Abstract
Methylpyridine derivatives serve as crucial components and intermediates in agrochemical products, particularly synthetic auxin herbicides. Accurate structural elucidation of these compounds is required for regulatory compliance and successful product registration. While mass spectrometry (MS) techniques are commonly employed for structural analysis, their effectiveness decreases when dealing with isomeric compounds. In this study, we propose an integrated workflow that combines gas chromatography–infrared spectroscopy (GC-IR) and density functional theory (DFT) calculations to enable structural elucidation of methylpyridine derivatives. GC-IR provides temporally resolved infrared (IR) spectra, enabling qualitative and quantitative analysis without compound isolation. GC-IR can effectively distinguish isomeric compounds by comparing IR spectra of energy-minimized structures calculated using DFT, even when the spectral differences are subtle. Our comprehensive approach leverages GC-MS, GC-IR, and DFT calculations, facilitating robust structural characterization of methylpyridine derivatives utilized in the production of diverse agrochemical products.
This is a visual representation of the abstract.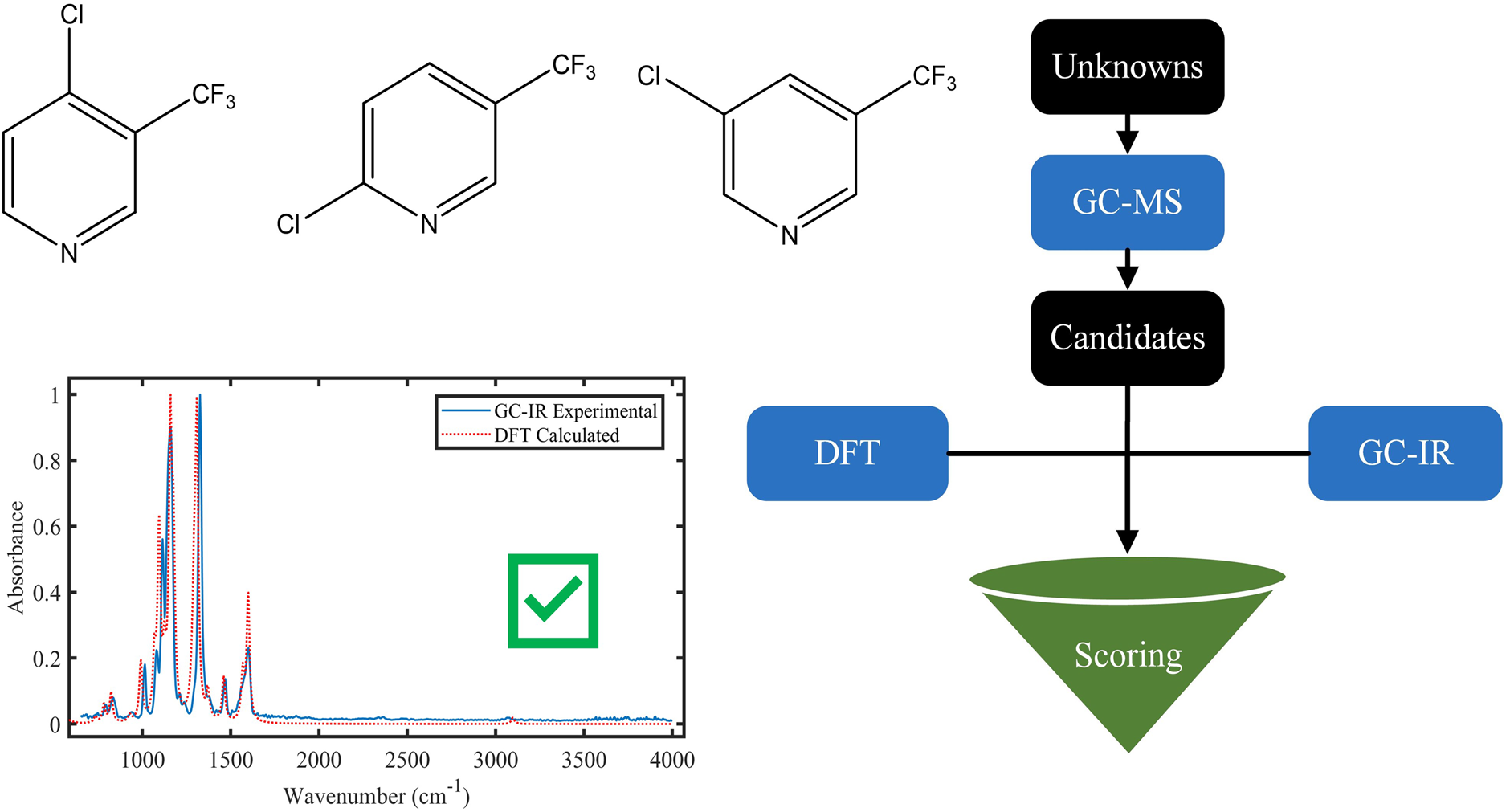
Keywords
Introduction
Methylpyridine derivatives play an important role in the agrochemical industry, serving as both active ingredients and intermediates. 1 Their widespread utilization stems from the scalability and cost-effectiveness of the chemical processes involved in their production. 2 The incorporation of trifluoromethyl pyridine enhances the bioavailability of these compounds compared to trifluoromethyl phenyl analogs.3–5 In this context, several agrochemical products harness the trifluoromethyl pyridine functionality, including sulfoxaflor (Isoclast) and picoxystrobin (Onmira). Additionally, synthetic auxin herbicides, a cornerstone of weed control, have relied on methylpyridine-derived compounds for over five decades. Notable examples include picloram, clopyralid, and aminopyralid.
Changes in the physiochemical properties of trifluoromethyl pyridine-containing compounds, such as basicity and lipophilicity, can be attributed to the exchange of a C=C bond with a C=N bond and an electron-withdrawing trifluoromethyl group. Chlorinated trifluoromethyl pyridines are another common moiety used to alter the physiochemical properties of active ingredients and act as an easily substituted nucleophile to incorporate trifluoromethyl pyridines into active ingredients. 6 The synthesis of trifluoromethyl pyridine typically involves sequential chlorination followed by fluorination, and the original reported synthesis was published in 1947. 7 By adjusting reaction conditions, a range of chlorinated trifluoromethyl pyridine products can be produced for specific applications.
Understanding the chemical structure of process impurities is pivotal in constructing robust and efficient chemical processes. Impurities can persist from intermediate stages to the final material, potentially leading to off-target effects or toxicity concerns. 8 Moreover, the structural elucidation of small molecule impurities and metabolites is a prerequisite for registering agrochemical products and active pharmaceutical ingredients, as mandated by U.S. Environmental Protection Agency and Federal Drug Administration guidelines. 9
Analyzing methylpyridine-related impurities relies heavily on gas chromatography (GC) and associated detection methods, including flame ionization detection, electron capture detection, and mass spectrometry (MS). While isolations for purification and enrichment facilitate alternative elucidation techniques such as Fourier transform infrared spectroscopy (FT-IR), ultraviolet visible, nuclear magnetic resonance (NMR), and X-ray crystallography, obtaining quantities of adequate purity can be time-consuming. 10 Leveraging MS offers distinct advantages due to its sensitivity and specificity.
The continued development of various high-resolution and tandem MS techniques has allowed for structural elucidation with higher levels of confidence; however, proposing definitive structures based solely on mass spectrometric data can still be challenging, especially for isomeric compounds.5,11 GC–infrared spectroscopy (GC-IR) provides temporally resolved IR spectra that can be used for qualitative and quantitative analysis without the need for isolation. The IR spectrum of a compound provides information about molecular connectivity and functional groups that are often lacking in mass spectral data. GC-IR provides a complementary structural elucidation technique in which components do not need to be isolated prior to spectroscopic analysis.12–17 Furthermore, IR spectra can effectively distinguish regioisomers, although these spectral differences may be subtle. Electronic structure calculations, including ab initio and density functional theory (DFT) calculations, can be used to calculate the energy-minimized structures and their associated IR spectra, which allows for comparison and structural assignment of experimental spectra. 18
In this article, we present a workflow that integrates GC-MS and GC-IR for structural elucidation of methylpyridine derivatives. Additionally, we showcase an automated method for assigning structures to experimental GC-IR data through comparison with DFT-calculated spectra.
Experimental
Materials and Methods
Purchased from Combi-Blocks was 2-chloro-5-(trifluoromethyl)pyridine (98%); 3-chloro-5-(trifluoromethyl)pyridine (97%) was purchased from Sigma-Aldrich; 4-chloro-3-(trifluoromethyl)pyridine (97%) was purchased from Matrix Scientific; and acetonitrile, liquid crystal–MS grade (99.8%), was purchased from Fisher Scientific chemicals.
Gas Chromatography–Mass Spectrometry
Gas chromatography–mass spectrometry (GC-MS) measurements were recorded using a Thermo Trace 1310 GC coupled to a Thermo Scientific Q-Exactive mass spectrometer. Split mode was utilized with a split flow of 60 mL/min, a carrier flow of 1.2 mL/min, and a split ratio of 10:1. The inlet was held at 250 °C for the duration of the experiment. A TG-5MS, 15 m × 0.25 inner diameter × 0.25 µm film thickness column was heated at 40 °C for 0.2 min before being ramped to 310 °C at 15 °C/min and held for 1.8 min. The MS transfer line was set to 300 °C, and analytes were analyzed by electron ionization (EI) at 70 eV. Full MS experiments were conducted at a resolution of 15 000 at full width half-maximum (FWHM) resolution with a 50–500 m/z scan range. Data-dependent MS/MS experiments were also conducted at 15 000 FWHM resolution with an isolation window of 4 m/z and a collision energy of 15. A mixture of 2-chloro-5-(trifluoromethyl)pyridine, 3-chloro-5-(trifluoromethyl)pyridine, and 4-chloro-3-(trifluoromethyl)pyridine was prepared at 1 mg/mL in acetonitrile and 1 µL was injected. The total ion chromatogram used to generate the mass spectra shown in Figure 1 can be found in Figure S1 (Supplemental Material).

(a) Electron ionization–mass spectrometry (EI-MS) and (b) EI-MS/MS of 2-chloro-5-(trifluoromethyl)pyridine. (c) EI-MS and (d) EI-MS/MS of 3-chloro-5-(trifluoromethyl)pyridine. (e) EI-MS and (f) EI-MS/MS of 4-chloro-3-(trifluoromethyl)pyridine.
Gas Chromatography–Infrared Spectroscopy
Gas chromatography–infrared spectroscopy measurements were recorded using a Thermo Trace 1310 GC coupled to a Thermo Scientific Nicolet iS50 IR spectrometer. The FT-IR system was equipped with a mercury–cadmium–telluride detector. The split mode was utilized with a split flow of 60 mL/min, a carrier flow of 1.2 mL/min, and a split ratio of 10:1. The inlet was held at 250 °C for the duration of the experiment. A TG-5MS, 15 m × 0.25 mm inner diameter × 0.25 µm film thickness column was heated at 40 °C for 0.2 min before being ramped to 160 °C at 15 °C/min and held for 0.8 min. GC-IR traces were collected over the range of 650–4000 cm−1 at a spectral resolution of 8 cm−¹ with a 0.63 min sampling interval. All solutions were prepared as 1 mg/mL in acetonitrile and 1 µL was injected. The gas chromatogram used to generate the IR spectra shown in Figures 2 and 3 can be found in Figure S2 (Supplemental Material).

Comparison of experimental GC-IR spectra of 2-chloro-5-(trifluoromethyl)pyridine (top), 3-chloro-5-(trifluoromethyl)pyridine (middle), and 4-chloro-3-(trifluoromethyl)pyridine (bottom). Spectra were acquired at 8 cm−¹ resolution from 650 to 4000 cm−¹.

Peak-normalized GC-IR spectra with overlaid DFT-calculated FT-IR spectra of (a) 2-chloro-5-(trifluoromethyl)pyridine, (b) 3-chloro-5-(trifluoromethyl)pyridine, and (c) 4-chloro-3-(trifluoromethyl)pyridine.
Infrared Spectral Scoring Matlab Application
Experimental IR spectra were extracted from chromatograms using a custom Matlab graphical user interface (Figures S3 and S4, Supplemental Material), averaging all spectra collected throughout the analyte elution. A solvent spectrum was subtracted from each analyte spectra to obtain pure components. Baseline correction using an asymmetric least squares algorithm was applied to each experimental spectrum. Experimental and DFT-calculated spectra were trimmed to a common wavenumber axis and normalized using the eigenvector's unit length normalization (2-Norm), which is a weighted normalization procedure where larger intensities are weighted more heavily in scaling.19,20 The resulting DFT spectrum was interpolated using a spline method to match experimental wavenumber axis values.
Normalized spectral similarity scores (NS3) were then calculated as a sum of squares of both Euclidean distance (d) and spectral angle mapper (SAM or α),
21
where r represents the experimental spectrum and t is the DFT-calculated spectrum for all wavenumber indices from 1 to C. Euclidean distances are zero for identical spectra and can be any positive number. The SAM (α) is an angle from 0 to
Results and Discussion
The workflow described integrates the use of GC-IR, GC-MS, and DFT calculations to elucidate unknown structures. While MS holds distinct advantages over IR spectroscopy in terms of sensitivity and selectivity, it often struggles with the unique challenge presented by constitutional isomers. These isomers cannot be differentiated solely based on accurate mass. Unless tandem MS generates unique fragments that can be rationally explained, determining the absolute structure using only MS is a formidable task.
Figure 4 illustrates a novel workflow that initially employs GC-MS and GC-IR to draw preliminary conclusions about unidentified structures. Details such as accurate mass, fragmentation patterns, and probable substituents are utilized to narrow down the list of potential compounds. These data subsequently inform the selection of candidate structures. For these candidates, IR spectra are generated using DFT calculations (EDF2/6-31G*) and compared against experimental IR data. The candidate structure with the lowest score is then identified as the top candidate for the unknown structure. Recently, a similar approach was implemented for DFT spectroscopy (NMR and IR) integrated identification of terrorist compounds; however, this work still relied heavily on manual comparison against the calculated DFT spectra. 18

Schematic diagram of steps for elucidation of unknowns using GC-MS, GC-IR, and DFT modeling.
As proof of concept, we evaluated a mixture of three chloro-(trifluoromethyl)pyridine isomers (2-chloro-5-(trifluoromethyl)pyridine, 3-chloro-5-(trifluoromethyl)pyridine, and 4-chloro-3-(trifluoromethyl)pyridine). Each regioisomer was distinguished by the positional variation of the single chloro-substituent. These compounds serve as examples of isomers that could be produced in a methylpyridine derivatization process where off-target substitution has taken place.
The GC-MS experiments conducted were twofold. Initially, high-resolution MS data were collected using EI, followed by tandem MS conducted on the base peaks in each mass spectrum. Figure 1 presents the respective mass spectra (top) and collision-induced dissociation (CID) spectra (bottom) for each chloro-(trifluoromethyl)pyridine isomer.
While EI typically results in significant fragmentation, small molecule pyridine compounds generate stable radical cation species, based on the lone pair on the pyridine nitrogen and the charge stabilization due to conjugation. Given the high abundance of the molecular ion, the accurate mass and isotopic ratios can be confidently used to determine the elemental formula of the analyte. 22 For example, the M+2 isotope in the EI spectrum for all three isomers signifies a single chlorine substituent. The high-resolution, accurate mass data were used to determine the elemental formulas within 3 parts per million (ppm) of the theoretical m/z value for the radical cation of C6H3ClF3N.
Tandem MS was then conducted on the high-intensity molecular ion at m/z 180.9905. The precursor ion was isolated and then fragmented by CID before being analyzed. Using tandem MS, we can further justify the elemental formula and begin to elucidate the structures of the species. Two major fragment ions are consistent across all three product ion spectra, m/z 126.0154 (C6H2F2N) and 146.0216 (C6H3F3N). These fragments are a result of the loss of a chlorine radical that produces m/z 146.0216 followed by subsequent loss of HF to produce m/z 126.0154. The cluster of ions in the m/z 161 and 162 range represents the elimination of fluorine radical or HF from the molecular radical cation. Interestingly, ion–molecule reactions (IMRs) with residual water were observed for all three analytes. These IMR species are m/z 164.0320 (C6H5OF3N) and 104.0134 (C6H2ON). As they are present in both the MS and MS/MS spectra, it can be concluded that residual water is present in both the ion source and the MS/MS collision cell.
Using the accurate mass, we can predict the presence of either a pyridine or benzene molecule and the presence of a chlorine substituent. The absence of an indicative loss of NH3 or HCN allows us to deduce that the molecule is a pyridine. Additionally, the presence of m/z 68.9948 indicates a trifluoromethyl substituent. Thus, we can predict that each of these molecules is a pyridine with a trifluoromethyl group and a chloro group. Although MS provides substantial information about the molecular formula based on the exact mass, MS/MS does not provide sufficient information to differentiate the three regioisomers. The accurate mass and fragmentation data do offer enough information to reduce the number of candidate structures to 10 potential isomers that are listed in Table I.
Similarity scores comparing experimental chloro-trifluoromethyl pyridines to all DFT-calculated structural isomers. A lower number is a stronger spectral match for all scoring types shown.

Entries in bold indicate the lowest values and strongest spectral match.
Vapor phase IR spectra were collected using GC-IR to provide additional structural information and are shown in Figure 2. Detailed peak assignments for each of the three regioisomers are included in Table S1 (Supplemental Material). Experimental vapor phase peak positions were generally consistent with reference texts’ assignments of poly-substituted pyridines and aryl compounds,23,24 particularly when accounting for the presence of strong electron-withdrawing groups that tend to shift adjacent C=C vibrations to higher frequencies. 23 Pyridine–Cl and pyridine–CF3 stretches were 10–30 cm−¹ higher than the reported ranges for Ar–Cl and Ar–CF3 stretches. 23 This difference is supported by comparisons of experimental vapor phase spectra of 2-chlropyridine and 2-trifluoromethyl pyridine spectra with corresponding mono-substituted benzene compounds. 25 Similarly, the hydrogen out-of-plane deformation modes in the GC-IR spectra near 800 cm−¹ are about 60 cm−¹ higher than similarly substituted lutidine (dimethylpyridine) spectra. 26 Comparing spectra of pyridine with 2-chloropyridine shows a similar trend with a +40 cm−¹ shift of CH out-of-plane deformation modes. The overtone and combination bands near 1700–2000 cm−¹ can sometimes be useful in assigning substitution patterns for poly-substituted pyridines but are notably absent in the experimental GC-IR spectra likely owing to low signal intensity and the low corresponding signal-to-noise ratio.
The low-frequency regions from about 680–930 cm−¹ are dominated by out-of-plane ring deformation, CH out-of-plane deformation, in-plane ring deformation, CF3 deformation, and Ar–Cl stretching modes. The DFT simulations show the expected vibrational coupling among the in-plane ring deformation modes, the CF3 deformation modes, and the Ar–Cl stretching modes, while the out-of-plane ring and CH deformations are relatively pure. Aside from the strong overlap of the in-plane and out-of-plane modes near 720 cm−¹ for the 3-chloro-5-(trifluoromethyl)pyridine, most high-intensity modes are distinct, if not fully resolved. Though most of the modes remain within the expected regions, the complex interplay between the pyridine ring, the electron-withdrawing constituents, and the vibrational coupling in the regioisomers makes unambiguous peak assignments difficult without the aid of computational tools. The 1000–1400 cm−¹ region is composed of broad and overlapping bending and stretching modes form Ar–Cl stretches (1013–1120 cm−¹), various CF3 stretching modes (1013–1375 cm−¹), Ar–CF3 stretches (1150–1375 cm−¹), and CH in-plane bending modes (1013–1375 cm−¹). The 1400–1600 cm−¹ region is composed of ring stretching modes.
By comparing the three GC-IR spectra to one another and to similarly substituted dimethylpyridine spectra, 26 regioisomer assignments can be assigned, though at a low level of confidence: The 3-chloro-5-(trifluoromethyl)pyridine can be tentatively assigned using the strongly absorbing CH out-of-plane deformation (1,1,1 H) at 895 cm−¹ that absorbs at a higher frequency than the analogous (two adjacent H) modes of the other regioisomers. This regioisomer assignment is strengthened by the corresponding peak shape and locations of the 1592 and 1431 cm−¹ ring stretching modes as compared to the 3,5-dimethylpyridine vapor phase spectrum. 26 The 2-chloro-5-(trifluoromethyl)pyridine is assigned by comparing ring stretching modes at 1375, 1469, and 1600 cm−¹ to that of 2,5-dimethylpyridine 26 (1387, 1489, and 1599 cm−¹, respectively). Similarly, the 4-chloro-3-(trifluoromethyl)pyridine is assigned by comparing ring stretching modes to that of 4,3-dimethylpyridine (1412, 1474, and 1566 cm−¹ versus 1414, 1487, and 1591 cm−¹, respectively). The strong Ar–CF3 stretch near 1280 cm−¹ is also a diagnostic peak for these regioisomers. Mechanical coupling of the Cl and Ar–CF3 lowers the frequency of the C–F vibration by about 20 cm−¹ for the 4-chloro-3-(trifluoromethyl)pyridine (ortho) compared to the 3-chloro-5-(trifluoromethyl)pyridine (meta) regioisomer. This mode is absent for the 2-chloro-5-(trifluoromethyl)pyridine (para) regioisomer. The regioisomer-dependent peak shift and peak absence of this C–F mode are supported by DFT simulations of the ortho-, meta-, and para-substituted chloro-trifluoromethyl benzene isomers that behave similarly.
For assignments using purely computational results, DFT-calculated IR spectra were generated for all 10 candidate structures (Table I). Similarity scores were calculated using a custom Matlab program to compare the experimental vapor phase IR spectra to the calculated DFT spectra. Figure 3 displays overlays of DFT and experimental spectra for a subset of chloro-(trifluoromethyl)pyridine isomers after normalization and baseline correction described in the Experimental section. An overlay of the full IR spectra 650–4000 cm−1 with the DFT spectra is shown in Figure S5 (Supplemental Material).
Euclidean distance, SAM, and NS3 similarity scores for all candidate structures are shown in Table I. For these three quantities, a lower score indicates a better match between the spectra. 21 Scoring the experimental data with the DFT-calculated spectra accurately determined the correct structure in all three cases. However, in the case of 2-chloro-5(trifluoromethyl)pyridine, the DFT spectrum for 4-chloro-3(trifluoromethyl)pyridine yielded a score nearly identical to the correct match. This implies that despite the precision of the scoring system, further experimentation may be required to confidently assign structures.
Conclusion
We have developed a workflow that integrates GC-MS and GC-IR spectroscopy for the structural elucidation of methylpyridine derivatives. This workflow includes an automated method for comparing experimental GC-IR data with spectra calculated using DFT.
The accurate mass and fragment information provided by GC-MS was utilized to narrow down the number of candidate structures to 10 regioisomers. Following this, vapor phase IR data were collected using a GC-IR system and compared to the DFT-calculated IR spectra of all potential regioisomers using a custom Matlab program. The result was a similarity score for each candidate structure, with the lowest value indicating the closest match and, thus, the most likely structure. This approach successfully predicted the correct structures for each of the three test compounds, though closely scored candidates should be manually inspected for accuracy and consistency because this process will likely select incorrect structures in some cases. This process should narrow the potential structure candidates and is not intended as a substitution for vibrational spectral interpretation.
This multidimensional structural elucidation workflow is a robust tool that enhances confidence in structure assignments by utilizing MS, IR spectroscopy, and DFT modeling. Although demonstrated using a high-resolution GC-MS system, this workflow can be expanded to any GC-MS platform, as the MS data were primarily used to reduce the number of candidate structures prior to GC-IR scoring. While calculating DFT spectra is not computationally demanding for a limited number of small molecule structures, this step may become a significant challenge if molecular complexity increases or if the number of candidate structures becomes exceedingly high.
This workflow represents a significant advancement in integrating mass spectrometric and spectroscopic data for our structural elucidation process. However, this approach is limited to volatile or semi-volatile compounds, making it unsuitable for larger nonvolatile compounds, which constitute a significant portion of our work. Future research will explore additional use cases and expand our workflow to the liquid phase.
Supplemental Material
sj-docx-1-app-10.1177_27551857241302108 - Supplemental material for Combined Gas Chromatography–Mass Spectrometry, Gas Chromatography–Infrared, and Computational Approach to Differentiate Constitutional Isomers of Methylpyridine Derivatives
Supplemental material, sj-docx-1-app-10.1177_27551857241302108 for Combined Gas Chromatography–Mass Spectrometry, Gas Chromatography–Infrared, and Computational Approach to Differentiate Constitutional Isomers of Methylpyridine Derivatives by Joshua T. Johnson, Brett Marsh, Hannah Zimmerman-Federle, David Archer, Michael Bishop and Ziyan Fan in Applied Spectroscopy Practica
Footnotes
Acknowledgments
Microsoft Copilot Enterprise (2024). Copilot (Mar 01 version; large language model) was used for grammar and clarity. All content in this manuscript is original and was created by the authors. References 2 and ![]() were suggested by Microsoft Copilot and were verified for relevance and accuracy.
were suggested by Microsoft Copilot and were verified for relevance and accuracy.
Declaration of Conflicting Interests
The authors declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
All supplemental material mentioned in the text is available in the online version of the journal.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.



