Abstract
Cambodia has experienced exponential economic growth in recent years and is expected to graduate from least developed country (LDC) status within the next decade. Membership of the World Trade Organization (WTO) will require Cambodia to grant product and process patents for pharmaceuticals upon LDC graduation. This study aims to measure the impact of the WTO Agreement on Trade-Related Aspects of Intellectual Property Rights (TRIPS) on the price of HIV and hepatitis C medicine in Cambodia once it graduates from LDC status and is obliged to make patents available for pharmaceutical products and processes. Using scenarios based on likely outcomes of accession to the TRIPS Agreement, it measures the impact on the price of the HIV treatment program and compares that impact with the hepatitis C treatment program. Graduation from LDC status would be expected to result in a modest increase in the cost of the antiretroviral (ARV) treatment program and very large increases in the cost of the direct acting antivirals (DAA) treatment program. If annual treatment budgets remain constant, patent protection could see 1,515 fewer people living with HIV able to access ARV treatment and 2,577 fewer people able to access DAA treatment (a drop in treatment coverage of 93%).
Keywords
Cambodia's rapid economic growth in the decade preceding the COVID-19 pandemic made it one of the fastest-growing economies in the region. 1 This growth has significantly reduced the number of people living in poverty. 2 Cambodia is currently designated a least developed country (LDC) by the United Nations with expectations of graduating from LDC status in the next decade. 3 Cambodia's membership of the World Trade Organization (WTO) requires it to provide patent protection for pharmaceuticals that meet standard criteria upon the loss of LDC status. This obligation could potentially compromise Cambodia's access to affordable medicine 4 and its ability to achieve its goal of universal health coverage. 5 There is growing awareness of the need to balance Cambodia's intellectual property (IP) policies and regulation with public health priorities to ensure access to affordable lifesaving medicine, diagnostics, and vaccines to diagnose, manage, and treat a broad range of medical conditions. This research aims to measure the impact of granting patents on the price of medicine in Cambodia once it graduates from LDC status. Specifically, it measures the potential impact on the price of the antiretroviral (ARV) treatment program for HIV in comparison with the impact on the price of the hepatitis C treatment program.
The first section of the paper outlines Cambodia's economic and political situation, its obligations with regard to membership of the WTO, and its current patent law as it applies to pharmaceuticals. It explores what Trade-Related Aspects of Intellectual Property Rights (TRIPS) flexibilities are omitted in Cambodia's IP laws and what TRIPS-plus provisions are present. It then provides an overview of the HIV and hepatitis C epidemiology and treatment programs in Cambodia before describing the methods, results, discussion, recommendations, and conclusion.
Background
Cambodia's Socioeconomic Situation
Cambodia's economic growth moved it into lower middle income country status 1 in 2015. Cambodia has articulated a goal to attain upper middle-income status by 2030. 6 These economic gains, however, are fragile, and large swaths of the economy are still living in extreme poverty. Many Cambodians live just above the poverty line, making them highly vulnerable to economic shocks. 7 This vulnerability has been exacerbated by the COVID-19 pandemic. 8 Increasing gross domestic product will have implications for Cambodia's eligibility for aid for health care. Many donors such as the Global Fund to Fight AIDS, Tuberculosis and Malaria and the Vaccine Alliance have already begun to reduce their funding and will continue to do so in the coming years. 9
Cambodia's Membership in the World Trade Organization and TRIPS
Cambodia joined the WTO in October 2004. 10 As a member of the WTO, Cambodia is obliged to implement the 1995 WTO TRIPS Agreement, a comprehensive multilateral agreement on IP requiring baseline levels of IP protection.11,12 This agreement includes providing at least 20 years of patent protection for pharmaceutical products or processes that meet minimum standards of patentability, novelty, inventive step, and industrial applicability. 13 From its inception, TRIPS was fiercely opposed by many low and middle-income countries (LMICs) who feared it would compromise their development and ability to access essential medicine. 14 TRIPS flexibilities were included in the original text to mitigate some of the LMIC concerns. The Declaration on the Agreement TRIPS and Public Health of 2001 (The Doha Declaration) was subsequently adopted by the WTO with the aim to promote a balanced interpretation and implementation of the provisions of the TRIPS Agreement in a manner that is supportive of a WTO member's right to protect public health and promote access to medicine for all. 15 The Doha Declaration reaffirmed the right of WTO members to implement the TRIPS flexibilities to address public health concerns and specifically recognized that patents and prices were a barrier to affordable medicine. 15
One of these flexibilities is the transition period 2 for LDCs. This transition period frees LDCs, such as Cambodia, from TRIPS obligations related to patents or other IP rights on pharmaceuticals until 2033 or until they are no longer an LDC 3 . 16 Some other flexibilities available to all WTO Member States include the freedom to set patentability criteria domestically (e.g., to not make patents available for new use of known substances, methods and processes, and to use strict patentability criteria for examination of pharmaceutical patents to mitigate frivolous patents and “evergreening” opportunities); to enable pre- and post-grant opposition of patents; to use compulsory licensing and parallel importation; 4 and to provide a Bolar (early working) exception for research and experimental use.17,18
Cambodia met all three criteria for LDC graduation at the most recent review in 2021 19 and is expected to graduate from LDC status 5 in the coming years and, subsequently, will be required to grant product and process patents for pharmaceuticals. Cambodia already has some of the highest out-of-pocket expenditure on healthcare in Southeast Asia. 20 This change in Cambodia's status combined with the loss of external aid for health care could potentially negatively affect access to medicine.
There is a dearth of literature that investigates the impact or potential impact of LDC graduation on access to medicine in LDCs that have graduated, or where graduation is imminent. Two recent qualitative studies explored the implications of the loss of the TRIPS Agreement's LDC-specific flexibilities on the four WTO members (Cambodia, Djibouti, Senegal, and Zambia) that met the graduation criteria for the first time in 2021.21,22 These studies concluded that all four countries will need to reform their patent laws and policies to include all TRIPS flexibilities afforded them in order to minimize the impact of patent protection on access to medicine.21,22 These studies did not seek to quantify the impact of LDC graduation on medicine prices or access. They analyzed Cambodia's patent related laws and policies for only very select TRIPS flexibilities, such as compulsory licensing and the LDC TRIPS waiver, and not the full range of flexibilities afforded it.
Other studies of the impact of LDC graduation have exclusively focused on Bangladesh, given its looming 2026 graduation date and significant generic pharmaceutical industry.23–27 A recent study found Bangladesh's LDC graduation can be expected to significantly increase the price of insulin, which could result in a 15 percent decline in the welfare of households in Bangladesh with one or more members living with diabetes. 28 Most of the studies have focused on the implications of graduation for Bangladesh's generic industry rather than on outcomes for access to medicine in Bangladesh. Some of these implications include high production and export costs that could undermine the sustainability of the industry. 24
Systematic reviews of the literature that measure the impact of IP rules on access to medicine found that the introduction of patent protection leads to an increase in the price of medicine and a decrease in consumer welfare.17,29 Additionally, several studies from India have found that the introduction of product patent protection, following the TRIPS Agreement transition period, caused medicine prices to rise30,31 and resulted in large losses in consumer welfare and welfare losses for the Indian economy.31–33 However, there is scant research outside of India that quantifies the impact of the introduction of patent protection on access to medicine.
Cambodia's Patent Law
Cambodia's existing patent law of 2003, The Law on Patents, Utility Model Certificates and Industrial Designs, its amendment in 2017, and its implementing regulation will govern patent grants once Cambodia graduates from LDC status. It is, therefore, critical that it includes the public health-related flexibilities and safeguards afforded by TRIPS. Cambodia's patent-related legislation omits several key public health-related TRIPS flexibilities, although a Law on Compulsory Licensing for Public Health has recently been introduced. 34 Table 1 outlines the TRIPS flexibilities that Cambodia's patent law currently omits.
TRIPS Flexibilities Omitted in Cambodia's IP Laws and Policies.

Adapted from The UNDP 2010 Good Practice Guide: Improving Access to Treatment by Utilizing Public Health Flexibilities in the WTO TRIPS Agreement.
Additionally, Cambodia's government has adopted various measures to streamline and accelerate the patent registration process that go well beyond TRIPS requirements. They include allowing the accession to multiple patent agreements designed to expedite the granting of patents, 41 such as the Joint Statement of Intent with Japan, 42 the Memorandum of Understanding (MoU) on Intellectual Property Cooperation with China, 43 the MoU with Singapore, 44 the Patent Cooperation Treaty, 45 the MoU with South Korea, 46 a Worksharing Arrangement with the United States, 47 and the Patent Validation Agreement with the European Patent Organization. 48
Cambodian patent law contains other TRIPS-plus provisions including a mailbox system. 18 This system allows for the filing of patent product applications during the LDC transition period—despite no TRIPS requirement to do so. 35 In accordance with Cambodia's Rule 45 of the Prakas on the Procedure for Granting Patents and Utility Model Certificates, mailbox applications will not be examined as to their patentability until 2033, or when Cambodia ceases to be a LDC.49,50 Cambodia's WTO Accession Agreement also contains TRIPS-plus clauses including patent linkage and data exclusivity 51 although these provisions have not been incorporated into Cambodian legislation to date. Patent linkage refers to the conditional relationship between the granting of marketing approval of a generic medicine and the patent status of the originator medicine. 52 Data exclusivity provides a period of time in which generic pharmaceutical companies cannot rely on existing medicine trial data submitted to regulatory authorities to obtain market approval for a generic medicine. 53
HIV Epidemiology and Treatment in Cambodia
In 2020, Cambodia had an HIV prevalence of 0.6 percent and approximately 75,000 people living with HIV (PLHIV). 54 Cambodia has been extremely successful in preventing new HIV infections, which declined steadily from a high of 15,000 per year in 1995 and 1996 to 1,100 in 2020. 54 Cambodia's National Center for HIV/AIDS Dermatology and STDs rapidly scaled up the provision of ARV treatment from 2001. 55 By the end of 2020, a total of 63,338 PLHIV were accessing ARV treatment, representing 84 percent treatment coverage of PLHIV. 54 See Table 2 for the number of PLHIV on first-, second-, and third-line regimes and the ARVs included in these regimes. In 2020, 77 percent of ARV costs were covered by the Global Fund to fight AIDS, Tuberculosis and Malaria (GFATM) and 23 percent from Cambodian government sources. 56 Domestic funding for ARV treatment has steadily increased from $708,000 in 2015 to $1.25 million in 2020 (all dollar amounts in U.S. dollars), however Cambodia is still heavily reliant on external donor funding for its ARV treatment program. 56
ARV Treatment Regimen and Number of PLHIV on Each Regimen in Cambodia.

Compiled using National Center for HIV/AIDS, Dermatology and STD 2020 data.
Hepatitis C Epidemiology and Treatment in Cambodia
Hepatitis C is a blood-borne virus that, untreated, can lead to liver damage and diseases, such as cirrhosis and liver cancer, and death. 57 Although there is no official data on the number of people living with hepatitis C in Cambodia, Médecins Sans Frontières (MSF) estimates that 2–5 percent of the population is infected. 58 MSF, together with the Cambodian Ministry of Health, launched Cambodia's first free hepatitis C treatment program in May 2016 at Preah Kossamak hospital in Phnom Penh in line with the World Health Organization's (WHO) global goal to eliminate hepatitis C by 2030. 59 Treatment was fully funded by MSF and consisted of a 12-week course of direct acting antivirals (DAA), which has a cure rate of 97 percent. 59
In 2020, 2,776 people completed hepatitis C treatment provided by MSF/Ministry of Health. This treatment program relies heavily on generic medicines made possible by Cambodia realizing its right as a LDC to not grant patents for pharmaceuticals. The treatment regime consisted of two generic DAAs in combination, 400mg of sofosbuvir and 60mg of daclatasvir, given once a day for 12 weeks. They were sourced from the generic companies Mylan, Natco, Pharco, and Hetero. This pan-genotypic regime was chosen because most hepatitis C infections in Cambodia are genotype 1 and 6 and a small number are genotype 2, which all respond well to the sofosbuvir/daclatasvir regime and do not require genotype testing. The cure rate was also very high for patients with liver cirrhosis, with an estimated 90 percent cure rate for patients with compensated cirrhosis and 66 percent for patients with decompensated cirrhosis. 60
Aim and Research Questions
This study aims to measure the potential impact of the TRIPS Agreement on the price of medicines in Cambodia once it graduates from LDC status and is obligated to make patents available for pharmaceutical products and processes. Specifically, it measures the anticipated impact on the ARV treatment program where most ARVs are off-patent and compares that with the impact on the hepatitis C treatment program where many medicines are still under patent in high-income countries. Impact is measured in terms of the cost of each treatment program and the number of people able to access treatment. The research questions addressed are:
What is the potential impact of Cambodia's implementation of TRIPS following LDC graduation on access to ARV treatment for HIV, measured by the cost of the ARV program and the number of people able to access treatment? What is the potential impact of Cambodia's implementation of TRIPS following LDC graduation on access to DAA for hepatitis C, measured by the cost of the hepatitis C treatment program and the number of people able to access treatment? How do the impacts on these treatment programs compare, and how can any differences be explained?
Methods
Scenario Development
Three possible ARV scenarios and two possible DAA scenarios were designed to explore the range of potential impacts of the TRIPS Agreement on Cambodia's ARV and DAA treatment programs. Only two DAA scenarios were explored as it was assumed that there would be no difference between baseline and full TRIPS flexibilities as Scenario 1 used only generic medicine (see Table 3 for the scenarios and their assumptions).
ARV and DAA Scenarios and Their Assumptions.

NCHADS: National Center for HIV/AIDS, Dermatology and STD.
Data Collection
The number of people treated with ARVs in Cambodia in 2020 was sourced from National Center for HIV/AIDS, Dermatology and STD (NCHADS) and was a constant for all three scenarios. The price data for ARV Scenario 1 was also sourced from NCHADS. Price data for the second and third ARV scenarios was sourced from the MSF publication, “Untangling the Web: HIV Medicine Pricing & Access Issues, 2020”. 61 This periodic publication provides an update on the availability, price, and supplier information in relation to ARVs globally. It details the lowest generic ARV prices available globally, which was relevant for Scenario 3 and details what the originator brands would charge Cambodia for lopinavir/ritonavir, darunavir and dolutegravir, the three ARVs that would be patented under the assumptions outlined in Scenario 2.
The number of people treated with DAAs in Cambodia in 2020 was provided by MSF and was constant for Scenario 1 and 2. The price data for DAA Scenario 1 was also sourced from MSF. The DAA price data for Scenario 2 was sourced from the WHO publication “Accelerating Access to Hepatitis C Diagnostics and Treatment: Overcoming Barriers in Low and Middle-Income Countries – Global Progress Report 2020”. 62 This publication provides updated data on pricing, licensing, and regulatory status of hepatitis C virus (HCV) medicines from the main originator and generic companies producing DAAs for HCV treatment. The lowest reported originator price for daclatasvir was sourced from this publication and used in DAA Scenario 2. The reported low-income country price for sofosbuvir was used for Scenario 2 and was also sourced from this publication.
The ARV and DAA data were entered into an Excel spreadsheet (see Supplementary File 1).
The World Intellectual Property Organization's Patent Scope database and the European Patent Office website were scanned for possible patents on ARVs included in the first-, second-, and third-line regimes and for DAAs used in the 2020 Cambodian treatment program (see Tables 4 and 5). It was assumed that a patent would also be granted in Cambodia if there was an existing patent in Japan, the United States, the European Union, China, Singapore, or South Korea on any of the ARVs and DAAs listed in the Cambodian treatment program and that the application for this patent was in the mailbox system. Although existing patent agreements with the above countries may vary in detail, they all obligate Cambodia to accelerate or grant pharmaceutical product patents if they had been granted in one or more of these countries once Cambodia is required to grant patents and those existing patent agreements are still valid and implemented.
ARVs Under Patent in Countries with Which Cambodia has a Patent Agreement.
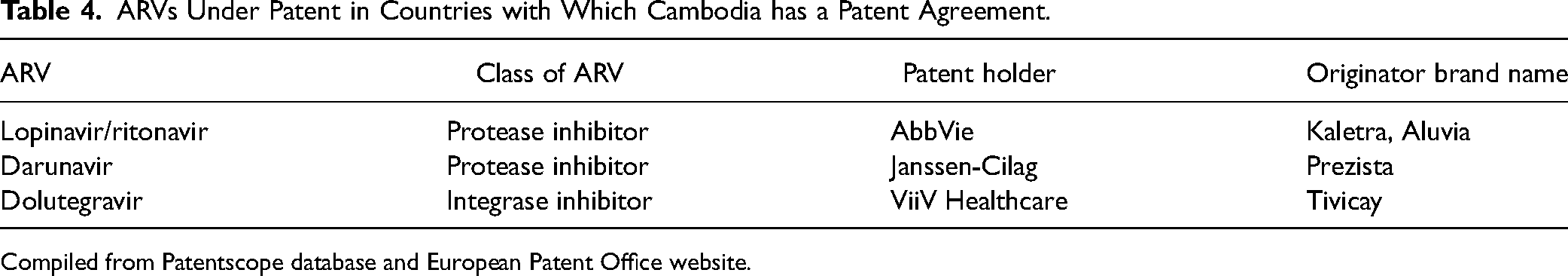
Compiled from Patentscope database and European Patent Office website.
DAA Under Patent in Countries with Which Cambodia has a Patent Agreement.

Compiled from Patentscope database and European Patent Office website.
Data Analysis
The cost of the ARVs and DAAs was calculated for each of the scenarios by multiplying the number of adults requiring a particular treatment regime by the unit cost of the medicine and adjusting for dosing (see Supplementary File 1 for full calculations). Using ARV Scenario 1 as a fixed total budget, the percentage of people who could access ARV treatment given the cost of each scenario was calculated. These percentages were compared for the ARV scenarios. The number of people able to be treated in each of the ARV scenarios using the budget for Scenario 1 was also calculated and compared. The cost of the DAA scenarios were calculated and compared. The percent change in the number of people able to access DAAs in Scenario 2 was calculated.
Results
Patent Status of ARVs and DAAs
ARVs found to be still under patent in any of the countries with which Cambodia has a patent agreement (European Union, China, Japan, Singapore, United States, and South Korea) are detailed in Table 4. The ARVs under patent (detailed in Table 4) form part of the second- or third-line regimens.
Both DAAs used in the treatment program in 2020 were found to be under patent in one or more of these jurisdictions. They are detailed in Table 5.
Costs Associated with Each Scenario
Table 6 details the cost of the three ARV scenarios. Detailed workings are shown in Supplementary File 1.
Costed ARV Scenarios.

ARV Scenarios
The most expensive ARV scenario is Scenario 2 ($5,039,461), which is marginally more expensive than Scenario 1 (baseline; $4,915,960). The least expensive scenario is Scenario 3 ($4,685,456), where the Cambodian government purchases the cheapest generics on the market. Treatment coverage would drop by 2 percent to 82 percent in Scenario 2 and increase by 4 percent to 88 percent in Scenario 3 if the existing Scenario 1 budget remained unchanged. Given the existing budget detailed in Scenario 1, Scenario 2 would see 1,515 fewer PLHIV able to access ARV. Scenario 3 would allow an additional 3,080 PLHIV to access ARVs given the baseline budget.
DAA Scenarios
The most expensive DAA scenario is Scenario 2 ($3,431,136). It is 14 times more expensive than Scenario 1 (baseline; $245,676). Given the budget of Scenario 1 and the cost of Scenario 2, the latter would see a 93 percent reduction in the number of people with hepatitis C able to access DAA treatment. Given the existing budget detailed in Scenario 1, Scenario 2 would see 2,577 fewer people able to access DAAs. Table 7 details the cost of the DAA scenarios.
Costed DAA Scenarios.

Abbreviations: SOF, sofosbuvir; DVC, daclatasvir.
Discussion
Impact of TRIPS
These results demonstrate that Cambodia's future TRIPS obligations to grant patents could potentially give rise to modest increases in the cost of the ARV program and significant increases in the cost of the DAA treatment program. Anticipated costs would be only marginally higher for the ARV treatment program under a patent protection regime as most ARVs in current use are older medicines and largely off-patent in most jurisdictions. Newer patented ARVs tend to only be used in second- and third-line regimens that have far fewer enrolments. These regimens are used only when a PLHIV has become resistant to a first-line regime or there are other clinical indications to avoid medication in a first-line regimen. However, more and more PLHIV are likely to transition to these regimens in the future. Additionally, it is possible that the current first-line regimen will be superseded by newer, more effective, or safer ARVs in the future. New ARVs are almost certain to be patented and, therefore, cost more than current off-patent first-line generics, thereby increasing the cost of the treatment regimen and possibly leading to fewer people being able to access treatment. These findings are consistent with studies that have explored the impact of the introduction of patents following the end of the transition period on medicine prices in India.30–33
Increased Cost to the ARV and DAA Treatment Program
Despite the small differences in coverage between the scenarios, the increased costs due to patent protection could result in 1,515 fewer PLHIV able to access treatment given the current budget. This reduction in coverage is not inconsiderable given that Cambodia's graduation from LDC status could also mean a reduction in external funding for the HIV program and ARVs, specifically. Eligibility for funding from the GFATM is determined by a country's income classification and disease burden. Cambodia is currently classified as having a high HIV disease burden and has recently been classified a LMIC by the World Bank. Cambodia will lose eligibility for GFATM funding, its only external funder for ARVs, if its HIV burden is not deemed high and if it transitions to upper-middle income country status.
Although ARVs are not ideal medicines to use to measure the impact of patent protection, they provide a useful comparison to newer and, therefore, more likely to be patented medicines, such as DAAs. The DAA scenarios did show considerable difference in cost given the high prices charged by the patent holders, Gilead and Bristol Myers Squibb (BMS). In fact, DAAs have proven to be incredibly lucrative for Gilead and BMS. When Gilead first launched sofosbuvir as Sovaldi in 2014, a 12-week course of treatment cost $84,000 to $94,000, or approximately $1,000 per pill. 63 In its first year on the market, Sovaldi generated $10.3 billion in sales for Gilead and came close to being the highest revenue-producing medicine in the world. 64 Cambodia may be forced to pay these inflated prices if it is unable to access generic alternatives legally. Generic competition has reduced the price of DAAs dramatically in jurisdictions where they are not under patent.
MSF's presence in Cambodia ended in December 2021 when they handed over financial and managerial responsibility of the hepatitis C treatment program to the Cambodian government. Unfortunately, the Ministry of Health is yet to secure a budget for DAA procurement; currently only those able to pay for treatment in the private sector can access DAAs. Treatment with generic DAAs is estimated to cost approximately $600 for a 12-week course. 65 The Ministry of Health hopes to be able to secure funding for DAA treatment in 2023. This is a critical time for Cambodia as looming patent protection may compel Cambodia to pay considerably more for DAAs at a time when it assumes full financial responsibility from MSF for the hepatitis C treatment program.
The Limits of Voluntary Licensing as a Remedy for Patent Barriers
It has been argued that IP barriers, such as the introduction of patent protection, can be addressed by remedial policies like voluntary licensing. 66 A voluntary license (VL) is an authorization given by a patent holder to a generic company, allowing it to produce a generic version of a patented pharmaceutical product. 67 VLs have been touted as a way to address the barriers that patents pose such as high prices and can substantially improve access to affordable generic medicines 68 ; however, current VL agreements would not provide sufficient access to the suite of medicines needed for Cambodia's HIV and hepatitis C treatment programs to alleviate the effects of Scenario 2 for each program.
It is possible that VL could enable Cambodia to access generic dolutegravir for HIV even if the company marketing it is granted patents under the conditions described in Scenario 2. The patent holder of dolutegravir, ViiV Healthcare, has signed VL agreements with the Medicines Patent Pool (MPP) that sublicenses generic manufacturers to allow them to produce and distribute generic versions of these products under the terms of the license. The MPP is a United Nations-backed public health organization working to increase access to and facilitate the development of life-saving medicine for LMICs via VL agreements. 69
There is also an MPP VL with the U.S. National Institutes of Health for patents related to the HIV medicine darunavir (DRV). This license does not include all patents related to DRV and it is therefore likely that Cambodia will still face IP barriers to accessing generic DRV. 70 The original AbbVie 2014 MPP license agreement for lopinavir/ritonavir included Cambodia but was limited to pediatric formulations. 71 The 2015 MPP AbbVie Agreement for the adult formulation of lopinavir/ritonavir was limited to African countries. 72 In March 2020, at the onset of the COVID-19 pandemic, the Israeli Ministry of Health issued a compulsory license for lopinavir/ritonavir as a possible treatment for COVID-19. The following day, AbbVie announced that it would cease to enforce its patents on lopinavir/ritonavir anywhere in the world highlighting the importance of TRIPS flexibilities in facilitating equitable access to medicine and ensuring Cambodia will be able to access generic lopinavir/ritonavir. 73
The VL picture is also patchy for hepatitis C medicine. BMS, the company that holds the patent for daclatasvir, also signed a VL agreement with the MPP in 2015, which enabled the generic manufacture and sale of daclatasvir in 112 LMIC. To date, one Bangladeshi and six Indian generic companies have signed sublicense agreements with BMS to manufacture and supply generic daclatasvir. 74 Although the VL agreement fortunately includes Cambodia, it covers only 65 percent of people living with HCV in LMICs and may exclude Cambodia once it graduates from LDC status.
In addition, Gilead, which holds the patent for sofosbuvir, has signed bilateral VL agreements with 11 Indian generic manufacturers and one each in Pakistan and Egypt that allow for the manufacture and sale of generic sofosbuvir in over 100 countries, 75 including Cambodia. It is not known what price these companies would charge Cambodia for sofosbuvir; however, it is unlikely to match the lowest price available globally. Graduation to middle-income status may change the eligibility of Cambodia to be included in some of the more restrictive VLs. Some VLs may only include LICs, and graduation from LDC status may exclude Cambodia from some of the more exclusive licenses at a time when it must implement patent protection for pharmaceuticals. For example, the ViiV dolutegravir license has been extended twice to expand the territories included, but the original 2014 agreement covered all LDC, low-income countries, and African countries royalty-free and only six LMICs were subject to royalty payments. 76
VLs have been criticized for their lack of transparency, restrictive conditions and higher prices than what can be achieved via compulsory licensing. 67 Access to the generics will depend on whether included generic manufacturers choose to register their products in Cambodia and what prices they command. VLs that lock in supply from specific overseas-based generic companies undermine opportunities for building domestic pharmaceutical manufacturing capacity.
Cambodia has some generic pharmaceutical companies with the potential for expansion. The COVID-19 pandemic has highlighted the need for countries to invest in their own pharmaceutical manufacturing capacity to ensure an affordable and sustainable supply of health products, including medicine. In response to supply and distribution concerns, Cambodian Prime Minister Hun Sen recently granted permission to a local generic company to produce molnupiravir (Lagevrio), an antiviral medicine used in the treatment of COVID-19. 77 In addition, the Cambodian generic pharmaceutical company, Cambodia Pharmaceutical Enterprise, has registered and is manufacturing generic versions of daclatasvir and sofosbuvir in Cambodia. 78 Despite these initiatives the authors could find no overarching strategy for domestic pharmaceutical manufacturing in Cambodia.
Cambodia's Need to Reform IP Laws
To ensure it can continue to protect access to medicine, Cambodia will need to make use of the TRIPS waiver transition period to update its IP-related laws to ensure that when it is compelled to grant patents, it has incorporated all flexibilities afforded it by the TRIPS Agreement. This will minimize the impact that patent protection will have on the cost of medicine and can facilitate access to affordable generics as evidenced by the relative affordability of ARVs in Scenario 3 (in comparison to the other two ARV scenarios) that used compulsory licensing to procure the cheapest generic ARVs on the market. The current Cambodian IP governance system lacks key provisions that could result in a greater number of poor-quality patents, less opportunity to oppose them, greater cost, and longer patent monopoly periods. 18
Cambodia's patent law unnecessarily provides for secondary patents, which add years to patent terms and delays the entry of more affordable generic medicine.79,80 Secondary patenting has led to increased medicine costs 81 and large social welfare losses. 82 The absence of a Bolar provision (which is omitted from Cambodian patent law) can add two or more years to regulatory approval times, which extends the effective monopoly period for originator medicine. 38 Studies have shown that without parallel importation, another important TRIPS flexibility, there would be an increase in governmental health care expenditure and a decrease in consumer welfare. 83 Parallel importation has also been shown to reduce prices for patented medicines.83–85 Although Cambodia has international exhaustion of patent rights that allows for parallel importation in their Law on Patents, Utility Model Certificates and Industrial Designs 2003, Cambodia's Law Concerning Marks, Trade Names and Acts of Unfair Competition includes a national exhaustion doctrine that could prevent parallel importation. 29
A lack of an administrative post-grant opposition procedure—also reflected in Cambodian law—can deter challenges to low-quality patents due to the necessary costs and legal representation required. This absence can lengthen the cost and time taken to revoke a patent granted in error and increase patent monopoly periods. Post-grant administrative procedures can decrease the number of low-quality patent applications. 86 Operating a mailbox system for the filing of patents without a requirement to do so will lead to patents being granted (from the filing date), which would otherwise not have been granted if pharmaceutical companies had to wait until the end of the TRIPS waiver period to submit an application. As a developing, but not least developed country, India was granted a ten-year transition period in which to implement the TRIPS Agreement and introduce patent protection for pharmaceutical products. Unlike Cambodia, India was legally required to operate a mailbox facility during this transition period. The Indian government designed Section 11A(7) of the Indian Patent Act to minimize the impact of a mailbox system. This legislation allows for the continued production of a patented product that was already in use or if there was significant investment in the production and marketing of a product prior to the introduction of patent protection, subject to “a reasonable royalty”. 87 , p.17 If Cambodia were to maintain the mailbox system, it could consider adopting an approach similar to India's to ensure it could maximize access to generic medicine already in use.
Limitations
Prospective studies have been shown to find larger negative effects of trade-related IP provisions on medicine prices and costs than retrospective studies. 29 As this is a prospective analysis, it is possible that assumptions made in this study overestimate the impact of the implementation of TRIPS obligations to grant patents for pharmaceuticals. Although it is likely that the obligation to grant patents for pharmaceuticals will impact the price of other patented medicines, the findings are valid for DAAs and ARVs only and not necessarily generalizable to other drugs. The effects of patent protection could be much greater for more expensive biologic drugs such as monoclonal antibodies and some vaccines. This is a limited analysis of the immediate effects of patent protection and does not measure the full impact of Cambodia's omission of key TRIPS Agreement flexibilities, such as parallel importation, Bolar provision, and post-grant opposition. These omissions could have more cumulative effects over time and their impact on the price of and access to medicine may not be immediately apparent. Further research is needed to better understand the impact of these flexibilities on access to medicine.
Recommendations and Conclusions
When the LDC waiver no longer applies to Cambodia, this country will be required to implement patent provision for pharmaceuticals in its patent law. The obligation to grant patents for pharmaceuticals can be expected to lead to modest increases in the cost of the ARV treatment program due to most ARVs being off-patent worldwide and very large increases in the cost of the DAA treatment program where many medicines are still under patent. Without an increase in annual treatment budgets, patent protection could see a 2 percent drop in treatment coverage, or 1,515 fewer PLHIV able to access treatment, and 2,577 or 93 percent fewer people able to access DAA treatment. These findings suggest that graduation from LDC status and the subsequent TRIPS obligation to provide patent protection can challenge a country's ability to access affordable medicine to maintain national treatment programs.
To minimize the impact of these potential cost increases and to achieve the Ministry of Health and WHO goal of hepatitis C elimination, it is recommended that Cambodia make careful use of its transition period to ensure its patent law is inclusive of all TRIPS flexibilities and reconsider membership in patent agreements and MoUs designed to expedite the granting of patents. It is recommended that Cambodia remove the mailbox facility to avoid granting retrospective patents and being overwhelmed by patent applications. If it decides to keep the mailbox facility, it should consider including a clause similar to the one in the Indian Patent Act that permits the continued production of a patented product where it is already in use or if there was significant investment in its production and marketing prior to the introduction of patent protection. Additionally, Cambodia should amend its Law Concerning Marks, Trade Names and Acts of Unfair Competition to allow for parallel importation and the Law on Patents, Utility Model Certificates and Industrial Designs 2003 to include provisions for Bolar and post-grant opposition and tighten its patentability criteria to exclude secondary and poor-quality patents. Although unlikely, Cambodia should consider delaying graduation from LDC status until 2033 to maximize the time for patent-free access to medicine.
Further research is needed to determine the potential impact of LDC graduation on access to medicine beyond ARVs and DAAs. Qualitative research to explore how countries can best navigate the complexities of the TRIPS/access to medicine nexus and preserve access to medicine in the context of economic pressures is also vitally important.
Supplemental Material
sj-docx-1-joh-10.1177_27551938241242602 - Supplemental material for Cambodia's Imminent Graduation from Least Developed Country Status: What Will be the Impact of the TRIPS Agreement on Access to HIV and Hepatitis C Medicines in Cambodia?
Supplemental material, sj-docx-1-joh-10.1177_27551938241242602 for Cambodia's Imminent Graduation from Least Developed Country Status: What Will be the Impact of the TRIPS Agreement on Access to HIV and Hepatitis C Medicines in Cambodia? by Brigitte Tenni, Joel Lexchin, Sovath Phin and Deborah Gleeson in International Journal of Social Determinants of Health and Health Services
Footnotes
List of Abbreviations
Acknowledgments
The authors would like to acknowledge and thank the MSF France and NCHADS for sharing their treatment data. Special thank you to Mickael Le Paih for sourcing the hepatitis C data.
Author's Contributions
BT designed the study with JL and DG. BT collected and analyzed the data. PS contributed expert knowledge to the data analysis. BT drafted the manuscript. JL, DG, and PS revised the manuscript for important intellectual content. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics
This study has ethics approval from Human Research Ethics Committee at La Trobe University (Ethics reference number HEC 21216).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Brigitte Tenni is the recipient of a Research Training Program scholarship from La Trobe University.
Supplemental Material
Supplemental material for this article is available online.
Notes
Author Biographies
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
