Abstract
Background:
Secondary immunodeficiency (SID) disorders are known to occur in patients with haematological malignancies (HM) due to immunosuppressive treatments. Recurring infections causing subsequent morbidity and mortality commonly occur in this patient cohort. Immunoglobulin replacement therapy (IgRT) benefits patients with primary antibody deficiencies. However, evidence supporting their therapeutic role is not as explicit in SID-associated antibody deficiencies, which raises the questions regarding its use in SID and the knock-on effects of this use on its access and availability more generally.
Objectives:
This study aimed to learn about the use of immunoglobulins in SID, identify themes concerning its use and access and suggest methods for improving access.
Design:
This study included a thematic analysis of a published data set of 43 articles concerning immunoglobulin use and access in SID.
Data Sources and Methods:
The data set used to perform the thematic analysis is based on research articles identified from Excerpta Medica Database (EMBASE) and PubMed databases, published as part of a systematic review and part 1 of this two-part publication series.
Results:
A thematic synthesis was conducted to identify recurrent themes. The three primary themes included (1) the context for IgRT prescription, which included patient characteristics and cost burden of IgRT administration, and its use in different countries; (2) factors contributing to inappropriate IgRT use, including health care professionals’ awareness of IgRT, disparity between guidelines and actual clinical practice, and the effect of shortages on prescription and chemotherapy-induced hypogammaglobulinemia (HGG); and (3) measures identified to improve IgRT use and access, which included multidisciplinary involvement, improved diagnostic tools and safer withdrawal and stewardship protocols.
Conclusions:
IgRT use is increasing in HM as a supportive therapy but without comprehensive clinical guidelines and appropriate prescribing recommendations, medication wastage may occur with consequences for immunoglobulin access.
Plain Language Summary
This study covers the use of immunoglobulins in SID, identifies themes concerning its use and access and suggests ways for improving both using a thematic analysis approach. The study identified that IgRT use is increasing in haematological malignancies as a supportive therapy but without comprehensive clinical guidelines and appropriate prescribing recommendations, medication wastage may occur with consequences for immunoglobulin access.
Introduction
Background
Antibody deficiency (AD) disorders are broadly classified as a group of immunodeficiencies whereby the individual is incapable of producing an effective antibody response to a pathogen. Clinically, AD is primarily characterised by recurring infections and can be diagnostically confirmed by a marked decrease in serum immunoglobulin G (IgG) level. 1 AD can be further indexed according to its aetiology as a primary or secondary AD (primary immunodeficiency (PID) or secondary immunodeficiency (SID), respectively) with significant predominant occurrence of the latter in general population. 2
PIDs and SIDs differ in several ways. PIDs are disorders originating from the innate or polygenic defects in the immune system in which a crucial component, immunoglobulin (Ig) synthesis and/or function, is impaired due to abnormalities in differentiation or function of B lymphocytes.1,3 The clinically significant PID includes rare diseases such as X-linked and autosomal agammaglobulinemia (XLA), more common and difficult to diagnose common variable immunodeficiency, among other diseases. In contrast, individuals with SID are not initially predisposed to AD, rather they develop this abnormality as a result of certain clinical conditions due to various aetiologies such as malnutrition, infections targeting the immune system, malignancy or due to specific medications negatively impacting the immune system.2,4 Pertaining to this review, both solid tumours and haematologic malignancies can result in low serum antibody levels, HGG, whether it manifests as disease-related condition, or iatrogenic secondary disorder caused by treatments such as B-cell-targeted therapies, steroids, immunosuppressive agents and radiation.2,5
In the absence of treatment, SID can lead to organ damage, immune dysfunction, recurring infections, morbidity and mortality. 5 The most common haematologic cancers such as multiple myeloma (MM) and chronic lymphocytic leukaemia (CLL) have high incidences of infection-related mortality, with reports of up to 65% in CLL being infection related. 6
Currently, first-line treatment of recurring bacterial infections in SID strongly favours prophylactic-antibiotic administration, such as macrolide antibiotics or co-trimoxazole. 7 However, a recent systematic review on the efficacy of antibiotic prophylaxis conducted by Egan et al. 8 has called for updated clinical guidelines to facilitate prophylactic-antibiotic decision-making. These findings coupled with the high rate of mortality in haematological cancers require close examination of alternative strategies, beyond antibiotics, to treat this vulnerable patient group.6,8
Immunoglobulin replacement therapy (IgRT) is a well-established treatment in PID patients with antibody deficiencies. Its mechanism of action is complex, modulating Fc receptor function, regulating T-cell and B-cell function and providing antibodies with a broad spectrum of specificities against various pathogens.9,10 To define the clinical outcomes of IgRT in SID, one must consider the following definitions utilised by the European Medicines Agency (EMA). The primary endpoint of Ig use is defined as a reduction in serious bacterial infections (SBIs); these are defined as ‘bacteraemia or sepsis, bacterial meningitis, osteomyelitis/septic arthritis, bacterial pneumonia or visceral abscess’ with secondary parameters relying on pharmacokinetic measures of serum Ig, reduction in antibiotic prescription, hospitalisation and all other infections. 11 Hence, this knowledge can be applied to SID as a determinant of the efficacy of treatment.
Despite SID being a licensed indication for IgRT by major regulators like the EMA, its utilisation in supportive cancer therapy and the validity of use in the current licensed indications is not sufficiently documented12–14; however, expert working groups have attempted to understand and characterise patient cohorts who would benefit from IgRT.15,16 In addition, some older and outdated research published more than 20 years ago in this area provides evidence supporting its benefit in HGG.17–19 Access to Ig therapies relies on availability of human plasma for its manufacture, meaning that production cannot be increased easily. The first article in this two-part series describes a scarcity of evidence concerning Ig access and describes that most available research on the topic concerns studies performed in the United States and Italy. With a projected 6%–8% annual increase in Ig usage, it is crucial to be informed about the best practices regarding the frequency of IgRT use and access in patients with haematological malignancies (HM). 20
Aim
To gain a better understanding of Ig use and access through thematic analysis of an available data set.
Objectives
To gain a broad understanding of IgRT use in haematological secondary immune deficiencies;
To understand issues related to IgRT access;
To discuss ways to improve access to IgRT.
Methodology
Overview
This data set consisted of 43 articles which were identified as part of published systematic review which was performed in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) statement 21 .
The present review integrated a qualitative approach to allow for a thorough exploration of themes concerning Ig use and access in SID. A thematic synthesis was drawn from the current body of evidence, and emergent themes were identified and discussed.
The primary endpoint of this review was to establish the current body of scientific knowledge on IgRT in HM in the previous 10 years (February 2012–February 2022), with a focus on use and access to therapies. The data set comprised of peer-reviewed articles concerning Excerpta Medica Database (EMBASE) and PubMed electronic databases. These articles were identified as a result of earlier published inclusion and exclusion criteria.
Data synthesis
The pre-published data set was analysed to create a narrative synthesis of the available knowledge base. Prior to this analysis, relevant quantitative data where possible were transformed into qualitative themes, such that statistical significance or numerical values could be explained in narrative terms. Any recurring quantitative measures were considered for statistical analysis. Synthesised data were interpreted under the main aims of this review, and counts of studies contributing to major themes were tabulated for clarity and ease of comparison.
Results
The thematic analysis of the articles in this systematic literature review has been broken down into three primary themes: (1) contextual factors, (2) access and inappropriate use and (3) improving use and access. These primary themes are then divided into sub themes as depicted in Figure 1.

Outline of qualitative thematic analysis.
Primary theme 1: Contextual factors
The first primary theme aligns with our first and second study objectives to gain a broad understanding of IgRT use in haematological secondary immune deficiencies. The theme of context, surrounding IgRT use and access in HM is further broken down into three subthemes:(1) patient characteristics for IgRT in HM, (2) awareness of IgRT in different countries, (3) health care practitioner (HCP) expectations and awareness of IgRT and (4) cost burden of intravenous immune globulin (IVIg) on patients and health care.
Subtheme 1: Patient Characteristics for Ig Treatment in HM
The patient characteristics covered in the following paragraphs include (1) patient populations suitable for IgRT, (2) tools to estimate patient population suitability for IgRT and (3) IgRT and infection.
Patient populations suitable for IgRT and Infection
In studies focusing on the most common HM requiring IgRT, the most commonly identified cancers requiring IgRT were MM, CLL and non-Hodgkin’s lymphoma (NHL).23–25 Seventeen studies aimed to describe patient characteristics for which IgRT administration would be warranted.12,23–38 Across these studies, there was great disparity between the ‘gold standard’ biochemical and physiological profile of a patient with HM requiring IgRT. It emerged that gender, age, histologic profile or early stage CLL were not characteristics associated with the need for IgRT.29,30 However, there were commonly mentioned characteristics that many clinician-reported and retrospective studies identified. A combination of low serum IgG levels, infections and risk of infection, failure to respond to vaccination or antibiotic prophylaxis and chemotherapy regimens can be used to build a physiological and biochemical profile for those requiring IgRT.12,23–26,28,30–32,34–38 This theme represents an overarching idea that a full clinical profile and holistically assessment of the patient is necessary before prescribing IgRT. The findings of these studies relating to patient characteristics are summarised in Table 1.
Significant findings to define patient parameters for immunoglobulin prescription.

Recurrent infections and SBI were a common characteristic for identifying those who require IgRT in the selected studies (n = 13). IgRT was prescribed to patients during or after a single severe infection or SBI in the majority of studies.12,23–26,30,32,34,35 However, many of these studies have different definitions and cut-off points for infection, with many physicians requiring two or more SBIs to initiate IgRT.23,25,35 Furthermore, bronchiectasis 37 or combined HGG with a single SBI or recurrent infection was seen as a ‘red flag’ and required initiation of IgRT. 12 , 25 ,31,32,39
Oftentimes, IgRT was initiated if there was a presumed risk of infection, as evident in qualitative studies on clinician opinion whereby HCPs refrain from the ‘watch and wait’ approach but rather initiate prophylactic IgRT in the absence of HGG or recurrent or SBIs.25,31,32,36 Many studies reported on the failure of antibiotic prophylaxis, B-cell–depleting therapy or failure to respond to vaccination as an indicator for IgRT.12,24,27,28,30,38
Tools to estimate patient population suitability for IgRT
Serum IgG levels and IgRT
Serum IgG monitoring was one of the most cited tools for defining patient characteristics eligible for IgRT. There was a lack of consensus in the literature regarding what the specific serum Ig level should be before treatment, however, where a defined level of HGG was noted, a range of <3.3 to <6 g/L with a mean of 4.68 g/L calculated based on data from articles in this study observing patient characteristics.12,23,28,30,36,37
Despite this, many studies reported not using serum IgG monitoring as a tool for identifying patients requiring IgRT. Van Winkle et al. 32 reported that only 30.55% of patients that had been administered IgRT had low serum IgG levels as a prerequisite for treatment. 32 Moreover, Svensson et al. 33 in their retrospective study on the prevalence of serum IgG subclass deficiency concluded that not all patients with HGG had an increased risk of infection and that it could possibly be a natural progression of CLL in later stages of the disease, and that subclass deficiency is not a useful tool by itself. This was presumed based on the identification of patients who had low serum IgG levels but no infection and those who had major infection in the absence of HGG. 33 This was expanded in a study by Mauro et al. 29 examining those with CLL Binet stage A; in this retrospective study of 899 patients, those who had low IgG levels at diagnosis did not have an increased risk of infection compared with those with physiologically ‘normal’ IgG levels. 29
An expansion of the concept of serum Ig deficiency was reported by Visentin et al. 38 who characterised combined antibody deficiency (CAD). CAD was defined as a reduction in not only serum IgG but also IgA and IgM, and this study reports that this cohort of patients had a significantly higher incidence of major infection than those without CAD. 38 However, Sánchez-Ramon et al. 40 highlighted that measurement of specific antibody response is ‘complex and requires diverse clinical considerations’ and suggested that utilising specific antibodies in SID is not without challenge, impeding its implementation. 40
Accordingly, there is no conclusive evidence on the value of serum IgG as a primary tool for identifying patients for IgRT, and further expansion of the theories presented by Visentin et al. 38 may define a combination of Ig levels as a determinant for IgRT initiation.
Subtheme 2: Awareness of IgRT in Different Countries
In this section, we cover the awareness of IgRT in both higher- and lower-income countries. In higher-income countries, 32 selected studies identified a geographical location for the study setting. Nine studies contextualised the global use patterns of IgRT. Na et al. 23 characterised clinicians’ opinions across seven countries in the Western hemisphere providing evidence on IgRT use in mainland Europe, the United States and Canada. In this study, there is a comparable use of IgRT among nations, with discrepancies only noted in areas such as serum IgG monitoring and route of administration. 23 Building on this, studies in France and Spain showed a preference for IgRT initiation in the presence of HGG and recurrent infection.24,31 This is mirrored in a clinician survey in New Zealand and Australia, where there is a preference towards IgRT for recurrent infections over antibiotic prophylaxis. 26 A German study reported IgRT initiation when patients had HGG without currently experiencing recurrent infections. 36 Both Na et al. 23 and De Meester et al. 41 acknowledged a rise in the use of subcutaneous immunoglobulin (SCIg) therapy in mainland Europe, this is, where there is a slow infusion through subcutaneous injection in contrast to the rapid infusion seen with IVIg. Both France and Belgium increased the frequency of ambulatory SCIg administration.23,41 Despite a preference for IgRT over antibiotic prophylaxis in both mainland Europe and Oceania, Duraisingham et al. 37 identified that the majority of UK clinicians would not initiate IgRT prior to a trial of antibiotic prophylaxis. The SIGNS study (assessment of Igs in a long-term non-interventional study) also concluded that antibiotic failure was a strong factor to be considered before initiation of IgRT and antibiotic prophylaxis was one of the key recommendations to arise from this study. 36
Concerning low- and middle-income countries, there were only three which addressed awareness and use of IgRT in Non-Western countries, that is, China. Ye et al.’s 42 work in the Zhejiang province of China in both a clinician survey and retrospective study has revealed that almost 65% of physicians did not consider IgRT treatment as an option for those with HM and SID. 27 Moreover, almost 92% of respondents indicated a lack of awareness or use of SCIg. A minor proportion of HCPs in China would consider IgRT use despite indicators such as low serum Ig levels or recurrent infections. Antibiotic prophylaxis was also reported to be underutilised, with only 3% of HCPs responding to the survey using it as a standard treatment of SID.42,43
This analysis shows an imbalance in awareness and use of IgRT between China and Western countries. However, this was without a comprehensive and expanded study of trends in administration globally. This theme assessed only awareness, not whether IgRT use is imbalanced worldwide. The findings of relevant studies in both higher- and lower-income countries are summarised in Table 2.
Significant findings in relation to awareness of IgRT in different countries.

Subtheme 3: HCP expectations and awareness of IgRT
There is also an imbalance in awareness of IgRT among HCPs globally. In the qualitative studies conducted in China, there was a poor understanding or utilising the benefits of IgRT in HM in health care professionals. 42 However, Benbrahim et al. 25 reported that physicians in France had high expectations of IgRT in improving patient outcomes, reducing infections and overall survival in all types of HM. Na et al. 23 found that immunologists adhere more closely to the guidelines than haematologists and other clinicians. A consensus study by Jolles et al. 12 that included immunologists and haematology and oncology experts also demonstrated the awareness and expectations of HCPs. 29 One retrospective study found that HCPs underutilised antibiotics due to increased awareness and fears of antibiotic resistance. 31 The administration of IgRT in all types of HM in the majority of studies was due to a belief that it would decrease mortality and increase overall survival which does not align with current clinical evidence. This is evident when compared with the outcomes of the systematic review by Raanani et al. 6 whereby lymphoproliferative disorders were not found to achieve benefit from IgRT in terms of mortality.
Subtheme 4: Cost burden of IVIg on patients and health care
IgRT is a limited resource and is associated with cost burden on both patients and health care systems as identified in several studies. The scarcity of its supply also drives up costs which is reflected in global imbalances of IgRT use.41,42 In a study regarding withdrawal of IgRT, Patel and Cowan 44 identified lifelong use as a cost burden to both patients and health care resources. The additional associated cost to patients, depending on the reimbursement scheme of the country, can reduce the availability of IgRT to patients who may benefit the most from the treatment.43,45 In their systematic review, Bonet et al. 46 highlighted that there are hesitations among physicians to administer IgRT due to the associated cost burden. However, it is noted that these costs may be off-set by a decrease in hospitalisation and antibiotic prescriptions when IgRT is utilised. 46 This underscores a need for cost-saving initiatives in IgRT prescription; De Meester et al. 41 praised the utilisation of Ig databases by the National Health Service (NHS), UK, and Australian BloodSTAR system as effective methods for preventing inappropriate prescription of IgRT and medication wastage, allowing a decrease in health care expenses. This system of monitoring would further facilitate identification of the more frequently used indications of IgRT and may encourage investment into resources for more prominently reported indications to overcome the limited supply of IgRT.
The route of administration is also a major determinant in health care costs; IVIg and hospital-administered SCIg require increased levels of staff training, increased patient clinic costs and more administrative fees. Hence, facilitated subcutaneous immunoglobulin (fSCIg) could be utilised as a tool in making IgRT more cost-effective as it not only allows for home infusion to decrease costs, but enables decreased interruptions to patients’ daily lives, which in turn reduces missed work-days and has a positive overall direct and indirect economic impact.45,47,48 Ultimately, it cannot be disputed that despite the associated cost burden, there is also a strong foundation of evidence here that highlights the ‘off-set’ costs when IgRT is safely and effectively prescribed. 46
Primary theme 2: Access and inappropriate IgRT use
The second primary theme aligns with our second study objective to understand issues related to IgRT access and is broken into three subthemes: (1) disparity between guidelines and current practice, (2) effect of shortages on access to IgRT and (3) cancer therapy–induced HGG requiring increased IgRT prescription.
Subtheme 1: Disparity between guidelines and current practice
Building on different countries’ treatment of SID in HM, there are also a variety of guidelines utilised in various parts of the world. Notwithstanding this, clinical practice has proven to differ from the guidelines in most screened studies. There is further cause for confusion when one considers that the guidance is currently not founded on a strong body of evidence which in addition to the sheer volume of guidelines available, and leads to disparities. Consensus studies like those from Jolles et al. 12 bring expert together to bridge the disparity between guidelines and practice. 29 Legendre et al. 31 found that less than 25.0% of IgRT prescriptions met the EMA guidelines and that there was a high incidence of IgRT use in patients with lymphoma. This was seen again in Germany, where oftentimes no infection is seen in patients prior to IgRT prescriptions despite German guidelines require a minimum of three SBIs before initiation of IgRT. 36 Agostini et al. 28 outlined the need to follow antibiotic prophylaxis guidance issued by the Infectious Diseases Society of America and utilise IgRT only in circumstances of antibiotic failure, a finding which was not reflected in a study of US clinician opinions by Na et al. 23 Ye et al. 42 identified the need for local guidelines due to the fact that HCPs failed to identify policies to utilise in IgRT treatment. The absence of policy was mentioned in one case study from Japan showing benefit of IgRT in rituximab (RTX)-induced HGG. 49 Wong et al. 26 denoted inconsistency between guidelines utilised by HCPs in Australia and New Zealand; despite a vast majority referring to Australian National Blood Authority Criteria, approximately 20% of clinicians did not utilise these guidance in informing clinical decisions. Casulo et al. 30 described that following the American Academy of Allergy, Asthma and Immunology, the recommendation to start prescription in ‘ patients who exhibited ⩾2 non-neutropenic infections in a 6-month period of time’, allowed for appropriate prescribing within the cohort of patients with RTX-induced HGG. Girmenia et al. 64 literature review of IgRT use in MM aligned with EMA guidelines to reserve use to those with serum Ig levels (<400 g/L) and recurrent life-threatening infections. Narrative literature syntheses published in France recommend only utilising IgRT in those for whom antibiotic prophylaxis has failed; however, it must be considered that this research is a lower level of evidence compared with systematic reviews or cohort studies.27,50
Subtheme 2: Effect of shortages on access to IgRT
Although many studies selected to emphasise that IgRT is a ‘limited resource’ and ‘prone to shortage’, a limited number of studies delve into the actual impact of this on how IgRT is allocated. De Meester et al. 41 and Derman et al. 51 both reported recent shortages in November 2020 and August 2019, respectively, with De Meester et al. 41 further contextualising the expected length of such a shortage to last more than 9 months In these shortages, those without a pre-approved indication, such as patients with SID, had limited access to IgRT, and access was only granted on a case by case basis dependent on supporting documentation and multidisciplinary review with hospital pharmacists at the forefront of the decision process.41,51 Evidently, during these periods of drug shortages, there are limited allotments allocated to hospitals and criteria issued from Departments of Health becomes more stringent, and these studies highlight that those with SID are often overlooked when it comes to prescribing during times of limited allocations.46,51,52 Characteristics of these studies are identified in Table 3.
Significant findings of studies relating to effect of shortages on access to IgRT.

Subtheme 3: Cancer therapy–induced HGG requiring increased IgRT prescription
One factor contributing to increased IgRT use is cancer-targeting agents having a higher prevalence of inducing HGG.
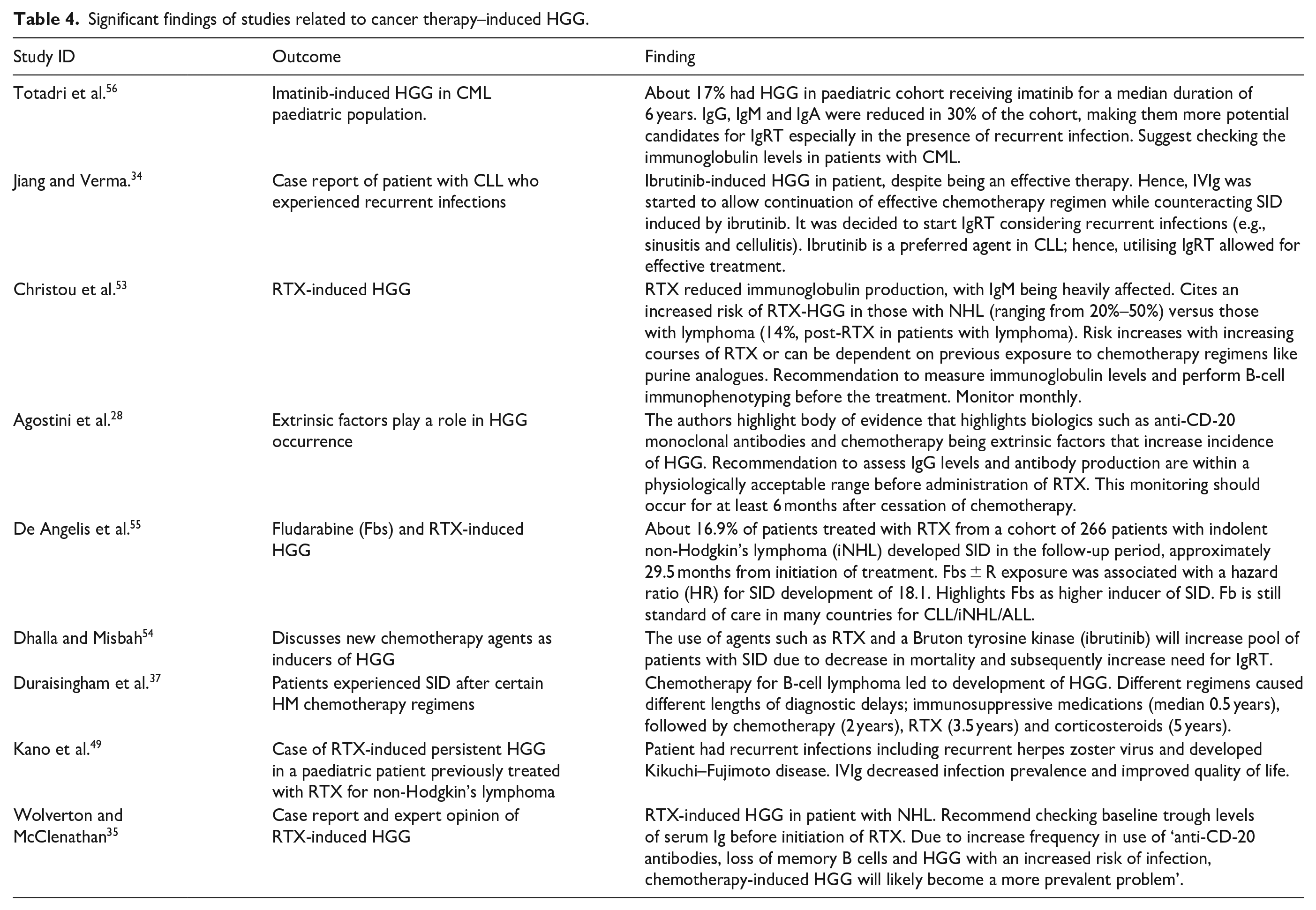
Approximately 20% of the selected studies identified new cancer-targeting agents as a factor increasing the prevalence of SID in HM (n = 9).28,34,35,37,49,53–56 Agostini et al. 28 highlighted the body of evidence whereby RTX, an anti-CD-20 monoclonal antibody, is shown as an extrinsic factor in development of HGG. Of nine studies, RTX is the most prevalently discussed therapy leading to HGG (77.7% of studies). The use of this therapy is associated with an increased frequency of recurrent infections like herpes zoster virus 49 and a reduction in Ig production. 53
Furthermore, the increased risk of HGG depends on the number of courses of RTX, previous chemotherapy, type of HM and baseline IgG levels.53,54 Christou et al. 53 stated that the mechanism of RTX-induced HGG cannot be fully described but that B-cell depletion and advancement of the HM may play a role. Three studies recommend performing a full patient work-up for Ig levels and antibody production, and that B-cell immunotyping should be monitored prior to, during and after RTX treatment.35,49,53 Other chemotherapy agents associated with inducing HGG included ibrutinib,34,54 corticosteroids, 37 CAR-T therapies such as tisagenlecleucel and axicabtagene, 40 fludarabine 55 and imatinib. 56 Different chemotherapy regimens had different SID diagnostic delays, for example, HGG associated with corticosteroids taking up to 5 years to become symptomatic. 37 A benefit of these studies is the revelation that IgRT allows for continuation of effective chemotherapy regimens whereby the normal levels of Ig return. This was reflected in a study by Jiang et al. 34 where a combination of IgRT and ibrutinib allowed for cancer remission with the efficacious chemotherapeutic agent and resolution of chemotherapy-induced HGG.
Overall, there is a comprehensive acknowledgement that with increased use of cancer-targeting agents, especially those specifically targeting B cells, for example, RTX and alemtuzumab, and an increased survival rate of patients with HM, the utilisation of IgRT will increase in addressing therapy-induced HGG. The major findings of these studies are summarised in Table 4.
Significant findings of studies related to cancer therapy–induced HGG.
Primary theme 3: Measures to improve rationale IgRT use and access
This theme aligns with our third study objective to understand ways to improve access to IgRT. It is further divided into three subthemes: (1) multidisciplinary involvement in IgRT decision-making process, (2) improved diagnostic tools for identifying HM patients with SID and (3) safe withdrawal of IgRT and increased stewardship.
Subtheme 1: Multidisciplinary involvement in IgRT decision-making processes
Na et al. 23 highlighted the involvement of a variety of HCPs in the decision-making process for IgRT prescription. Immunologists were described to have a significant impact on this process due to stricter adherence to guidelines, measurement of specific antibody response (94.0%), utilisation of immunisation tests and increased monitoring. Despite a higher usage of IgRT, which is likely due to referral of ‘complex cases’, immunologists had a greater propensity to closely monitor and adjust dosage regimens of those on IgRT. 23 One publication describes engagement of clinical pharmacists as frontline reviewers of all IVIg prescriptions. This procedure was implemented in a time of medication shortages and allowed stewardship over IVIg allocation. 51 A multidisciplinary approach with an expert panel of researchers, doctors, and HCPs from different health care settings in Italy was chosen to accurately construct recommendations for IgRT prescription in Italy. 34 This approach and the democratic consensuses reached allowed for accurate, evidence-based guidelines to be established. Correspondingly, a panel of immunologists and haematologists defined similar guidelines utilising a similar democratic consensus model and Delphi exercise. 12 Involvement of expert from different disciplines allowed for a starting point to be reached on guidelines in the prescription of IgRT and, hence, brought to light that a multidisciplinary approach may help to overcome challenges of access.
Subtheme 2: Diagnostic tools for identifying HM patients with SID
As evidenced in the variety of guidelines and standards for IgRT prescription, accurate diagnostic tools may reduce diagnostic delays and aid appropriate prescribing. A study on calculated globulin as a screening tool for SID had a positive predictive value for identifying HGG (82.5% accuracy). 57 Evaluation of Polysaccharide Typhim Vi Antibody proved an accurate tool in both diagnostic evaluation and a measure for IgRT cessation. 58 Beyond Polysaccharide Typhim Vi vaccinations, Sánchez-Ramón et al. 24 noted that it is likely that tests utilised for PID like vaccination response would have utility in SID. This would involve administration of pneumococcal and polysaccharide vaccinations and a measurement of response conducted at 4–6 weeks post-administration. 24 Conversely, pure IgG subclass screening was found to not be justified as a diagnostic tool without evidence of recurrent infections. 33
Subtheme 3: Safe withdrawal of IgRT and increased stewardship
Studies in the area of IgRT withdrawal and stewardship are limited. Implementation of an IVIg stewardship programme (ISP) resulted in a reduction in patients on IVIg and overall consumption of IVIg (4902 g in 86 patients before ISP to 1777 g in 55 patients after ISP). This study placed stringent controls on the criterion negating IVIg prescription and withdrew therapy in those failing to meet the current body of evidence. The guidance of a multidisciplinary team in the development of this stewardship programme resulted in a net decrease in inappropriate prescribing and increased access to patients at the greatest risk for SBIs. 51 Patel and Cowan 5 discussed the burden of lifelong IgRT prescription and, hence, recommended re-evaluation of IgRT need to be performed every 4–6 months. This study recognises that humoral immunity should not be evaluated until 5 months after cessation due to the half-life of IgG. 44 Implementation of new guidelines within the UK NHS spurred a retrospective evaluation of withdrawal of IgRT in HM; in a cohort of 20 patients who trialled cessation, none had an increased risk of infection, and further 40% of these patients remained stable despite persistently reduced IgG levels. 59
Discussion
This systematic review covers the access to and use of IgRT. The majority of studies recently published on this topic were on the ‘lower’ scale in terms of levels of evidence. 50 However, due to the relatively new emergence of SID in HM as an indication for IgRT and the benefit of retrospective data analysis, the lack of RCTs does not take away from the importance of this topic. In fact, retrospective data collection often overcame selection bias and allowed a longer follow-up period and patient cohort, and appropriate access to domains of this topic like serum IgG levels as one of determinants for initiation or discontinuation of IgRT.29–33,38,43,47,48,52,55,57–61
Theme 1: Context of IgRT use
In the context of IgRT use, the indications and patient cohorts varied, and there was heterogeneity between patient characteristics, guidelines, HCP expectations, cost and access. To create a standard patient profile for IgRT administration, it is important to build on studies that explored this theme. One should consider a holistic patient profile, which would include but not solely rely on serum IgG levels or history of SBI, and adopt a combined approach. Further assessment and exploration of diagnostic tools may expose a new ‘gold standard’ method of SID diagnosis.
Currently, there is a lack of clarity regarding which patients should be treated with IgRT and those who are actually prescribed IgRT in a clinical setting. Malignancies like lymphomas are not mentioned in guidelines, despite being one of the most common condition treated off-label with IgRT in Europe. 31 This indicates a potential for inaccurate guidelines which do not reflect real-world use. There was no clear agreement between the key stakeholders in specifying patient’s characteristics for SID diagnosis, and prescription and termination of IgRT. While patient characteristics vary and the context of IgRT use diverges depending on the setting, country and HCP involvement, a biochemical and physiological profile can be built using a combination of evidence as indicated in the reviewed studies.
Analysis reveals no real evidence that IgRT is restricted to higher-income countries despite the majority of published work being restricted to Europe. However, the work by Ye et al.42,43 has shown that IgRT is not commonly utilised in Asia where HCP awareness of this treatment is limited. Recent publications have placed the United States as the main user and supplier of Ig therapies, accounting for 46% of global Ig sales. 62 In contrast, a population-dense Asia-Pacific region has a much smaller Ig consumption. Nonetheless, there is an expectation of Asia-Pacific to become the fastest-growing region in terms of Ig market shares.20,63 This suggests that despite a limited awareness of HCPs about IgRT therapy in SID, as found in our study, there is an expectation in projected increase in Ig need globally, which means that a sufficient supply is needed beyond just higher-income countries. Consequently, to address this imbalance between Ig usage and awareness in the higher-income countries versus regions like China, the appropriate steps should be taken for dissemination of guidance on IgRT use in SID not just in Europe, the United States or Oceania, but globally to allow for equality in health care and distribution of polyvalent Igs.23,24,26,31,36,37,42,43 Steps could also be taken to develop training material to improve the awareness and understanding of IVIg as a scarce and valuable treatment.
Although there have been advancements in SID research in recent years, many randomised control trials focus on PID as a main indication for IgRT, and there are limited trials in comparison relating to SID. This may further perpetuate the incidence of inappropriate prescribing in the absence of sufficient unbiased evidence. This is seen where clinicians have heightened expectations of IgRT success or high incidences of off-label IgRT prescription HM.6,23,25
Research is needed regarding the economic impact of IgRT in HM. There is conflicting evidence about whether it is more cost-effective to use IgRT which would decrease hospitalisation, or if IgRT is too precious and expensive a resource to utilise in SID.41,44,46 One suggestion is the incorporation of secondary and ambulatory administration of IgRT through patient-taught self-administration of SCIg that would ultimately decrease the cost burden of frequent hospital appointments for IVIg administration.41,44 IgRT places a significant economic burden on countries and patients alike, and increased utilisation of SCIg and monitoring trends in IgRT supply could allow for countries to ensure appropriate allocation of IgRT to the most vulnerable patients. 41
Theme 2: Access and inappropriate IgRT use
This study identified that most publications shared the main conclusion highlighting the need for clear guidelines on the use of Ig to prevent inappropriate prescription. There is a global trend in clinical practice that does not reflect clinical guidance. Clinicians rely on a combination of regional guidance, guidance set out by regulatory bodies like the EMA and professional organisations like the American Academy of Allergy, Asthma and Immunology.23,26,28,30,31,43,64 This incongruity in practice is increasing the rate of ‘off-label’ prescribing as there is no globally accepted standard guideline identified from the variety of studies assessed. The problem of IVIg access goes beyond inappropriate prescription as inappropriate supply impacts IVIg allocation decisions in times of limited supply. There was an underlying theme that there is an urgent need to establish scientifically based guidelines for IgRT use in HM. Failure to establish criteria could mean that those with HM, especially those outside the scope of guidelines, for example, lymphoma, will be overlooked in times of limited supply.
Cancer-targeting novel therapies inducing HGG are on the rise in treatment of HM.28,34,35,37,49,53–56 These therapies prolong and increase the survival rate, and as a consequence, many more HM patients may need IgRT in their lifetime. Some studies recommend a full assessment of patient’s health status, including immunological parameters, before administering anti-cancer therapies with the propensity to induce HGG. In addition, they highlight the need for the development of clear parameters justifying initiation and discontinuation of IgRT administration in this cohort.
Theme 3: Improving rationale IgRT use and access
To ensure appropriate prescribing and address factors leading to unnecessary IgRT use, three approaches were found to be commonly considered. The introduction of Stewardship programmes that assessed by Derman et al. 51 may allow for vigilant allocation of IVIg therapy in times of restricted availability. This experience of Ig prescribing to more vulnerable patient cohorts, with adherence to criteria incorporating appropriate and agreed upon laboratory and clinical patient profile, would ensure continuing access. Bousfiha et al. (2017) suggested that having appropriate criterion in place for prescription of IgRT will enhance access and improve global equality of supply. Evidently, IgRT shortages occur globally but this theme is beyond the scope of this study.
Analysis shows that multidisciplinary involvement in decision-making related to appropriate use of IgRT therapy is a positive step forward. However, it must be stated that multidisciplinary involvement in decision on IgRT use is more likely to take place and be effective in large teaching hospitals and less likely in small less professionally diverse health care settings. Specialist clinical pharmacists are at the forefront of medication supply in hospital settings and with a high level of knowledge of dosing, administration routes and efficacy of treatment, their inclusion in the prescription process may enhance rational use according to established national and international guidelines and recommendations by independent scientific multi-speciality key opinion leaders. This promotion of the rational use of IVIg may have a positive effect on IgRT availability and access.51,66 Utilisation of other HCPs in the wider use and administration of IgRT, such as immunologists, haematologists, nurses and researchers, may help key opinion leaders to generate more rounded evidence-based discussion and help regulatory bodies to reach more progressive guidance with a lower risk of bias.23,13,64 It is evident that the prescription of IgRT should rely on a multidisciplinary decisions, as it has been shown that even doctors, depending on their area of expertise, reach different conclusions regarding the appropriate dosing of IgRT in different patient cohorts. In the absence of informed guidance sufficiently describing parameters for initiation and cessation of IgRT, it can be difficult for a treating physician to make the best decision. It has been demonstrated that withdrawing IgRT in patients who fulfil certain criteria is a practical solution that may help in improving access to some extent. Further steps should be taken to build on this evidence and transform consensus into guidelines, with tapering schedules and monitoring procedures clearly outlined.24,44,51
Limitations
This thematic analysis had several limitations. First, it was limited to articles contained within the original data set which were written in English only and covered the timeframe of the previous 10 years. Comparisons of studies were hindered in many aspects due to heterogeneity between the HM assessed, definitions of HGG and patient characteristics. Although this study reported on disparities between countries in terms of IgRT awareness, western countries represented the majority, possibly since IgRT is more commonly utilised there. Furthermore, this study was restricted to examination of use and access and did not necessarily consider clinical benefits, patient-reported outcomes and statistical analyses of IgRT success in all studies.
Conclusion and recommendations
This thematic analysis explored three primary themes: (1) the contextual factors surrounding IgRT use and access in HM, (2) access and factors contributing to inappropriate IgRT use and (3) ways to improve access to IgRT in cohort of HM patients with secondary immunodeficiencies. In terms of IgRT patient characteristics, a combination of serum IgG levels, infection history and patient response to first-line treatments like antibiotics need to be assessed when deciding on the appropriate patients for IgRT administration. There is also a need for improved education internationally regarding IgRT use in HM to raise awareness and expectations among the public and health care professionals.
When it comes to inappropriate use of IgRT, there is sufficient evidence that in many cases there is a disparity between the clinical practice and guidelines which are limited in scope. To improve the rationale, use and access to IgRT, stewardship protocols closely examine the distribution of IgRT to patients and assess the criteria for which IgRT should be advanced and more thoughtfully implemented.
Further studies are needed to assess an appropriate use of IgRT in HM. These studies should aim to further assess benefits of involvement of HCP of various specialties in creating recommendations and guidelines that will include information not only on appropriate administration schedules but also on therapy withdrawal practices. The role of Ig stewardship programmes could also be explored on a local level with the expectation of publishing the outcomes of stewardship interventions. Further studies exploring ‘gold standards’ of IgRT treatment and the decision to administer IgRT versus other treatments like antibiotics would also improve our understanding of IgRT, its use and access. While the prophylactic benefits of IgRT in HM have been discussed by Raanani et al., 6 more recent systematic reviews and meta-analyses would also be beneficial to examine IgRT clinical benefit in specific HM to reflect current practice.
Finally, the newer efficient cancer-targeting therapies are increasing the survival rate of patients with HM, which is likely to impact the clinical need for IgRT. The analysis included in this report is aimed to set the stage for more studies to address the global access and appropriate use of IgRT in HM. HCPs, regulators, payers and policy-makers should realise that this is an ever-growing and ever-changing field of medicine and that the prevalence of HGG in HM is only likely to increase. Hence, now is the time to invest resources into better understanding IVIg use, so that patients can continue to access IgRT as a supportive cancer treatment in HM. The ultimate goal is to achieve that elusive ‘gold standard’ of treatment and continuity of access to IgRT for all patients.
