Abstract
Objective
To describe changes observed in case characteristics and patient outcomes before and after ambulance service participation in a ‘Leave Behind’ take-home naloxone (THN) dispensing/distribution program.
Methods
This before-and-after study included emergency ambulance attendances for patients experiencing an opioid overdose one year before 1 October 2021 (pre-participation phase: 1 October 2020–30 September 2021) when St John Western Australia (SJWA) participated in the THN program and those one year after the date (post-participation phase: 1 October 2021–30 September 2022).
Results
There were 945 and 1240 opioid overdoses among 221,451 and 222,480 emergency ambulance attendances in the pre- and post-participation phases (0.43% vs 0.56%, p < 0.001). The number of ambulance attendances for overdose in the post-participation phase was not significantly different from that predicted based on the ambulance attendances in the pre-participation phase. No statistically significant differences in patient age, sex, and geographical location were identified between the two phases. Compared to the pre-participation phase, more patients had naloxone administered prior to ambulance arrival (10.7% vs 15.1%, p = 0.003), and more patients were discharged at the scene (21.2% vs 29.8%, p < 0.001) in the post-participation phase. No difference was found in mortality within one day (3.4% in the pre-participation phase vs 3.5% in the post-participation phase, p = 0.30).
Conclusions
After implementation of a ‘Leave Behind’ take-home naloxone dispensing/distribution program, more patients had naloxone administered prior to ambulance arrival, and the likelihood of discharging a patient at the scene significantly increased without affecting patient survival rates.
Introduction
Opioid overdose rates have been increasing steadily in many countries over the last decade and remain a public health concern around the world. 1 Fatalities due to opioid use in Australia more than doubled between 2002 and 2019, increasing from 2.5 fatalities per 100,000 individuals in 2002 to 5.0 fatalities per 100,000 individuals in 2019. 2 Not surprisingly, ambulance attendance for opioid (mostly heroin and pharmaceutical opioids) overdose has also been reported to have increased by some ambulance services in Australia. However, many opioid users refuse to be transported to a hospital for further treatment when experiencing an opioid overdose, 3 despite patient transport refusal being associated with higher risks of future emergency services encounters due to a subsequent non-fatal opioid overdose. 4 Recent research in Western Australia (WA) suggests that the cost of ambulance transport can be a deterrent for consumers who are already marginalised and might prevent them from seeking additional support. 5
Naloxone is an opioid antagonist that can reverse the effects of an opioid overdose with minimal adverse side effects.6,7 It is widely regarded as a ‘safe, effective, and easy-to-administer drug’ 8 and its wider distribution has been recommended by the World Health Organization to help reduce opioid-related deaths. 9 Recent research conducted in Australia and internationally suggests that THN programs are effective in increasing knowledge of drug users and peers about risk factors and management of opioid overdose and can reduce fatalities.10,11
‘Leave Behind’ naloxone programs allow emergency medical services (EMS), including paramedics/ emergency medical technicians to distribute naloxone kits to people who are at risk of further opioid overdose, as well as their family members or friends, to help manage subsequent overdose events. 3 Prior to the introduction of these programs, such patients might have been previously left on the scene without any additional help. 3 It has been estimated that a large proportion of people who might be at risk of an opioid overdose do not engage with alcohol and drug services. As a result, leave-behind naloxone programs have been implemented internationally,3,12–15 in an effort to reach consumers at higher risk of overdose. 15
Emergency medical ‘Leave Behind’ naloxone programs seem to be effective in increasing the availability of naloxone in urban settings, with relatively low barriers and are considered ‘a promising method to help save lives and reduce future overdose events’. 13 Recent research suggests that ‘Leave Behind’ naloxone programs can also play an important role in connecting vulnerable community members to follow up services and emphasise the role of the patients’ family members and social network in doing so.13,16 A recent study aiming to analyse the outcomes of an emergency medical service ‘Leave Behind’ naloxone program in the United States of America (USA), found that naloxone kits had been distributed to approximately 50% of the people who were present at the scene of an opioid overdose between June 2018 and June 2019. 16 The majority of patients who had experienced an opioid overdose were admitted to hospital (82%), while transport to hospital was refused by 18% of patients. 16 Sixty-one percent of these patients had accepted a naloxone kit on scene. 16 The naloxone kits had been provided to family members (48%), patients themselves (37%), or a friend who was present (14%). 16 Patients whose naloxone kit had been distributed to their family members or a friend were respectively 5.16 times and 3.69 times more likely to be connected to a peer support specialist, compared to patients who did not accept the kit. 16
In Australia, THN distribution and training programs have been in place in various cities since 2012 17 ; to help prevent overdose deaths by training potential overdose witnesses to respond to overdose situations. Naloxone has been available over the counter in Australia since 2016 without prescription and as a pre-filled syringe, 18 while naloxone nasal spray (Nyxoid®) was listed as a subsidised medicine in November 2019. 19 Following a successful THN pilot program that provided free naloxone in various settings (e.g., participating pharmacies, approved medical practitioners, alcohol and drug treatment centres, needle and syringe programs, hospital emergency departments and other health centres) in several Australian states, from the 1st of December 2019 to the 30th of June 2022, 10 the program was expanded to provide naloxone at no cost across the country from July 2022. 20 Coincident with the THN pilot program, the supply of naloxone in various settings continued to expand in Australia. 10 This included settings which were not subsidised by the pilot funding, including the current St John Western Australia (SJWA) ‘Leave Behind’ take-home naloxone dispensing/distribution program 5 and the WA Police Force Naloxone Pilot, whereby trained police could carry and administer naloxone to opioid overdose victims. 21
As of 1 October 2021, SJWA enabled its paramedics, ambulance officers, and volunteer emergency medical technicians throughout WA to dispense/distribute intranasal THN (Nyxoid®) to patients who had experienced a suspected opioid overdose, or bystanders who had witnessed an opioid overdose. While this capability had been previously available to SJWA Clinical Support Paramedics (CSPs) from August 2021, CSP coverage is confined to a smaller geographical area of the major city of Perth, Western Australia. The intended primary purpose of SJWA dispensing THN was to provide a safety-net for opioid overdose patients who refused transport to a hospital after being administered naloxone by SJWA or bystanders. This safety-net relates to the short half-life (30–90 min) of naloxone, 22 whereby the initial dose(s) may wear off, with a risk of respiratory depression unless additional naloxone is administered. In addition, if not used for the patient's current overdose, SJWA's dispensing of THN may also be beneficial in being available for future overdoses.
Study objectives
The study objectives were: (1) to describe the number of ambulance attendances for opioid overdose, case characteristics and patient outcomes, before and after the introduction of THN by SJWA on 1 October 2021; (2) to compare characteristics and outcomes between cases where THN was and was not dispensed in the post-participation phase and (3) to explore reasons why ambulance personnel did not dispense THN for opioid overdose patients who were discharged at the scene in the post-participation phase.
Because the COVID-19 pandemic overlapped with the study periods, the association between the number of COVID cases and the occurrence of opioid overdose was examined.
Methods
Study setting
This before-and-after study was conducted in WA, geographically the largest state of Australia, with 2.5 million square kilometres of land area and a population of 2.6 million in 2021. 23 The majority of the population (80%) lives in the Perth metropolitan area, which accounts for only 0.2% of the landmass of WA.
St John WA provides a state-wide emergency ambulance service, accessed through the national emergency phone number ‘000’. In the metropolitan area, ambulances are staffed by registered paramedics and ambulance officers (student paramedics). In rural areas, ambulance crew composition can include a mix of registered ambulance paramedics and volunteer emergency medical technicians, or volunteer-only crews.
Intervention
The ‘Take-Home-Naloxone’ (Nyxoid ®) nasal spray consisted of one spray (100 µL); with each dose of 100 µL containing 1.8 mg naloxone. The total dispensed amount was 3.6 mg (2 × 1.8 mg Sprays) in a Box with Instructions. 24 The SJWA Clinical Practice Guidelines 24 stated that for patients who have required Naloxone, every attempt should be made to convince the ‘at risk’ patient to be transported to a healthcare facility. Where such attempts proved futile, IN Naloxone – Nyxoid® could be left with the patient. This was to avoid the risk of opioid related respiratory depression due to the short half-life of Naloxone. In such circumstances ambulance personnel were required to document leaving Nyxoid® with the person or a responsible other person in the electronic patient care records (ePCR). 24
Data sources
Data were extracted from the SJWA electronic patient care record (ePCR). Ambulance personnel record patient characteristics and interventions provided in the ePCR using a tablet with specialised data entry software. The ePCR data was linked with the WA death registry to confirm the survival outcome of patients after ambulance attendance. For this linkage, probabilistic linkage software named Fril (version 2.1.5, Emory University and Center for Diseases Control and Prevention, Atlanta, Georgia, U.S.) was used, supplemented by Python Record Linkage Toolkit (version 0.14.0) using names, dates of birth, and residential addresses of patients as key identifiers. The performance of the Fril has elsewhere been reported to be 99% for precision (positive predictive value) and 95% for recall (sensitivity) to identify the same individuals. 25
Participants
All emergency ambulance attendances from 1 October 2020 to 30 September 2022 were identified, encompassing the 12 months before and 12 months after the date when THN became available for use by SJWA ambulance personnel throughout WA. The primary unit for reporting and analysis was each individual case of opioid presentation. Thus, the total number of cases could exceed the number of individuals due to repeat opioid presentations by individual patients. Among all emergency ambulance attendances, opioid overdose cases were identified in three ways, as follows: 1) attendances where ambulance personnel recorded in the ePCR that overdose was the reason that an ambulance was requested; 2) those cases where the use of naloxone was recorded; and/or 3) those cases where any of the keywords related to opioid overdose (e.g., opioid, opiate, heroin, IVDU (i.e., intravenous drug use), naloxone, Nyxoid, Narcan) were documented in the free-text in ePCR. The true opioid overdose cases were then selected by reading free-text information in the ePCR, in which ambulance personnel documented the patient's history, complaints, conditions, outcomes and clinical interventions. This screening process was performed by one of the authors (HT), who is a specialist in emergency medicine with extensive research experience in emergency medicine/prehospital care and significant familiarity with SJWA ePCR data. Non-emergency attendances were excluded, such as interhospital transfers, booked transports, cases where a patient absconded before EMS engagement, and where ambulance personnel could not locate a patient. Finally, all opioid overdose cases were classified into two groups according to the phase of this study: the pre-participation phase (1 October 2020 to 30 September 2021) and the post-participation phase (1 October 2021 to 30 September 2022).
Variables
The following information was extracted from the linked data: patient age, sex, date of ambulance request, geographical location (metropolitan or rural area), dispatch urgency as determined by ambulance dispatcher, ranging from 1 to 3 (1 for most urgent and 3 for least urgent), transport urgency determined by attending ambulance officers ranging from 1 to 5 (1 for most urgent and 5 for least urgent), scene interval (time from arrival at scene to departure from the scene), transport interval (time interval from departure from scene to arrival at hospital), mission interval (time from a call for an ambulance to end of attendance), police attendance, administration of naloxone prior to and after SJWA arrival, type of person who administered naloxone prior to SJWA arrival (friend/bystander, family member, health professional, patient self, police/security, or unknown), dispense of take-home naloxone by ambulance personnel, disposition from the scene (discharge at the scene or transport to a hospital), re-attendances of an ambulance within 24 h after the end of attendance, mortality within one day after the end of ambulance attendance. For cases where THN was not dispensed among those discharged at the scene in the post-participation phase, the reasons why THN was not dispensed were explored by reviewing the free-text variable in the ePCR. When patients had multiple ambulance attendance for opioid overdose, the information above was collected separately for each incident. For example, if a patient's age changed between one case of opioid presentation and another, a different age was recorded for the two cases.
Statistical analysis
The variables above were compared between the pre- and post-participation phases using a chi-square test or Fisher's exact test for categorical variables, a Mann-Whitney U-test for continuous variables if skewed, or a student's t-test if not skewed. The daily attendance for opioid overdose in the post-participation phase was predicted by a seasonal autoregressive integrated moving average (SARIMA) model using daily attendance in the pre-participation phase. 26 Using a grid-searching algorithm, hyperparameters of the SARIMA model that minimised the Akaike information criterion were selected. 27 The observed daily attendance in the post-participation phase was graphically compared with the 95% prediction interval, and the daily number of coronavirus disease (COVID-19) patients graphed for reference. Also compared were the above variables for attendances where THN was and was not dispensed among those discharged at the scene during the post-participation phase. Statistical significance was based on α=0.05. Python version 3.8.11 was used with the statistical libraries (statsmodels version 0.13.1 and Scipy version 1.7.3) for data management, time-series analysis and statistical tests.
Ethics approval
This study was approved by Curtin University Human Research Ethics Committee as a sub-study of HR 128/2013 and the SJWA Research Governance Committee.
Results
During the study period, 945 opioid overdose cases were identified among 221,451 emergency attendances (0.43%) in the pre-participation phase and 1240 opioid overdose cases among 222,480 attendances (0.56%) in the post-participation phases (p < 0.001). Daily attendance for opioid overdose fluctuated during the study period and had a slightly increasing trend (Figure 1). Predicted daily attendance in the post-participation phase also fluctuated (shaded area), and actual attendance in the period was within the predictions (Figure 1).
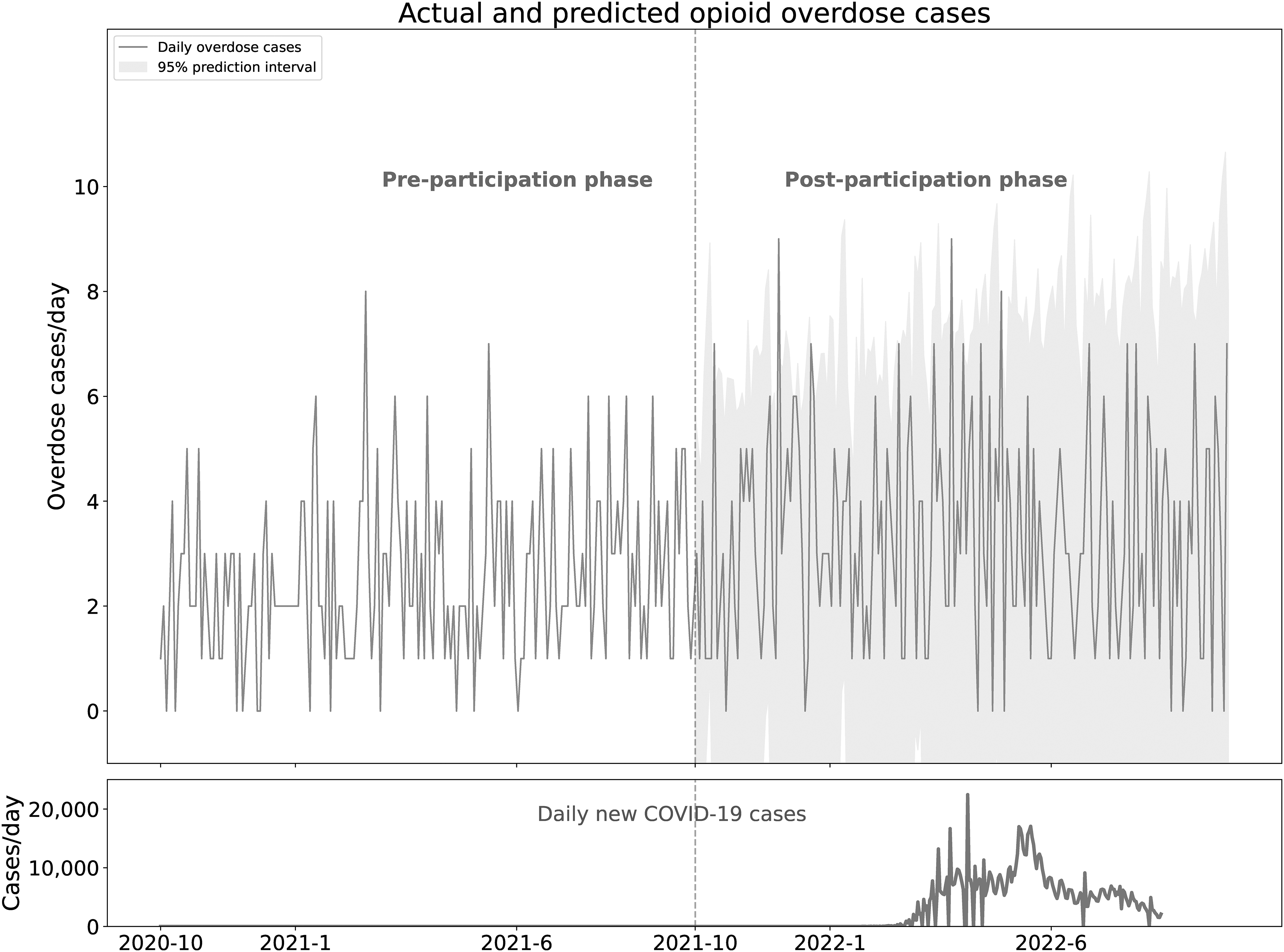
Daily attendance to opioid overdose cases. The grey area in the post-participation phase indicates the 95% prediction interval of daily attendance predicted by the seasonal autoregressive integrated moving average model using daily attendances in the pre-participation phase. Actual daily attendance (grey line) was mostly within the prediction interval.
Most characteristics did not differ significantly between the two phases (Table 1). The median age was 40.5 years (Interquartile range [IQR] 39.4–41.5 years) for the pre-participation phase and 41.0 years (IQR 40.1–41.9 years) post-participation (p = 0.49); males were predominant throughout (65.3% for the pre-participation phase; 66.8% post-participation phase, p = 0.50) (Table 1). There was a statistically significant difference in scene interval, with 25 min (IQR 17–38 min) pre-participation, compared to 30 min (IQR 19–47 min) post-participation (p = 0.001); and in mission interval, with 109 min (IQR 90–132 min) pre-participation, compared to 112 min (IQR 94–140 min) post-participation (p = 0.005) (Table 1).
Case characteristics in the pre- and post-participation phases
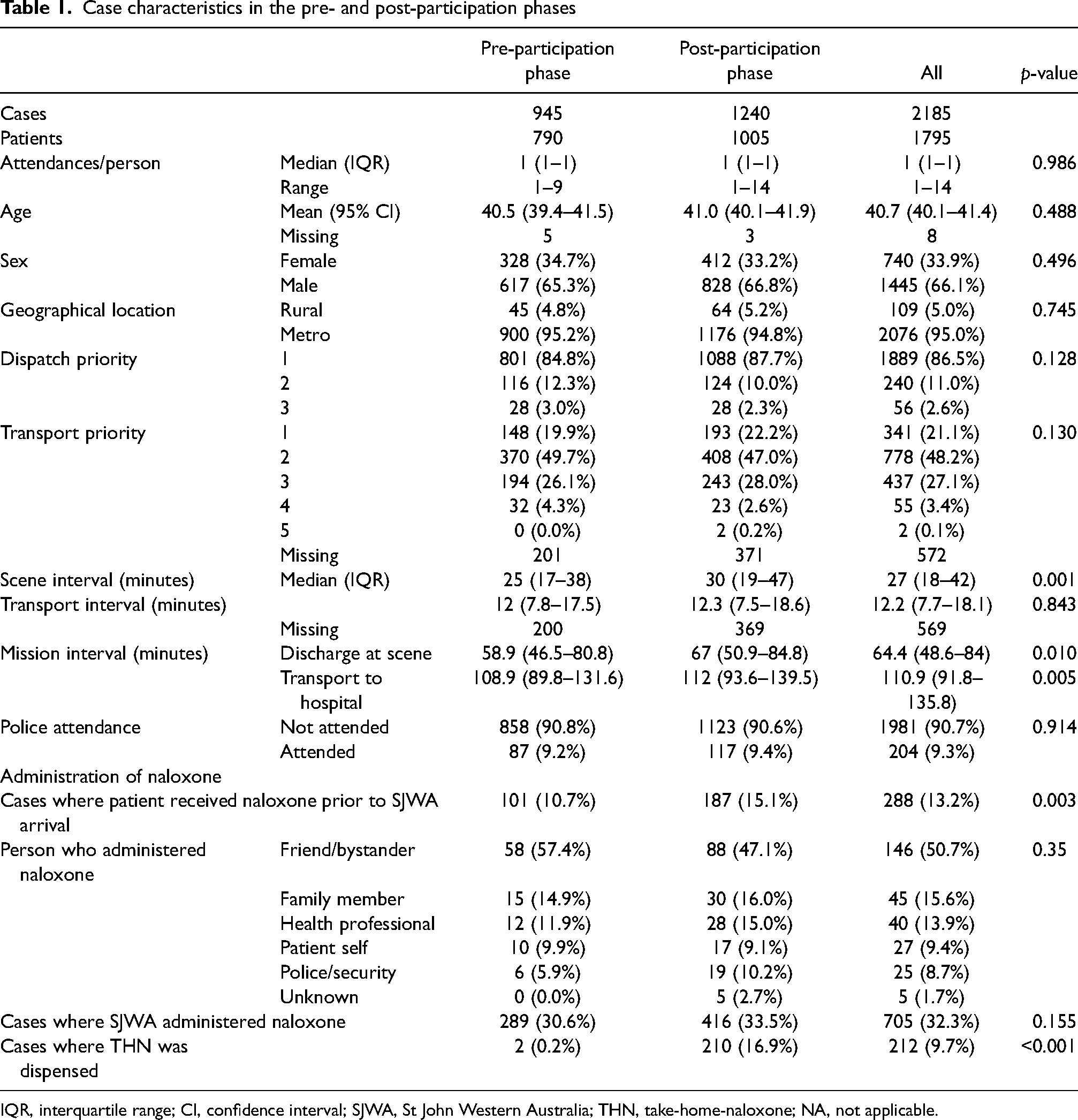
IQR, interquartile range; CI, confidence interval; SJWA, St John Western Australia; THN, take-home-naloxone; NA, not applicable.
The administration of naloxone prior to ambulance arrival was more likely in the post-participation phase (15.1%) than in the pre-participation (10.7%) (p = 0.003) (Table 1). Friends/bystanders most frequently administered naloxone in both phases (57.4% for the pre-participation phase and 47.1% for the post-participation phase) among cases where naloxone was given before EMS arrival (Table 1). On-scene time was significantly longer post-participation than pre-participation (median 30 min vs. 25 min, p < 0.001) (Table 1). Overall (for all pre-participation and post-participation cases combined) on-scene times were significantly longer for patients discharged at the scene (median 52 min, IQR 37–73) than for patients transported to hospital (24 min, 16–33) (p < 0.001).
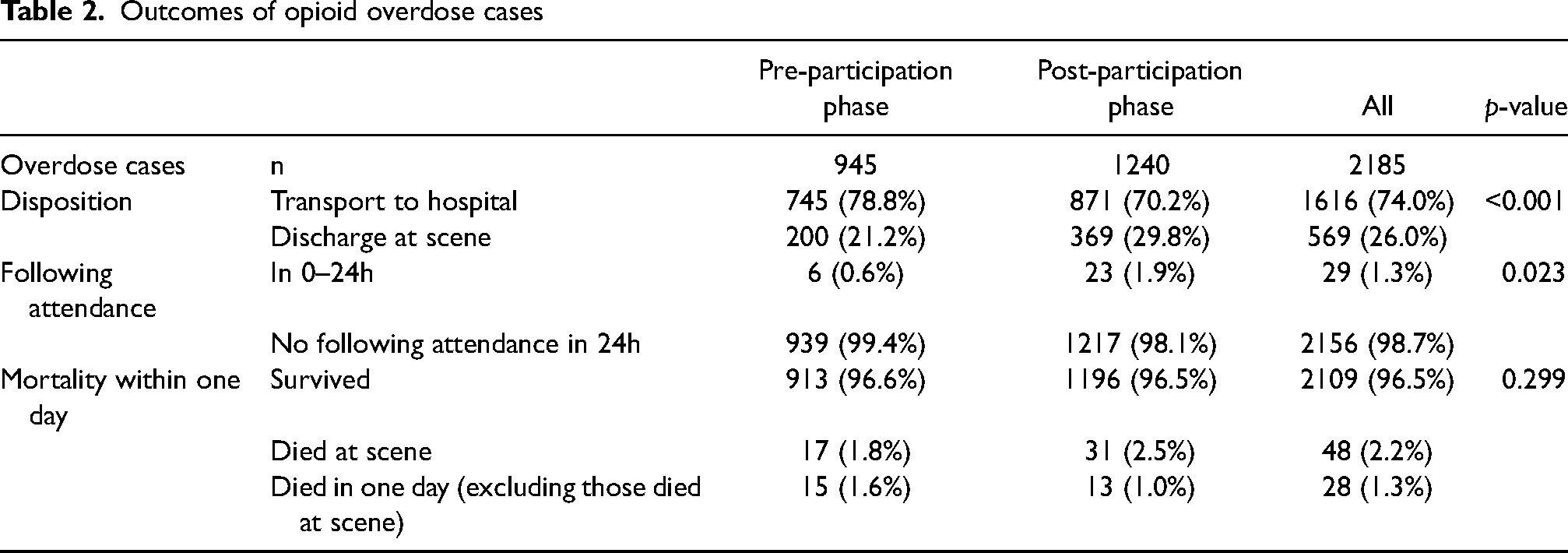
Patients were more likely to be discharged at the scene in the post-participation phase than in the pre-participation phase (21.2% pre-participation vs 29.8% post-participation, p < 0.001) (Table 2). More patients requested an ambulance within 24 h of the preceding opioid attendance in the post-participation phase than in the pre-participation phase (0.6% for the pre-participation phase vs 1.9% for the post-participation phase, p = 0.023) (Table 2). There was no statistically significant difference in one-day mortality rates (3.4% for the pre-participation phase vs 3.5% for the post-participation phase, p = 0.30) (Table 2).
Outcomes of opioid overdose cases
There were 369 cases where a patient was discharged at the scene during the post-participation phase (Table 3). Cases where THN was dispensed (n = 183/369) were more likely: to be in the metropolitan area (p = 0.04), have fewer police attendances (p < 0.001), have naloxone administered by ambulance officers (p < 0.001) and survive for at least one day (p < 0.001) than those where THN was not dispensed (n = 186/369) (Table 3). The reasons why ambulance personnel did not dispense take-home naloxone to the n = 155 patients discharged at the scene in the post-participation phase are shown in Table 4.
Characteristics and outcomes of cases where take-home naloxone was and was not dispensed in the post-participation phase
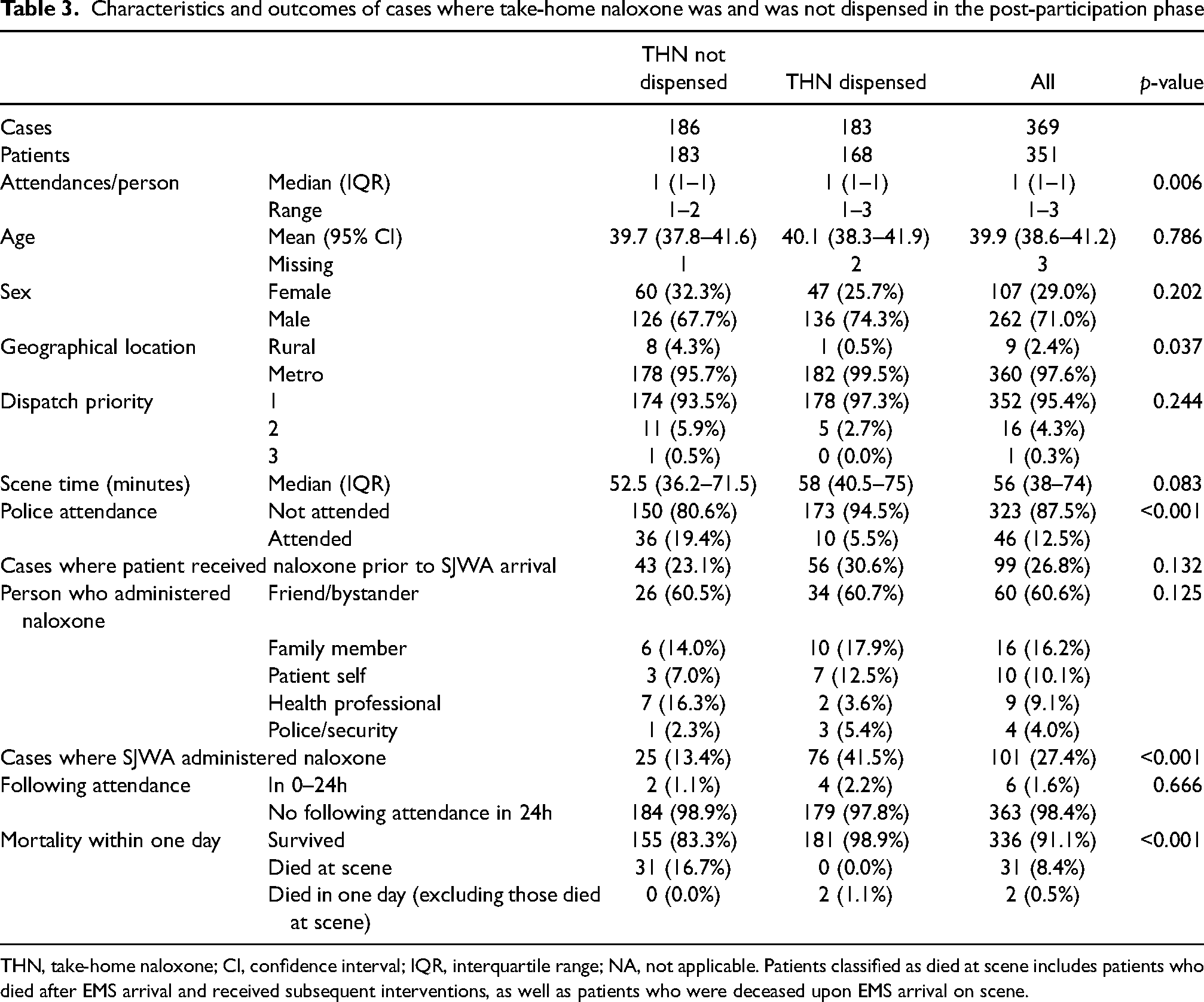
THN, take-home naloxone; CI, confidence interval; IQR, interquartile range; NA, not applicable. Patients classified as died at scene includes patients who died after EMS arrival and received subsequent interventions, as well as patients who were deceased upon EMS arrival on scene.
Discussion
This observational study described case and patient characteristics before and after the commencement of a ‘Leave Behind’ take-home naloxone dispensing/distribution program by ambulance personnel. The observed number of daily ambulance attendances for opioid overdose in the post-participation phase was not statistically significantly different from the number predicted from the underlying trend. Ambulance personnel spent longer on the scene and patients were more likely to be discharged at the scene in the post-participation phase than in the pre-participation phase, without an increase in mortality rate. Safe discharges at the scene mean less workload for the already crowded emergency departments (ED), 28 as well as enabling ambulance availability, at a time of increasing ambulance demand. 29
However, the commencement of THN provision by ambulance personnel did not change the number of call-outs for opioid overdose. This is consistent with results reported by McAuley et al. in Scotland. 30 They found that the national naloxone program, similar to the program in this study, did not have an impact on ambulance attendance to opioid overdose patients. These results are also consistent with other studies conducted in Australia, which found that the number of ambulance call-outs following the implementation of various THN programs was not statistically significant pre and post interventions.10,31 It is unclear why these THN programs have had no apparent impact on ambulance attendance. One possibility, is that the dispensing of naloxone via ambulance personnel may have had a modest coverage of the at-risk population, at least within the 12-month period post-intervention.
Another potential factor that could explain the lack of impact on ambulance attendance is the adherence to the guidelines received during naloxone training and/or brief education. In fact, many people who had received naloxone training and/or brief education in WA were instructed during the training to call an ambulance after administering naloxone and wait with the person until the ambulance arrives, to ensure the person does not overdose again and remains safe once naloxone wears off. 5 THN distribution and training programs have been available in Australia since 201217and seem to be fairly well-known among at the at-risk population. For example, a recent study conducted among a group of people who regularly inject drugs in WA (where the current study was conducted) found that eight in ten participants (83%) had heard about naloxone, nearly three-quarters (72%) had heard of THN programs, and 54% had been trained in naloxone administration. 32
Furthermore, other factors resulting from COVID-19, such as the restrictions on travel, border closure, and lockdowns might have had an impact on the supply of illicit drugs in Australia.33,34 For instance, during the period of 2020 and the first half of 2021, the availability and purity of heroin in the illicit drug market in WA declined, while its priced increased, 33 which might have led to reduced opioid overdose rates. A retrospective study analysing the impact of COVID-19 on the emergency department overdose presentations in WA found that there was a statistically significant reduction in the number of overdose presentations during COVID-19 compared to before the pandemic. 35 It was also found that there was a noticeable (but non-statistically significant) decrease in the number of people arriving by ambulance, from 88% pre-pandemic to 69% during COVID-19, and an increase in the number of patients arriving by other ways (e.g., private transport), from 12% pre-pandemic to 31% during COVID-19. 35 Further research is needed to determine the specific impacts on ambulance call outs following the implementation of a THN program.
Among cases where a patient was discharged at the scene in the post-participation phase, THN was dispensed in about half of the cases. In the other cases, refusal of treatment from ambulance personnel was the main reason why THN was not dispensed. These results are in line with a recent study analysing the outcomes of an emergency medical service ‘Leave Behind’ naloxone program in the USA. 16 Indeed, this study found that naloxone kits had been distributed to approximately 50% of the people who were present at the scene of an opioid overdose. 16 These people included the patient's family members (48%), followed by the patient themself (37%), a friend (14%), or to an unknown source (2%). In this study 82% patients were transported to the hospital, while 15% refused transport to hospital. 16
Bergstein et al. explored reasons for the refusal of transport by patients following opioid overdose. 36 They reported three main reasons for the refusal: the patients cannot tolerate withdrawal symptoms; the patients anticipate inadequate care at a hospital; and perceptions of stigma from ambulance personnel. 36 To increase the number of people who accept THN dispensations by EMS providers, ambulance personnel need to be cognisant of the fear of harsh withdrawal symptoms and communicate with patients more empathetically. 36 Even though transport to hospital seems the be the gold standard practice, previous research5suggests that the cost of ambulance transport in WA may actually serve as a major deterrent for some individuals. This is likely exacerbated for those in financial difficulties and struggling to meet their basic needs in daily life, and especially for those with existing outstanding debts for past ambulance transports. Further research should examine the specific reasons for the refusal of transport by people who might be at risk of opioid overdose.
In the current study, longer on-scene times were found in the post-participation phase than pre-participation phase (median of 30 min vs. 25 min). This may be attributable to the greater proportion of patients discharged on-scene post-participation than pre-participation (30% vs 21%), and the finding that patients discharged on scene had longer on-scene times than patients transported to hospital (median of 52 min vs. 24 min). When a patient expresses refusal of ambulance treatment or transport, ambulance personnel are required to assess the patient to determine if the patient is competent to make that decision. 37 If the patient is found to have the capacity to make the non-transport decision, the ambulance personnel must complete forms to record information given to the patient, including risks of non-transport, possible consequences after discharge, and advice as to what to do in case of any deterioration. Effort is made to obtain the signature of the patient, carer, bystander, and/or witness, to verify the content recorded on the forms. 37 These processes might have prolonged the stay on the scene in case of non-transport, resulting in longer on-scene intervals in the post-participation phase.
While it was found that the total number of attendances for opioid overdose was consistent with the number predicted from the underlying trend, the post-participation period had a significantly higher proportion of cases re-representing with an opioid overdose within the next day (1.9% of cases post-participation versus 0.6% pre-participation). The reasons for this are unclear. There was no evidence in the ePCR notes to suggest that the supply of naloxone from the previous presentation was a factor that promoted the subsequent overdose.
Limitations
This study has limitations related to the retrospective nature of the study design. The availability of hospital and emergency medical services may differ between the pre- and post-participation phases because the number of COVID-19 cases increased in the post-participation phase. We acknowledge that we cannot discount other unmeasured factors that may have changed over time, that could have potentially influenced the study results.
Another limitation was that it was not possible to accurately differentiate intentional versus unintentional overdose. As stated in the Australia's annual overdose report, 38 (p. 24) ‘There is no systematic definition to differentiate intentional from unintentional death, and coroners may not make a finding on intent for various reasons. Care should therefore be taken in interpreting figures relating to intentional self-harm’. However, it is likely that the majority of cases would have been unintentional drug overdoses. According to the recently published National Illicit Drugs Indicator Project (NIDIP) report 2 analysing trends in overdose and other drug-induced deaths in Australia, the majority of deaths caused by opioid overdose in 2021 were unintentional, making up 76% of the total number of deaths (763 deaths). On the other hand, 19% of deaths (196 deaths) were determined to be intentional, while 4.9% (49 deaths) had an undetermined intent. 2 According to the authors, this pattern has remained quite consistent over the years in Australia. 2
In the current study, a decision was made to focus on mortality within 1 day of the EMS attendance. The mortality outcome in overdose literature varies greatly among studies, ranging from 0 to 365 days. For example, Ashburn et al. (2019)
39
examined mortality up to one year after out-of-hospital naloxone administration encounters, including mortality at day 0,
Finally, the results of this study may not be generalisable to other EMS, with different staffing profiles and/or patient casemix.
Conclusions
Daily ambulance attendance rates for opioid overdoses did not differ between the pre- and post-participation phases, after the commencement of THN dispensation by ambulance personnel. The proportion of discharges at the scene increased, without any statistically significant increase in mortality of ambulance-attended overdose patients, either at the scene or within 24 h. The program may contribute to the decreased likelihood of transports to a hospital, resulting in improved availability of ambulances.
Reasons why ambulance personnel did not dispense take-home naloxone to patients discharged at the scene in the post-participation phase

Footnotes
Declaration of conflicting interests
RB, CN, and JB are employees of St John Western Australia (SJWA); DB was an employee of SJWA at the time of the study. JF and SB have adjunct research positions with SJWA; and JF receives research funding from SJWA.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
