Abstract
Objective:
To investigate the effects of the Coronavirus disease 2019 (COVID-19) virus on seminal fluid analysis (SFA) parameters in patients who have had the infection.
Background:
COVID-19 is caused by severe acute respiratory syndrome coronavirus 2 and has had a significant global impact. Recent studies suggest a potential relationship between COVID-19 and male reproductive health, particularly in its effects on semen quality.
Methods:
A prospective cohort study was conducted, and the trial had 50 participants outset (6 participants were lost to follow-up later, and the final analysis involved 44 participants). The study was conducted at the Baghdad Medical City Complex, and it lasted from August 2020 to August 2022. Patients who had received any COVID-19 vaccination were excluded.
Results:
In the present study, 44 male participants were included in this study, their mean age is 31.8 ± 5.1 years ranging from 22 to 42 years, their mean body mass index is 24.7 ± 3.4 kg/m2, and about 77.3% were smokers. All SFA parameters were significantly reduced from baseline to 3-month visits, while from 3 to 6 months, there was a significant increase in all SFA parameters.
Conclusion:
COVID-19 infection has a negative effect on SFA parameters during the first 3 months of post-viral infection. Parameters of SFA recovered to levels comparable to baseline SFA between 3 and 6 months after viral infection.
Introduction
Coronavirus disease 2019 (COVID-19) is a highly contagious viral illness mainly caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). It had a disastrous effect globally, resulting in more than six million deaths worldwide as of March 2022; it became the most significant global health crisis since 1918 when the influenza pandemic took place.1,2 The first cases of coronavirus illness were reported in December 2019 in Wuhan, Hubei Province, China. The disease had spread across the world rapidly in a short period, which made the World Health Organization (WHO), on 11 March 2020, to announce this disease as a global pandemic. Since being announced a global pandemic, the virus has affected many countries worldwide and has posed significant challenges to their healthcare systems. 3
The prolonged shutdowns during the COVID-19 pandemic led to a high rate of loss of livelihoods, and the economic factors have significantly affected the world. Many countries have faced and experienced second or even third outbreak waves of COVID-19, and this may be explained by the characteristics of this virus to the emergence of mutant variants. Despite the substantial progress in COVID-19 clinical research has resulted in a better SARS-CoV-2 understanding and the management of this respiratory illness, the spread of the virus has been continuing and the concerns of the pandemic increased, as SARS-CoV-2 continues to wreak havoc globally.4–6
One of the major concerning issues is that SARS-CoV-2 and other RNA viruses have a specific adaptation characteristic, including the most important being that it is prone to genetic evaluation with mutations development over time, causing mutant variants that may involve a new different characteristic than the original ancestral strains.7–9 As the impressive efforts and the short time to develop a vaccine to prevent the illness and infection caused by COVID-19 and the efforts including boosters of vaccine, the presence of the new SARS-CoV-2 variants is a major concern for a new spread of this virus.10–12
Infertility is medically defined as the inability to establish clinical pregnancy following 12 months of regular, unprotected sexual intercourse or impairment of a person’s capacity to reproduce either as an individual or with a partner. Depending on various factors, the intervention for fertility may start before the 12th month; these factors include the medical, sexual, and reproductive history; age; physical finding; and diagnostic testing. The terms “subfertility” and “infertility” can be used interchangeably.13,14
Subsequently, evaluation and treatment may be delayed due to many reasons. The male factor is identified along with the female factor; the male factor is identified in 35% of couples with infertility, while approximately in 10% of cases the male factor is the only identifiable cause. 15
Male infertility may be due to different underlying etiologies, such as a man having a decreased sperm cell number during ejaculation (oligozoospermia) or even no sperm cells in the ejaculate (azoospermia), and there are some infertile men with normal sperm cell number. Most cases are associated with low concentration of sperm and inadequate or insufficient sperm quality. Poor sperm quality is represented by a decrease in the motility of the sperm (asthenozoospermia) and/or abnormal morphology, with an increased incidence of teratozoospermia in spermatozoa, while the minority of cases of infertile men may have normal concentration of the sperm and a normal morphology and motility. 16
It is known that the main two transmission routes of COVID-19 are through respiratory droplets or close contact, but recently, scientists think that maybe there is another route of transmission, which is through sexual intercourse due to the presence of SARS-CoV-2 traces in samples of urine and feces. It is significant to understand about the transmission pathways of this virus. However, there is a possibility of the negative impact of this virus on human fertility, leading to a negative impact on it.17,18
As this subject garnered special interest from scientists and researchers, many research projects were done to resolve the conflict and ensure the suspicion of the relationship between COVID-19 and the male reproductive system. All the investigations were done by researchers to detect the presence of SARS-CoV-2 in a semen sample of a patient who was diagnosed with COVID-19 infection. The results are variable. Some research investigations found that there are no traces of SARS-CoV-2 in infected persons and suspect there is no evidence of COVID-19 effect on male fertility, while some research resulted in a positivity presence of SARS-CoV-2 traces in a semen sample in infected patients. Another concern is that the expression of angiotensin-converting enzyme 2 (ACE2) is represented as a major molecule receptor that binds with SARS-CoV-2 and enters the host cells, leading to an increase in the rate of infection by this virus, which then consequently effects the male reproductive system and become significant.2,3,17,18
This subject should have a special focus by researchers. This study aims to evaluate the impacts of SARS-CoV-2 on the male semen count, concentration, morphology, quality of motility, and volume by investigating the seminal fluid and analyzing the effects of COVID-19 on it. To the best of our knowledge, this is the first study to investigate the role of SARS-CoV-2 on male fertility potential for 6 months.
Methods and patients
Study design
A prospective cohort study was conducted, and the trial had 50 participants at outset (6 participants were lost to follow-up later, and the final analysis involved 44 participants). Reverse transcriptase polymerase chain reaction or PCR was used to confirm COVID-19 infection in all the subjects (RT-PCR) who had male partners; those who had female partners were not tested for COVID-19. According to the most recent Iraqi guidelines approved by the Iraqi ministry of health in 2021 (June), range of severity of COVID infections is represented as mild, moderate, severe, or critically ill patients. 19 All participants were followed up in two visits after the baseline visit, each 3 months apart (total three visits), and seminal fluid analyses (SFAs) were performed in each visit.
Following the initial assessment, based on clinical evaluation, patients with COVID-19 were divided into four groups: mild, moderate, severe, and critical cases. Patients were categorized as mild cases if they have no pneumonia on a CT scan; moderate cases when patients do have pneumonia on a CT scan; severe cases if patients have pneumonia on a CT scan, oxygen saturation 93%, or respiratory rate 30 breaths per minute; and finally, the critical cases group includes patients with respiratory failure/need for mechanical ventilation. In the current study, only people with mild and moderate illness were enrolled. All patients received care in accordance with the WHO treatment protocol, which is based on the severity of the condition. 20 Although the sixth edition of the WHO manual was published in 2021, the fifth edition was used for the duration of this study, which commenced in 2020, to maintain consistency in methodology and interpretation.
Study settings
The study was conducted at the Baghdad Medical City Complex, and it lasted from August 2020 to August 2022.
Inclusion criteria
Newly diagnosed COVID-19 infection in male participants’ partner only
Mild and moderate COVID-19 infection
Apparently healthy young male (normal secondary sexual characteristics and normal testicular sizes)
High-risk group for infection (health care professionals)
Exclusion criteria
History of infertility
History of inguinal and scrotal surgeries (like varicocele surgeries, epididymal cyst surgery, and orchiopexy), mumps infection, and scrotal trauma
History of chemotherapy, radiation exposure, or hormonal treatment
COVID-19 vaccination
Semen analyses
Masturbation was used to gather semen samples. Semen parameters were evaluated in accordance with the guidelines in the laboratory manual of the WHO. Baseline semen samples were collected after confirmation of SARS-CoV-2 infection using RT-PCR and prior to any treatment initiation. All samples were processed under strict infection-control standards, including PPE use, biosafety cabinets, and disinfection protocols according to WHO guidelines.
All semen parameters were measured using an automated semen analysis system shown in Figure 1.

Automated SFA measurement device.
Semen parameters were evaluated in accordance with the fifth edition of the WHO Laboratory Manual for the Examination and Processing of Human Semen (2010).
Ethical consideration
Ethical consideration was one of the priorities of this study. Every procedure in this study that involves human participants was carried out according to the ethical standards of the institutional and/or national research committee. A written consent was obtained from every participant of this study.
The reporting of this study conforms to the STROBE statement 21 (Supplemental Material).
Statistical analysis
All continuous variables followed normal distribution, paired t-test was used to analyze the difference between each pair of time, and p-value is considered to be significant if <0.05. All analyses were carried out using SPSS version 24.0.
Results
In the present study, 44 male participants were included in the study, with a mean age of 31.8 ± 5.1 years ranging from 22 to 42 years, and their mean body mass index (BMI) was 24.7 ± 3.4 kg/m2, and about 77.3% were smokers, as illustrated in Table 1.
Assessment of demographical data.

Y: year, SD: standard deviation, kg: kilogram, BMI: body mass index.
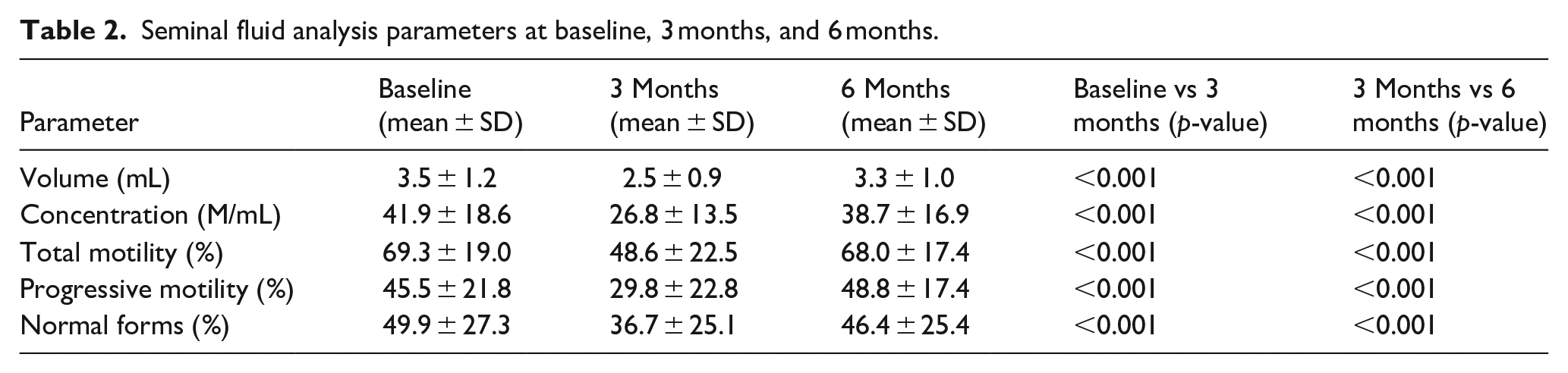
All SFA parameters were significantly reduced from baseline to the 3-month visit, as illustrated in Table 2 and Figures 2–6.
Seminal fluid analysis parameters at baseline, 3 months, and 6 months.

Assessment of seminal volume throughout the study.

Assessment of seminal concentrations throughout the study.

Assessment of seminal total motility throughout the study.

Assessment of seminal progressive motility throughout the study.
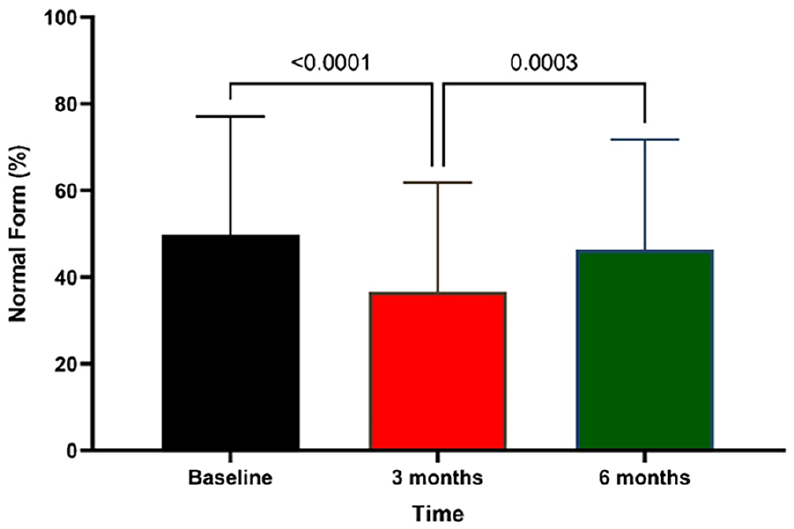
Assessment of seminal normal form throughout the study.
All SFA parameters were significantly increased from 3 months to 6 months visit, as illustrated in Tables 3 and 4 and Figures 2–6.
Assessment of SFA parameters according to WHO criteria during the study period.

This table shows the classification of SFA parameters based on WHO criteria.
SFA changes by COVID-19 severity group (n = 44).

Only patients with mild (n = 26) and moderate (n = 18) disease were included.
Discussion
Several viral illnesses have been described in the literature to negatively impact spermatogenesis, including Mumps virus, Zika, HBV, and HCV.22–24 According to Lorusso et al, 25 patients who tested positive for HBV and HCV had significantly worse sperm motility, concentration, viability, and morphology than those in the control group. Total sperm count and total count of motile sperms were reduced by around 50% on day 60 compared to day 7 after the onset of symptoms, according to Joguet et al. investigation into the impact of the Zika virus on semen parameters. Patients with COVID-19 in the study by Joguet et al. 23 had significantly lower mean total motile sperm counts and total sperm motility. The complication of epididymo-orchitis affects 15%–30% of males who contract mumps. In the majority of cases, epididymo-orchitis affects just one testicle, causing a temporary decrease in sperm parameters (such as count, morphology, and motility). However, 15%–30% of cases are bilateral, resulting in atrophy of testis and eventual infertility.24,26
In the current work, we have documented the sperm characteristics in a cohort of 44 COVID-19-infected patients and, to the best of our knowledge, have conducted a longitudinal evaluation of spermatogenesis both during and after COVID-19 recovery. Currently there is no study comparing the COVID-19 pandemic effects on seminal fluid analysis parameters from 3 to 6 months after infection (Table 2).
In the present study, seminal fluid volume, concentrations, both total and progressive motility, and normal form significantly changed during our assessment. During the first 3 months after infection, the SFA were significantly reduced for all these parameters, while in an unexpected manner, the next 3 months revealed an increase in these parameters’ value statistically similar to the baseline value, indicating recovery of SFA from 3 to 6 months after the initial infection (Table 4). This table shows the classification of SFA parameters based on WHO criteria.
In a recent study, Holtmann et al. (involved 34 participants and semen obtained within 54 days after the absence of symptoms in addition to baseline semen parameters) found that COVID-19 has a negative effect on sperm quality in four recovered patients who required hospital care during the COVID-19 course, but not in 18 recovered patients with mild symptoms who could receive care at home. However, the cohort size was small, and the effects of COVID-19 on spermatogenesis are still unknown. Here, our results provide concrete evidence of transient decreases in total sperm concentration, sperm count, and the total of motile and progressive motile sperm in patients who had recovered from the virus, pointing to a negative but potentially reversible effect of the virus on testicular function. 27
In a study by Guo et al., 28 (the study involved 41 patients, semen analysis obtained after a median of 56 days after hospital discharge, and they were followed again 29 days later) total sperm concentration, sperm count, and the total of motile and progressive motile spermatozoa in the patients were all safely lower after recovery from COVID-19 than those of the control group (patients with no COVID-19 infection followed up to the same duration as those with active COVID-19 infection). The total sperm concentration, sperm count, and the total count of motile and progressive motile sperm per ejaculate clearly increased, and at the second sampling, the morphologically abnormal sperm percentage reduced. The increases in the percentages of motile sperms were not statistically significant, suggesting that either a longer period may be required for the recovery of sperm motility or that these patients already had low sperm motility prior to COVID-19. These results show that the aftereffects of the virus may persist for one spermatogenic cycle given that the human spermatogenic cycle is thought to last roughly 74 days,29,30 which is consistent with our observations.
The results of a different cohort research with people with mild symptoms showed that mild COVID-19 significantly reduced overall sperm motility and motile sperm count (Table 4). The drop in sperm concentration was noticeable as well, although it was not statistically significant. 31
It is possible that SARS-particular CoV-2’s pathogenic mechanism is not the only cause of patients’ transient sperm parameter changes. COVID-19’s effects on male reproduction have other potential explanations. A large retrospective cohort study also revealed that COVID-19 was linked to higher rates of mental health problems, 32 and the patients may have experienced emotional and mental stress as a result of infection by the SARS-CoV-2. Meanwhile, clinical data currently available suggest a negative correlation between psychological stress and spermatogenesis. 33
Second, given that the majority of patients typically present with a fever, some reports have noted temporarily lowered semen parameter values in individuals who had previously been ill with a febrile illness.34,35 A recent study found that recovered patients with fever symptoms during the COVID-19 course is associated with lower sperm concentration, sperm count, and motile sperm number than recovered patients without fever. However, only 18 patients were included in the study for this comparison. Future studies must therefore be conducted on a wider scale to determine the influence of fever. 27 High fever causes degeneration of germ cells. 36
Viral invasion, secondary inflammation, or an immune reaction caused by viral illnesses can have a direct impact on spermatogenesis and testicular endocrine function. 37
In addition, recent studies have explored the broader impact of SARS-CoV-2 on male reproductive health, reinforcing the transient nature of these effects and the potential involvement of endocrine disruption and inflammation pathways.21,38
Conclusions
COVID-19 infection has a negative effect on SFA parameters during the first 3 months after the viral infection. During the 3- to 6-month period after viral infection, recovery of SFA parameters to values almost similar to the baseline SFA was observed. More studies with a larger sample size are required to assess the possible molecular mechanism, which will help to deduce a possible treatment regimen to counter this temporary subfertility.
Supplemental Material
sj-docx-1-map-10.1177_27550834251366837 – Supplemental material for COVID-19 pandemic effects on seminal fluid analysis parameters: A prospective cohort study
Supplemental material, sj-docx-1-map-10.1177_27550834251366837 for COVID-19 pandemic effects on seminal fluid analysis parameters: A prospective cohort study by Ibrahim Qasim Madab, Nibbras Ibrahim Hamid, Montadhar Hameed Nima, Hashim Manea, Shireen Qasim Madab and Israa Habeeb Naser in The Journal of Medicine Access
Footnotes
Ethics approval and consent to participate
Ethical approval was provided by the ethical committee in the university of Baghdad, college of medicine with the number (BMC-004). Written informed consent was obtained from all study participants before enrollment.
Author contributions
Shireen Qasim Madab: Data curation, Investigation, Writing—review & editing.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Competing interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
Data will be available on reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
