Abstract
Objectives:
Telehealth treatment with medication for opioid use disorder (teleMOUD) was made possible with regulations following the COVID-19 pandemic that permitted prescribing buprenorphine without an in-person visit. This study evaluates the self-reported outcomes of patients treated by teleMOUD using the Brief Addiction Monitor (BAM), a 17-question tool that assesses drug use, cravings, physical and psychological health, and psychosocial factors to produce 3 subset scores: substance use, risk factors, and protective factors.
Methods:
Patients treated by a teleMOUD provider group operating in >30 states were asked to complete an app-based version of BAM at enrollment and at 1 month. Patients who completed both assessments between June 2022 and March 2023 were included.
Results:
A total of 2556 patients completed an enrollment BAM and 1447 completed both assessments. Mean number of days from baseline BAM to follow-up was 26.7 days. Changes were significantly different across most questions. The substance use subscale decreased from mean 2.6 to 0.8 (P < .001), the risk factors subscale decreased from mean 10.3 to 7.5 (P < .001), and the protective factors subscale increased from mean 14.3 to 15.0. (P < .001). Substance use and risk factor subscale changes were significant across all sex and age groups, while protective factors subscale did not improve for those <25 and >54 years. Patient reports of at least 1 day of illegal use or misuse decreased, including marijuana (28.1% vs 9.0%), cocaine/crack (3.9% vs 2.6%), and opioids (49.8% vs 10.5%).
Conclusions:
Among patients treated by teleMOUD who completed assessments at enrollment and 1 month, there was improvement in drug use, risk factor, and protective factor scores.
Introduction
Telehealth treatment with medication for opioid use disorder (teleMOUD) was facilitated during the COVID-19 pandemic with relaxation of the Ryan Haight Act, which otherwise mandated an in-person evaluation prior to prescribing buprenorphine. 1 Buprenorphine is one of 3 medications approved in the United States for treatment of OUD and is the only one that can be currently prescribed in the outpatient setting and dispensed at commercial pharmacies for at-home use. 2 The convenience of telehealth combined with the ability to obtain the prescription at a pharmacy is notable 3 and telehealth services have proliferated in response.4,5
Prior studies evaluating the efficacy of teleMOUD are largely limited to descriptive articles and studies reporting rates of treatment retention. The results of these studies have so far been promising and suggest that teleMOUD is associated with significantly higher rates of retention.6,7 Recently, there has been an increase in data evaluating outcomes such as overdose among patients receiving teleMOUD; for example, in one study conducted among Medicare beneficiaries, patients treated in telehealth versus in-person visits had similar rates of medically treated overdose. 8 However, a quantitative assessment of patient improvement with teleMOUD is missing, as is a description of outcomes on a broad variety of patients (ie, not just Medicare, Veterans Affairs, or commercially insured). As the federal government and states weigh permanent changes to permit teleMOUD, evidence to evaluate its efficacy may be helpful.
In this article, we evaluate a cohort of patients treated by a teleMOUD provider using the Brief Addiction Monitor (BAM). BAM is a tool used to systematically monitor patient progress during substance use disorder (SUD) treatment. 9 BAM was created in 2013 and was used initially to evaluate veterans undergoing SUD treatment. 10 In our practice, a large telehealth provider with over 10 000 active patients seen in >30 states, we administer the BAM at enrollment and every 30 days. We hypothesized that patients would improve on the 3 domains tested by the BAM in the 30 days following commencement of teleMOUD treatment.
Methods
Included patients were seen by a teleMOUD provider group that sees patients almost exclusively by telemedicine. The model has been described elsewhere, but briefly, it entails regular visits with a continuity provider (either the physician, physician assistant, or nurse practitioner), the provision of buprenorphine treatment for documented OUD, and drug screening at least monthly. 11 Therapy (both group and 1:1 with a licensed clinical social worker or other licensed therapist), peer-support groups (run by other non-licensed staff), and peer recovery coaching services are also available and encouraged but not required. The group has contracts with public and commercial insurers in multiple states, although approximately 40% of patients self-pay for the treatment. Importantly, individuals engaged in teleMOUD must meet several enrollment requirements, including access to internet-connected technology, a safe place to store buprenorphine, absence of any uncontrolled co-occurring condition that would seriously limit medication adherence, and the ability to provide a drug-screening test at random at a minimum of every 30 days, on average.
BAM is a 17-question tool that asks questions about drug use, cravings, physical and psychological health, and psychosocial factors. The BAM results in 3 summary subscales: (1) substance use, including alcohol and/or drug use (3 questions with 12 points possible), (2) risk factors asking about physical and mental health and cravings (6 questions with 24 points possible), and (3) protective factors asking about social supports that may mitigate drug use (6 questions with 24 points possible). Lower substance use and risk factor scores are favorable, while higher protective factor scores are favorable. Therefore, the total score is less meaningful than the score of the 3 subscales. Two questions, one asking about daily use of specific substances (“street drugs” in the verbiage of the tool) and another asking about satisfaction with progress toward recovery goals, are not included in the BAM subscales. For the subscales, scores of ≥1 on the substance use scale or ≥12 on the risk factor scale indicate a need for more intensive treatment. Conversely, protective factor scale scores of ≤12 indicate the need for more intensive social work to augment the patient’s social support structure. 12 Supplemental Appendix 1 contains the list of questions, point values, and the scoring schema used in this study.
The workflow for administering the BAM in this teleMOUD practice is as follows: once a patient is checked out from their first visit, instructions to complete the BAM are surfaced in the group’s dedicated telehealth app, and the survey is completed electronically by the patient on their own within the app. Patients are instructed to complete the BAM within 10 days or else it expires and is no longer available. Following that, when a follow-up visit is booked for the same patient, if the date of the follow-up visit was 23 days or more from the first visit, instructions to complete the follow-up BAM will be surfaced to the patient 5 days before the follow-up visit. The follow-up BAM is available from that point until 5 days after the follow-up appointment. To reflect the possibilities permitted by this algorithm, BAM questionnaires that were completed 18 to 40 days after the initial visit were considered as the 1-month measurement. If a patient completed more than 1 BAM 18 to 40 days after their first BAM, we analyzed only the first follow-up submission within the window. Patients eligible for baseline had the BAM questionnaire surfaced in their app within the first 10 days of care. A patient is eligible for month 1 questionnaire if they (1) had a visit on treatment day 23 or after and (2) the BAM questionnaire is surfaced during the month 1 window (18-to 40-day window).
This study took place from June 15, 2022 (the first date when the BAM process was fully implemented) to March 15, 2023 (the time of decision to analyze the data). The study was approved by the Western Copernicus Group (WCG) Institutional Review Board. Descriptive analysis was conducted to describe sex, age category, and appointment payment method (insurance vs self-pay used to cover the teleMOUD service) for patients who completed baseline BAM questionnaires and those who completed the baseline BAM and the follow-up BAM questionnaire. Chi-squared test was used to determine the differences between the baseline and follow-up sample. Age categories were defined as 18 to 24, 25 to 34, 35 to 44, 45 to 54, and 55 years old and older.
As patients are clustered by their primary provider, we explored using linear multilevel models to adjust for clustering. We found very low intracluster correlation coefficient (ICC) results when using random intercept models with exposure to our program and BAM sub-scores. The ICC score was 0.026 for the risk factor subscale, 0.019 for the usage subscale, and 0.043 for the protective factor subscale. These results indicated that analyses that do not account for clustering are appropriate for our dataset.
We calculated the mean score and standard deviation by question at baseline and month 1 among patients who completed baseline and month 1 measures. A paired t-test, a Wilcoxon sign-rank test, and chi-squared tests were used to determine if the follow-up score was different from the baseline score, as appropriate based upon the distribution of the data. In addition to describing changes for each question, we described self-reported use of “illegal or street drugs or abuse of any prescription medications” (verbiage from BAM) at baseline and month 1 among patients who completed both measures.
To assess changes in sub-scores over time and to determine if patient characteristics altered the relationship between enrollment in the program and change in BAM scores, we used paired t-tests and signed-rank tests to determine statistical significance of change in score over time. Patient characteristics included age category, sex, and appointment payment method. In addition to MOUD by the primary provider, patients are offered behavioral health services, and are considered exposed to behavioral health services if they attended at least one therapy session. To determine if exposure to behavioral health services and payment method type impacted the change over time in score, a Wilcoxon signed-rank test or independent samples t-test (where appropriate) was used to compare the change in sub-scores by exposure status and payment type.
Results
During the study period, 3782 patients were prompted to complete an initial BAM, of which 2556 (67.6%) completed and submitted the questionnaire. Of the 2556 patients who completed baseline, 1739 were eligible for month 1 BAM. Of the eligible patients, 1447 (83.2%) completed the month 1 BAM. Table 1 describes the characteristics of patients who completed baseline BAM and those who completed month 1 BAM and Table 2 describes patients eligible for month 1 BAM, and those who completed month 1 BAM. There was no significant difference between sex and appointment payment method. Patients who completed both baseline and month 1 BAMs varied by age for those 45 to 54 years old (more completed month 1 than expected) and 18 to 24 years old (fewer completed month 1 than expected). The mean number of days from baseline to follow-up BAM was 26.7 days, with a standard deviation of 5.8 days and a range of 18 to 40 days. Our final study sample included the 1447 patients who completed both assessments.
Demographics of Patients who Completed Baseline Versus Completed Baseline and One-Month Brief Addiction Monitor Assessments.
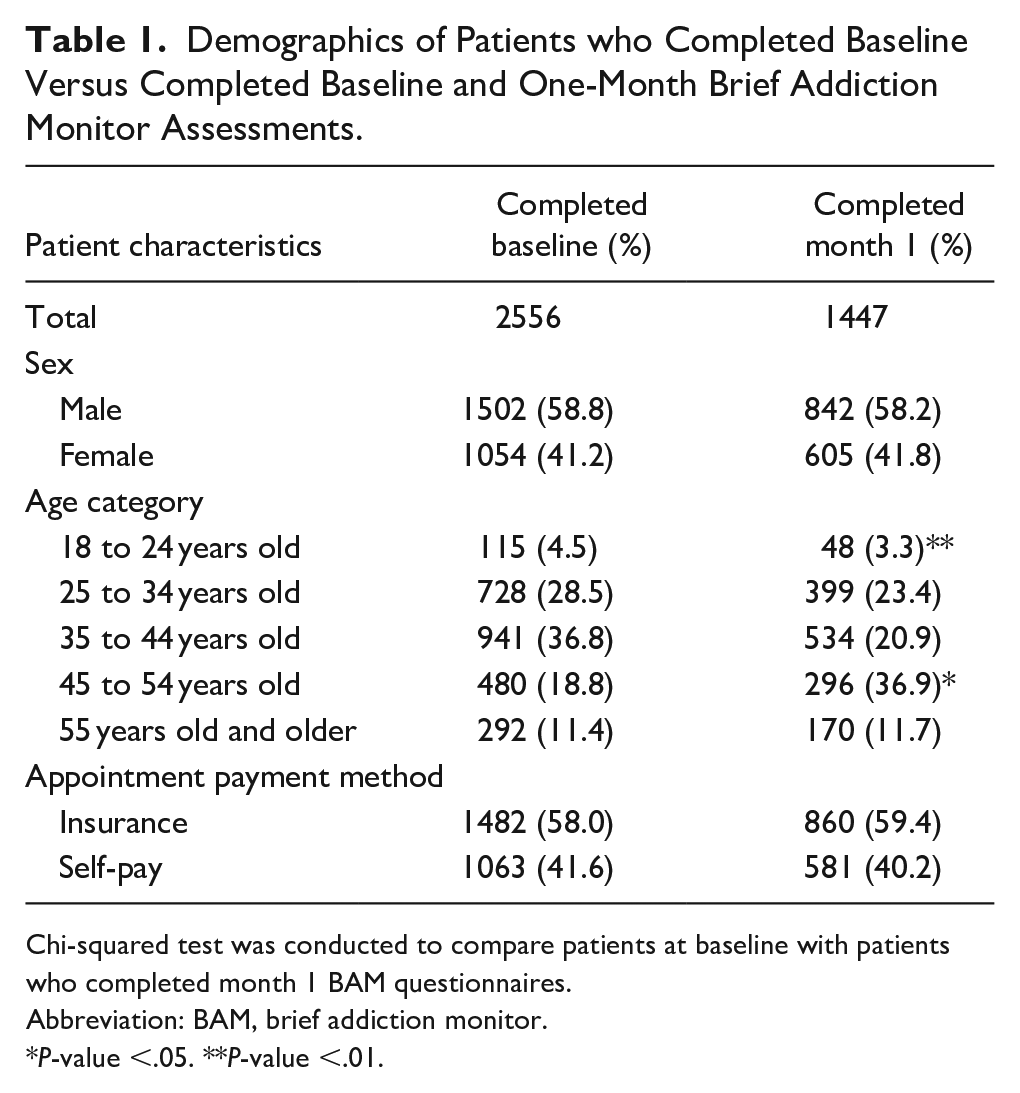
Chi-squared test was conducted to compare patients at baseline with patients who completed month 1 BAM questionnaires.
Abbreviation: BAM, brief addiction monitor.
P-value <.05. **P-value <.01.
Demographics of Patients Who Were Eligible for One-Month versus Those Eligible and Completed One-Month Brief Addiction Monitor Assessment.
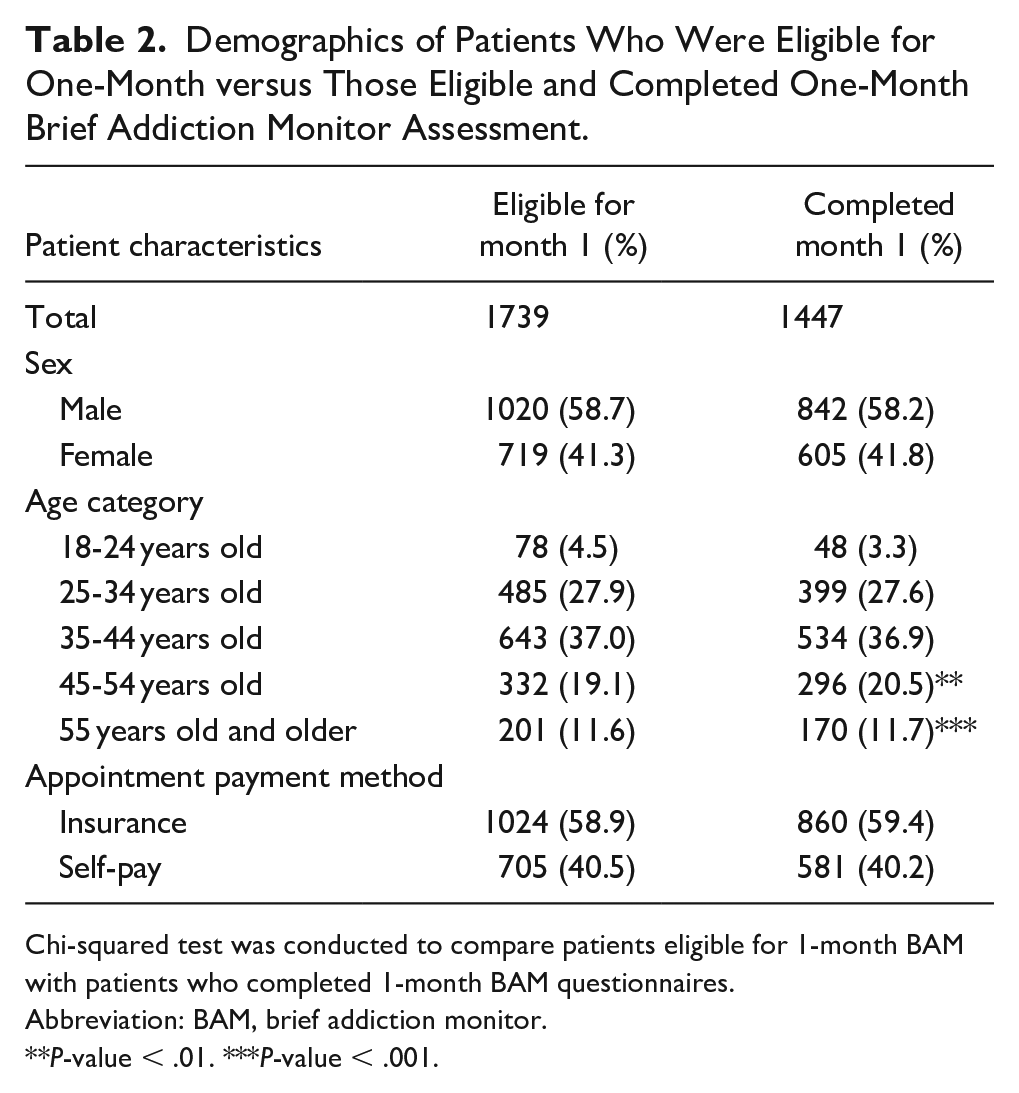
Chi-squared test was conducted to compare patients eligible for 1-month BAM with patients who completed 1-month BAM questionnaires.
Abbreviation: BAM, brief addiction monitor.
P-value < .01. ***P-value < .001.
Table 3 indicates mean scores by question. Key findings are the subset scores: substance usage decreased from 2.61 to 0.81 (P < .001), risk factors decreased from mean 10.32 to 7.49 (P < .001), and protective factors increased from mean 14.27 to 14.99 (P < .001). Each question had statistically significant improvement, including satisfaction with progress toward achieving recovery goals, mean 2.91 to 3.23 (P < .001). Table 4 describes a follow-up to question 6, which asks about the number of days using illegal/street drugs or “abuse” of any prescription medications in the past 30 days. Among those who completed both baseline and month 1, there were 809 patients (55.9%) who reported use initially, while at 1 month, 231 (16.0%) reported use. Those reporting 0 days of use increased for multiple drug categories, including marijuana (71.9% vs 91.0%), sedatives/tranquilizers (84.3% vs 95.8%), cocaine/crack (96.1% vs 97.4%), opioids (50.2% vs 89.5%), and stimulants (88.3% vs 95.7%). For those reporting more than 0 days of any drug use, the number of days of use decreased across multiple categories as well.
Mean Brief Addiction Monitor Scores at Enrollment versus One Month.
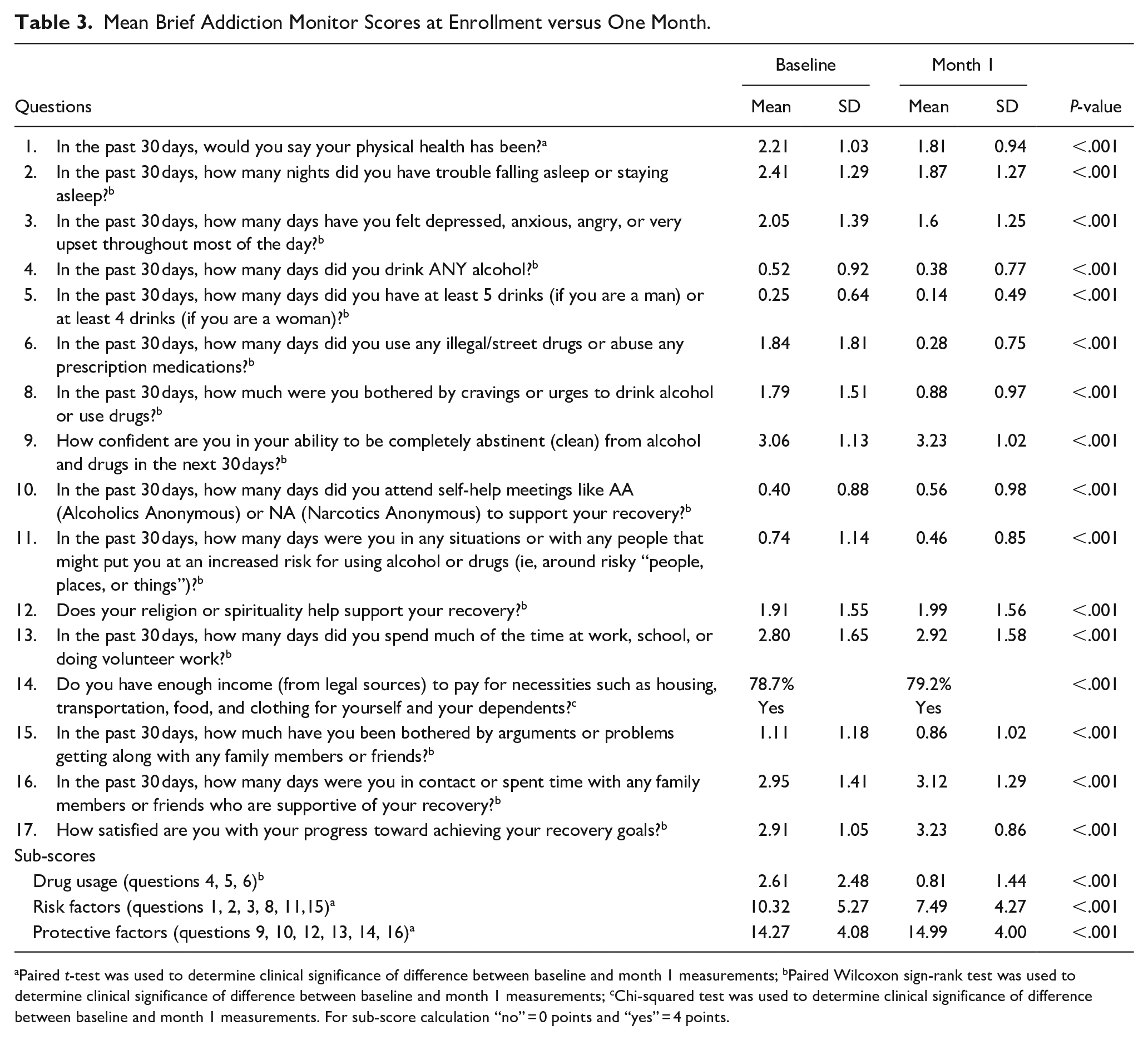
Paired t-test was used to determine clinical significance of difference between baseline and month 1 measurements; bPaired Wilcoxon sign-rank test was used to determine clinical significance of difference between baseline and month 1 measurements; cChi-squared test was used to determine clinical significance of difference between baseline and month 1 measurements. For sub-score calculation “no” = 0 points and “yes” = 4 points.
Self-Reported Illicit Drug Use or Prescription Drug Misuse in the Past 30-Days Among Patients Who Completed Both an Enrollment and One-Month Brief Addiction Monitor Assessment. a
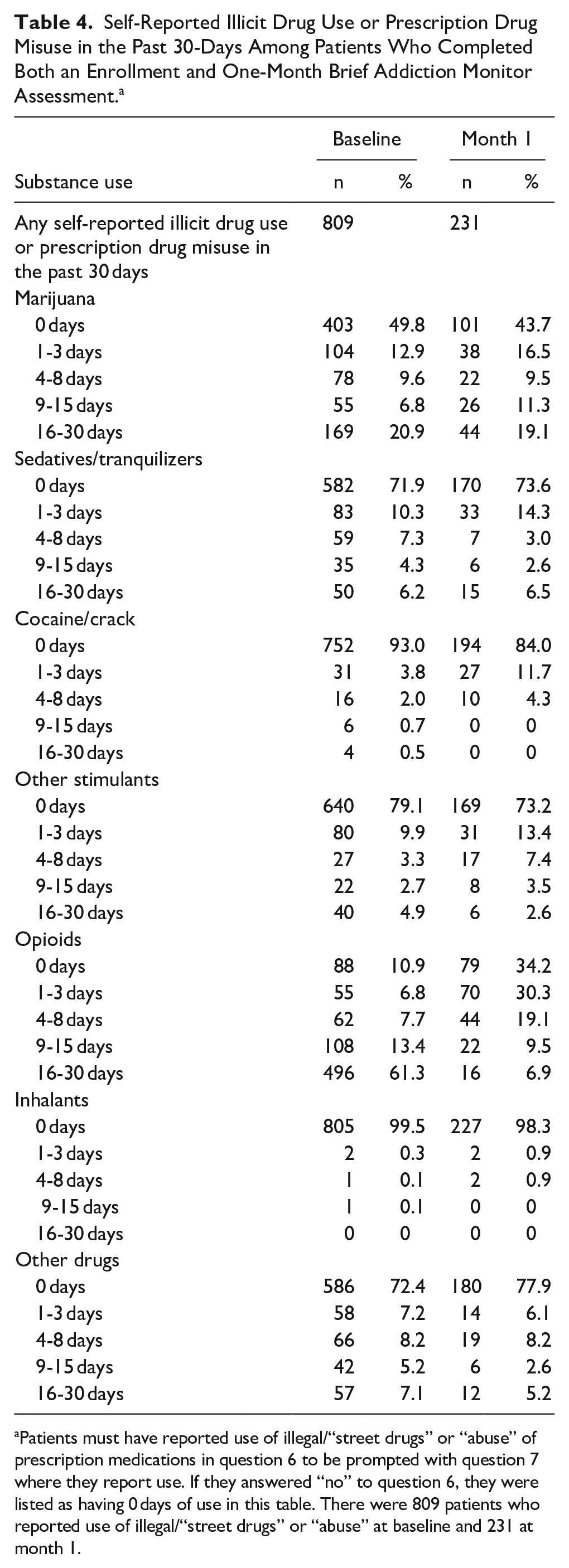
Patients must have reported use of illegal/“street drugs” or “abuse” of prescription medications in question 6 to be prompted with question 7 where they report use. If they answered “no” to question 6, they were listed as having 0 days of use in this table. There were 809 patients who reported use of illegal/“street drugs” or “abuse” at baseline and 231 at month 1.
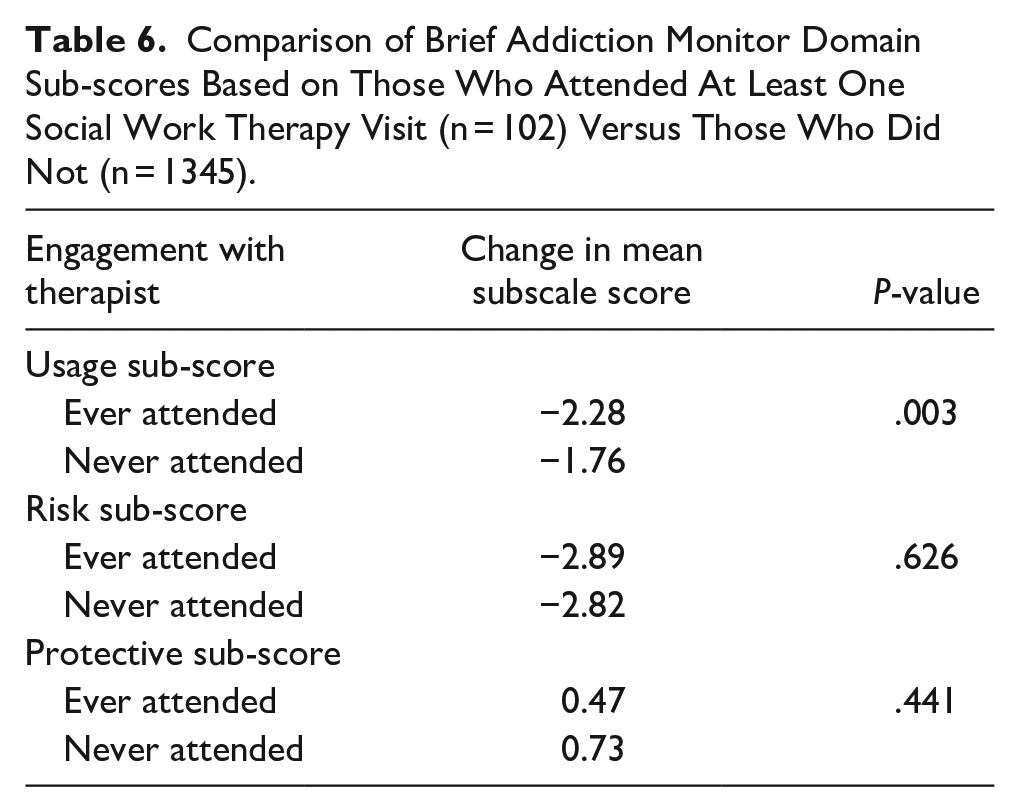
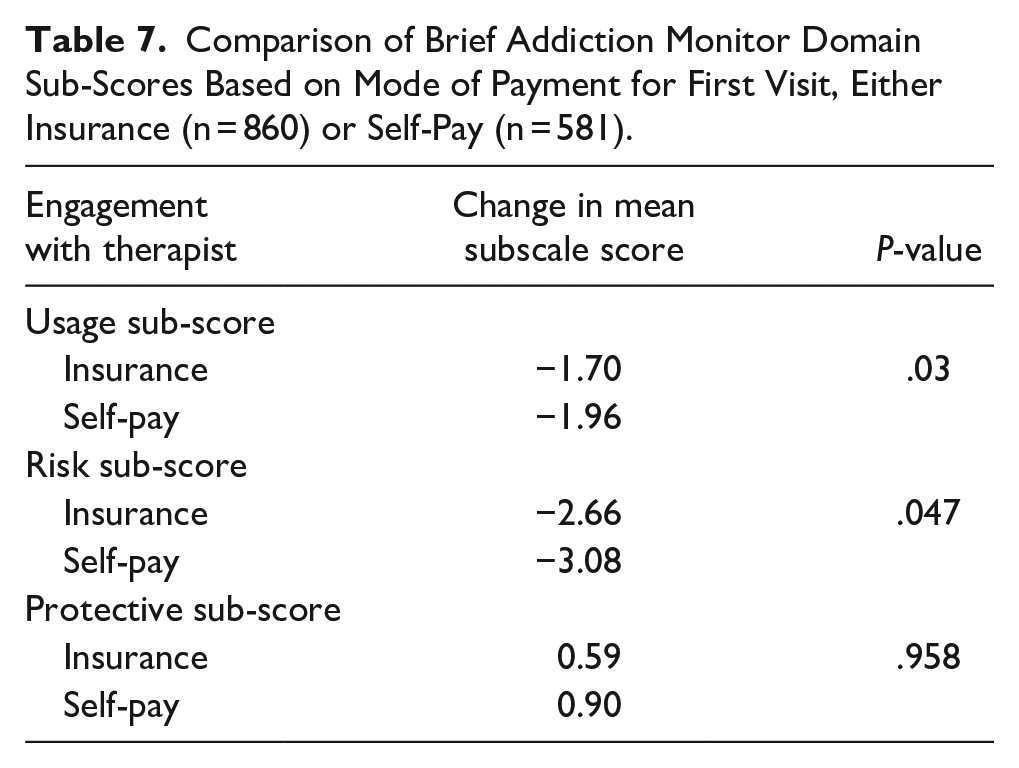
Table 5 shows the statistical significance of change in all 3 subscales overall, by sex and age category. Usage and risk factor scores were significantly improved for all groups. While protective factors were improved for all categories, the improvement was not statistically significant for those 18 to 25 years and 55 years or older. Table 6 shows the change in sub-score by behavioral health treatment exposure status. Patients who were exposed to therapy had, on average, a larger change in mean sub-scores compared to patients who were not exposed. When comparing change over time by exposure status, the usage sub-score was the only comparison that was statistically improved. Table 7 describes the change in subscale scores by appointment payment method. Self-pay patients had statistically significant larger changes in scale sub-scores for usage and risk factor subscales.
Change in Mean Brief Addiction Monitor Subscales Stratified by Overall, Sex, and Age Category.
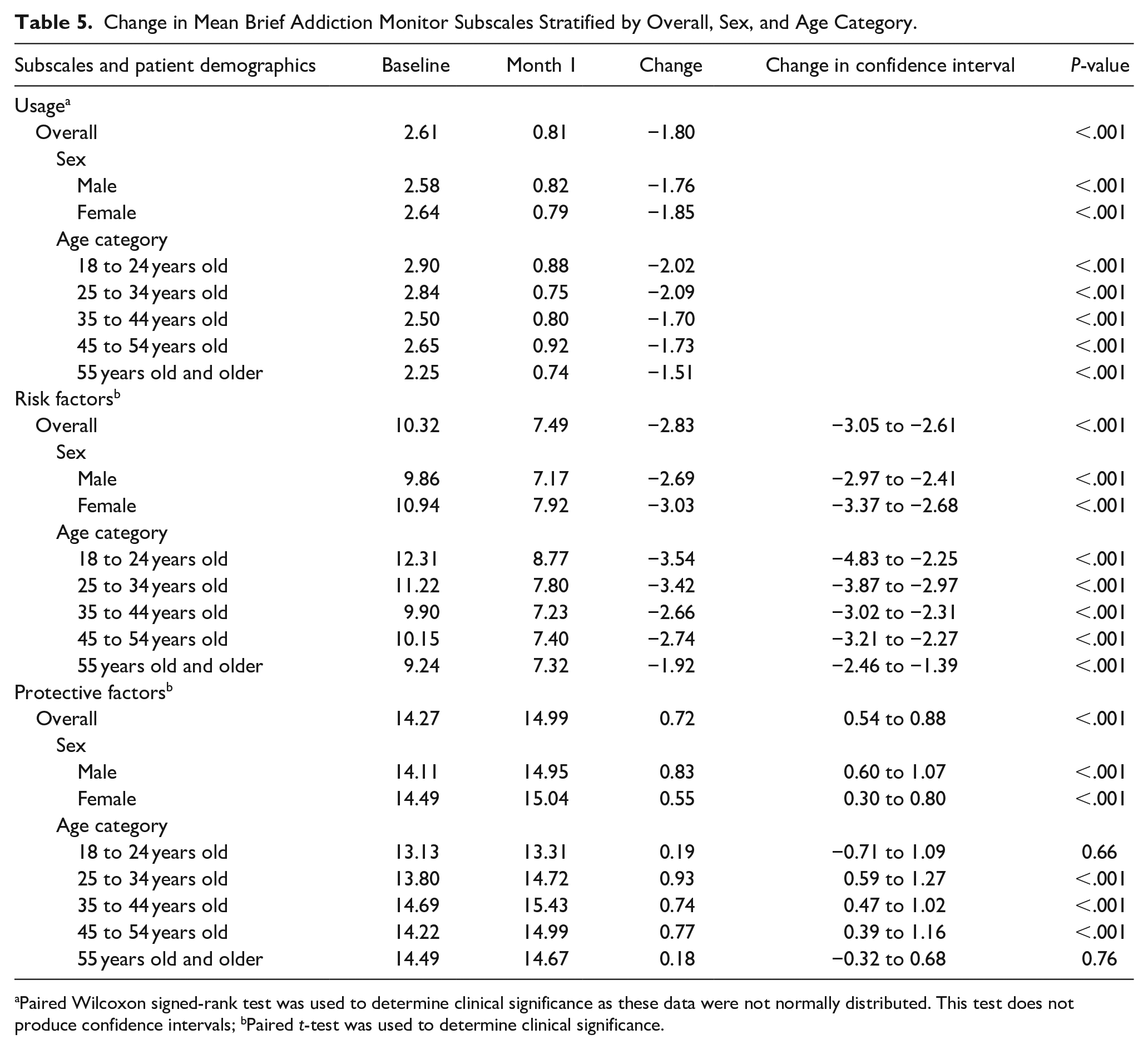
Paired Wilcoxon signed-rank test was used to determine clinical significance as these data were not normally distributed. This test does not produce confidence intervals; bPaired t-test was used to determine clinical significance.
Comparison of Brief Addiction Monitor Domain Sub-scores Based on Those Who Attended At Least One Social Work Therapy Visit (n = 102) Versus Those Who Did Not (n = 1345).
Comparison of Brief Addiction Monitor Domain Sub-Scores Based on Mode of Payment for First Visit, Either Insurance (n = 860) or Self-Pay (n = 581).
Discussion
This study evaluating patient self-reported progress on the BAM instrument found significant improvement in all 3 subscales (substance usage, risk factors, and protective factors) about 1 month after commencing treatment with a single teleMOUD provider. Other recent studies have provided evidence that teleMOUD increases retention in treatment 6 and improves outcomes 13 but have not explored the mechanism underlying this improvement. Our results indicate that substance use drops substantially during the first month of treatment and overall risk factors decrease as well. However, although protective factors increased significantly, the clinical relevance of a mean increase in scores from 14.3 to 15.0 is likely unsubstantial.
Once an individual starts buprenorphine, they have decreased cravings to use other opioids as their receptors are now occupied by buprenorphine. 14 This, in turn, reduces illicit substance use and likely the risky behavior associated with that, and the effect would be expected to begin even shortly after treatment. 15 Protective factors, though, are more difficult to address and likely require more time than just 1 month of treatment. Protective factor questions on the BAM ask about religion/spirituality, time spent at work, school and volunteering, income to pay for necessities, and support from family members and friends. In our experience, these protective factors are skills that are worked on after the patient is stable on buprenorphine and no longer using illicit or non-prescribed opioids.
We also found that these protective factors were not significantly improved for individuals 18 to 25 years old, or for individuals 55 years and older. This finding may indicate that more up-front support is needed for patients in these age groups. A study from another teleMOUD provider found no difference in retention in teleMOUD services by geography or race/ethnicity, but patients under the age of 30 were less likely to have 180-day retention in treatment, suggesting that the younger age group may be most vulnerable to relapse. 16 It is not clear from this research why the older age group experienced no improvement. However, we did discover that patients who self-paid for their OUD treatment had larger decreases in their usage and risk factor scores, possibly indicating a greater effect of treatment for more vulnerable patients.
Although not a primary goal of the study, findings about self-reported past 30-day drug use were also notable. At baseline, of patients who reported “illegal/street drug” use or misused prescription medications, about 30% reported use of marijuana, about 15% used sedatives/tranquilizers, about 4% used cocaine or crack, and about 10% used other stimulants. These findings suggest that use of other drugs with misuse potential is high among our cohort of patients. Our practice is focused primarily on the treatment of OUD, yet we also address other substances, especially stimulants, as patients may not be aware that stimulants may be adulterated with fentanyl. 17 Furthermore, it was reassuring to see that use in all of these categories decreased concurrently with their OUD treatment, indicating that treatment of one use disorder may also reduce use of other substances.
There may be other reasons why we observed improvement in 1 month. The clinical model is not limited to buprenorphine, but also includes unlimited group therapy sessions which are frequently offered throughout the week, as well as peer recovery coaching, case management, and one-on-one therapy with a social worker. We described an even greater reduction in drug use among those who also participated in therapy sessions, although those patients may have been more motivated and self-selected therapy. Patients are also required to submit urine toxicology tests immediately upon entering treatment with the group and typically frequently (about every 14 days) near the beginning of treatment. Providing urine toxicology tests may increase accountability and decrease a patient’s desire to use other substances, although falsification of results is possible. 18
Limitations
There are several limitations to this study that must be considered. The teleMOUD model is not appropriate for all patients. Patients either need insurance that covers the service or the ability to self-pay as well as a safe place to secure their buprenorphine and access to technology, which excludes some of the most vulnerable patients struggling with opioid use disorder. There was selection bias as not every patient completed the baseline BAM, and 57% completed both a baseline and 1-month assessment. Patients who relapsed, abandoned the program, or were struggling with recovery would leave treatment and not be eligible for month 1 BAM. Although we were able to ascertain that patients were well-matched on sex and appointment payment method, patients who completed the 1-month assessment skewed toward the older age groups. There are also certain to be other undetected differences in these groups. Completion of the BAM is encouraged but is not a requirement to be treated. The version of BAM we used is simpler to complete on a phone app, but a revised version may be more predictive. 19 Although we have routinely used BAM in our practice for several years, newer evidence suggests that it may not perform well in a non-veteran population. 20
Core requirements for the program are taking buprenorphine as prescribed, attending scheduled visits, and providing on-demand urine toxicology tests. However, patients who engaged with individual, group, or peer-support therapy may have had improved outcomes due to either self-selection and different characteristics or a treatment effect. The substances that patients in the group use are variable, ranging from fentanyl and heroin to nonmedical use of prescription opioids to other substances like kratom. Outcomes may vary depending on substances used. Additionally, because teleMOUD is more patient-centered and convenient, 3 many patients enter the practice already on buprenorphine. These patients would lead to underestimation of the differences in BAM as they may already be in recovery. There are likely to be differences in efficacy based on provider-level variations. To address this, we did perform a sensitivity cluster analysis and did not detect any significant differences. Finally, our teleMOUD platform uses its own app and unique processes; outcomes may be different in other teleMOUD programs.
Conclusions
Based on initial and 1-month assessments with the BAM, there was improvement in drug use, risk factor, and protective factor scores in the teleMOUD model. By determining the categories by which patients report improvement or in which they need more assistance, this nascent model of care delivery can evolve to provide even more effective care. Further research is needed to ascertain BAM scores beyond 1 month, and also which responses on the BAM predict retention or, conversely, which patients are at risk of treatment discontinuation and are in need of more intensive treatment.
Supplemental Material
sj-docx-1-saj-10.1177_29767342231212790 – Supplemental material for An Assessment of the One-Month Effectiveness of Telehealth Treatment for Opioid Use Disorder Using the Brief Addiction Monitor
Supplemental material, sj-docx-1-saj-10.1177_29767342231212790 for An Assessment of the One-Month Effectiveness of Telehealth Treatment for Opioid Use Disorder Using the Brief Addiction Monitor by Barbara Burke, Brian Clear, Rebekah L. Rollston, Emily N. Miller and Scott G. Weiner in Substance Abuse
Footnotes
Acknowledgements
Preliminary results from this study were presented at the American Telehealth Association Annual Conference and Expo, San Antonio, TX, March 2023.
A preprint server was not used.
Author contributions
Substantial contributions to conception or design of the work, or the acquisition, analysis, or interpretation of data for the work: BB, BC, RLR, SGW. Drafting of the work or revising it critically for important intellectual content: All authors. Final approval of the version to be published: All authors. Agreement to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved: SGW.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Ms. Burke, Dr. Clear, Ms. Miller and Dr. Weiner are employed by Bicycle Health, and Dr. Clear and Dr. Weiner also have equity interest. Dr. Rollston is a former employee and consultant of Bicycle Health. Outside of this work, Dr. Weiner is also on the acute pain committee of Vertex Pharmaceuticals, Inc and scientific advisory board of Cessation Therapeutics, Inc.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: There was no external funding for this work. Outside of this work, Dr. Weiner is funded by National Institutes of Health grants 5R01DA044167, 5R01HS026753, and 1R01DA058315.
Compliance,Ethical Standards,and Ethical Approval
The study was approved by the Western-Copernicus Group (WCG) Institutional Review Board.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
