Abstract
Background:
Biologics and small-molecule therapies are commonly used for long-term maintenance in patients with inflammatory bowel disease (IBD), including Crohn’s disease (CD) and ulcerative colitis (UC). Dose escalation is often required to maintain treatment response, but real-world data on its frequency and contributing factors are limited.
Objectives:
To evaluate patterns of dose escalation during maintenance treatment and identify patient- and treatment-related factors associated with dose escalation in IBD.
Design:
Retrospective, population-based cohort study.
Methods:
Using data from the Korean Health Insurance Review and Assessment Service (HIRA), we identified adults with CD or UC who initiated maintenance treatment with biologics or small-molecule agents between 2018 and 2022. Dose escalation was defined as two consecutive doses ⩾50% higher than the expected daily dose within 1 year of treatment initiation. Multivariable logistic regression was used to assess associated factors.
Results:
Among 2,186 patients with CD and 1,806 with UC, dose escalation occurred in 20.9% and 14.1%, respectively. In CD, ustekinumab had the highest escalation rate (69.1%), compared with infliximab (16.4%), adalimumab (21.2%), and vedolizumab (23.2%). For UC, adalimumab was most frequently escalated (21.8%), followed by tofacitinib (19.5%), ustekinumab (17.9%), and infliximab (10.0%). Steroid use, later lines of therapy (third- and fourth-line) were significantly associated with increased odds of dose escalation.
Conclusion:
Dose escalation is a common occurrence in IBD maintenance therapy, with substantial variation across treatment agents and patient profiles. These findings emphasize the importance of individualized treatment strategies to improve long-term disease control.
Keywords
Introduction
Inflammatory bowel disease (IBD), encompassing Crohn’s disease (CD) and ulcerative colitis (UC), is a chronic, immune-mediated condition marked by relapsing and remitting gastrointestinal inflammation.1,2 Due to its prolonged course and frequent co-occurrence with comorbidities, effective long-term disease control is essential.3,4 This typically involves sustained therapeutic strategies aimed at inducing and maintaining remission, minimizing complications, and improving quality of life.5,6
For patients with moderate-to-severe CD or UC who do not respond to conventional therapies, biologics and small-molecule agents—collectively referred to as advanced therapies—are recommended.7–9 Advanced therapies are generally indicated for patients with an inadequate response or intolerance to conventional treatments and include biologic agents targeting specific inflammatory pathways (anti-TNFα, anti-integrin α4β7, anti-IL12/23, and anti-IL23) as well as small-molecules such as Janus kinase (JAK) inhibitors.7,8 These therapies have demonstrated improved efficacy in inducing and sustaining remission and reducing corticosteroid dependence. These therapies not only improve disease control but also enhance quality of life, and reduce disease burden through sustained remission.10,11
During maintenance therapy, dose adjustments—such as increasing the dose or shortening administration intervals—are commonly employed to address loss of response.12,13 Reported rates of dose escalation among users of biologics or small-molecules range from approximately 10% to over 50%, depending on the specific agent and patient population studied.14,15 While such strategies may help re-establish disease control, they also raise concerns about increased healthcare costs and the potential for ongoing inadequate clinical benefit.16,17
To support evidence-based decision-making in IBD care, it is critical to understand when and in whom dose escalation occurs. Despite its clinical relevance, real-world evidence on dose escalation patterns—particularly in relation to individual therapies and patient characteristics—remains limited. Identifying predictive factors for escalation may help guide personalized treatment plans and optimize resource use.
This study aimed to evaluate the patterns of dose escalation during maintenance therapy with biologics and small-molecule drugs in patients with IBD, and to identify patient- and treatment-related factors associated with escalation across clinical subgroups.
Methods
Data source
We conducted a population-based cohort study utilizing the Korea Health Insurance Review and Assessment Service (HIRA) database (HIRA-M20230415001), covering the period from January 1, 2012, to September 30, 2022. The HIRA database includes approximately 98% of the entire South Korean population, and is derived from the National Health Insurance (NHI) system—a universal public health insurance program operated by the Korean government. 18 The database covers detailed healthcare utilization information, including patient demographics, history of disease with diagnostic code of the International Classification of Diseases, 10th Revision (ICD-10), patients’ use of health resources, including procedures, prescriptions, and other resources for treatment.
Study design and population
We conducted a retrospective cohort study to identify adult patients with CD or UC who received maintenance treatment with advanced therapies between January 1, 2019, and September 30, 2021. Eligible patients were those diagnosed with CD (ICD-10 K50.xx) or UC (ICD-10 K51.xx) between January 1, 2018 and September 30, 2022.
Patients were required to be 18 years of age or older at the initiation of therapy and to have remained on continuous treatment with the same advanced agent for 1 year without discontinuation or switching. Individuals younger than 18 years or those who discontinued or switched therapies during the one-year follow-up period were excluded from the final cohort.
A two-step indexing process was used to define induction and maintenance treatment. Patients who initiated advanced therapy—infliximab, adalimumab, vedolizumab, ustekinumab, golimumab, or tofacitinib—were first identified. The first index date was defined as the date of the first prescription of an advanced therapy during the study period, indicating the initiation of the induction period. Among these patients, those who completed the induction period were further indexed for maintenance treatment. The second index date was defined as the date of their first prescription for maintenance treatment following the induction period of advanced therapy. The induction periods were determined based on the approved drug labels. 12 For CD, the induction periods were as follows: 2 weeks for infliximab, 16 weeks for adalimumab, 16 weeks for vedolizumab, and 16 weeks for ustekinumab. For UC, the induction periods were 16 weeks for infliximab, 14 weeks for adalimumab, 14 weeks for vedolizumab, 16 weeks for ustekinumab, 16 weeks for golimumab, and 16 weeks for tofacitinib. To account for potential delays in prescription refills, a 14-day grace period was applied after the induction phase. More detailed information on study design is available in Supplemental Figure 1.
The discontinuation of advanced therapy was defined with the interruption of treatment with a gap of 90 days or more between the expected dosing date, based on the approved treatment schedule per label, and the subsequent prescription within one-year of follow-up. 19 If the expected dosing date occurred before the end of follow-up and no subsequent prescription was issued within 90 days, the patient was classified as discontinued. Switching of treatment was defined as the prescription of a different advanced therapy than the drug of interest.
Study outcomes
The primary outcome of this study was the proportion of patients who experienced dose escalation within 1 year after initiating maintenance treatment. Dose escalation was defined as the administration of two consecutive prescriptions in which the average daily dose was at least 50% greater than the expected maintenance dose, as determined by the approved Korean product labels.15,17 This threshold was selected based on label guidance for agents that permit ⩾50% increases under specific clinical conditions. To assess robustness, we additionally conducted sensitivity analyses using alternative thresholds of 20%, 100%, and 200%. The assessment period for dose escalation spanned 1 year from the index date, defined as the start of maintenance treatment. Patients prescribed tofacitinib at 10 mg twice daily at the initiation of maintenance treatment were excluded from the dose escalation analysis. Although this dosage exceeds the standard maintenance dose, it was not considered dose escalation in this study due to its frequent use as an initial strategy followed by subsequent dose reduction. The expected daily dose during the maintenance treatment was determined based on the Korean approval label (Supplemental Table 1). For infliximab, the first maintenance dose was used to determine the standard dose based on body weight. The magnitude of dose escalation, also considered a primary outcome, was estimated by comparing each patient’s cumulative annual dose to the expected cumulative standard dose based on label-recommended maintenance treatment regimens.
To explore potential factors associated with dose escalation, we examined a range of patient-level demographic and clinical characteristics. These included age, sex, Charlson comorbidity index (CCI), and specific comorbid conditions such as small bowel cancer, extraintestinal manifestations, anxiety, and depression.12,20,21 Treatment history variables included prior use of conventional therapies (5-aminosalicylic acid, immunosuppressants, and corticosteroids) and treatment line (i.e., first-line, second-line, or later).21,22 Opioid use was also included as a covariate, as it is often associated with more severe disease and poor clinical outcomes in patients with IBD.22,23 All covariates were evaluated at baseline within 365 days before the first index date (Supplemental Table 2 for more details).
Statistical analysis
Descriptive analysis was used to summarize baseline characteristics. Continuous variables were presented as means with standard deviations (SDs) or medians with interquartile ranges (IQRs), and categorical variables were summarized as frequencies and percentages. To compare the proportion of patients who experienced dose escalation across different therapies, we used the chi-square test. Time to dose escalation was measured from the second index date (date of starting maintenance treatment) to the date of the first occurring dose escalation. We performed multivariable logistic regression analysis to evaluate the likelihood of dose escalation in both the CD and UC patients. Odds ratios (ORs) and 95% confidence intervals (CIs) were reported. Covariates included age, gender, CCI, the presence of comorbidities, opioid use, and line of treatment before the index date.12,24 A p-value of less than 0.05 was considered statistically significant.
Sensitivity analysis was conducted to evaluate variations in the proportion of patients undergoing dose escalation, contingent upon the definition of the dose escalation threshold. These thresholds were established based on percentages reported in previous studies.15,17,21 We examined dose escalations at thresholds of 20%, 50%, 100%, and 200% above the expected daily dose specified on the product label.
All analyses were conducted using SAS Enterprise Guide 9.4 (SAS Institute Inc., Cary, NC, USA). The manuscript adheres to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines for reporting of observational studies (Supplemental Note). 25
Results
Baseline characteristics
We identified 2,186 patients with CD and 1,806 with UC under maintenance treatment between January 1, 2019, and September 30, 2021 (Supplemental Figures 1 and 2).
The baseline characteristics of these patients are summarized in Table 1. Among the CD cohort, 72.28% were male with a mean age of 32.03 years (SD: 12.55). The UC cohort was also mostly comprised of males (67.22%) and had a higher mean age than CD, with a mean age of 41.28 years (SD: 16.00). Among CD patients, 21.68% had previously received at least second-line treatments or advanced therapy before initiating advanced therapy, compared to 34.82% of UC patients. Regarding prior medications, most patients in the CD and UC groups had received 5-ASA (75.94% in CD; 92.75% in UC), corticosteroids (65.69% in CD; 74.14% in UC), and immunosuppressants (81.38% in CD; 64.34% in UC).
Baseline characteristics of patients with Crohn’s disease and ulcerative colitis with advanced therapy in Korea between January 2019 and September 2021.

Values were presented in numbers (%) or means (standard deviations) unless stated otherwise.
CCI, the Charlson comorbidity index.
Dose escalation in maintenance treatment
Among the 2186 patients with CD, 457 (20.91%) experienced dose escalation within 1 year of maintenance treatment initiation. As shown in Figure 1, dose escalation was the most common observation in patients receiving ustekinumab, significantly differing from other advanced therapies (p < 0.0001), with 69.07% (n = 67) of patients experiencing an increase. For other therapies, the proportions were 16.4% (n = 184) for infliximab, 21.19% (n = 150) for adalimumab, and 23.24% (n = 56) for vedolizumab.

Proportion of patients with dose escalation during maintenance treatment by disease type and treatment.
In the UC patients, 255 patients (14.12%) experienced dose escalation within 1 year of maintenance treatment initiation. The proportion of patients with dose escalation varied significantly across advanced therapies (p < 0.0001). Among these patients, adalimumab has the highest proportion of dose escalation (21.75%), followed by tofacitinib (19.46%), ustekinumab (17.87%), and infliximab (10.02%). The proportion of dose escalation varied across specific therapies within the CD and UC patients.
In sensitivity analyses applying alternative thresholds (20%, 50%, 100%, 200%), the overall proportion of patients experiencing escalation varied by definition (Supplemental Table 3).
Magnitude of annual cumulative dose during maintenance treatment
As shown in Figure 2(a), most patients with CD received a cumulative dose increase of ⩾50%. Among those with 50–100% escalation, the highest proportion was seen with ustekinumab (76.1%), followed by adalimumab (57.3%), vedolizumab (55.4%), and infliximab (52.2%). Escalation to 100%–200% of the standard dose was most frequent with infliximab (26.6%), which was also the only agent associated with escalations exceeding 200%.
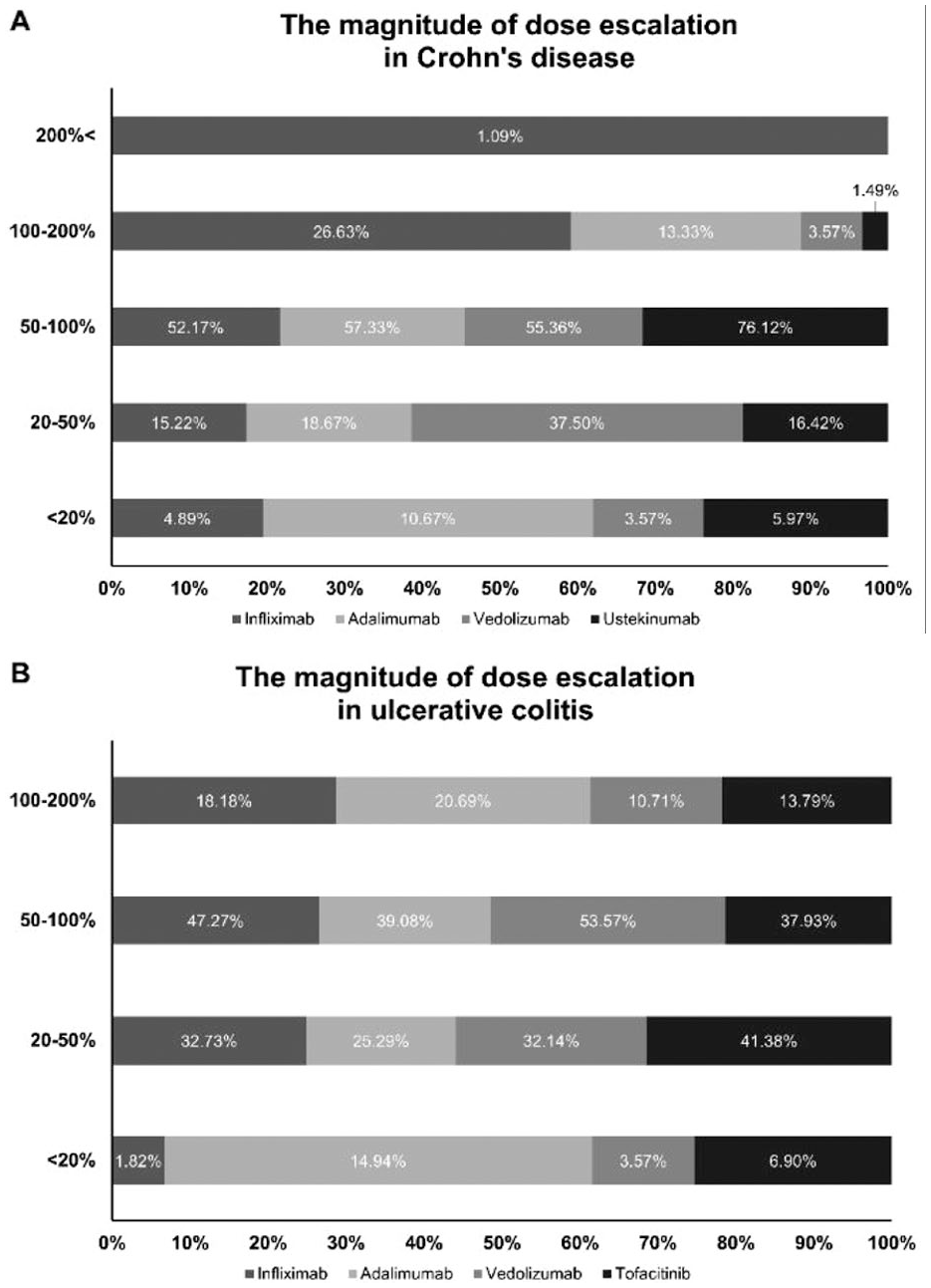
The magnitude of dose escalation during maintenance treatment across advanced therapies in (a) Crohn’s Disease and (b) ulcerative colitis.
In UC patients (Figure 2(b)), over half had dose increases above 50%. Tofacitinib had the highest share in the 20–50% range (41.4%). For 100–200% escalation, adalimumab (20.7%) and infliximab (18.2%) were the most common. No UC patients exceeded 200% escalation.
The average annual cumulative dose during maintenance treatment differed by disease type. In CD, the average annual doses per patient were 2,806 mg for infliximab, 2,289 mg for vedolizumab, 1,242 mg for adalimumab, and 572 mg for ustekinumab. In UC, the corresponding averages were 4,271 mg for tofacitinib, 2,546 mg for infliximab, 2,267 mg for vedolizumab, 1,276 mg for golimumab, 1,217 mg for adalimumab, and 540 mg for ustekinumab.
Time to dose escalation during maintenance treatment
Table 2 presents the average time to dose escalation during maintenance treatment for patients with CD and UC. CD Patients treated with ustekinumab experienced the shortest interval to dose escalation, with a mean of 2.61 months (95% CI, 1.87–3.36), suggesting a more rapid adjustment compared to other treatment options. In contrast, infliximab was associated with the longest time to escalation (6.24 months; 95% CI, 5.78–6.69), followed by vedolizumab (5.20 months; 95% CI, 4.23–6.17) and adalimumab (4.97 months; 95% CI, 4.44–5.50).
Average time to dose escalation during maintenance treatment (unit: month).
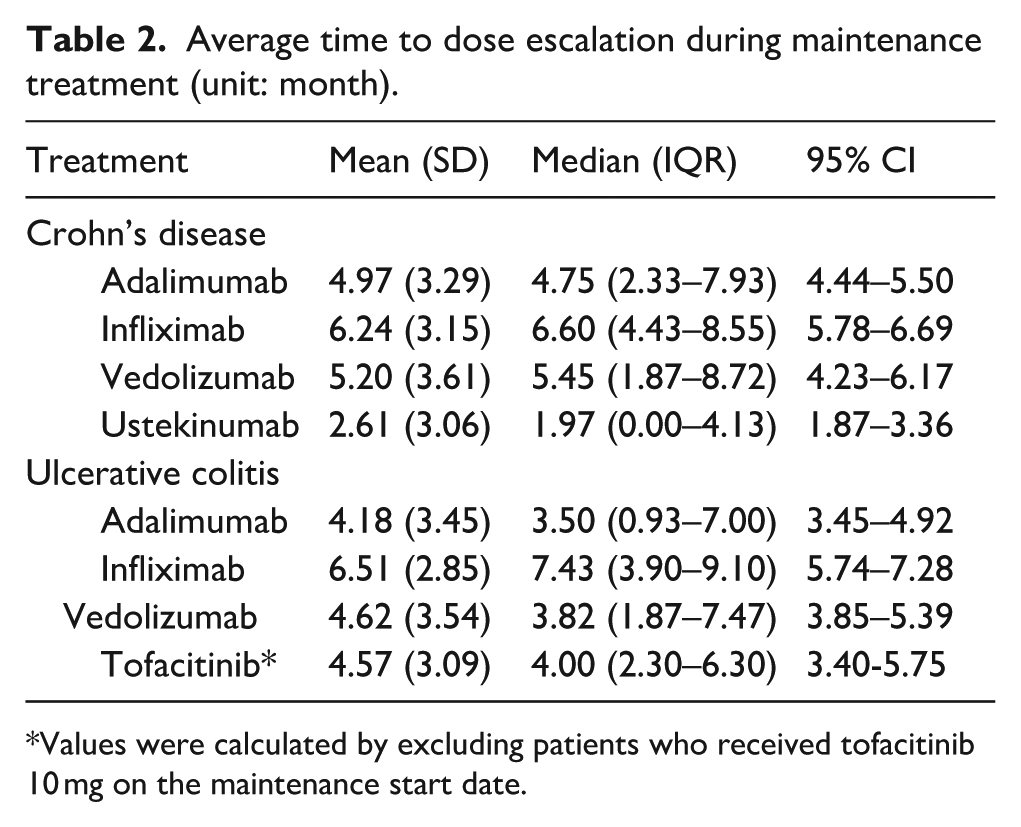
Values were calculated by excluding patients who received tofacitinib 10 mg on the maintenance start date.
Among patients with UC, infliximab again demonstrated the longest average time to escalation at 6.51 months (95% CI, 5.74–7.28). Shorter mean time to escalation was observed with vedolizumab (4.62 months; 95% CI, 3.85–5.39), tofacitinib (4.57 months; 95% CI, 3.40–5.75), and adalimumab (4.18 months; 95% CI, 3.45–4.92). It is important to note that the analysis of tofacitinib excluded patients who began maintenance therapy with a 10 mg dose, to minimize confounding by initial treatment intensity.
Factors associated with dose escalation in IBD treatment
In the CD patient, several clinical characteristics were significantly associated with an increased likelihood of dose escalation during maintenance treatment. Notably, the presence of anxiety was linked to a higher odds of escalation (OR, 1.714; 95% CI, 1.155–2.543). Corticosteroid use also emerged as a significant factor (OR, 1.444; 95% CI, 1.124–1.855), suggesting that patients with more active disease were more likely to require treatment intensification. Specifically, the odds ratios increased with each later treatment line: second line (OR, 3.197; 95% CI, 2.435–4.198), third line (OR, 5.612; 95% CI, 3.755–8.388), and fourth line (OR, 10.754; 95% CI, 4.405–26.254) (as detailed in Table 3).
Multivariable logistic regression analysis of factors associated with dose escalation in Crohn’s disease and ulcerative colitis.

The OR could not be calculated due to zero events in UC. This reflects the absence of patients experiencing the outcome in this subgroup, which prevents a reliable estimate of the effect size.
CI, Confidence interval; CCI, Charlson comorbidity index; EIM, extraintestinal manifestations.
Among patients with UC, similar patterns were observed. Additionally, patients receiving third-line (OR, 1.823; 95% CI, 1.163–2.857) or fourth-line treatment (OR, 2.706; 95% CI, 1.359–5.389) had elevated odds of escalation compared to those on first-line regimens.
Other variables, including sex, CCI score, and opioid use, were not significantly associated with dose escalation in either CD or UC populations.
Discussion
This population-based cohort study examined real-world patterns of dose escalation in patients with IBD receiving biologics and small-molecule agents during maintenance treatment. Our findings contribute to a deeper understanding of current treatment dynamics and may inform individualized therapeutic strategies.
One key strength of this study is the inclusion of recent nationwide data and a broad range of agents, including tofacitinib, which has not been widely evaluated for dose escalation in prior research. This allows for a more comprehensive and contemporary assessment of real-world treatment response patterns. Furthermore, we explored factors associated with dose escalation, such as corticosteroid use, advanced lines of therapy, and comorbid conditions, providing clinically relevant insights for personalized care.
This study evaluated dose escalation patterns during maintenance treatment in patients with CD and UC. During the 1-year maintenance follow-up period, more than 14% of patients in both IBDs experienced dose escalation. Among CD patients, ustekinumab exhibited the highest dose escalation rate at 69.07%, compared to other advanced therapies. A previous study also reported that dose escalation was most frequent with ustekinumab in CD patients, which was similar to our findings. 12 However, other studies have reported that infliximab (65.9%), vedolizumab (53.3%), and adalimumab (47.4%) over 3 years of follow-up. 14 The reason for the different results in CD patients between countries was due to differences in dosing regimens. Notably, in Korea the approved ustekinumab maintenance dosing interval was every 12 weeks, which was longer than the 8-week schedule used in other countries. Therefore, if there was no effect within a long period, earlier dose escalation of ustekinumab may be considered to improve effectiveness. In UC patients, adalimumab had the highest dose escalation rate, which was similar to previous studies.19, 20 ,26 However, in a recent study, vedolizumab and infliximab were shown to have higher dose escalation rates compared to adalimumab, which is thought to be due to the different definition of dose escalation in that study. 27
We defined dose escalation using a ⩾50% increase from the expected dose, providing a conservative threshold that prioritizes clinically significant changes over minor or routine adjustments.12,14,15,17,20 Sensitivity analyses using lower thresholds (e.g., ⩾20%) resulted in higher observed escalation rates, particularly among ustekinumab users in CD and adalimumab users in UC. Our threshold thus enhances the clinical relevance of our findings and highlights substantial, decision-driven escalations rather than administrative or minor changes. The time to dose escalation also varied by agent. Ustekinumab in CD and adalimumab in UC were associated with the shortest times to escalation, while infliximab showed the longest. This likely reflects its weight-based dosing and individualized titration, complicating direct comparisons with fixed-dose therapies. Mean time to escalation in our cohort (approximately 4–5 months) aligns with a previous study reporting similar timelines for inadequate response. 28
Several clinical factors were associated with an increased likelihood of dose escalation. Corticosteroid use and the administration of third- or fourth-line therapies were strongly linked to escalation, possibly reflecting more severe or refractory disease. Notably, anxiety was also associated with escalation in CD patients. While previous studies have explored the influence of mental health comorbidities on IBD outcomes, findings have been inconsistent.20, 21 Given the high prevalence of anxiety and depression in IBD, 29 further research is needed to clarify whether these comorbidities directly affect treatment response or primarily reflect underlying disease severity.
Patients undergoing second-line or later therapies were significantly more likely to escalate dosing. This may result from prior treatment failure or loss of response, prompting more aggressive management.12,26 Recent American Gastroenterological Association (AGA) guidelines further acknowledge that patients with prior biologic exposure or partial responses to standard dosing may require prolonged induction regimens or higher maintenance doses to achieve or sustain clinical response.30,31 In this context, dose escalation during maintenance treatment may represent an adaptive strategy to optimize effectiveness in patients with increased disease complexity rather than necessarily an isolated indicator of drug failure. In addition, the presence of anti-drug antibodies (ADAs) in patients receiving biologics may contribute to reduced efficacy and dose escalation. ADAs can lead to diminished drug levels or hypersensitivity reactions, often requiring switching agents. Patients with prior ADA development may also be predisposed to immunogenicity with subsequent biologics,32,33 underscoring the need for more precise, individualized therapeutic strategies. Furthermore, biologics have been reported as a major contributor to healthcare costs in Korea, and international evidence shows that dose escalation further increases drug-related expenditures, suggesting the potential socioeconomic implications of treatment intensification.34–36
This study has several limitations. First, due to the characteristics of claims data, we lacked clinical and laboratory parameters such as body weight, disease activity indices, and biomarkers, which limited our ability to directly assess disease severity or response. Instead, we used relevant proxies such as corticosteroid use, treatment lines, and comorbidity profiles; however, residual confounding may remain. Second, we did not distinguish between treatment-naïve and experienced patients, which may influence escalation patterns given previously reported heterogeneity in efficacy of advanced therapies according to prior biologic exposure. 37 Differences in real-world prescribing practices, including treatment line and initial dosing strategies, may also have contributed to the observed patterns. Treatment line was included as a covariate to partially address this limitation. Third, adherence data were unavailable, particularly for self-administered therapies, which could introduce discrepancies between prescribed and actual use. Fourth, a potential limitation of this study is selection bias. To ensure assessment of dose escalation within a consistent maintenance regimen, patients who discontinued or switched therapy during the maintenance period were excluded by design. Finally, although small bowel cancer was retained as a covariate due to its potential clinical relevance, its very low frequency in our cohort limits the interpretability of the regression results.
Conclusion
This population-based cohort study observed the incidence of dose escalation among IBD patients. Several factors, including the presence of comorbidities and prior treatment history, influenced the likelihood of dose escalation. These findings underscore the importance of strategic treatment approaches that account for the interactions between different therapies in the long-term management of IBD. By tailoring efficient therapeutic strategies to the specific disease type and each patient’s treatment journey, we can potentially reduce the socioeconomic burden of the disease.
Supplemental Material
sj-docx-1-taj-10.1177_27558428261419710 – Supplemental material for Patterns and factors associated with dose escalation during maintenance treatment for inflammatory bowel disease: a population-based cohort study
Supplemental material, sj-docx-1-taj-10.1177_27558428261419710 for Patterns and factors associated with dose escalation during maintenance treatment for inflammatory bowel disease: a population-based cohort study by Dahye Ryu, Hyun Jin Han, Junghyun Na, Seoyeon Choi, Young Mi Park and Hae Sun Suh in Sage Open Chronic Disease
Supplemental Material
sj-docx-2-taj-10.1177_27558428261419710 – Supplemental material for Patterns and factors associated with dose escalation during maintenance treatment for inflammatory bowel disease: a population-based cohort study
Supplemental material, sj-docx-2-taj-10.1177_27558428261419710 for Patterns and factors associated with dose escalation during maintenance treatment for inflammatory bowel disease: a population-based cohort study by Dahye Ryu, Hyun Jin Han, Junghyun Na, Seoyeon Choi, Young Mi Park and Hae Sun Suh in Sage Open Chronic Disease
Footnotes
Acknowledgements
We appreciate the Korean Health Insurance Review and Assessment Service (HIRA) for providing the research data (M20230415001). The authors declare that the results do not reflect the views of HIRA or the Ministry of Health and Welfare.
Ethics approval and consent to participate
This study was approved by the Kyung Hee University Institutional Review Board (KHSIRB-23-114, 10 April 2023), which granted exemption from full ethical review. The requirement for informed consent was waived because the analysis was based solely on a de-identified secondary database.
Consent for publication
Not applicable.
Author contributions
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by funding from AbbVie and by a grant from the Ministry of Food and Drug Safety, Republic of Korea (grant number: 21153MFDS601).
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Hae Sun Suh reports financial support was provided by Abbvie Korea and the Ministry of Food and Drug Safety. If there are other authors, they declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Data availability statement
The dataset used in this study is not publicly available due to privacy and ethical restrictions. Access was limited to researchers who received approval and was facilitated through the National Health Insurance internal network system.
Supplemental material
Supplemental material for this article is available online.
