Abstract
Background:
Chronic low back pain (CLBP) is a leading cause of disability, often managed with opioids despite risks. Cannabis, containing tetrahydrocannabinol (THC) and cannabidiol, offers a potential alternative, but long-term data are limited.
Objective:
To evaluate the efficacy, safety, and opioid-sparing effects of cannabis therapy over 10 years in CLBP patients.
Methods:
In a prospective study (2013–2023), 168 adults (135 males, mean age 47 ± 19 years) with CLBP (≥1 year) and anatomical abnormalities (e.g., disc herniation, 83%) received cannabis (20% THC, 50 ± 10 g/month) at a specialized orthopedic clinic. Baseline treatments included opioids (91%) and surgery (47%). Outcomes—pain (Visual Analogue Scale, VAS), disability (Oswestry Disability Index, ODI), quality of life (SF-12), and opioid use—were assessed at baseline, 1, 3, 12, 24, 60, and 120 months. Statistical analysis used ANOVA, t-tests, and Kruskal–Wallis; missing data were imputed via Last Observation Carried Forward.
Results:
At 10 years (n = 159), VAS decreased from 84.6 ± 14.8 to 7.0 ± 11.0 (p < 0.0001), ODI from 61.7 ± 13.7 to 4.0 ± 6.0 (p < 0.0001), and SF-12 Physical and Mental Component Summaries improved by 21.1 points each (p < 0.0001). Opioid use dropped from 91% to 8.2% (morphine equivalent dose: 30 ± 12 to 1 ± 3 mg, p < 0.001). Six patients (3.6%) discontinued due to adverse effects; nine died.
Conclusions:
Long-term cannabis therapy significantly reduced pain, disability, and opioid dependence in CLBP, with sustained quality-of-life gains, supporting its role in chronic pain management.
Introduction
Chronic low back pain (CLBP) affects approximately 25% of the global population, contributing to substantial personal, societal, and economic burdens. 1 Traditional treatments—non-steroidal anti-inflammatory drugs, muscle relaxants, and opioids—often fail to provide sustained relief and carry significant risks, notably opioid dependency. 1 Given these limitations, cannabis-based therapies are increasingly recognized for their potential in managing chronic pain and reducing opioid reliance, leveraging compounds like delta-9-tetrahydrocannabinol (THC) and cannabidiol (CBD) that interact with the endocannabinoid system (ECS).2,3
Cannabinoids modulate pain by acting on CB1 and CB2 receptors within the ECS, altering pain perception and reducing inflammation—a distinct mechanism from opioids. 4 Studies demonstrate THC’s efficacy in reducing pain intensity in chronic conditions, including CLBP, 5 while CBD is under investigation for its analgesic potential. 6 However, the diversity in individual responses and cannabis product heterogeneity necessitates comprehensive evaluation of its long-term therapeutic value. 7 Short-term data suggest promise: a 2-week study of 249 CLBP patients found THC-dominant edibles significantly reduced pain, 8 and observational studies link cannabis to decreased opioid use.9–17
Despite these findings, long-term evidence remains scarce, and complexities arise with cannabis use disorder (CUD), which may increase risks during spinal surgeries, such as prolonged hospital stays and complications.18,19 These risks highlight the need for personalized approaches, particularly given CLBP’s demographic and clinical variability. 20 Systematic reviews note a paucity of controlled trials specific to cannabis for back pain, underscoring gaps in understanding its sustained efficacy and safety. 7
This prospective study addresses these gaps by evaluating cannabis therapy over 10 years in 168 CLBP patients with anatomical causes (e.g., disc herniation, spinal stenosis) at a specialized orthopedic clinic since 2013. We assessed pain severity (Visual Analogue Scale, VAS 21 ), disability (Oswestry Disability Index, ODI 22 ), quality of life (SF-12 23 ), patient perception (PGIC 24 ), and opioid dependence, offering the longest follow-up to date. We hypothesized that cannabis would deliver durable pain relief, enhance function, and reduce opioid use, with an acceptable safety profile, contributing critical data to its role as a sustainable pain management option.
Materials and Methods
Study design and participants
This prospective cohort study enrolled 168 adults (135 males, 33 females; mean age 47 ± 19 years, range 18–92) with CLBP (≥1 year) at a cannabis-therapy-specialized orthopedic clinic from 2013. Inclusion required unsuccessful prior therapy (e.g., physiotherapy, ≥3 analgesics including opioids) and imaging-confirmed pathology (e.g., disc herniation 83%, spinal stenosis 41%). Exclusion criteria included pregnancy, CUD, or lack of anatomical cause. Informed consent was obtained; the study was approved by the Institutional Review Board (612–16 and 807–21), adhering to the Declaration of Helsinki (2013 revision).
Intervention
Patients received cannabis (20% THC, mean 50 ± 10 g/month, range 30–100 g) via inhalation or edibles, titrated individually. Baseline treatments included opioids (153/168, 91%), paracetamol (95%), and surgery (79/168, 47%). Inclusion criteria required stable anatomical causes of CLBP not requiring surgical intervention, with no planned or performed surgeries during the study. Patients were recommended to strengthen core muscles and perform Tai Chi. In addition, new adjunctive treatments such as additional physical therapy, acupuncture, reflexology, and hydrotherapy were allowed. Invasive procedures such as surgery, discoplasty, nucleotomy, and spinal injections were not allowed during the study to minimize confounding variables.
Compliance was monitored monthly via self-reported drug diaries (tracking daily consumption in grams), return of unused cannabis for verification, and cross-referencing with prescription refill data from the licensed provider. Doses were adjusted as needed for tolerance in 37/168 patients (22%), with mean increases of 10–15 g/month.
Data collection
Patient-reported outcome measures were collected at baseline, 1, 3, 12, 24, 60, and 120 months: VAS (pain, 0–100), 21 ODI (disability, 0–100), 22 PGIC (perceived improvement, 1–7), 24 and SF-12 v1 (Physical Component Summary, PCS; Mental Component Summary, MCS) at baseline and 120 months. 23 Imaging (CT/MRI) confirmed pathology at baseline and as needed. All participants provided written informed consent for study participation and publication of findings.
Statistical analysis
The statistical analysis used Excel Analyse-it 5.90 (2024). Continuous variables (VAS, ODI, SF-12 PCS, SF-12 MCS) were analyzed with ANOVA for multiple timepoints and t-tests for pairwise comparisons (e.g., opioid use). PGIC (ordinal) was assessed with Kruskal–Wallis. Tukey-Kramer post-hoc tests evaluated continuous variable differences. Missing data from living patients were imputed using Last Observation Carried Forward (LOCF), deemed more accurate than mean substitution or regression prediction; 25 deceased patients (n = 9) were excluded from later timepoints. The per-protocol set included subjects at 120 months (n = 159); the complete dataset comprised all treated subjects (n = 168) with LOCF imputation. Significance was set at p < 0.05.
Results
Of 168 subjects (135 males, 80.4%; mean age 47 ± 19), females were older (50.3 ± 22 vs. males 42 ± 18.1, t-test, p < 0.025). Anatomical causes included disc herniation (139/168, 83%), spinal stenosis (70/168, 41%), vertebral fracture (20/168, 12%), and post-tumor resection (5/168, 3%) (Tables 1 and 2). Baseline VAS was 84.6 ± 14.8, ODI 61.7 ± 13.7, SF-12 PCS 36.5 ± 6.3, and MCS 36.6 ± 11.1 (Table 3).
Study Cohort at Different Time Points
Data were analyzed after imputation of missing values using the Last Observation Carried Forward method (LOCF).
Gender-Based Age and BMI Summary at Baseline
Baseline Measurements Summary
SD, Standard Deviation; VAS, Visual Analogue Scale; ODI, Oswestry Disability Index; SF12, Version 1 Short Form 12; PCS, Physical Component Summary; MCS, Mental Component Summary; PGIC, Patient Global Impression of Change.
VAS decreased significantly (ANOVA, p < 0.0001; Fig. 1). From 84.6 ± 14.8, it fell to 18.3 ± 12.1 at 24 months (difference 66.3, Tukey-Kramer, p < 0.0001), 17.4 ± 11.8 at 60 months (difference vs. 24 months 0.9, p = 0.09), and 7.0 ± 11.0 at 120 months (difference 77.6, p < 0.0001). ODI mirrored this trend (ANOVA, p < 0.0001; Fig. 2). From 61.7 ± 13.7, it dropped to 9.8 ± 7.9 at 24 months (difference 51.9, p < 0.0001) and 4.0 ± 6.0 at 120 months (difference 57.7, p < 0.0001); 24-to-60-month change was nonsignificant (p = 0.87).
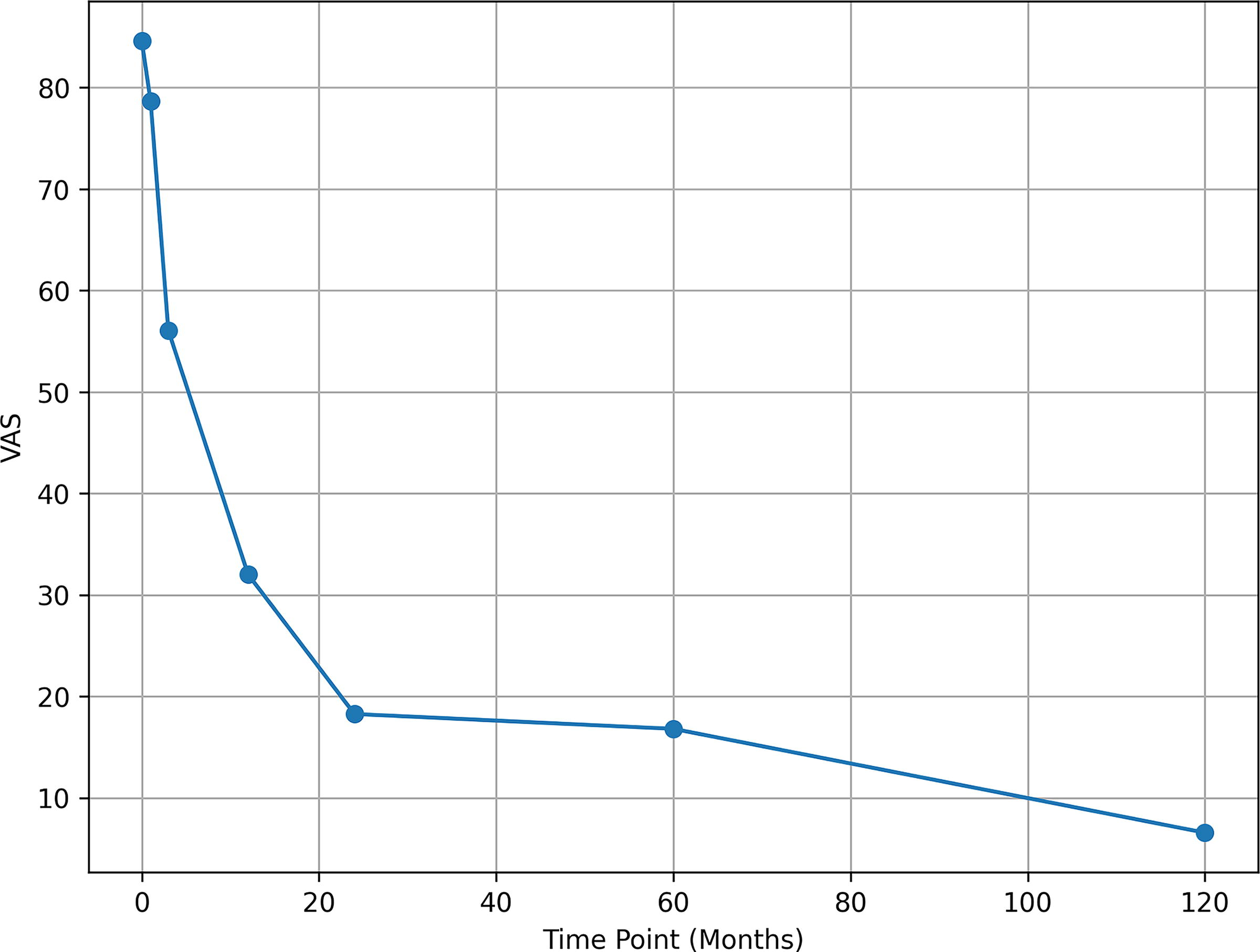
VAS scores variation over time (n = 168), LOCF of missing data.

ODI scores variation over time (n = 168), LOCF of missing data.
PGIC rose from 1.0 ± 0 at baseline to 6.1 ± 0.9 at 120 months (Kruskal–Wallis, H = 751.48, df = 6, p < 0.0001; Fig. 3), with major gains by 3 months (5.8 ± 1.0). SF-12 PCS improved from 36.5 ± 6.3 to 57.6 ± 9.2 (difference 21.1, ANOVA, F[1,325] = 604.81, p < 0.0001; Figure 4), and MCS from 36.6 ± 11.1 to 57.7 ± 10.5 (difference 21.1, F[1,325] = 322.83, p < 0.0001; Figure 4), based on 327 observations (168 baseline, 159 at 120 months post-LOCF).
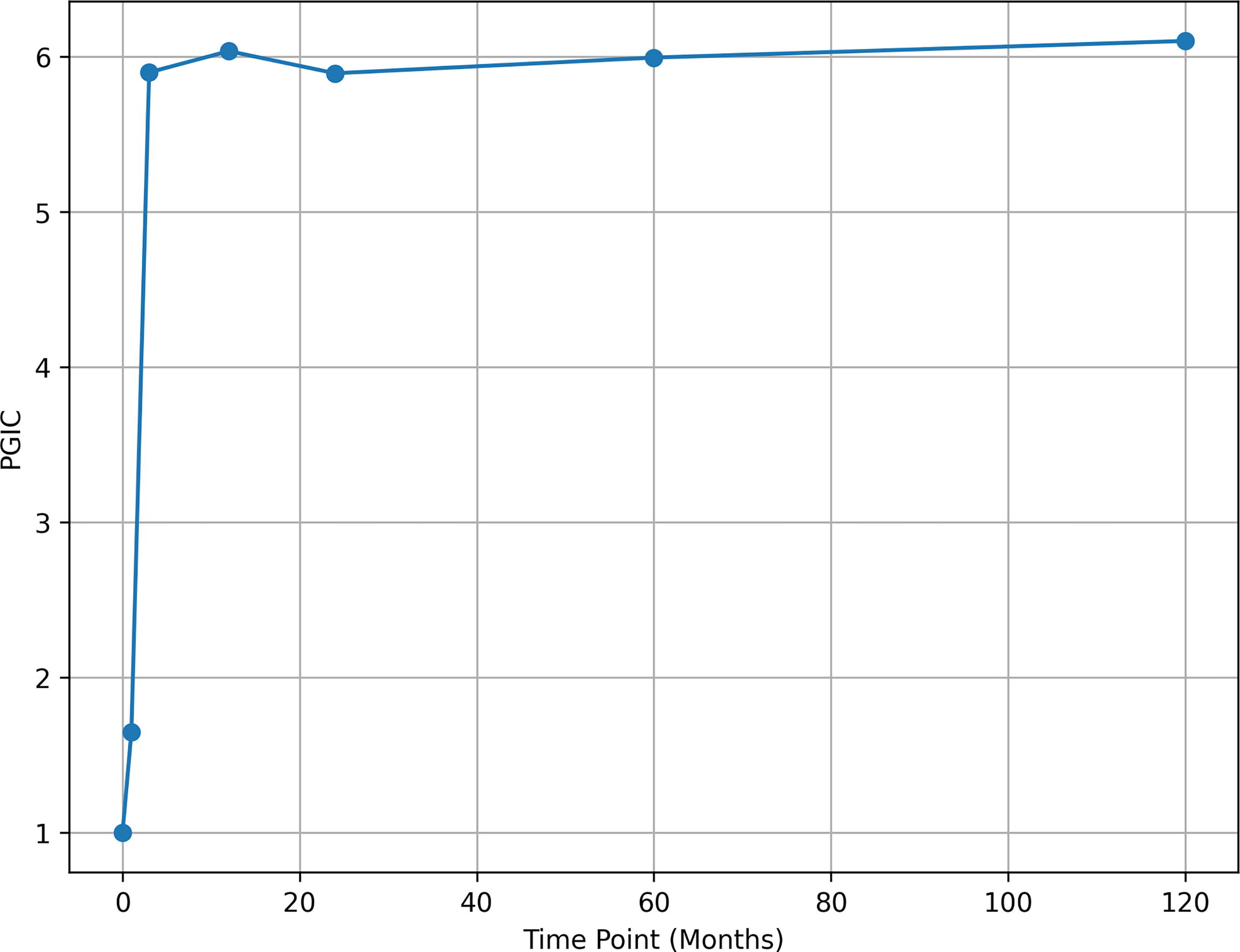
PGIC scores variation over time (n = 168), LOCF of missing data.

SF-12 PCS comparison of timepoint 0 with timepoint 120 months (n = 168), LOCF of missing data (left). SF-12 MCS comparison of timepoint 0 with timepoint 120 months (n = 168), LOCF of missing data (right).
Baseline opioid use (153/168, 91%; morphine equivalent daily dose, MEDD 30 ± 12 mg) declined to 13/159 (8.2%; MEDD 1 ± 3 mg) at 120 months (t-test, p < 0.001), with 5 on fentanyl (100 µg), 4 oxymorphone, and 4 oxycodone; their VAS (6.5 ± 10.8) matched the cohort. Six patients (3.6%) discontinued due to adverse effects (e.g., dizziness); 9 died; tolerance required adjustments in 22% (37/168). Principal component analysis showed ODI as the dominant factor, with the first component explaining 86.5% of variance (Fig. 5).

Principal component analysis indicates ODI is responsible for most over-time variation (n = 168), LOCF of missing data.
Discussion
This 10-year study demonstrates that cannabis therapy (20% THC, 50 ± 10 g/month) significantly reduces pain (VAS: 84.6 ± 14.8 to 7.0 ± 11.0), disability (ODI: 61.7 ± 13.7 to 4.0 ± 6.0), and opioid use (91% to 8.2%) in 168 CLBP patients, with sustained quality-of-life improvements (SF-12 PCS: 36.5 ± 6.3 to 57.6 ± 9.2; MCS: 36.6 ± 11.1 to 57.7 ± 10.5). These findings extend short-term evidence 8 and align with 12-month observational data, 9 positioning cannabis as a viable, long-term alternative to traditional analgesics.
THC’s effects, mediated by CB1 receptors, 4 likely drove pain relief, while mental health gains (MCS) suggest broader benefits. The opioid reduction from 91% (153/168) to 8.2% (13/159; MEDD: 30 ± 12 to 1 ± 3 mg) corroborates shorter-term studies 9 and systematic reviews advocating cannabis as an opioid-sparing option. 7 This is critical amid the opioid epidemic, as cannabis targets distinct pain pathways without the addiction or overdose risks of opioids. 4
Integration with current knowledge reinforces these results. Recent meta-analyses suggest moderate-quality evidence for cannabinoids in chronic non-cancer pain, including CLBP, 7 through direct long-term CLBP studies are limited. 4 Our findings of sustained pain and functional improvement align with observational reports of inhaled 5 and CBD-focused 6 therapies, supporting cannabis’s role in personalized medicine. CLBP’s complexity—evident in our cohort’s anatomical diversity (e.g., disc herniation 83%) and prior treatments (e.g., surgery 47%) 1 —underscores the need for tailored approaches, which cannabis’s variable pharmacokinetics may facilitate. 20
Safety considerations include tolerance (dose adjustments in 22% by 60 months) and adverse effects leading to discontinuation in 6 patients (3.6%), such as dizziness or paranoia, consistent with broader literature. 7 Unlike opioids, no addiction-related mortality occurred, though CUD studies caution of surgical complications,18,19 emphasizing patient selection and monitoring. Psychiatric risks (e.g., psychosis) warrant screening, particularly with THC-dominant therapy. 7 The slower improvement from 24 to 60 months (VAS: 18.3 ± 12.1 to 17.4 ± 11.8; ODI non-significant change) suggests a plateau, possibly due to CB1 receptor desensitization, 8 necessitating strategies like periodic breaks.
While improvements were sustained over 10 years, we cannot exclude contributions from natural history or unreported minor adjunctive interventions, though protocol adherence minimized these risks.
Limitations include the single-center design, lack of a control group, and LOCF imputation, which may overestimate effects. 25 The focus on 20% THC limits CBD-specific insights, though CBD’s anti-inflammatory and anxiolytic roles are noted elsewhere. 6 A primary limitation is the lack of a control group, which precludes definitive attribution of improvements solely to cannabis therapy. Natural disease progression, placebo effects, or regression to the mean could contribute to observed outcomes, though the sustained reductions over 10 years suggest a treatment-specific effect beyond spontaneous recovery.
Reliance on self-reported diaries and returns introduces possible recall bias or underreporting, though refill records provided objective corroboration.
Nine deaths (unrelated to therapy) and 7 discontinuations (6 adverse effects, 1 unspecified) reduced the cohort to 159 by 120 months, yet per-protocol analysis supports robustness.
Future research should prioritize large-scale randomized controlled trials (RCTs) to confirm efficacy, compare administration methods (e.g., inhalation vs. edibles 5 ), and refine dosing to mitigate tolerance and adverse effects. 7 Detailed pharmacodynamic analyses and multimodal integration (e.g., with physiotherapy 1 ) could optimize outcomes. Given cannabis’s opioid-sparing potential, 9 studies in diverse populations, including Arabic cultures, 20 could broaden accessibility and application.
In conclusion, this study supports cannabis’s therapeutic potential for CLBP management, offering significant pain relief, functional improvement, and opioid reduction over 10 years. Future efforts should refine protocols to ensure safe, effective, and tailored pain management strategies.
Conclusions
Ten years of cannabis therapy (20% THC) yielded profound reductions in pain (VAS: 84.6 ± 14.8 to 7.0 ± 11.0), disability (ODI: 61.7 ± 13.7 to 4.0 ± 6.0), and opioid dependence (91% to 8.2%) in 168 CLBP patients, with significant quality-of-life gains (SF-12 PCS: 36.5 ± 6.3 to 57.6 ± 9.2; MCS: 36.6 ± 11.1 to 57.7 ± 10.5). These findings highlight cannabis as a viable long-term alternative to traditional analgesics, supporting its role in chronic pain management. While safe and effective for most, tolerance (dose adjustments in 22%) and psychiatric risks (discontinuation in 3.6%) require careful monitoring. Larger RCTs are essential to validate these results, compare administration methods, and optimize dosing to mitigate adverse effects. Multimodal approaches integrating cannabis with physiotherapy could further enhance outcomes.
Footnotes
Authors’ Contributions
Authors Roles according to Contributor Role Taxonomy (CRediT): D.R.: Conceptualization; data curation; formal analysis; methodology; writing—original draft. M.K.: Investigation; methodology; writing—review and editing. M.E.: Investigation; writing—review and editing. M.Y. Writing—review and editing.
Author Disclosure Statement
No competing financial interests exist.
Funding Information
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
