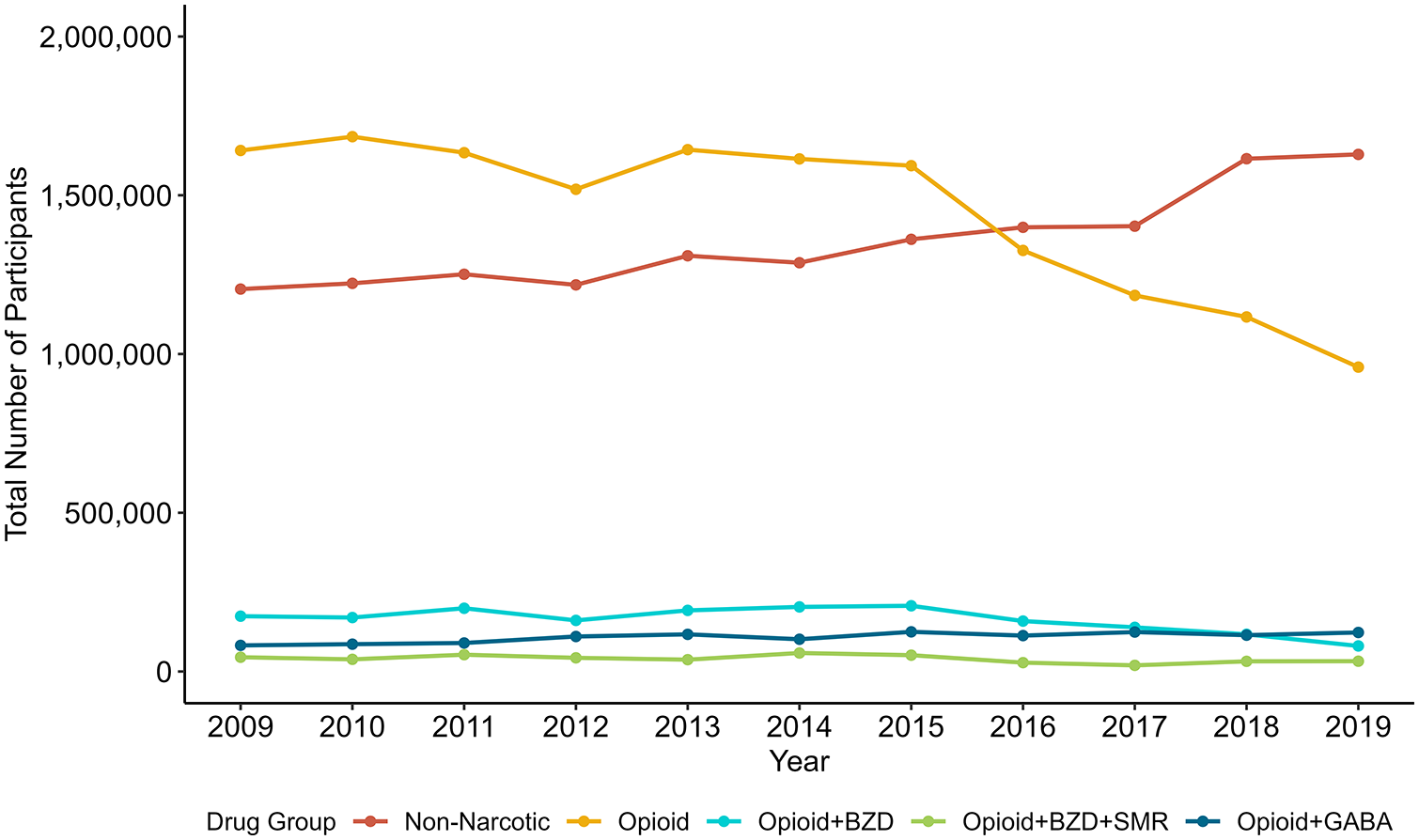
Number of unique respondents per drug group per year, 2009 to 2019.
Cohort Demographics, Weighted.
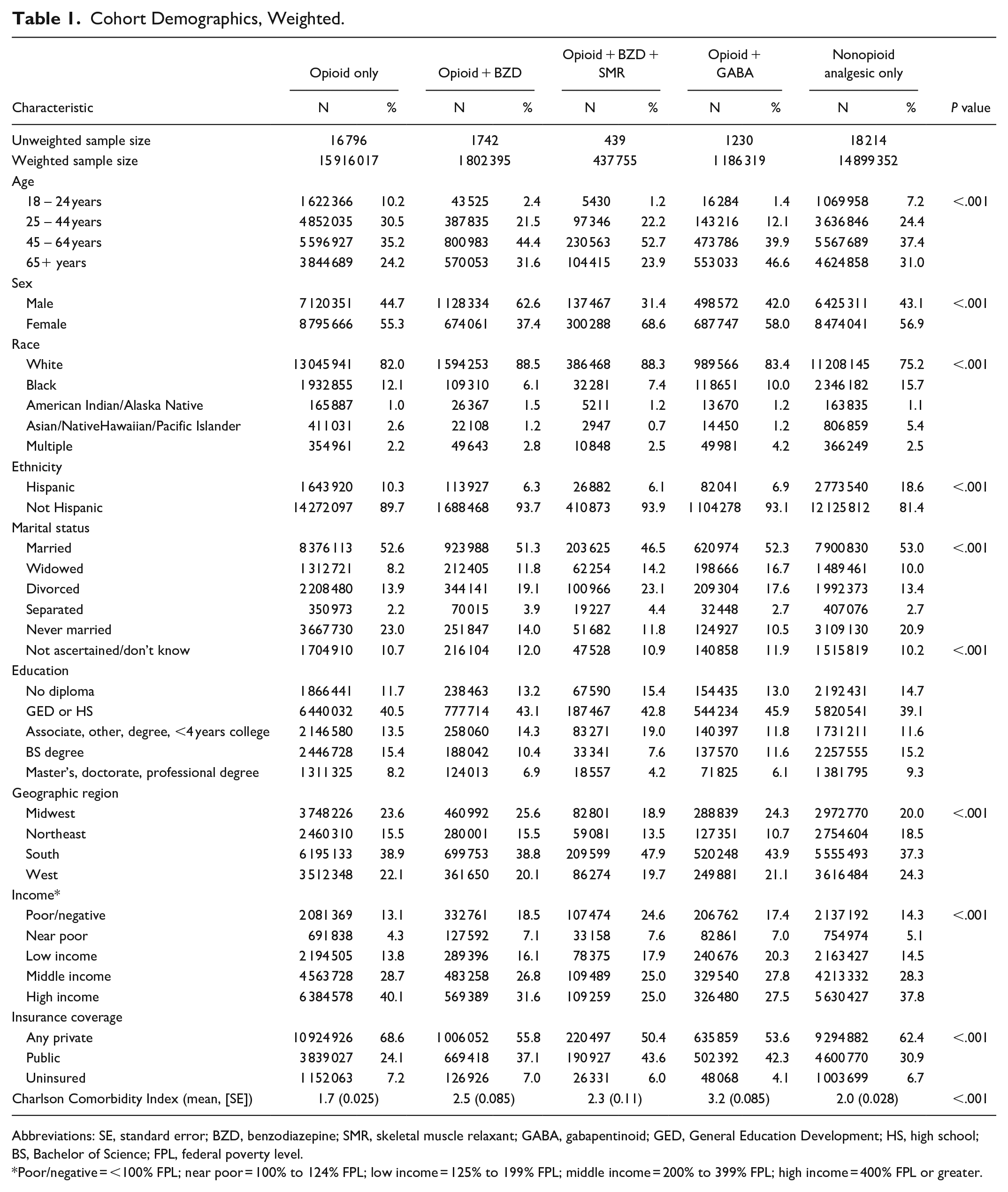
Abbreviations: SE, standard error; BZD, benzodiazepine; SMR, skeletal muscle relaxant; GABA, gabapentinoid; GED, General Education Development; HS, high school; BS, Bachelor of Science; FPL, federal poverty level.
Poor/negative = <100% FPL; near poor = 100% to 124% FPL; low income = 125% to 199% FPL; middle income = 200% to 399% FPL; high income = 400% FPL or greater.
Healthcare Costs and Resource Utilization
Between 2009 and 2019, opioid-gaba respondents had the highest average annual total medical costs ($22 129) compared to opioid-only ($10 813), double-threat ($16 317), triple-threat ($17 084), and nonopioid analgesic only respondents ($6929). This trend was also observed in average inpatient hospitalization costs, outpatient costs, office-based costs, and prescription drug costs (Figure 2). Opioid-gaba respondents had the highest average number of inpatient hospitalizations, outpatient visits, office-based visits, and prescription drugs. Triple-threat respondents had the highest average ED costs ($5699) and average yearly number of ED visits (Figure 2). Adjusted for covariates, opioid-gaba respondents also had the highest incremental total annual medical costs (+$11 684, 95% CI: +$8633, +$13 571) of the study groups when compared to nonopioid analgesic respondents. Opioid-only respondents had the highest incremental effect on outpatient costs (+$950, 95% CI: +$856, +$1064; Table 2) and the lowest incremental effect in other cost categories compared to nonopioid analgesic respondents. Double-threat respondents had a higher incremental effect on total medical costs (+$8789, 95% CI: +$6893, +$5471), driven by inpatient costs and prescription drug costs compared to nonopioid analgesic respondents. Double-threat respondents also had higher incidence rates of inpatient hospitalizations (IRR: 2.69, 95% CI: 2.37, 3.04) and ED visits compared to nonopioid respondents. Triple-threat respondents had a similar result to double-threat respondents, with a high incremental effect on total medical costs compared to nonopioid analgesic respondents (+$9876, 95% CI: +$6527, +$12 240).
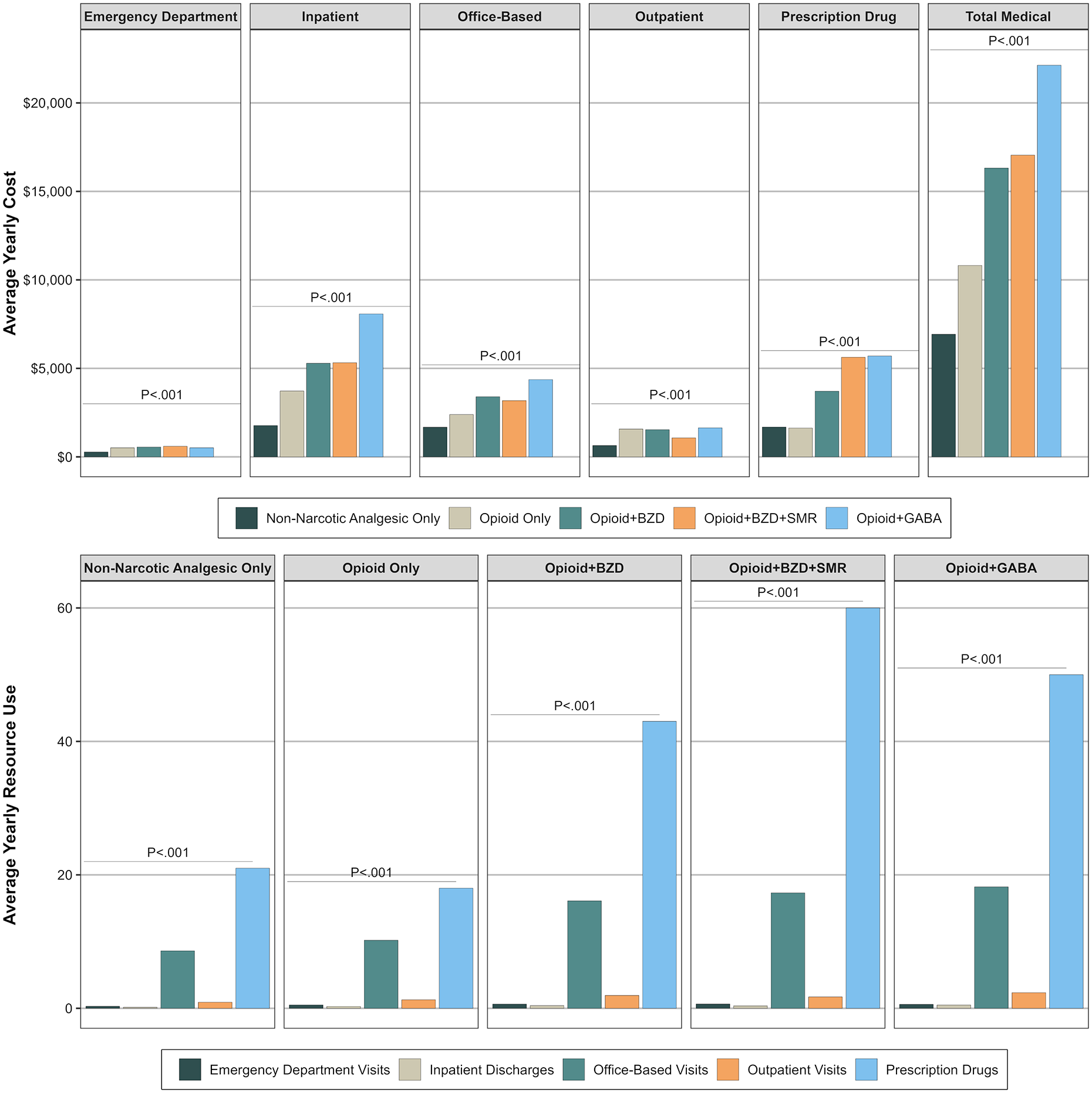
Average yearly costs and resource use by drug group, 2009 to 2019. The top half of the figure illustrates unadjusted average yearly cost by cost category compared across nonnarcotic analgesic, opioid-only, opioid/BZD, opioid/BZD/SMR, and opioid-gaba respondents. The bottom half illustrates unadjusted average yearly resource use by resource use category compared across the 5 drug groups of interest.
Estimated Average Annual Differences in Healthcare Costs and Resource Utilization by Treatment Group, 2009 to 2019.
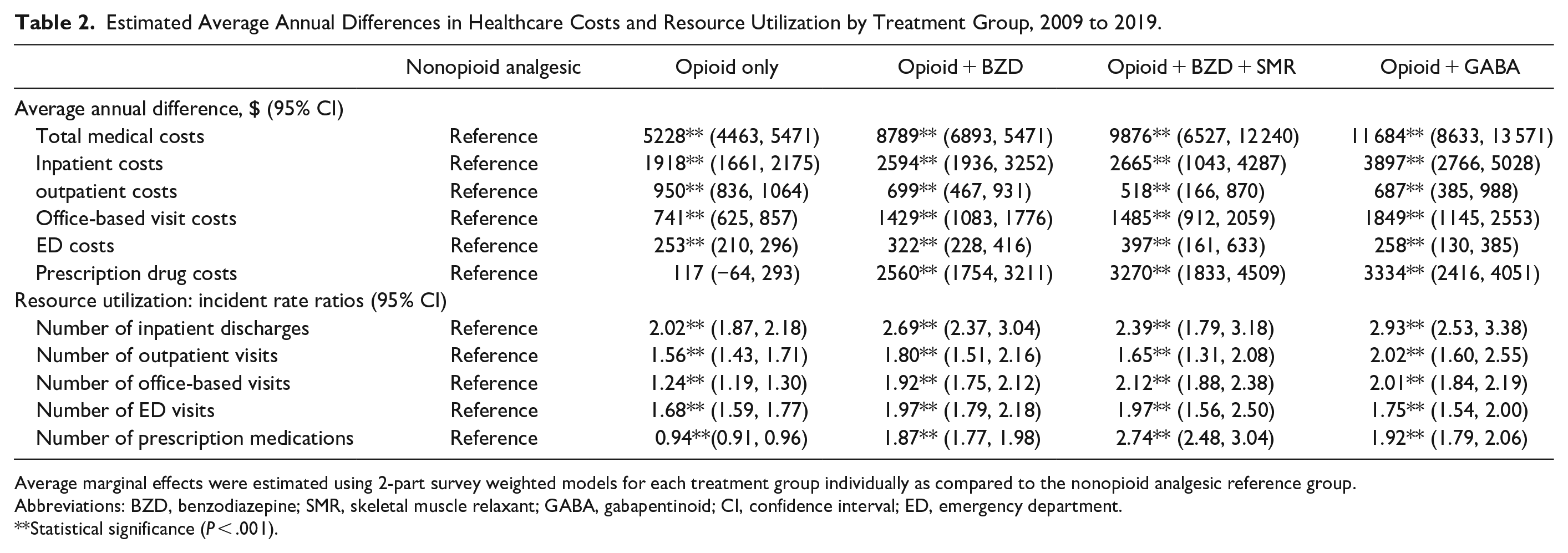
Average marginal effects were estimated using 2-part survey weighted models for each treatment group individually as compared to the nonopioid analgesic reference group.
Abbreviations: BZD, benzodiazepine; SMR, skeletal muscle relaxant; GABA, gabapentinoid; CI, confidence interval; ED, emergency department.
Statistical significance (P < .001).
Annual Trends in Healthcare Costs and Resource Utilization
Table 3 describes the yearly trends in costs and resource utilizations for the study drug groups compared to the nonopioid analgesic group. All groups had a significantly higher average annual increase in total medical costs compared to nonopioid analgesics, with the opioid-gaba group reporting the highest average annual increase of all pairs (+$1237, P < .001). Opioid-only respondents had a significant average annual increase in outpatient costs (+$98.84, P < .001), office-based costs (+$106.14, P < .001), and prescription drug costs (+$53.99, P = .03) compared to nonopioid analgesic respondents. Opioid-only respondents also had significantly higher average annual increases in outpatient visits (+0.042, P = .03) and office-based visits (+0.22, P < .001) compared to nonopioid analgesic respondents. Triple-threat respondents had significantly higher average annual increases in office-based costs compared to nonopioid respondents (+$189.36, P = .02). Double-threat respondents had significantly higher average annual increases in prescription costs (+$317.42, P < .001), number of inpatient hospitalizations (+0.030, P < .001), number of outpatient visits (+0.17, P = .04), and number of ED visits (+0.042, P = .02) compared to nonopioid analgesic respondents.
Average Annual Differences in Costs and Resource Utilizations Trends of Groups Compared to Nonopioid Analgesic Only Respondents, 2009 to 2019.
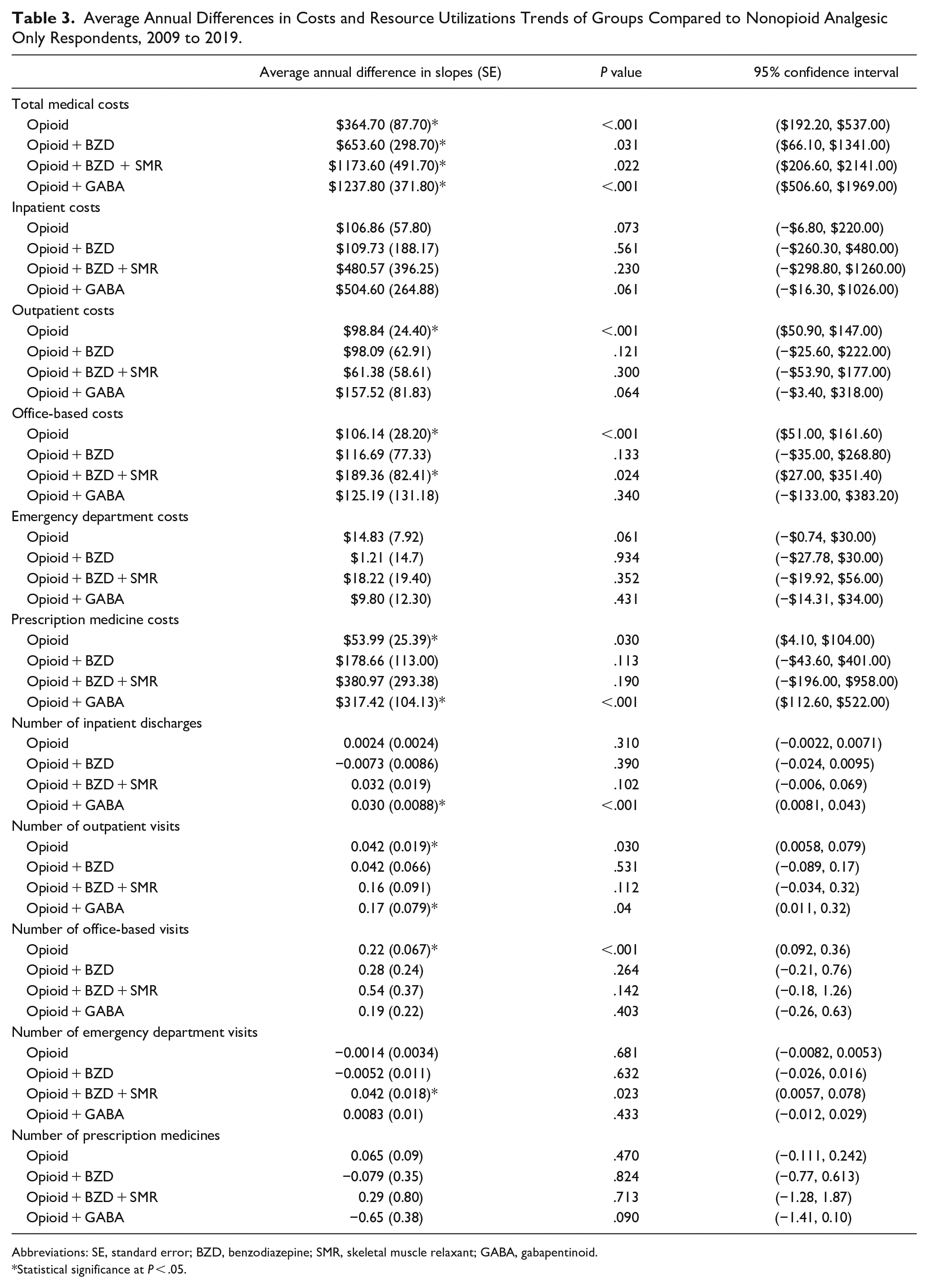
Abbreviations: SE, standard error; BZD, benzodiazepine; SMR, skeletal muscle relaxant; GABA, gabapentinoid.
Statistical significance at P < .05.
Discussion
In this analysis, we found that double- or triple-threat respondents demonstrated greater increases in the growth of inpatient hospitalizations, ED use, office-based visits, and total medical costs over time compared to nonopioid analgesic respondents. Participants in these two groups were older and had a higher comorbidity burden compared to nonopioid analgesic respondents, and tended to fall into lower socioeconomic strata with higher rates of public insurance. Our findings indicate that there is an association between double- and triple-threat status and healthcare expenditures; however, the underlying mechanism for this is unclear.11,39,40 We believe this may be due to several potential hypotheses. First, double- or triple-threat respondents may have complicated clinical presentations that include the presence of multiple comorbidities, such as anxiety or depression in addition to chronic pain. 41 Second, the association between these drug combinations with increased inpatient hospitalizations and ED use may suggest that these patients have a higher risk of experiencing compounded adverse events, such as respiratory depression. This is particularly concerning, as participants in these groups were, on average, older than the nonopioid analgesic group. Respiratory depression in these patients may lead to a higher risk of negative consequences, such as opioid-related overdose compared to opioid-only respondents.10,42 Of the five study groups, respondents in the triple-threat group had the greatest increase in growth of inpatient hospitalizations, ED costs, and number of ED visits compared to nonopioid analgesic respondents.
Of the study drug group comparators, opioid-gaba respondents had the highest total medical costs in total and in growth over time when compared to nonopioid analgesic respondents. Opioid-gaba respondents also had greater inpatient hospitalizations and prescription drug costs and use compared to nonopioid analgesic respondents over time. These findings are similar to other studies that indicate opioid prescribing rates are greater among individuals who are also prescribed gabapentinoids.43 -46 The steady increase in gaba use may reflect prescriber’s perception that gabapentinoids are a safer option for chronic pain management. 47
We hypothesize that the effect of gabapentinoids potentiating the effect of opioids may be responsible for the increase in growth of inpatient hospitalization costs and use over time for opioid-gaba respondents. While these higher inpatient expenditures have been noted and somewhat mitigated through policy changes by certain regulatory bodies, such as the Centers for Medicare and Medicaid Services and third-party payers, it may not be enough to stave off the rising trends noted here.48 -50 Gaba and pregabalin are foundational in the use of multimodal analgesia to treat chronic pain. 51 To continue encouraging safe use of these agents concurrently with opioids, investigators should focus on identifying factors associated with inpatient events and costs among opioid-gaba users.
We noted higher healthcare costs across various resource categories among double-threat, triple-threat and opioid-gaba respondents in total and over time. While we hypothesized that these results may potentially be due to a higher rate of severe adverse events commonly observed with these medication combinations (eg, respiratory depression, etc), we cannot rule out other potential causative factors. For example, the triple-threat and opioid-gaba groups were older than the other study groups and had a higher comorbidity burden. It is possible that healthcare costs were simply higher in these groups due to higher disease burden, which would require more intensive care. However, literature suggests that the negative effects of concurrent opioid-CNS depressant use persist even among cohorts matched on opioid use/nonuse. Hawkins and colleagues reported a higher rate of all-cause mortality and opioid overdose with concurrent opioid/BZD use among Veterans Affairs patients with posttraumatic stress disorder compared to matched groups of opioid-only or BZD-only patients. 52 Weisberg et al reported similar results among a population of long-term prescription opioid and/or BZD users with HIV infection. 53 Finally, Sharma and colleagues found that concurrent use of opioids and BZDs resulted in a higher risk of hospitalization/ED visits and mortality over a matched group of opioid-only users in a Canadian sample. 13 Although our analyses did not utilize matching, we posit that the overall increase in healthcare expenditure among triple-threat and opioid-gaba respondents noted here may be, in part, associated with a higher use of resources linked to opioid overdose and related events.
Compared to nonopioid analgesic respondents, the number of opioid-only respondents decreased over time. This downward trend supports other published national trends starting in 2012 for opioid prescriptions reported by the CDC and other sources.54,55 We hypothesize the reason for this decline to be multifactorial. Primarily, US policies at both the federal and state level aimed at increasing scrutiny on opioid use may have played a strong role in our observed reduction of opioid-only respondents. For example, state prescription drug monitoring programs (PDMP), an increase in the amount and quality of provider education, and the release of clinical guidelines aimed at restricting the use of opioids for noncancer pain may have contributed to the decline noted here.6,17,56 -61
There was an increase in the use of outpatient and office-based services and costs over time among opioid-only respondents compared to nonopioid analgesic respondents. Although the average annual increases in outpatient visit, office-based visit, and prescription drug costs among opioid-only respondents may seem modest, third-party payers tasked with covering healthcare expenses for thousands of such patients should remain mindful of these gradually increasing costs, which can have an impact on patients due to increased opportunity costs. Opioid-only respondents also had lower inpatient hospitalizations, ED use, and ED costs when compared to nonopioid analgesic respondents. We hypothesize that various policies aimed toward expanding behavioral health services and increasing access to medications used for opioid use disorder (MOUD) may have driven these observations. For example, Medicaid expansion after implementation of the Affordable Care Act has been associated with reductions in opioid-related hospital use and an increase in access to buprenorphine prescriptions for MOUD.62 -64 Federal policies have also greatly expanded access to treatment for opioid use disorder, such as the Comprehensive Addiction and Recovery Act (CARA) of 2016, the 21st Century Cures Act, the Substance Use-Disorder Prevention that Promotes Opioid Recovery and Treatment (SUPPORT) for Patients and Communities Act of 2018, and the Consolidated Appropriations Act.65 -68 While these are only hypotheses for the underlying reasons for the trends among opioid-only patients noted here, future research may want to focus on these trends with the advent of newer legislation, such as elimination of the X-waiver, which was previously a required training and registration process by physicians to prescribe buprenorphine. 69
Limitations
Our study had limitations of note. Some MEPS data components may be subject to recall bias, including reported demographic characteristics. We did not distinguish between chronic or acute opioid respondents. Therefore, our results cannot be extrapolated to one subgroup or the other. Nonopioid analgesic respondents may have used their medications for reasons other than for alleviating pain, such as low-dose aspirin for the use of cardiovascular disease prevention. Similarly, respondents reporting use of benzodiazepines or gabapentinoids may have been using their medication for psychiatric conditions (eg, anxiety), or alcohol withdrawal. Accordingly, we cannot assume that our results apply only to patients who were using these medications for pain. We could not ascertain whether opioid respondents also had substance use disorder, or whether these respondents were prescribed their medications from 1 physician versus several (eg, “doctor shopping”). We could not be certain whether MEPS respondents who reported purchase of prescription medications took those medications as directed or whether use of medications in concurrent categories (double threat, triple threat, opioid-gaba) overlapped in any 1 round. Our study groups were also significantly different from each other with regard to demographic characteristics, which may have affected our findings. For example, the opioid-gaba group was, on average, the oldest group, which may have played a role in their high use of inpatient hospitalizations. However, we did control for this bias to a certain extent by adjusting for age in our models. Because we could not be certain of outpatient or office-based physician specialty type, we also could not determine whether the increase in use of these services among opioid-only respondents was due to an increase in the use of behavioral health services related to substance use disorder or not. We could not ascertain the primary reason for each MEPS respondent’s reported healthcare use event (eg, inpatient hospitalization, outpatient visit, etc). Therefore, we could not ascertain whether resources utilized were primarily used for pain, or for another reason. While we utilized mutually exclusive categories to define our study groups, there may have also been some cases of chronic opioid respondents picking up a single (“one time”) concurrent CNS depressant (eg, a benzodiazepine). The effects of a singular concurrent prescription may be less impactful than the effects of a chronic, long-term concurrent prescription, which may have affected our results. Finally, we did not include other medications used for pain that may also have been used for other indications (eg, SSRIs, SNRIs, etc). This may have resulted in an underestimation of individuals in the “non-opioid analgesic” group.
Conclusions
Compared to nonopioid analgesic respondents, opioid-only respondents had a greater use of outpatient or office-based services. Double- or triple-threat respondents and opioid-gaba respondents demonstrated increased total medical costs and use of inpatient hospitalizations and ED services over time compared to nonopioid analgesic users. Policymakers and third-party payers should orient future legislation, regulation, and education on emphasizing and rewarding safe and continuous oversight from physicians and pharmacists for patients taking concurrent opioid combinations to stagnate the rising risk of negative clinical consequences.
Supplemental Material
sj-docx-1-saj-10.1177_29767342241247372 – Supplemental material for Association of Opioid and Concurrent Benzodiazepine, Skeletal Muscle Relaxant, and Gabapentinoid Usage on Healthcare Expenditure and Resource Utilization: A Serial Cross-Sectional Study, 2009 to 2019
Supplemental material, sj-docx-1-saj-10.1177_29767342241247372 for Association of Opioid and Concurrent Benzodiazepine, Skeletal Muscle Relaxant, and Gabapentinoid Usage on Healthcare Expenditure and Resource Utilization: A Serial Cross-Sectional Study, 2009 to 2019 by Aryana Sepassi, Meng Li, Kangho Suh, Britney Stottlemyer and Mark Bounthavong in Substance Use & Addiction Journal
Footnotes
Author Contributions
AS: Conceptualization, data curation, formal analysis, investigation, methodology, project administration, resources, software, validation, visualization, roles/writing—original draft, writing—review and editing.
ML: Conceptualization, methodology, roles/writing—review and editing, supervision.
KS: Conceptualization, methodology, roles/writing—review and editing, supervision.
BS: Conceptualization, methodology, roles/writing—review and editing.
MB: Conceptualization, methodology, roles/writing—review and editing.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Compliance,Ethical Standards,and Ethical Approval
The Ethics Committee of the University of California, Irvine, waived the need for ethics approval and the need to obtain consent for the collection, analysis, and publication of the retrospectively obtained and anonymized data for this noninterventional study.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
