Abstract
The traditional ST-segment elevation myocardial infarction (STEMI) paradigm has directed the identification and treatment of acute coronary occlusion (ACO) in the in-hospital and out-of-hospital (OOH) environment. Growing evidence suggests that it fails to identify a substantial proportion of ACO cases, leading to delays in intervention and missed treatment opportunities. The occlusion myocardial infarction (OMI) paradigm has emerged as a more inclusive diagnostic framework, emphasising the recognition of ACO regardless of ST-segment elevation. While OMI offers a more accurate classification, its adoption in OOH settings presents challenges, particularly due to the complexity of ECG interpretation and variability in clinician expertise. Artificial intelligence (AI) has the potential to address these limitations by providing real-time ECG analysis, improving diagnostic accuracy, and reducing inter-rater variability. AI-driven models, particularly artificial neural networks, have demonstrated superior sensitivity in detecting ACO compared to clinician interpretation using the traditional STEMI criteria, with the ability to identify occlusion earlier and more consistently. Additionally, AI integration with telemedicine could facilitate remote expert consultation, ensuring timely decision-making, particularly in resource-limited settings. By enhancing diagnostic accuracy and enabling earlier intervention, AI has the potential to improve prehospital cardiac care, and ultimately, patient outcomes. Future research should focus on optimising AI models, integrating them into OOH workflows, and validating their real-world effectiveness in acute coronary syndrome management.
Introduction
Acute myocardial infarction (AMI), most often caused by acute coronary occlusion (ACO), remains a leading cause of morbidity and mortality worldwide, with ischaemic heart disease causing nearly 9 million deaths in 2021. 1 To achieve optimal outcomes, rapid identification and management are required, with delayed or missed diagnosis leading to poorer outcomes.2–4 Due to limited diagnostic tools, out-of-hospital (OOH) identification of ACO is traditionally based on the presence of ST-segment elevation myocardial infarction (STEMI) criteria on the electrocardiogram (ECG). Recently, there has been a growing voice to expand the diagnostic criteria for ACO beyond the STEMI criteria, 5 with occlusion myocardial infarction (OMI) proposed as a potential replacement. 6 Often as first medical contact (FMC), 7 the introduction of OMI as a diagnostic category has significant implications for OOH care, where early and accurate diagnosis is important for determining management, including hospital destination decisions and reperfusion pathways. Although it is difficult to precisely quantify the population-level impact of the OMI introduction, the potential to reclassify high-risk patients earlier offers a significant opportunity to improve care delivery. The challenge in identifying OMI on the ECG lies in its complexity, with often subtle ECG characteristics, underscoring the potential of artificial intelligence (AI) as a supportive tool. In this paper, we suggest that AI-assisted ECG interpretation is the next step towards addressing the diagnostic limitations of the STEMI paradigm in OOH care.
Shifting paradigms from STEMI to OMI
ECG identification of ACO has evolved significantly since the early 20th century, when Harold Pardee first identified ST-Elevation (STE) in a patient with chest pain, 8 laying the groundwork for subsequent advances. The introduction of the STEMI criteria in 2000 9 was driven by trials demonstrating the mortality benefit of fibrinolytics in suspected AMI patients with STE. 10 This represented a significant improvement over the preceding Q-wave/non-Q-wave MI classification, with the STEMI criteria being used as a surrogate for ACO. However, despite adjustments for age and sex, the STEMI criteria have significant limitations and will miss a significant minority of ACO. 11 By definition, the STEMI criteria focus on one aspect of the ECG to identify ACO: the presence of STE. This leads clinicians to ask the incorrect question, “is it a STEMI?.” Rather, the correct question should be “is there an ACO?.”
This isolated focus on STE leads to several ECG characteristics of ACO being ignored, including hyperacute T-waves, 12 subtle reciprocal findings, down-up T-waves, inverted reciprocal hyperacute T-waves, ST depression in V1-V4 13 and terminal QRS distortion. 14 These and other ECG features are key to identifying ACO under the OMI paradigm. 15 The STEMI criteria also ignore the concept of proportionality, which involves evaluating whether the ST segment or T wave is appropriately scaled to the overall size of the QRS complex that it follows. Proportionality plays an important role in detecting ACO in the setting of abnormal repolarisation patterns, such as left bundle branch block, 16 differentiating anterior myocardial infarction from left ventricular aneurysm, 17 and distinguishing benign early repolarisation from left anterior descending (LAD) occlusion. 18 A recent meta-analysis reported a sensitivity of 43% for the diagnosis of ACO using STEMI criteria. 19 Smaller studies also reported STEMI criteria-associated false positive rates of up to 36% for physicians,20,21 and this inconsistency extends to paramedics,22–28 while false positives remain a concern due to artefact and non-cardiac causes of STE, false negatives pose a quieter challenge. Importantly, false-negative cases often go unrecognised in the prehospital setting, limiting opportunities for feedback and system-level quality improvement. Despite this, the simplicity and historical precedence of the criteria have sustained its widespread use in clinical practice.
Studies have demonstrated that 25% to 34% of NSTEMI patients have an ACO, either TIMI 0 or 1 flow on angiogram,29,30 and have higher in-hospital and long-term mortality compared to NSTEMIs with no ACO.31,32 Moreover, NSTEMI patients with confirmed ACO demonstrate similar infarct size, troponin levels and angiographic findings to patients with STEMI, but experience longer times to angiography. 33 This suggests that the NSTEMI cohort comprises two groups: those with occlusion (STEMI-negative OMI) and those without (non-occlusive MI).
Current international guidelines recommend an early invasive strategy (<2 h) for very-high-risk NSTEMI patients, many of whom display ECG features that align with those included in the OMI paradigm.34–36 However, one study found that only 6% of such patients received treatment within the recommended timeframes, 37 highlighting a potential focus for improving the delivery of patient care. Although this single-country finding may not represent all practice settings, it highlights an opportunity to improve the timely identification and management of ACO in the OOH setting, given the known benefits of prehospital early notification.38–40
Research has shown that applying the OMI paradigm ECG analysis could reclassify up to 30% of STEMI-negative patients with ACO, 31 and that OMI identification may complement, rather than replace, current practice. Importantly, identifying OMI in the OOH setting is not intended to dictate in-hospital treatment decisions but rather to support the early recognition of patients who may benefit from urgent intervention, including direct transport to PCI-capable centres.
OMI represents a more refined and nuanced approach to ACO identification, an extension of the STEMI criteria, with the aim of improving diagnostic accuracy. The definition of OMI in the literature uses thrombolysis in myocardial infarction (TIMI) flow grades of 0–2 or TIMI 3 flow accompanied by markedly elevated cardiac biomarkers, typically, high-sensitivity troponin T levels greater than 1000 ng/L (reference range <14 ng/L), or high-sensitivity troponin I levels greater than 5000-10,000 ng/L (∼>10.0 ng/mL with 4th-generation assays). 6 Incorporating TIMI 3 with high troponin aims to capture patients with significant and prolonged ACO to cause myocardial injury with spontaneous reperfusion after the ECG is recorded, but before PCI is performed. OMI diagnosis in-hospital uses a combination of ECG, troponin, echocardiographic, and angiographic data. However, in the OOH setting, only the ECG is available in real time. While the ECG may not provide the diagnostic accuracy of multimodal testing that would occur in-hospital, expert ECG interpretation using predefined OMI criteria has been shown to detect ACO an average of three hours earlier than STEMI criteria. 41 In this context, the ECG remains the most rapid and practical tool for guiding decisions in the OOH environment.
The challenges of paramedic ECG interpretation under the OMI paradigm
Education challenges
Despite the potential of OMI ECG characteristics to improve ACO identification, consistent and accurate ECG interpretation remains a barrier. In contrast to the uniform ECG changes often seen in STEMI, recognising ACO changes under the OMI paradigm is significantly different. 15 Recognising subtle ECG features makes interpretation nuanced and demanding, and these subtle changes may go unnoticed, particularly in unpredictable OOH environments, where clinicians must make rapid decisions under pressure.
The challenge is compounded by the fact that these ECG patterns rely on a different mindset than counting millimetres of STE and require significant initial and ongoing education. While paramedics receive training in ECG interpretation, detecting nuanced changes under OMI demands a depth of expertise that may not be fully developed, especially among junior clinicians or those working in rural or remote areas with limited exposure. When accounting for the financial costs of training, combined with the potential that clinicians may still not be proficient, the introduction of OMI across larger ambulance services may prove to be impractical and cost prohibitive.
Cognitive load and inter-rater reliability
Time pressure and cognitive load in the OOH environment exacerbate the challenges of ECG interpretation. The urgency of ACO treatment requires clinicians to rapidly assess patients and initiate care, often prioritising STE 42 while potentially overlooking more subtle indicators. Even among experienced paramedics, STEMI identification remains subjective, with variability influenced by fatigue, stress and experience, leading to poor interrater reliability. This is well recognised across medical specialties,20,43 and may be more pronounced under the OMI paradigm. While interpretation by those familiar with OMI ECG characteristics has shown high levels of agreement, 41 reliance on expert interpretation in the literature limits generalisability and poses a significant challenge to widespread implementation. Without an objective or automated diagnostic aid, subjectivity increases the risk of over- and under-triaging of patients, potentially impacting patient outcomes. In this setting, ECG interpretation should be supported by clinical features readily available to paramedics, such as ongoing ischaemic chest pain, haemodynamic instability, or signs of myocardial irritability, all of which increase the pre-test probability of occlusion.
Systems-based challenges
A significant challenge in adopting the OMI paradigm is its limited acceptance and implementation in the in-hospital environment. STEMI remains the dominant framework guiding ACO management, and if prehospital clinicians are using OMI in isolation, their management may not align with in-hospital care pathways. This misalignment could lead to inconsistencies in patient care, delays in treatment, and difficulties in securing timely access to reperfusion therapy.
Despite these challenges, there is still a potential benefit to OOH adoption of OMI. Unlike STEMI, OMI identification in hospitals is not solely an ECG diagnosis but incorporates other diagnostic tools such as troponin, echocardiography, and angiography. Paramedic recognition of OMI could help guide transport decisions to PCI facilities, where further testing could confirm the diagnosis. By identifying patients with potential ACO, paramedics can prioritise transport to a hospital with PCI capabilities rather than non-PCI, reducing secondary transfers and minimising treatment delays. 44 While full alignment between out OOH and in-hospital systems is ideal, implementing OMI in OOH care may still improve patient outcomes by facilitating improved triage and ensuring that high-risk patients receive rapid, in-hospital assessment. This decision can be further informed by clinical indicators of high-risk presentation, which may support transport to PCI-capable centres even when classic ECG findings are absent.
The role of AI in ACO identification
Given the challenges of human-based OMI ECG identification, there is potential for AI to enhance diagnostic precision and use as a decision-support tool. 45 AI can assist clinicians by providing real-time ECG interpretation, identifying subtle ischaemic changes and reducing inter-rater variability. Machine learning algorithms, particularly deep learning models such as artificial neural networks (ANNs), have been demonstrated to be at least non-inferior to physician comparators and potentially superior, regardless of the specialty or years of experience, 46 making them promising for OOH use.
AI in ECG interpretation
ECG interpretation is largely based on pattern recognition, making it well-suited for AI applications. Conventional ECG software uses predefined rules to interpret waveforms, whereas AI models learn from large datasets and can detect complex, non-linear relationships that may not be immediately obvious to human interpreters. 47 This is particularly relevant in the context of OMI, where ECG changes may be subtle. Different AI architectures offer unique strengths in ECG analysis. Convolutional neural networks are effective at recognising visual features such as wave shape and amplitude. 48 Recurrent neural networks, including long short-term memory variants, 49 track how ECG signals evolve over time. More recently, transformer-based models have shown promise by detecting patterns within specific leads and across the overall ECG, allowing for more accurate and nuanced interpretation. 50
Recent studies have highlighted the potential of ANNs in the detection of ACO. Herman et al. identified ACO nearly three hours earlier than the STEMI criteria, with comparable specificity (93.7% vs 97.7% for STEMI), but superior sensitivity (80.6% vs 32.5%) and AUC (0.938 vs 0.651). 51 Similarly, Al-Zaiti et al. 52 developed an ANN model to identify STEMI-negative OMI on OOH ECGs, outperforming commercial ECG interpretation algorithms. When combined with clinician judgement, the model correctly reclassified one-third of the patients. In addition, AI has been shown to have increased ability to detect LAD occlusion on the initial ECG, when compared to serial ECGs using the STEMI criteria, 53 as well as superior accuracy in catheterisation laboratory activation when compared to emergency department physicians and cardiologists. 54
One of AI's most compelling advantages is its ability to continuously improve over time. Unlike human clinicians, who require formal retraining to enhance their ECG interpretation skills, AI models can be refined continuously through exposure to new data. This allows the AI to adapt to the emerging ECG criteria and enhance its diagnostic abilities.
AI integration
Another advantage of AI is its ability to provide consistent and standardised analysis. Unlike human interpretation, which can be influenced by factors such as fatigue, stress or experience, AI delivers objective assessments. This standardisation minimises variability in ECG interpretation across different paramedics, agencies and regions, ultimately leading to more reliable and equitable patient care. Additionally, AI has the potential to facilitate telemedicine and remote consultation. By integrating AI analysis with telemedicine systems, clinicians can transmit ECGs to a central location where AI-based interpretation can be reviewed alongside expert consultation. This approach could be particularly valuable in regions with limited access to specialist cardiology input, ensuring that all patients receive the highest level of diagnostic support.
If validated in prospective studies, AI tools that can identify OMI earlier than standard ECG interpretation could help expedite intervention and shorten reperfusion times. Whether through prehospital thrombolysis or direct transport to a PCI centre, early recognition of OMI has a potential role in minimising myocardial damage and improving long-term outcomes. By integrating AI into OOH care, the potential exists to enhance ACO management and ensure that more patients receive timely and appropriate treatment. Not only can AI-based decision aids be used for earlier notification to hospitals, but they can also be used to reduce the amount of false-positive hospital notifications, leading to better utilisation of limited ambulance resources. 55
Despite the potential, it should be noted that many studies into OMI and AI tools are retrospective and often limited to single-centre data, which may not generalise well to other populations. Prospective multicentre validation trials have commenced 56 and the results will be crucial in understanding how best to support broader implementation and foster clinician trust.
Regulation and ethics
Important regulatory, ethical, and medico-legal issues must be considered in this context. As AI ECG systems are considered medical devices, they must undergo rigorous validation and regulatory approval before being used in practice. In Australia, the Therapeutic Goods Administration (TGA) requires risk classification and listing. In the United States, the Food and Drug Administration (FDA) assesses them through specific pathways, and in Europe, a CE Mark under medical device laws is required. All three frameworks require rigorous validation, continuous evaluation, and clearly defined clinician oversight. The use of AI tools is legally complex, and it is essential that regulation keeps pace with the rapid evolution of AI models. 57
While AI models offer several advantages, such as objectivity, resistance to fatigue, and the ability to be retrained, they should not be viewed as replacements for clinical expertise. Consistent with the FUTURE-AI international consensus framework, 58 AI should be positioned as a complementary tool to support clinicians by highlighting high-risk ECGs. This framework also emphasises the importance of clearly defining clinical responsibilities, ensuring that ultimate decision-making remains with human clinicians. However, safe deployment requires clear clinical governance structures with defined oversight, accountability and appropriate clinician engagement. This approach helps ensure that AI augments rather than undermines decision-making, balancing innovation with patient safety and professional trust.
Clinician trust
Another barrier is clinician trust and usability of AI-based tools. While commercial interpretation algorithms for STEMI have benefits, there are several well-known limitations, 59 with false-positive STEMI activations often a result of ECG artefact or non-ischaemic causes of chest pain 60 which results in a lack of trust. AI adoption success will require clinicians to find AI systems reliable, interpretable, and easy to integrate into their workflows. If AI outputs lack transparency with unclear reasoning or frequently contradict clinical judgment, clinicians may be hesitant to rely on them. 61 Addressing this requires user-friendly interfaces, clear decision explanations and opportunities for paramedics to engage with AI tools during training. One of the benefits of AI-based tools in ECG analysis is the use of Explainable AI (XAI), specifically saliency maps (Figure 1). Saliency maps are common in AI-based interpretation, and in this setting, they can show paramedics which features of the ECG contributed most to the AI model's decision, ensuring transparency. The algorithm overlays colour on the tracing to highlight which features of the ECG most influenced its verdict. By revealing the rationale behind the output, XAI allows clinicians to verify that the model focuses on clinically meaningful features, thereby improving trust. In the paramedic context, saliency maps and other XAI features can also serve an educational role by helping clinicians understand which ECG features indicate occlusion and reinforce learning through real-time feedback in the field or during training scenarios. AI should be positioned as a support tool that enhances rather than replaces clinical expertise. Ensuring that AI aligns with OOH care workflows and reinforces ECG interpretation skills will be key to increasing trust and successful implementation.
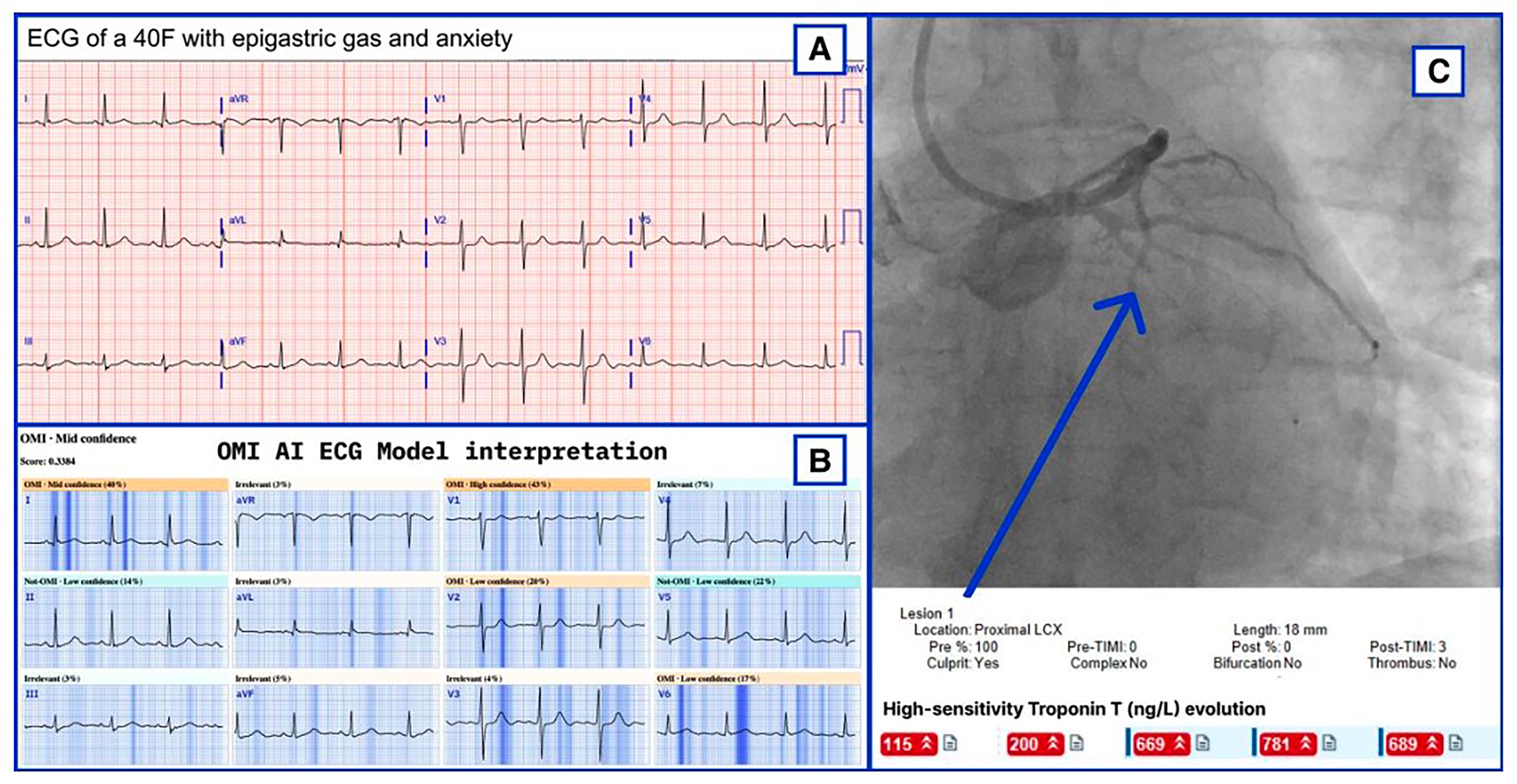
Example of a saliency map generated by the PMcardio AI model from Herman et al. 51 Darker blue areas on the ECG highlight the most influential to the model classification of OMI.
Practical limitations
Despite the potential benefits of AI-based ECG interpretation, several challenges remain before it can be widely deployed. AI interpretation tools have already been packaged as smartphone apps, 51 allowing the algorithm to run locally on a device that many crews already carry. A parallel approach is to embed the model directly into next-generation cardiac monitors. Both approaches may be reasonable, but each requires rigorous validation on the chosen platform and in real-world ambulance conditions. Even when inference occurs on the handset, most app or cloud-based workflows still depend on a short burst of mobile data to upload the tracing or return the result. Coverage gaps are common in rural settings, tunnels and high-rise areas and can delay or interrupt analysis and limit the benefit of early AI support. Appropriate digitisation of the ECG is essential for AI interpretation. 62 Ambulance motion, vibration, CPR and electrical noise introduce artefact that can degrade model accuracy, and robust preprocessing and noise-tolerant training data are essential. Deploying AI on a large scale also requires licence fees, device upgrades, data plans and staff training, with cost-effectiveness studies needed to justify the expenditure. For AI to be truly feasible in OOH care, the model must either run reliably on a mobile app platform or be fully integrated into the ECG monitor or defibrillator ecosystem while remaining accurate, fast and affordable, even when connectivity is suboptimal.
Implications for out-of-hospital care and future directions
The shift from STEMI to OMI represents a critical advancement in OOH cardiac care. However, subtle and variable ECG findings present diagnostic challenges that require more advanced and consistent tools. Several priority areas have been identified to guide research and system improvement (Table 1).
Future directions for the OMI paradigm in OOH care.

Looking ahead, AI in ECG interpretation integration will likely be required if the OMI paradigm is broadly adopted. Currently, there are 12 different ECG criteria that fall under OMI,15,63 with research ongoing. As OMI ECG characteristics increase in quantity and complexity, the ability of paramedics to learn and maintain competency will drastically decrease. AI offers a potential solution for supporting consistent and accurate ECG interpretation, especially in rural or resource-limited environments.
Conclusion
A key limitation of the STEMI paradigm is that it encourages clinicians to ask the wrong question, “does the patient have a STEMI?” instead of the more clinically relevant “does the patient have an acute coronary occlusion?” The shift to the OMI framework aims to improve the early identification of ACO, particularly in OOH settings where paramedics are the FMC. AI-supported ECG interpretation may help bridge this gap by providing decision support for clinicians, improving sensitivity and reducing diagnostic variability. The implementation of AI requires overcoming challenges in algorithm validation, paramedic education and system integration. Although early studies are promising, prospective data confirming improved patient outcomes with AI-guided OMI recognition are not yet available. If these challenges are addressed, AI-guided identification of OMI may assist with improved triage, shorten treatment delays and improve patient outcomes; however, prospective evidence is still needed.
Footnotes
Author contribution(s)
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
