Abstract
Background:
West Virginia entered an institution for mental disease Section 1115 waiver with the Centers for Medicare & Medicaid Services in 2018, which allowed Medicaid to cover methadone at West Virginia’s nine opioid treatment programs (OTPs) for the first time.
Methods:
We conducted time trend and geospatial analyses of Medicaid enrollees between 2016 and 2019 to examine medications for opioid use disorder utilization patterns following Medicaid coverage of methadone, focusing on distance to an OTP as a predictor of initiating methadone and conditional on receiving any, longer treatment duration.
Results:
Following Medicaid coverage of methadone in 2018, patients receiving methadone comprised 9.5% of all Medicaid enrollees with an opioid use disorder (OUD) diagnosis and 10.6% in 2019 (P < 0.01). In 2018, two-thirds of methadone patients either had no prior OUD diagnosis or were not previously enrolled in Medicaid in our observation period. Patients residing within 20 miles of an OTP were more likely to receive methadone (marginal effect [ME]: −0.041, P < 0.001). Similarly, patients residing in metropolitan areas were more likely to receive treatment than those residing in nonmetropolitan areas (ME: −0.019, P < 0.05). Metropolitan patients traveled an average of 15 miles to an OTP; nonmetropolitan patients traveled more than twice as far (P < 0.001). We found no significant association between distance and treatment duration.
Conclusions:
West Virginia Medicaid’s new methadone coverage was associated with an influx of new enrollees with OUD, many of whom had no previous OUD diagnosis or prior Medicaid enrollment. Methadone patients frequently traveled far distances for treatment, suggesting that the state needs additional OTPs and innovative methadone delivery models to improve availability.
Keywords
Highlights
West Virginia added Medicaid coverage of methadone in 2018 as part of a federal waiver
We examine geographic proximity as a predictor of methadone use and treatment duration
Those within 20 miles of an Opioid Treatment Program (OTP) had increased odds of receiving methadone
Additional strategies are needed to improve methadone availability across the state
Introduction
West Virginia has the highest drug overdose death rate in the United States (84.1 per 100,000). 1 Increasing access to medications for opioid use disorder (MOUD)—methadone, buprenorphine, and naltrexone—is a vital component in effectively addressing the state’s overdose crisis. Medicaid is an important source of coverage for opioid use disorder (OUD) treatment in West Virginia. This trend accelerated with the Medicaid expansion under the Affordable Care Act in 2014. Over the last decade, West Virginia has been among the fastest growing states in the share of residents covered by Medicaid. 2 A prior study of West Virginia Medicaid found that in the first three years of the Medicaid expansion, the share of patients with diagnosed OUD increased rapidly, and the increase was most rapid for those treated with medications. 3 During this time period, the state Medicaid program covered buprenorphine and naltrexone, but had not yet started covering methadone treatment. 4
Starting in 2018, West Virginia entered an institution for mental disease (IMD) Section 1115 waiver with the Centers for Medicare & Medicaid Services (CMS), which in part allowed Medicaid to cover methadone at West Virginia’s nine opioid treatment programs (OTPs) for the first time. 5 Prior research on IMD waivers using national facility data finds that provisions modestly increased access to medications in outpatient settings. 6 The potential impact of such IMD waivers is likely to be greatest in the states (e.g., Idaho, Indiana, Kentucky, Nebraska, Oklahoma, West Virginia) that did not previously cover methadone but agreed to do so as a condition of the IMD waivers. However, West Virginia has a limited supply of OTPs and is constrained in opening new facilities due to a statewide moratorium which may limit the reach of Medicaid coverage of methadone treatment. The moratorium began in 2007 (West Virginia Code §16-5Y-12) and disallows the issuing of new certificates of need to OTPs, prohibiting new programs from obtaining licensure to operate under current state law. While there have been recent legislative efforts to overturn the moratorium, it remains in effect.
Medicaid coverage of methadone treatment was required by all states under the Substance Use Disorder Prevention that Promotes Opioid Recovery and Treatment (SUPPORT) for Patients and Communities Act as of October 1, 2020, though this coverage was implemented earlier through IMD waivers in states such as West Virginia, which began in 2018. While methadone access has been studied in ecological studies, we are unaware of prior studies examining take-up of methadone following changes in coverage for methadone under state or federal policy. West Virginia is an ideal context to measure take-up of methadone, given the state’s policy under its IMD waiver predates the national policy change, given the share of the state population enrolled in the Medicaid program, and given the prevalence of OUD in the population. Our primary objective is to examine methadone treatment initiation, duration, and characteristics of patients who began methadone treatment following Medicaid coverage of methadone in West Virginia.
Understanding utilization of methadone among enrollees who received Medicaid-covered methadone for the first time following implementation of West Virginia’s policy change is important to inform future policy decisions addressing disparities in access to MOUD. In West Virginia, Medicaid represented the first major source of public funding for OTP services—federal block grants had previously not covered OTP services. 7 Thus, many people covered by Medicaid would likely have either been new to methadone treatment or have been self-paying for methadone treatment prior to the waiver, given there was no coverage of OTP services through Medicaid or Medicare prior to 2018 and 2020, respectively, though this has not been explored. It is also unknown whether Medicaid coverage of OTP services would have different impacts for those already recognized as needing treatment (e.g., enrolled with an OUD diagnosis) compared to those without. Additionally, while expanding Medicaid coverage of OTP services removed the cost barrier, prior studies have identified that distance from an OTP reduces methadone use and treatment duration,8 –14 and there are wide disparities in access to OTPs across the United States, particularly in nonmetropolitan areas.10,15,16 To that end, we also examine whether these known barriers (i.e., travel distance and residing in a nonmetropolitan area) impacted methadone initiation and treatment duration following Medicaid coverage of methadone treatment.
Methods
Data
Our study used data from two main sources. First, we analyzed records of individuals with at least one OUD diagnosis claim (ICD-10 code F11.X) between 2016 and 2019 who were enrolled in the Medicaid expansion program for ten or more consecutive months in a calendar year (N = 43,981 person-years). Second, we recorded the addresses of OTPs from the Substance Abuse and Mental Health Services Administration’s Opioid Treatment Program Directory. 17 The final dataset included 43,288 observations. Observations were excluded due to one of the following reasons: (1) the patient resided in a ZIP code that was not mappable to a ZIP code tabulation area (ZCTA) due to administrative errors in the claims data or (2) the patient resided in a ZIP code outside of West Virginia. Patients in our sample resided in 700 of 708 ZCTAs in West Virginia.
Measures
Our study had two main outcome variables from the Medicaid claims data, including (1) a dichotomous measure that took a value of “1” if a patient received any methadone services from an OTP (procedure code H0020; and “0” otherwise) in 2018 or 2019 and (2) a count variable that calculated treatment duration as the total number of days of methadone treatment using H0020 codes (in West Virginia, methadone is billed on a weekly basis, so we imputed seven days for every billed service).
We compared MOUD use among patients receiving methadone and patients who never received methadone using Medicaid claims among patients with an OUD diagnosis. We created dichotomous measures for receipt of MOUD (e.g., any MOUD, any buprenorphine formulated for OUD, any naltrexone, and any methadone); variables took a value of “1” if a patient was enrolled in Medicaid with an OUD diagnosis and received a given medication, and “0” otherwise. Some patients had claims for multiple MOUD in a given year.
Using our summary variables, we created mutually exclusive groups characterizing the prior year for patients receiving methadone in 2018 or 2019: (1) not previously enrolled in the Medicaid expansion program in the prior year, (2) previously enrolled without an OUD diagnosis, or (3) previously enrolled with an OUD diagnosis in the prior year receiving other forms of MOUD (or no MOUD). Variables took a value of “1” if a patient was enrolled in Medicaid in the prior year with MOUD prescription fills prior to methadone treatment in 2018 or 2019, and “0” otherwise. For example, patients who received buprenorphine in 2017 and then received methadone in 2018 were coded as “1” for “previously in Medicaid with an OUD diagnosis and received buprenorphine” (and “0” otherwise).
We created indicator variables for patient characteristics that have been measured in prior research examining utilization and duration of MOUD treatment,3,18 –20 including enrollee age, sex, race, and ethnicity (Non-Hispanic White, Non-Hispanic Black, Hispanic, and Other). Like prior research, we used International Statistical Classification of Diseases and Related Health Problems (ICD-10) codes to create dichotomous indicator variables for the number of comorbidities common among Medicaid patients with OUD.3,18
We used Esri’s ArcGIS Network Analyst extension to estimate the distance in miles to the nearest OTP from the centroid of each ZCTA. 21 We assigned unmatched ZIP codes to the nearest ZCTA using the Health Resources Services Administration, John Snow, Inc., and the American Academy of Family Physicians’ Uniform Data System (UDS) Mapper crosswalk. 22 We compared the average miles from an OTP by type of MOUD received (any MOUD, buprenorphine, naltrexone, or methadone). We created a categorical variable to indicate proximity to an OTP based on driving distance in miles (less than ten miles, between ten and 20 miles, and over 20 miles). We created the distance categories based on the median distance (i.e., 50th percentile) to an OTP across the full sample. Last, an indicator variable was created that specified whether a patient resided in the same ZCTA as an OTP.
Similar to our hypothesis that distance to the nearest OTP would be associated with methadone use, we hypothesized that county classification as a metropolitan or nonmetropolitan area would influence methadone initiation and duration. We stratified and examined the receipt of methadone by county designation as metropolitan or nonmetropolitan county using the US Department of Agriculture 2013 rural–urban continuum (RUC) codes. 23 County Federal Information Processing System (FIPS) codes were assigned to each patient ZIP code using the US Department of Housing and Urban Development’s USPS ZIP code crosswalk files; 24 ZIP codes that spanned multiple counties were assigned to the county seat. We then created a dichotomous variable indicating whether a patient resided in a metropolitan (RUC codes 1-3) or nonmetropolitan area (RUC codes 4-9).
Descriptive Analysis
We calculated descriptive statistics and conducted two-sample t-tests to compare the characteristics of patients receiving Medicaid-covered methadone in 2018 or 2019 to patients who never received methadone over the study period for all study variables. We also examined prior enrollment and MOUD use among patients who ever received methadone.
We calculated the mean and median driving miles to the nearest OTP for patients who ever received methadone compared to patients who never received the medication. We also drew a choropleth map in ArcGIS showing driving distance to the nearest OTP of West Virginia’s nine OTPs at the ZCTA level. Additionally, we examined miles to the nearest OTP by patient residence in a metropolitan or nonmetropolitan area and miles to the nearest OTP among patients residing in the same ZCTA as the OTP.
Regression Analysis
We conducted a two-part regression analysis estimating whether distance to the nearest OTP impacted our two main outcome variables of interest. First, we used logistic regression to estimate the odds an enrollee received any Medicaid-covered methadone following the new coverage. Conditional on receiving any methadone, the second model used poisson regression to estimate duration in treatment (measured as days of methadone treatment). Unadjusted analyses examined the associations between distance to the nearest OTP and county classification as a metropolitan or nonmetropolitan county on our 2 main outcomes: (1) receipt of any methadone treatment and (2) conditional on any methadone treatment, duration in treatment. Adjusted analyses controlled for patient characteristics, including age, sex, race and ethnicity, and presence of chronic conditions. Additional regression analyses stratified patients by residence in a metropolitan or nonmetropolitan county, and sensitivity analyses examined distance alone without the inclusion of metropolitan status. Regression analyses were conducted using the glm logit and poisson commands in StataCorp, LLC’s Stata version 17.0. This study was determined to be exempt by the Johns Hopkins University Bloomberg School of Public Health Institutional Review Board.
Results
Descriptive Analysis
Table 1 reports sample characteristics for methadone and non-methadone patients over the full observation period (methadone was measured for the years 2018 and 2019). Following Medicaid coverage of methadone in 2018, 9.5% of patients with OUD received Medicaid-covered methadone; with a significant increase to 10.6% by 2019 (P < 0.01). Buprenorphine was the most common form of MOUD received (54.2%). Naltrexone use was limited over the observation period, with 9.8% receiving the medication. On average, patients initially receiving methadone were older (P < 0.001), resided in metropolitan areas (P < 0.001), and had fewer chronic conditions than patients receiving other MOUD or no medications (P < 0.001). Male patients received other forms of MOUD more often than methadone, whereas female patients were more likely to receive methadone (both P = 0.021).
MOUD Utilization, Methadone Duration, and Demographic Characteristics by Receipt of Methadone, 2016 to 2019.
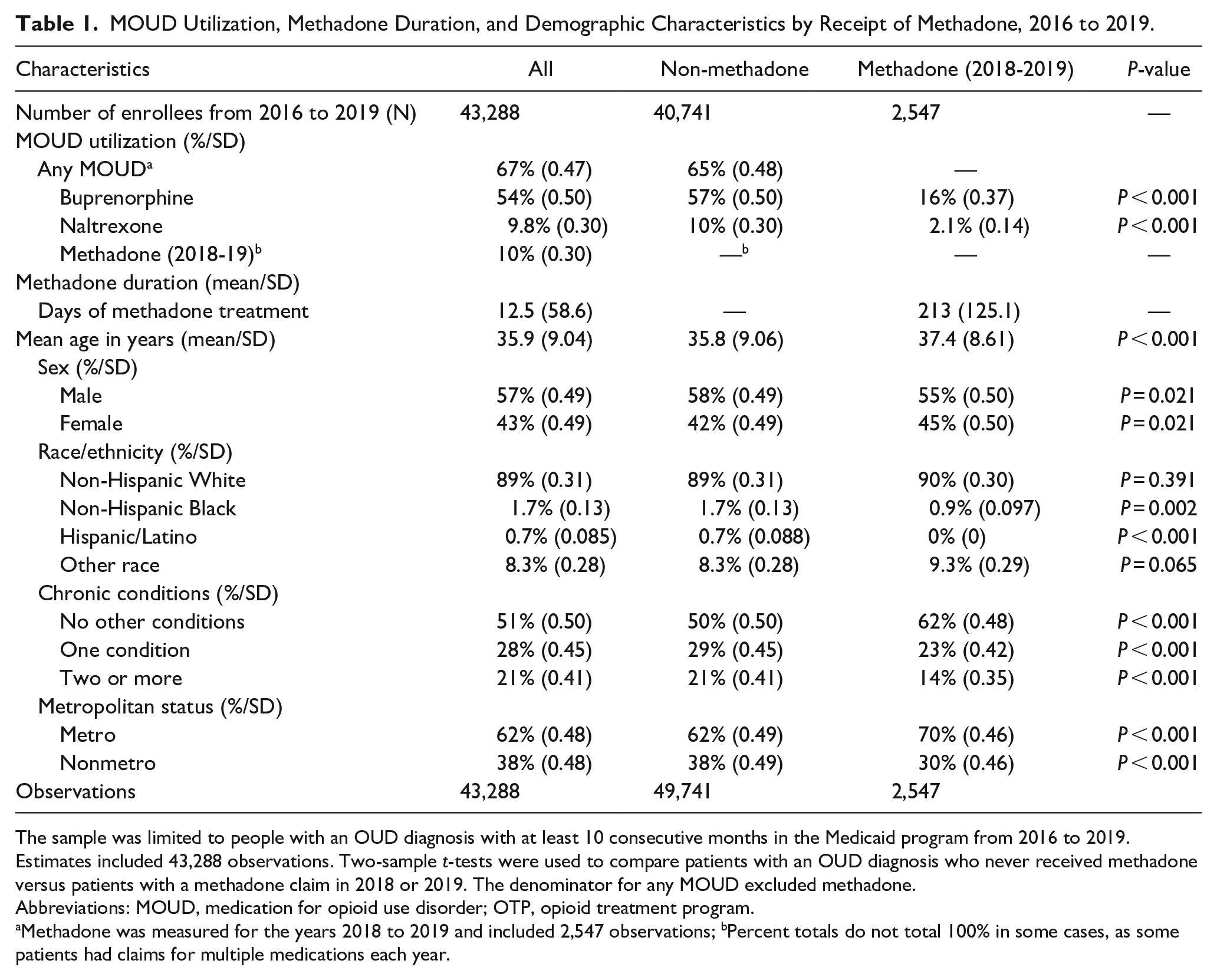
The sample was limited to people with an OUD diagnosis with at least 10 consecutive months in the Medicaid program from 2016 to 2019. Estimates included 43,288 observations. Two-sample t-tests were used to compare patients with an OUD diagnosis who never received methadone versus patients with a methadone claim in 2018 or 2019. The denominator for any MOUD excluded methadone.
Abbreviations: MOUD, medication for opioid use disorder; OTP, opioid treatment program.
Methadone was measured for the years 2018 to 2019 and included 2,547 observations; bPercent totals do not total 100% in some cases, as some patients had claims for multiple medications each year.
We hypothesized that proximity to OTPs is one factor predicting take-up of methadone. In 2019, 57.7% of our sample receiving methadone resided within the same ZCTA as an OTP, while 42.3% of enrollees traveled to another ZCTA for methadone treatment (Figure 1; Table 2).
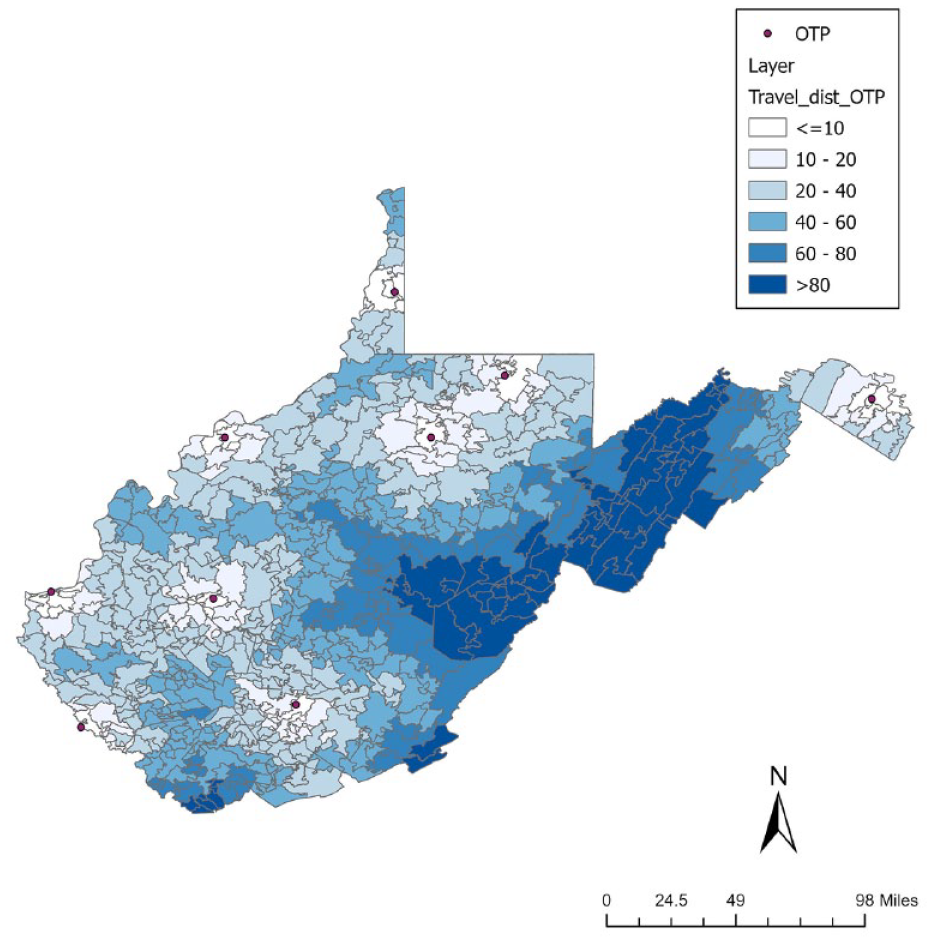
Driving distance to the nearest Opioid Treatment Program (OTP). The map of distance to the nearest OTP was drawn using the Network Analyst extension in ArcGIS. Driving distance in miles is shown from ≤10 miles to >80 miles, with a range of 2 to 142 miles.
Distance to the Nearest OTP by Receipt of Methadone, 2018 to 2019.
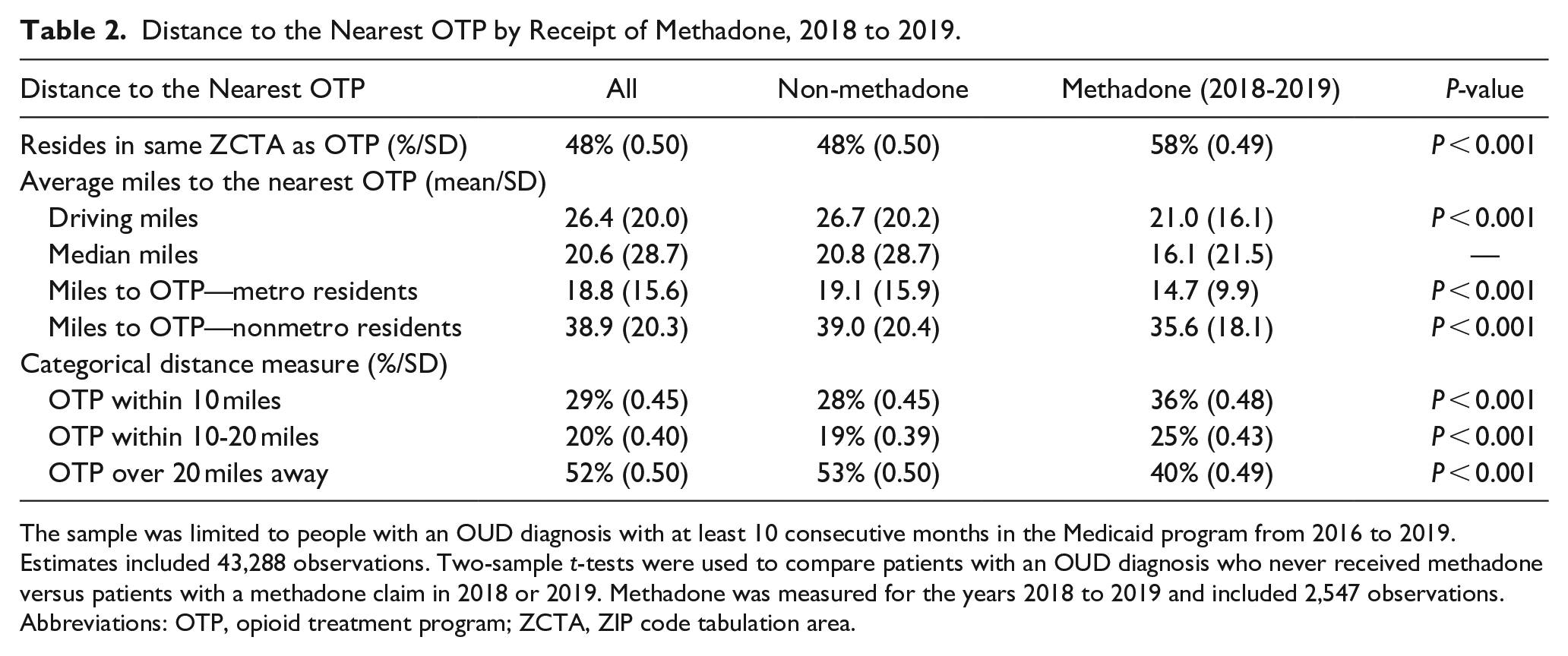
The sample was limited to people with an OUD diagnosis with at least 10 consecutive months in the Medicaid program from 2016 to 2019. Estimates included 43,288 observations. Two-sample t-tests were used to compare patients with an OUD diagnosis who never received methadone versus patients with a methadone claim in 2018 or 2019. Methadone was measured for the years 2018 to 2019 and included 2,547 observations.
Abbreviations: OTP, opioid treatment program; ZCTA, ZIP code tabulation area.
On average, patients receiving methadone lived 21.0 miles from the nearest OTP (P < .001). In comparison, enrollees receiving other MOUD or no MOUD lived an average of 26.7 miles from an OTP. The majority of patients who received methadone resided 20 miles or less from an OTP (60.1%).
We also hypothesized that enrollees who initiated Medicaid-covered methadone would be more likely to reside in metropolitan areas; around 70.0% of patients receiving methadone resided in metropolitan areas. Metropolitan residents traveled an average of 14.7 miles to the nearest OTP compared to 35.6 miles for nonmetropolitan enrollees receiving methadone. Of note, two of West Virginia’s 9 OTPs are in urban areas (nonmetropolitan areas, RUC codes 4-7) and seven are in metropolitan areas (RUC codes 1-3); none are located in completely rural areas (RUC codes 7-9).
Finally, we hypothesized that proximity to an OTP would be associated with treatment duration. On average, enrollees receiving methadone between 2018 and 2019 received 213 days of methadone per calendar year with comparable levels of duration across distance categories. Patients who resided ten miles or less from an OTP had an average duration in treatment of 197.7 days, compared to 203.5 days for those ten to 20 miles away and 232.5 days for those over 20 miles from an OTP.
Figure 2 shows the MOUD utilization history among Medicaid patients that initiated methadone in 2018 and 2019 using full Medicaid claims. Analyses differentiated methadone patients who were not previously enrolled in the prior year and initiated methadone and those who were previously enrolled in Medicaid without an OUD diagnosis. Results showed a large initial influx of methadone patients in the first year of coverage (see Supplemental Material 1 for 2014-2019 enrollment trends). Results showed that a quarter (n = 279/24.8%) of the 1,126 patients receiving methadone in 2018 were not previously enrolled in the Medicaid program at all in 2017 and 45.1% (n = 508) were previously enrolled without an OUD diagnosis. Most of the new patients (n = 118/42.3%) in 2018 resided more than 20 miles from an OTP. Among patients who were previously in Medicaid with an OUD diagnosis in the year prior to initiating methadone (n = 339), more than half were previously receiving buprenorphine (n = 174/51.3%). Many patients on methadone in 2018 who were previously enrolled in Medicaid with an OUD diagnosis had no prior MOUD utilization in Medicaid (n = 148/43.7%). Few had prior use of naltrexone (n = 17/5.0%) before initiating methadone in 2018.
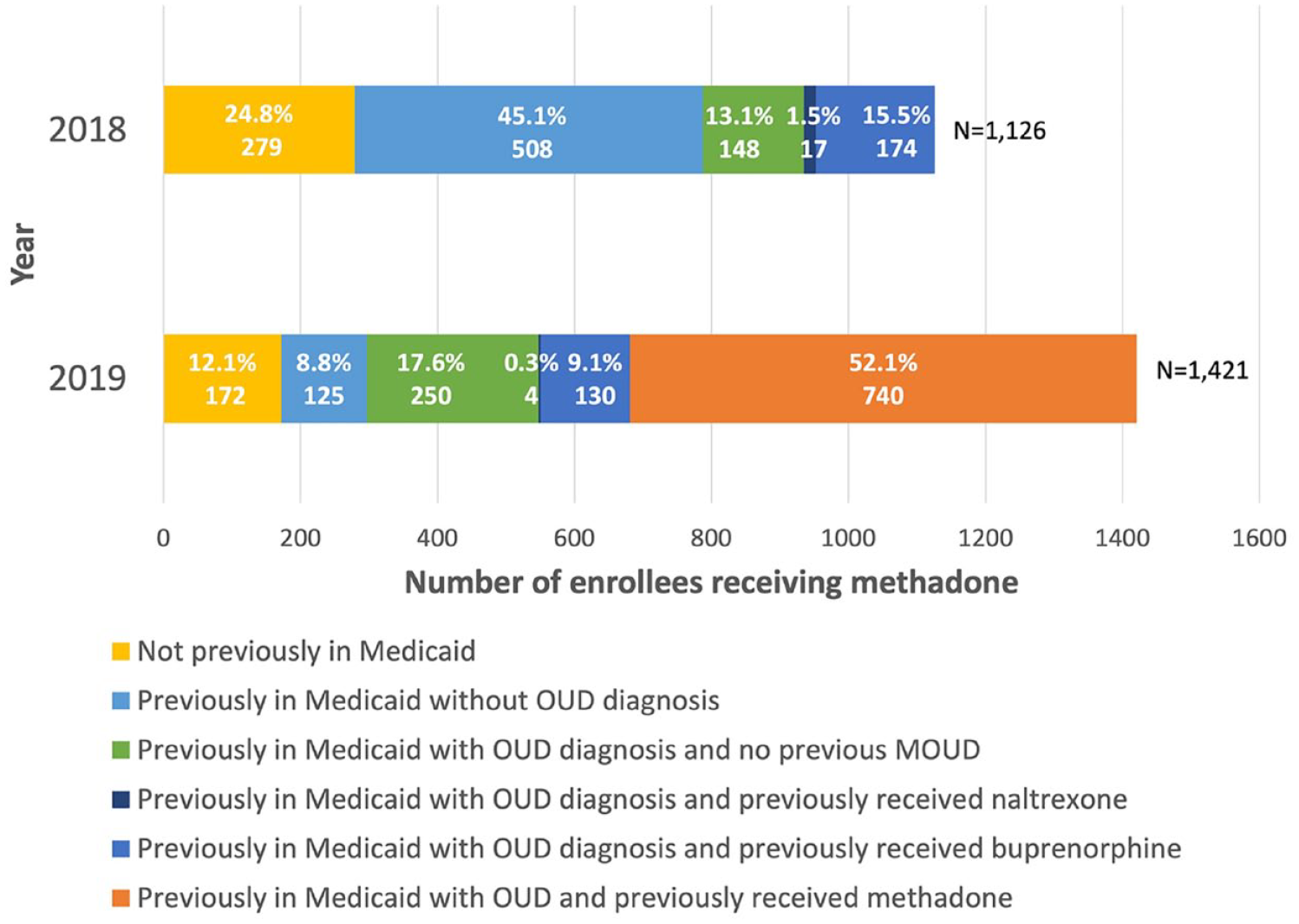
Prior Medicaid enrollment and MOUD utilization among patients with an OUD diagnosis who received methadone, 2018 to 2019. The figure reports prior MOUD utilization among methadone patients in 2018 or 2019. Prior enrollment and medication utilization was measured for the previous year (e.g., the 2018 bar reflects Medicaid enrollment and MOUD utilization in 2017). Prior methadone receipt is only reported in 2019 as the medication was first covered in 2018. Some patients received multiple MOUD in the prior year; patients who received any methadone in the previous year regardless of any other MOUD are represented in the “previously in Medicaid with OUD and previously received methadone” group. Patients who received buprenorphine alone or with naltrexone are represented in the previous buprenorphine group. Patients who received naltrexone alone without any prior buprenorphine or methadone are represented in the previous naltrexone group.
In the second year of Medicaid coverage of methadone, the majority (n = 740/65.7%) of the 1126 patients that received methadone in 2018 continued the medication in 2019. Compared to methadone patients in 2018, a much smaller share of patients who received methadone in 2019 were not enrolled at all in the previous year (n = 172/12.1%) or were enrolled in the prior year without an OUD diagnosis (n = 125/8.8%). In 2019, 37.8% of new patients traveled more than 20 miles for treatment. Compared to 2018, a smaller share of methadone patients were previously enrolled with an OUD diagnosis and receiving buprenorphine (n = 215/19.1%), naltrexone (n = 4/0.4%), or no MOUD (n = 250/22.2%) in 2019.
We also examined overall MOUD utilization among all patients and new patients in 2018 and 2019 to compare take-up of methadone or other MOUD following the implementation of the waiver (see Supplemental Material 2). We found that patients with OUD in their first year of Medicaid enrollment were less likely to use any MOUD but had similar rates of methadone use specifically.
Regression Analysis
Adjusted regression model results are reported in Table 3; unadjusted and adjusted model results were comparable. Logistic regression results were consistent with the descriptive analyses finding that enrollees with an OUD diagnosis that resided more than 20 miles from an OTP had a decreased probability of receiving any methadone compared to those residing within 10 miles of an OTP ([marginal effect] ME: −0.041; P < 0.001). This effect was driven by enrollees in metropolitan areas, as nonmetropolitan residents (ME: −0.019; P < 0.05) were marginally less likely to receive any methadone treatment. Older age and fewer chronic conditions were associated with receipt of any methadone. Non-Hispanic Black patients were less likely to receive methadone (ME: −0.060, P < 0.001) than patients of other races. Contrary to our hypothesis, poisson model results did not show a significant relationship between distance to an OTP and methadone treatment duration. Also contrary to our hypothesis, patients who resided in nonmetropolitan counties were marginally more likely to remain engaged in treatment than patients living in metropolitan areas (ME: 24.3; P < 0.05). See Supplemental Material 3 for additional regression results stratified by metropolitan or nonmetropolitan county residence and Supplemental Material 4 for a sensitivity analysis that excludes metropolitan status from the model.
Marginal Effects of Regression Predicting Initiation and Duration of Medicaid-Covered Methadone.
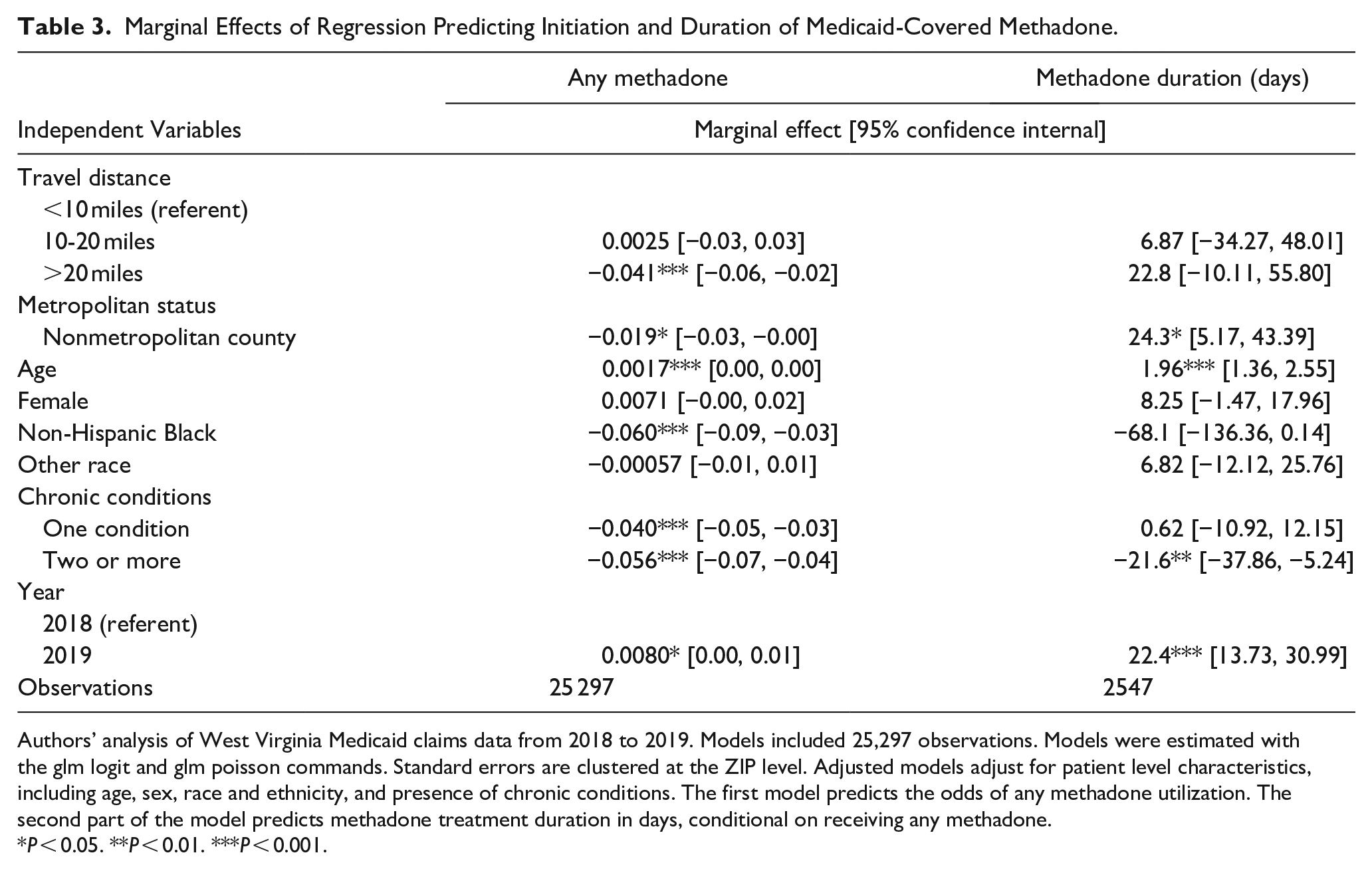
Authors’ analysis of West Virginia Medicaid claims data from 2018 to 2019. Models included 25,297 observations. Models were estimated with the glm logit and glm poisson commands. Standard errors are clustered at the ZIP level. Adjusted models adjust for patient level characteristics, including age, sex, race and ethnicity, and presence of chronic conditions. The first model predicts the odds of any methadone utilization. The second part of the model predicts methadone treatment duration in days, conditional on receiving any methadone.
P < 0.05. **P < 0.01. ***P < 0.001.
Discussion
Providing access to all forms of MOUD is a critical strategy in the opioid overdose crisis. West Virginia was relatively late to cover methadone in Medicaid and has done so in the context of stringent limits on service availability, given the moratorium of new OTPs. Our analysis finds that the population initially receiving Medicaid-covered methadone skewed toward older female enrollees residing in metropolitan areas, with a somewhat lower burden of chronic disease. Non-Hispanic Black patients were less likely to access methadone than patients of other races. Methadone patients residing in metropolitan areas traveled around 15 miles to treatment, compared to nonmetropolitan residents who traveled over twice as far. When methadone was added to Medicaid coverage, the number of enrollees using the service increased in the first 2 years. The majority of patients who received Medicaid-covered methadone in 2018 continued the medication in 2019, indicating high levels of treatment retention.
Around one-quarter of patients that received methadone in 2018 were not enrolled in the prior year, raising the question of whether methadone access itself may have encouraged enrollment. Once methadone coverage took effect, perhaps patients sought treatment for a previously undiagnosed OUD, indicating that methadone is either a preferred treatment option and/or an OTP is more conveniently located than a provider prescribing other MOUD. It is also possible that patients who previously received methadone were not covered by Medicaid but, following the policy change, may have sought care through their Medicaid coverage.
Many methadone patients were previously enrolled in Medicaid but did not have an OUD diagnosis, especially in the first year of methadone coverage (45.1% in 2018 and 8.8% in 2019). This is consistent with literature documenting that the prevalence of OUD in the United States may be substantially undercounted. 25 Patients who were previously enrolled with an OUD diagnosis were potentially better positioned to initiate methadone; previously enrolled patients were perhaps more likely to learn about the new coverage or more readily able to receive a referral to treatment than patients with a previously undiagnosed OUD.
Contrary to our hypothesis, we did not find a significant association between distance to an OTP and treatment duration in our regression analysis, and we found a positive, marginally significant association between residence in a nonmetropolitan county and duration of treatment. The average duration of methadone treatment in West Virginia from 2018 to 2019 was 213 days, or 7 months, and duration was largely consistent across distance categories. Our results are consistent with the recommended minimum range of 6- to 12-months duration in methadone treatment,26,27 though for many patients, remaining on methadone beyond one year is optimal. 27
Still, a key finding is that take-up of methadone was highest among enrollees residing in the counties with an OTP (58% of methadone patients lived in the same ZCTA as an OTP), which is not surprising since travel distance is a known barrier to access and retention. Our findings show that patients residing within 20 miles of an OTP may be more likely to utilize methadone than other forms of MOUD, consistent with prior research documenting fewer missed doses of methadone for those residing within closer proximity to an OTP. 8 However, as noted previously, West Virginia only has nine OTPs. Thus, the impact of West Virginia’s policy change may be blunted because of the limited capacity for methadone treatment. Our results seem to reflect this given the initial large influx and increasing number of methadone patients in the initial years, from 0% coverage for methadone before 2018 to 9.5% by the end of 2018, followed by a modest but statistically significant increase to 10.6% of all Medicaid enrollees diagnosed with OUD receiving methadone by 2019. Methadone utilization in the state may be capped at around 10% of enrollees in the Medicaid program who need treatment, given the state’s current capacity restraints. Additionally, distance barriers may be especially salient for people with OUD in West Virginia, where the mountainous terrain, winding roads, and limited public transportation in some areas prohibit access.
Given that enrollees with OUD have access to affordable treatment, additional patients could receive services. Of all forms of MOUD, most patients new to Medicaid with an OUD diagnosis between 2018 and 2019 received buprenorphine (44.4% in 2018 and 45.6% in 2019). Access to the full range of MOUD is important given that, for many patients, methadone is the optimal treatment. Following methadone coverage, still over a third of enrollees diagnosed with OUD received no MOUD in 2018 or 2019.
Ideally, the methadone access gap might be overcome by lifting the moratorium to serve more areas in the state. There was legislation under consideration in 2023 that would have repealed the moratorium, though it was not passed. 28 Ultimately, stigma and political barriers continue to limit access to MOUD, particularly methadone. The recent expansion of mobile methadone through a rule from the US Drug Enforcement Administration could also expand OTP services and improve treatment duration, though to our knowledge there have not been any mobile units initiated in West Virginia, which may be related to limitations posed by state law.
Finally, the study brings into perspective a key aspect of the IMD waivers. While the study was not designed to assess the causal effect of the policy change, it is likely that the waiver increased treatment availablity. This is important because IMD waivers continue to be a strategy for CMS to influence the array of services available to Medicaid enrollees. As states approach the end of their initial waiver period, it is worth considering whether there are further terms or provisions of waivers that could further serve to expand methadone access. For example, CMS may impose more expansive requirements to require states to ensure that access to methadone is the standard of care at all points in the service continuum, including residential and outpatient substance use treatment programs. Continued innovative methadone delivery models should be prioritized to further eliminate affordability and travel-related treatment barriers.
Limitations
Limitations should be considered. First, descriptive analyses differentiated patients who were previously in Medicaid without an OUD diagnosis from patients who were completely new to the program and MOUD in our observation period, though it is possible some patients were previously in Medicaid and received MOUD before the year observed or received MOUD through other forms of payment. As noted, we do not observe whether patients received methadone prior to 2018, given that this would have occurred outside of the Mediaid program, likely through self-payment. Additionally, our observation period ends in 2019; the dynamics of methadone treatment and patient characteristics may have changed in the years following our observation period.
Conclusion
The implementation of the IMD waiver may have been associated with an influx of enrollees. Many individuals who initiated treatment with Medicaid-covered methadone had no prior diagnosis of OUD or prior Medicaid enrollment in our observation period and potentially were existing uninsured OTP patients, underscoring that Medicaid coverage of methadone may bring underserved populations to the program. At minimum, Medicaid coverage of methadone reduces affordability barriers to OTP services for patients who otherwise were not receiving MOUD. Still, methadone patients in West Virginia Medicaid frequently traveled over 20 miles to an OTP, suggesting that the state needs greater methadone availability.
Supplemental Material
sj-docx-1-saj-10.1177_29767342231208516 – Supplemental material for Utilization of Medications for Opioid Use Disorder Among West Virginia Medicaid Enrollees Following Medicaid Coverage of Methadone
Supplemental material, sj-docx-1-saj-10.1177_29767342231208516 for Utilization of Medications for Opioid Use Disorder Among West Virginia Medicaid Enrollees Following Medicaid Coverage of Methadone by Samantha J. Harris, Rachel K. Landis, Wenshu Li, Bradley D. Stein and Brendan Saloner in Substance Abuse
Footnotes
Author Contributions
BS, SJH, and RKL originated the project. BS supported this work with funding and supervision. SJH led the article preparation and data analysis. RKL and WL contributed in data preparation and analysis. BDS provided subject matter expertise. All authors contributed to interpretation of the results, writing and editing, and approved the final article draft for submission to Substance Abuse.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The National Institute on Drug Abuse provided funding to support this work (K01DA042139, PI: Saloner).
Compliance,Ethical Standards,and Ethical Approval
Institutional Review Board approval was not required.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
