Abstract
Background and Rationale:
Paired vagus nerve stimulation (VNS) has emerged as a safe and effective therapy to reduce upper limb (UL) motor deficits and improve motor function in individuals with chronic ischemic stroke with moderate-to-severe arm impairment. VNS paired with rehabilitation motor tasks led by an occupational or physical therapist is typically done in-clinic during initial treatment. Attending regular outpatient sessions can be difficult for individuals with limited mobility/transportation.
Objectives:
The primary objective of the VNS-REHAB At-Home study is to assess the feasibility, safety, compliance, and therapist/patient acceptance of delivering up to 36 hours of therapist-activated VNS paired with UL rehabilitation in a home setting. Secondary objectives include evaluating improvements in UL impairment and function.
Trial Design:
The acute stage is therapist-activated VNS therapy paired with at home rehabilitation in 1- to 2-hour sessions up to 5 times per week for up to 18 weeks, with a maximum of 36 total hours of therapist visit duration. The long-term stage is a follow-up period of up to 2 years during which participants continue self-activated VNS at home using a magnet during functional tasks.
Study Setting:
Therapist-activated Paired VNS will be done in the participant’s home.
Eligibility Criteria:
Participants implanted or scheduled to be implanted commercially with the Vivistim Paired VNS System and assessed as able to receive therapist-activated VNS in the home setting.
Interventions:
The study will use mobile therapists in the participant’s home instead of a clinic for the initial treatment phase. Paired VNS will be done according to the manufacturer’s instructions for use.
Outcomes:
The primary endpoint is the number of individuals who complete at least 24 hours of therapy provided in the home setting by a therapist within an 18-week period. Secondary endpoints are change from baseline in assessment measures and patient-reported outcomes.
Sample Size:
Up to 150 participants.
Introduction
In the United States, about 800 000 people experience new or recurrent stroke each year. 1 Among survivors, upper limb (UL) impairment is both common and often persistent, with few regaining full function. UL impairment is one of the strongest predictors of long-term disability following a stroke and most people continue to experience significant limitations in their daily activities despite intensive rehabilitation.2,3 As individuals progress beyond the early recovery window, few evidence-based options are available to address these lasting impairments. Traditional rehabilitation often yields limited gains in the chronic phase where further improvement is unlikely, and strategies focus on compensation rather than restoration. New treatments are required to improve the quality of life (QOL) and daily functioning of patients with post-stroke UL impairment.
Paired vagus nerve stimulation (VNS) has emerged as a safe and effective therapy to improve UL outcomes in this population.4,5 At least 6 months following an ischemic stroke, eligible patients are implanted with a VNS device, which allows pairing of VNS during practice of task-specific UL movements. Following 2 pilot studies,6,7 a 108-patient pivotal trial, VNS-REHAB, was conducted at 19 sites in the United States and United Kingdom.4,8 VNS-REHAB was a triple-blinded study in which participants were randomized to intense rehabilitation paired with active VNS (VNS group) or intense rehabilitation paired with sham VNS provided for study blinding (control group). Improvement in the Fugl-Meyer Assessment Upper Extremity (FMA-UE) score and other assessments was significantly greater with Paired VNS compared to sham VNS, and results persisted through 1 year of follow-up.4,9 The pivotal study supported efficacy and safety of Paired VNS for UL improvement after a stroke, and the Vivistim® System (MicroTransponder, Inc.; Austin, TX, United States) was approved for use by the FDA in August 2021 and began commercial distribution in May 2022. 10
The standard Paired VNS therapy protocol involves pairing VNS during both therapist-supervised rehabilitation and individual functional practice outside the clinic. During rehabilitation sessions, the therapist delivers stimulation with a wireless controller at key moments of active movement during functional tasks. Beyond therapist-supervised sessions in-clinic, individuals can initiate a cyclical VNS session using a handheld magnet either prior to performing prescribed exercises or before engaging in daily activities or hobbies involving active use of the affected UL (eg, meal preparation, dishwashing, and gardening). These self-activated sessions deliver a brief pulse of stimulation every 10 seconds over a 30-minute period and can be performed up to 8 times within a 24-hour period.
While safety and efficacy have been established, Paired VNS Therapy is typically delivered in clinic-based settings during the initial treatment phase. For individuals with limited mobility or transportation options, attending regular outpatient sessions can be difficult due to barriers related to accessibility, scheduling, caregiver burden, and treatment adherence.11,12 These challenges are often exacerbated by broader social determinants of health, such as geographic location and socioeconomic status, which contribute to disparities in equitable access to care.11,12 Outpatient therapy services delivered in the home by an occupational or physical therapist, informally referred to as “mobile therapy,” may offer a more practical, sustainable, and convenient alternative for patients and their caregivers.
The VNS-REHAB At-Home study will evaluate the feasibility of delivering therapist-activated Paired VNS exclusively in the home environment to help overcome these challenges. Mobile therapists trained in Paired VNS Therapy will provide intensive, structured rehabilitation sessions paired with VNS in the patient’s home. This model is designed to replicate the quality and consistency of clinic-based therapy while reducing access barriers and enabling functional training in the patient’s daily environment.
The primary objective of the VNS-REHAB At-Home study (ClinicalTrials.gov, NCT05691023) is to assess the feasibility, safety, compliance, and patient acceptance of delivering up to 36 hours of therapist-activated VNS paired with rehabilitation in a home setting. Other objectives include evaluating changes in UL impairment and function. This approach aims to generate real-world evidence supporting home-based Paired VNS therapy as a practical, patient-centered strategy to advance motor recovery after stroke.
This article presents the clinical protocol approved by the Western Institutional Review Board (WCG IRB) and detailed methodology for the ongoing trial. It provides a foundation for future outcomes reporting and supports transparency and reproducibility in implementing this home-based therapeutic model.
Methods
Study Design
VNS-REHAB At-Home is a single-arm, postmarket study enrolling a maximum of 150 participants, with up to 50 mobile therapists, serving as independent study investigators, conducting up to 36 hours of at-home therapy per participant. The study will assess at-home therapy for participants who are commercially implanted with the Vivistim device. Participants will be offered participation in VNS-REHAB At-Home before or after implantation of the VNS device per the indications for use, following ischemic stroke. 13 Study participation includes therapist-activated Paired VNS in the participant’s home, self-activated VNS using a magnet, and scheduled outcome assessments. Paired VNS implantation (or scheduled implantation) is a criterion for eligibility to enroll in VNS-REHAB At-Home; therefore, the implant procedure is not part of this study.
This study has 2 stages (Figure 1). The acute stage (Stage 1) is a treatment period of up to 18 weeks of Paired VNS with rehabilitation for UL motor deficits provided at home by a therapist supplemented with self-activated VNS. The long-term stage (Stage 2) is a follow-up period of up to 2 years during which participants continue self-activated VNS at home using a magnet during therapist-prescribed functional tasks. Participants who complete at least 24 hours of therapist-activated home rehabilitation sessions will be included in the study analysis.

Stage 1 of the VNS-REHAB At-Home Study includes the collection of outcome measures at 5 timepoints: Screening, Baseline, and after performing 12, 24, and 36 hours of therapy. Therapy will begin within 3 days of the baseline assessments. Therapist-activated therapy consists of up to 36 hours of Paired VNS delivered by mobile therapists over a 6- to 18-week timeframe. Self-activated Paired VNS occurs concurrently with therapist-activated sessions and continues for the study duration. Stage 2 outcome measures are assessed at 3 months, 1 year, and 2 years. During this period, therapists also complete regular check-ins (in person or remotely) to monitor progress and compliance with self-activated sessions.
Study Objectives and Endpoints
The primary objective of the study is to assess safety, compliance, and acceptance of Paired VNS at-home therapy to determine feasibility. The primary outcome measure is the total hours of therapist-activated in-home therapy sessions completed. In previous Paired VNS studies,4,6,7 participants were considered compliant if they completed at least two-thirds of scheduled therapy sessions. Accordingly, the primary feasibility endpoint for this study is the proportion of individuals who complete at least 24 session hours (ie, two-thirds of the total possible 36 hours) within 18 weeks. Based on experience from the previous Paired VNS studies,4,6,7 we expect that at least 90% of participants will achieve 24 or more hours of completed therapy.
Other objectives include measuring improvements in UL impairment and function that may support benefits of fully home-based Paired VNS delivery and will be summarized as secondary exploratory endpoints.
Participant Population
Inclusion criteria are as follows:
Participant implanted or scheduled to be implanted commercially with the Vivistim System
Participant assessed as able to receive therapy in their home
Exclusion criteria are as follows:
Participant aged 18 years or younger
Pregnant women
Aphasia and other cognitive deficits are not exclusionary provided that participants are sufficiently able to understand the potential risks and benefits of the study, to personally provide informed consent, and to understand and cooperate with the treatment.
As this is a postmarket, open-label, longitudinal study, there are no medication-related exclusion criteria. However, all medications will be documented, including the use of botulinum toxin for spasticity management. Injection schedules will follow clinical recommendations made by the treating therapist and the participant’s health care provider. Study visits may be rescheduled to accommodate these treatments, provided the full 36 hours of therapy are completed within the allowed 18-week timeframe.
Study Treatment
Study Device
The Vivistim Paired VNS System is intended to be used to stimulate the vagus nerve during rehabilitation therapy to reduce UL motor deficits and improve motor function in chronic ischemic stroke individuals with moderate to severe arm impairment. 13 The Vivistim System consists of an implantable pulse generator (IPG), lead, and electrode; non-implanted system components include a wireless push-button controller that the therapist uses to synchronize therapy with the IPG; and a Stroke Application Programming Software system (SAPS) to provide clinician control of settings. The implantation procedure and safety profile have been previously described. 14
Study Intervention and Setting
The study intervention pairs VNS, delivered via the implanted device, with high-repetition, task-specific rehabilitation in participants with UL ischemic stroke deficits. The study will use mobile therapists in the participant’s home instead of in-clinic delivery of therapist-activated Paired VNS.
At each session, the therapist will use the software and a remote or keyboard press to trigger a brief burst of VNS during goal-oriented UL functional tasks, thus pairing VNS and rehabilitation (Figure 2a). The device will be programmed to standard commercial settings but may be adjusted based on individual participant tolerance at the clinician’s discretion. Typical device settings include an output current of 0.8 mA delivered over 0.5 seconds, a pulse width of 100 µs, and a stimulation frequency of 30 Hz, consistent with the parameters established in the VNS-REHAB pivotal study and previous trials.4,6,7,15 At the first therapy visit, participants will also be given a magnet and taught to swipe it over the IPG prior to performing prescribed functional UL tasks and daily activities for self-activated VNS (Figure 2b-d). Magnet settings may be adjusted, if necessary, though the system limits daily self-activated VNS use to a maximum of 4 hours for safety. The IPG records a log of both therapist-activated Paired VNS and self-activated VNS sessions.
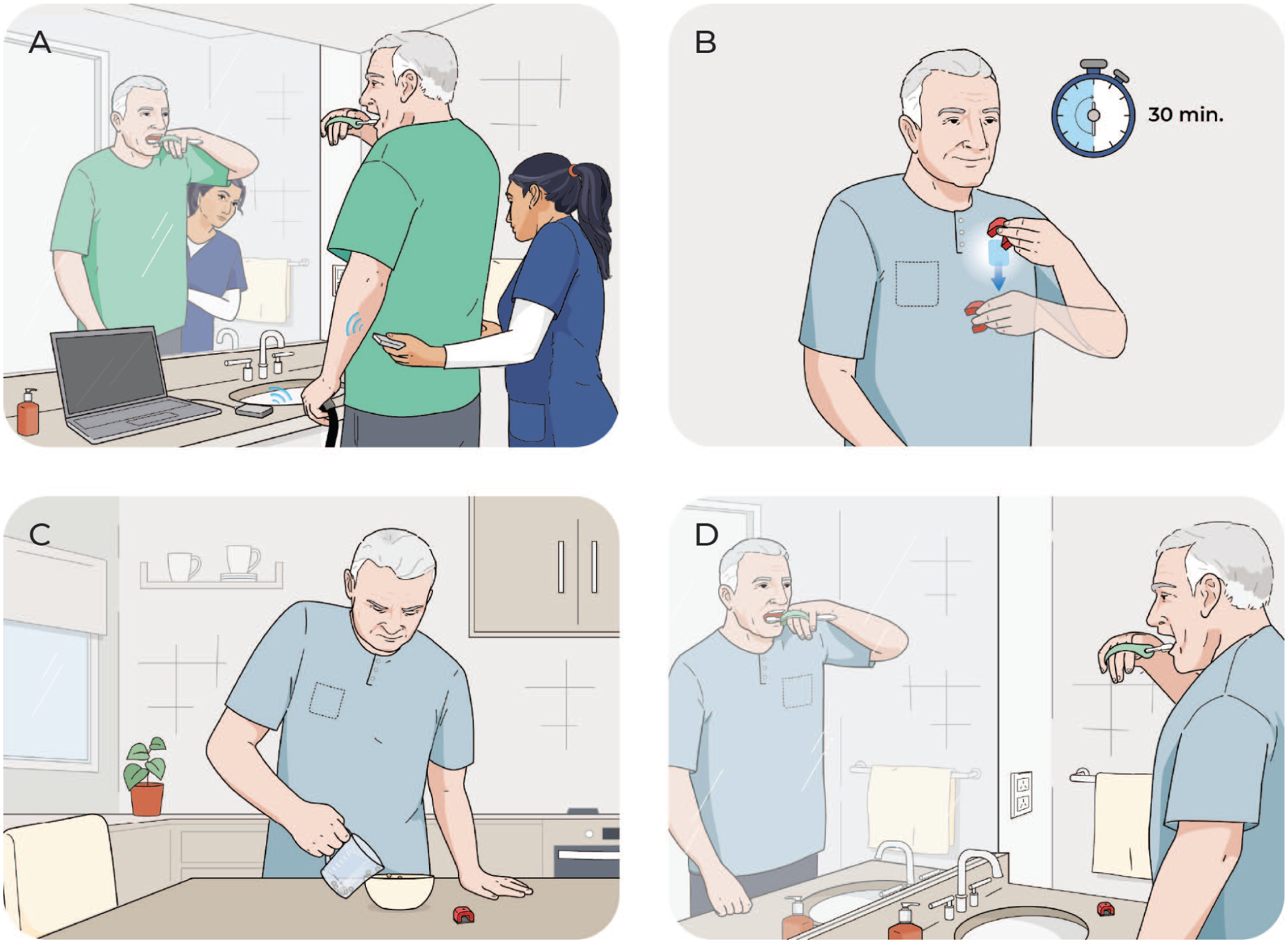
Therapist-activated sessions pair VNS with goal-oriented, challenging, high-repetition functional tasks to promote neuroplasticity-based upper extremity motor improvement. Therapists trigger VNS with a handheld remote during key active upper extremity movements for a given activity. Participants complete up to 36 hours of therapist-activated Paired VNS therapy in the home environment during this trial. (a) An example functional task (toothbrushing) during therapist-activated Paired VNS. The therapist may trigger VNS during the key movement of bringing the toothbrush to the mouth. Additional key movements may also be stimulated during this task. (b) Self-activated Paired VNS via a magnet swipe initiates a 30-minute session of cyclical VNS. After a magnet swipe, participants perform a therapist-prescribed (c) structured home-exercise program comprised of functional tasks (eg, pouring beans into a container) and/or (d) daily activities.
Therapist-Activated Paired VNS at the Participant’s Home
At enrollment, the therapist will evaluate the home environment to assess the feasibility of conducting therapist-activated Paired VNS sessions and assessments safely in the home.
Participants have 6 to 18 weeks to complete up to a total of 36 hours of therapist-activated Paired VNS sessions. Therapists are encouraged to schedule 1.5 hour (90 minute) sessions 3 times per week as implemented in previous Paired VNS studies4,6,7 and in typical commercial use. However, the duration and number of sessions per week may be adapted to accommodate scheduling availability and the participant’s tolerance for high-repetition sessions. Session duration may range from 1 to 2 hours, with the total number of sessions per week varying from 1 to 5, provided that 2-hour sessions do not exceed 3 times per week. The flexibility in session scheduling is designed to reflect real-world considerations for mobile therapy and promote accessibility. Table 1 highlights examples of potential scheduling options for therapist-activated sessions.
Potential Scheduling Options for Therapist-Activated Paired VNS Sessions.

Paired VNS therapy is grounded in established principles of motor learning and experience-dependent neuroplasticity.8,16 The approach emphasizes active movement, with stimulation delivered during volitional efforts while the individual performs task-specific, goal-directed, and meaningful tasks using the affected upper limb. The therapist designs tasks that are salient, engaging, and appropriately challenging to the individual to optimize motor learning. Sessions include high-repetitions based on the principles of massed and variable practice, that is, focusing on repeated task performance within a block of time while varying task context across repetitions or sessions to maintain engagement and promote generalization.
The number of tasks and time taken to do each activity will depend on the participant’s level of impairment, tolerance for high-repetition therapy, and overall session duration. Consistent with evidence-based rehabilitation guidelines and previous studies, it is recommended that the participant performs at least 3 to 6 functional tasks during each scheduled therapist visit and at least 30 repetitions per task to reach a minimum goal of 300 total movement repetitions paired with VNS.4,6 -8 The therapist will trigger stimulation during target movements within a repetition (eg, deliver stimulation while participant attempts to grasp a toothbrush). For more complex movements, stimulation may be delivered multiple times within a single repetition (eg, deliver stimulation while participant grasps toothbrush and again while participant brings it to the mouth, Figure 2a).
The use of adjunct strategies such as splinting, taping, or functional electrical stimulation, is permitted at the therapist’s discretion, in alignment with real-world clinical practice and stroke rehabilitation clinical practice guidelines. 17 Guidance will be provided to support the appropriate integration of VNS with adjunctive modalities when applicable.
Self-Activated VNS Using Magnet
Participants will also receive a magnet in the first at-home therapy session. The participant will be provided with verbal and written instructions on how to activate the device by swiping the magnet over the chest wall (where the VNS device is implanted; Figure 2b). The therapist will prescribe similar, repetitive functional tasks that were done during sessions (Figure 2c) and daily activities based on the participants’ short and long-term goals (Figure 2d). Functional goals can change during therapy and functional activities will be adapted accordingly.
On days or times that the participant is not engaging in therapist-activated Paired VNS sessions, they are instructed to do at least 1 hour (typically 2 magnet sessions) of self-activated VNS with the therapist-prescribed home program (Figure 2b-d). Prior to task practice, the participant will use the magnet to activate 30 minutes of cyclical VNS – a 0.5-second pulse of VNS every 10 seconds during the 30-minute session. These settings were shown to be effective based on clinical studies; therefore, stimulation parameters are typically not adjusted unless the participant experiences discomfort. Many participants do not perceive stimulation at 0.8 mA, so therapists will explain that therapy is still being delivered even if it is not felt. While participants will be instructed to self-activate VNS at least twice daily on the days they are not receiving therapist-activated Paired VNS, they can initiate additional sessions up to a maximum 4 hours in a 24-hour period. Participant adherence to the self-activated component of the study will be monitored via the device IPG log. Strategies to promote participant adherence, in addition to the individualized exercise program, may include ongoing education, behavioral contracting, and reinforcement for the participant and care partner.
Safety
Vagus nerve stimulation has a well-established safety profile with a low incidence of serious adverse events. 14 While the safety of Paired VNS delivered in a home environment is expected to be comparable to previous trials4,6,7,14 the study investigators (mobile therapists) will document and report any adverse events (AEs) and device complications that may occur from the start of therapy through study completion. Study investigators have the responsibility for the safety of the participants under their care and will be instructed to use clinical judgment to support participants’ tolerance of therapy and maintain safety.
Adverse events will be analyzed descriptively, and study oversight will include assessment of causality and severity of AEs by a physician safety monitor and the study director. Any unexpected or serious AEs will be reviewed immediately and an external physician reviewer may be consulted to determine further action, if warranted.
Study Assessments and Conduct
Good Clinical Practices and Role of the Sponsor
This study will be conducted in accordance with the Declaration of Helsinki and ISO 14155:2020. All enrolled participants will provide informed consent, and the protocol has been approved by the WCG IRB.
The study sponsor developed the protocol and will oversee data collection, management, and analysis. Study investigators include independent mobile therapists who are directly responsible for participant enrollment, delivery of interventions, and data collection. Publications reporting study outcomes will be led by independent clinicians and investigators from participating sites, comprising the VNS-REHAB At-Home Study Group. Lead authors will have full access to the study data and will assume primary responsibility for data interpretation, manuscript preparation, and submission.
Therapist Training
All mobile therapists will be trained as study investigators and will complete training in research ethics and regulations as required by the IRB. They will participate in 5 remote training sessions (1 hour each), and a 5-hour in-person session focused on standardized assessment administration, applying motor learning principles, delivering Paired VNS therapy, and using the SAPS system. All therapists will become certified to administer the Montreal Cognitive Assessment (MoCA).
The in-person training includes hands-on practice with motor assessments and Paired VNS Therapy. Therapists receive assessment manuals and standardized kits. They will independently score video-recorded FMA-UE and Action Research Arm Test (ARAT) motor assessments as part of study training. Refresher calls and protocol reviews will be conducted as needed. Administrative feedback will be provided to ensure adherence to procedures and documentation standards.
Study Schedule
The schedule of activities is described in Table 2.
Schedule of Activities from Enrollment through Study End.

Abbreviations: 9-HPT, 9 Hole Peg Test; AE, adverse event; ARAT, Action Research Arm Test; BBT, Box & Block Test; BDI, Beck Depression Inventory; E, Enrollment; EQ-5D, EuroQol-5 Dimensions; FMA-UE, Fugl-Meyer Assessment - Upper Extremity; GIC-C, Global Impression of Change-Clinician; GIC-P, Global Impression of Change-Patient; MAL, Motor Activity Log; MAS, Modified Ashworth Scale; MoCA, Montreal Cognitive Assessment; NA, not assessed; PRN, as needed; SAE, serious adverse event; SIS, Stroke Impact Scale.
The acute stage of the VNS-REHAB At-Home study (Figure 1) includes screening and consent, pre-therapy baseline evaluation, and Paired VNS provided in the participant’s home by a therapist. Assessments will be conducted after 12, 24, and 36 hours of at-home therapy.
Although the implant procedure is not part of the study, any procedural adverse events will be documented.
The study will be done in the participant’s home with a qualified and trained occupational or physical therapist. Study staff will train therapists on Paired VNS and assessments. The therapist will visit the participant’s home at least twice a week for up to 18 weeks, such that up to 36 hours of therapist visit duration occur. Participants are encouraged to swipe their magnet daily. However, on days when the therapist does not visit, participants are asked to complete at least 2 self-activated VNS sessions (or approximately 1 hour).
The long-term stage includes assessments at 3 months, 1 year, and 2 years after completion of 36 hours of therapist-activated sessions, as well as ongoing self-activated Paired VNS during therapist-prescribed activities (Figure 1). Follow-up will continue through 2 years. Additionally, therapists will have regular check-ins with participants, in person or remotely, to evaluate progress and compliance with self-activated sessions. Participants may opt to receive additional at-home therapist-activated sessions or in-clinic therapy sessions beyond the study through their insurance or self-pay. Therapists will document any additional therapy provided, including the type, frequency, and duration.
Assessments
The therapist will perform assessments at the participant’s home. The following assessments will be used to capture secondary endpoints and quantify change in impairment, function, and participation. The therapist will administer all tests. FMA-UE is video-recorded in all visits. Video recording of other motor tests is optional. Therapists are also encouraged to record their first and last session, as well as anytime they want to document a specific task.
The assessment schedule and details are summarized in Tables 2 and 3.
Assessment Details.
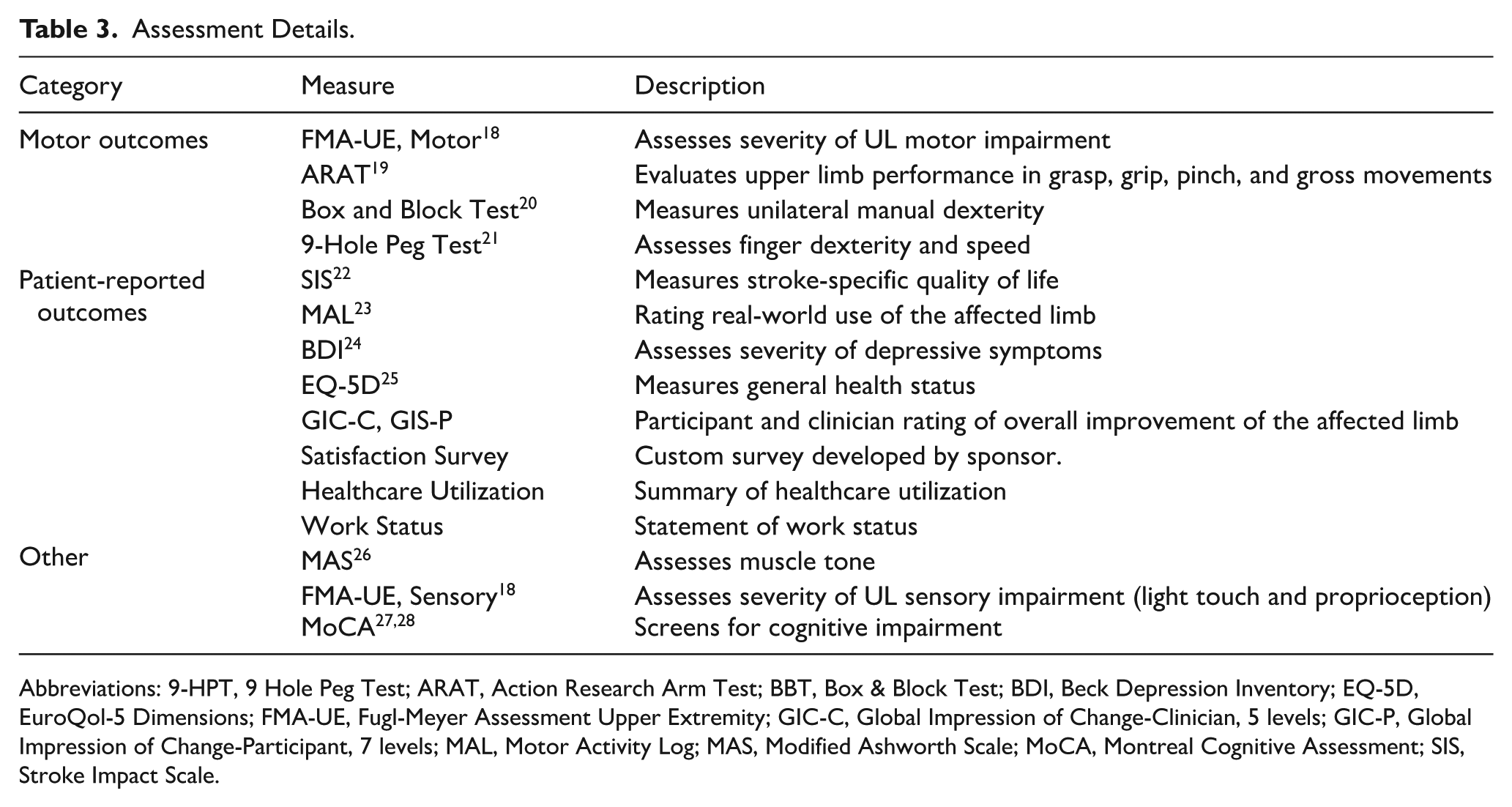
Abbreviations: 9-HPT, 9 Hole Peg Test; ARAT, Action Research Arm Test; BBT, Box & Block Test; BDI, Beck Depression Inventory; EQ-5D, EuroQol-5 Dimensions; FMA-UE, Fugl-Meyer Assessment Upper Extremity; GIC-C, Global Impression of Change-Clinician, 5 levels; GIC-P, Global Impression of Change-Participant, 7 levels; MAL, Motor Activity Log; MAS, Modified Ashworth Scale; MoCA, Montreal Cognitive Assessment; SIS, Stroke Impact Scale.
FMA-UE
FMA-UE is a quantitative impairment measure widely used in stroke rehabilitation trials.18 -33 The motor component assesses 33 items on an ordinal scale, with a total possible score of 66. In general, scores of 0 indicate the participant “cannot perform,” 1 denotes “can perform partially,” and 2 signifies “can perform fully.” The sensory component of the FMA-UE, which evaluates light touch and proprioception, will also be administered. Each item is scored from 0 (anesthesia/no sensation) to 2 (normal) for a maximum total score of 12. Excellent inter-rater and intra-rater reliability and construct validity have been demonstrated, and evidence suggests that the FMA-UE is responsive to change, with established minimally clinically important difference (MCID) in the literature.18-33
ARAT
The ARAT is a functional outcome measure to assess UL performance.19,31,34 Items comprising the ARAT are categorized into 4 subscales (grasp, grip, pinch, and gross movement) and arranged in order of decreasing difficulty, with the most difficult task examined first, followed by the least difficult task. 35 Each item is rated on a 4-point scale from 0 to 3, reflecting the quality of grasp, arm movement, body posture, and the ability to complete the task within 5 seconds. Both arms are tested separately and scores are summed to yield a total of up to 57 per arm. The outcome has excellent reliability and validity along with established MCID cut-offs.34,36
Other Measures
The Box and Block Test will be performed to assess unilateral gross manual dexterity.20,31 It measures the total number of blocks a participant can move from 1 compartment to another within 1 minute, tested separately with each arm.
The 9-Hole Peg Test evaluates unilateral finger dexterity by measuring the total time it takes to place 9 pegs into 9 holes and then remove them, with each hand tested separately. 21
The MoCA is a brief, 30-question assessment to detect cognitive impairment.27,28 Therapists will administer the MoCA to evaluate a potential participant’s cognitive status and ability to understand and actively engage in study activities as part of the enrollment visit. The MoCA will also be administered at the 36-hour assessment timepoint. Additionally, the modified Ashworth scale (MAS) is a muscle tone assessment scale used to assess the resistance experienced during passive range of motion. 26
Patient-Reported Outcomes
Patient-reported outcomes will be collected using standardized assessments. The Motor Activity Log-14 (MAL-14) assesses the individual’s perception of how much and how well 14 daily tasks are performed by the affected arm. 23 The Stroke Impact Scale (SIS) has been developed to assess 8 different domains of health-related QOL: strength, memory, emotion, communication, activities of daily living, mobility, hand, social role (participation), and general recovery. 22 The EQ-5D-5L is a standardized instrument for use as a general measure of health outcome that provides a simple descriptive profile and a single index value for health status. 25 The Beck Depression Inventory (BDI) is a 21-item self-report questionnaire that measures characteristic attitudes and symptoms of depression. 24 The Global Impression of Change (Improvement only, Patient [GIC-P, 7 levels]/Clinician [GIC-C, 5 levels]) is a brief observer-rated scale that measures symptom global improvement or change. The study also includes custom surveys developed by the sponsor to assess satisfaction, healthcare utilization and work status.
Withdrawal From Study
Participants may withdraw from the study at any time. The therapist or safety reviewer may also withdraw a subject for any reason, after consultation with the sponsor. Participants who have their VNS system removed will be withdrawn from the study at the time of device removal.
Data Analysis
Sample Size
The sample size calculation is based on the feasibility endpoint, defined as the proportion of completed therapy hours, with data from previous Paired VNS trials serving as the benchmark.
In VNS-REHAB, participants were considered compliant if they completed at least two-thirds of their scheduled therapy sessions. 4 Applying this same definition, compliance in the current study is defined as completion of at least 24 hours of therapist-activated Paired VNS in-home sessions (ie, two-thirds of the total 36 hours).
Previous Paired VNS studies showed that 90% of subjects received their expected therapy dose.4,6,7 Consistent with those experiences, the expected outcome in the present study is that at least 90% of participants will achieve 24 or more hours of completed therapy. The feasibility threshold is set at 70%, which is slightly higher than the 66% (24 out of 36 hours) required for protocol compliance. Therefore, if the expected rate is 90% (P1) and the actual rate is 70% (P2), a sample size of 33 subjects provides sufficient power to demonstrate that the observed proportion of compliant participants is no worse than the predefined feasibility threshold (70%).
Statistical Analysis Plan
The primary analysis will be based on the proportion of participants successfully completing at least 24 hours of therapist-provided, at-home therapy. It is expected that at least 70% of participants will be successful in the primary endpoint. For participants not completing at least 24 hours of therapist-activated sessions, reasons for not meeting the primary endpoint will be tabulated and reported descriptively.
Secondary endpoints, measured in changes from baseline in assessments and patient-reported outcomes, will be analyzed descriptively.
All data will be summarized with descriptive statistics, means, and standard deviations for continuous data, and counts and percentages for categorical data. Confidence intervals will also be provided where appropriate. All unscheduled assessments will be excluded from summary tables. Data from unscheduled assessments will be displayed in listings.
All efficacy and safety analyses will be performed on the intent-to-treat population, defined as all participants who consent and receive at least 1 day of Paired VNS Therapy. Data analyses and statistical testing will be conducted using SAS Version 9.4 or higher. Unless specified for a specific test, a significance level of α = .05 will be used.
Safety endpoints include adverse events and device complications, which will be analyzed descriptively.
Analyses outside of this protocol may be performed to supplement results or for research purposes at the discretion of the study sponsor.
Discussion
Paired VNS therapy was successful in improving outcomes in participants with post-stroke UL impairment wherein rehabilitation sessions were conducted at an outpatient clinical site. 4 Conducting these sessions in the participant’s home may substantially improve availability and convenience of Paired VNS, potentially overcoming significant barriers associated with clinic attendance. The home-based model may offer enhanced patient accessibility, improved adherence rates, and reduced logistical barriers, thereby amplifying the potential benefits of Paired VNS rehabilitation. Multiple systematic reviews of clinical trials have found that home-based, therapist-led UL rehabilitation is as effective as clinic-based interventions.37-40 The existing evidence base on providing home-based post-stroke rehabilitation for UL impairment justifies the approach that will be explored in the VNS-REHAB At-Home study.
Study Limitations
This single arm, open-label postmarket study is intended to assess the feasibility of at-home therapist-activated Paired VNS sessions. Limitations inherent to the design should be acknowledged. First, the study design precludes direct comparisons with control groups, potentially limiting definitive conclusions regarding efficacy. Second, home-based therapy introduces variability related to individual environmental and motivational factors, potentially affecting therapy consistency and adherence. Future studies can address these limitations through randomized controlled designs, and inclusion of broad demographic and clinical profiles.
Conclusions
The VNS-REHAB At-Home study will provide valuable insight into the provision of therapist-activated Paired VNS sessions outside a traditional clinic environment. If the study findings indicate that this delivery method is feasible, Paired VNS could become a more convenient and accessible option for individuals with chronic post-stroke UL impairment, while also supporting a more sustainable, patient-centered model of care. Results from the study may offer crucial real-world evidence to inform broader implementation of home-based, mobile neurorehabilitation with Paired VNS, ultimately improving treatment access and quality of life for stroke survivors.
Footnotes
Acknowledgements
The authors thank David Pierce for his contribution to protocol development, and Shelley Coppola, Lionel Dacpano, Brooks Fowler, Diana Hansen, and Andrew Schmid for their administrative and technical support. They also thank Brent Tarver for editorial assistance and Tim Peoples for medical writing support.
Ethical Considerations
This study will be conducted in accordance with the Declaration of Helsinki and ISO 14155:2020. The study protocol was approved by the Western Copernicus Group Institutional Review Board (approval #: 0226640) on December 20, 2022.
Consent to Participate
All enrolled participants will provide informed consent prior to participating in the study.
Consent for Publication
Not applicable.
Author Contributions
SD: Conceptualization, Project Administration, Writing – Original Draft, and Writing – Review & Editing. RP: Investigation, Project Administration, Conceptualization, and Writing – Review & Editing. LG: Investigation, Project Administration, and Writing – Review & Editing. SZ: Conceptualization, Investigation, Project Administration, and Writing – Review & Editing. KA: Investigation, Project Administration, and Writing – Review & Editing. AD: Investigation, Project Administration, and Writing – Review & Editing. RH: Conceptualization, Formal Analysis, and Writing – Review & Editing. NE: Conceptualization, Formal Analysis, and Writing – Review & Editing. CP: Conceptualization, Investigation, Formal Analysis, Supervision, and Writing – Review & Editing.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: MicroTransponder, Inc.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: RP and SZ have served as consultants for MicroTransponder, however, no consulting fees were provided for research activities or preparation of this manuscript; SD, RH, NE, and CP are employees of MicroTransponder, Inc; LG, KA, and AD have no disclosures to report.
Data Availability Statement
Data sharing requires a signed data access agreement with specific outside funding explicitly stated to access the database without any support from investigators or MicroTransponder. Requests should be sent to
