Abstract
Background:
Patients admitted to neurological critical care unit (NCCU) frequently require ventilator support and tracheostomy. This prospective longitudinal observational study collated outcomes following tracheostomy amongst patients admitted to NCCU.
Methods:
In this prospective longitudinal observational study, we collected a predetermined set of clinical data for all patients admitted to the NCCU who underwent tracheostomy, until they were discharged from the unit. Exploratory data analysis of the tracheostomy outcomes was performed to identify the clinical parameters influencing decannulation.
Results:
Fifty four (5.4%) of 999 consecutive patients admitted to NCCU required tracheostomy. The mortality among people who had tracheostomy was 11.1% (n = 6) and decannulation rate was 59.3% (n = 32). Tracheostomies were more commonly done in men, those admitted with GCS of 3.0 and had cerebrovascular accidents. Thirty (55.6%) patients had sialorrhea during their NCCU admission which demonstrated significant associations with the total length of NCCU admission, time taken for decannulation, admission GCS, Fiberoptic Endoscopic Evaluation of Swallowing (FEES) referrals, need to recannulate following decannulation, pulmonary infections, and oral candidiasis. Incidence of chest infections was higher prior to the institution of pharmacological management of oral secretions. Prevalence of oral candidiasis was higher among patients who received pharmacotherapies for oral secretion.
Conclusion:
Oral secretion issues increase incidences of pulmonary infections, delay decannulation, increase the need for recannulation, and prolong the stay of patients in NCCU. Pharmacological management of oral secretions was associated with a reduced incidence of chest infections and a shorter duration to achieve decannulation.
Introduction
Patients admitted to neuro-critical care unit (NCCU) frequently require prolonged periods of mechanical ventilation. The insertion of tracheostomy is a common procedure in NCCU, undertaken in approximately 10% of ventilated patients in both the United Kingdom and the United States. 1 Tracheostomy alleviates respiratory workload, enhances patients’ vocalisation, and ensures a secure airway for patients with diminished alertness, impaired deglutition, and compromised coughing.1 -3
Tracheostomy is usually done at a point in time when prognostication and eventual outcomes remain uncertain. 2 The need for tracheostomy increases with age, lower GCS, pupillary abnormalities, extra-cranial injuries, and early secondary insults. 4 Evidence suggests that tracheostomy is associated with reduced hospital stays among patients with stroke and traumatic brain injury. 1 Patients with neurological disorders account for one-fifth of all tracheostomies. They also exhibited higher mortality than those without neurological disorders. Those requiring tracheostomy following stroke have higher mortality rates compared to individuals with other neurological disorders. 5
Studies have identified the clinical predictors for successful decannulation. A study from Italy involving 351 patients with severe acquired brain injury (sABI) found that the absence of pulmonary infections, normal tracheal anatomy during fibro bronchoscopy, and a better level of consciousness at admission increased the likelihood of successful decannulation. 6 Another retrospective cohort study analysing 463 patients with sABI identified younger age, absence of salivary aspiration, the aetiology of ABI, absence of a vegetative state, and higher scores for both voluntary and reflex coughing as key predictors of successful decannulation. 7
This study aims to synthesise evidence on the clinical course and outcomes of patients who had tracheostomy following admission to NCCU of a tertiary neuroscience centre in the UK. It also seeks to understand the role of oral secretion management following tracheostomy.
Methods
Setting
This was a single-centre, prospective, longitudinal observational study on patients admitted to neuro-critical care unit (NCCU) at a tertiary teaching hospital in England and required tracheostomy from July 2020 to December 2021. The project was reviewed by both the Clinical Research and Innovation Office (CRIO) and Clinical Effectiveness Unit of Sheffield Teaching Hospitals NHS Foundation Trust (STH 20830) and was approved as a service evaluation which does not require informed consent.
We adopted a non-probability sampling technique (single-stage purposive), defined by specific inclusion and exclusion criteria. The inclusion criteria were (a) over 16 years of age, (b) admission to the neuro-critical care unit, and (c) required tracheostomy after admission to NCCU. We excluded patients who already had tracheostomy prior to their admission to NCCU.
Data and Outcome Measures
Patients’ clinical and sociodemographic information were obtained from the electronic patient information system and tracheostomy ward round notes. Data was collected by 2 Speech and Language Therapists (SLTs) and was recorded on electronic Case Report Forms (CRFs). Basic clinical and sociodemographic data were recorded upon recruitment. The SLT team then reviewed the patients once weekly and captured their clinical information for the week, until discharge from NCCU or death. Outcome measures collected are shown in Table 1. A preliminary pilot study (n = 6) was conducted to ensure robust standardisation measures were in place.
Outcome measures.

Tracheostomy Weaning and Decannulation
A consultant neuro-anaesthetist, Speech and Language Therapist (SLT), physiotherapist, and the nursing team conduct tracheostomy ward rounds twice a week. During these rounds, the team identified patients who had undergone tracheostomies and discussed their management plans, including the commencement of weaning from tracheostomy. The treating teams made all the clinical decisions including the presence of infections and use of antibiotics. Our institutional criteria for commencing weaning are shown in Box-1.
Tracheostomy weaning criteria. 8

Swallowing evaluations were performed by a member of the SLT. This involved an assessment of oropharyngeal swallow function, ability to manage oral secretions, and review for commencement of cuff deflation trials. In addition to bedside swallowing assessments, some patients were referred for a fibreoptic endoscopic evaluation of swallowing (FEES) to inform recommendations for pharmacological management of oral secretions and therapeutic interventions for swallowing.
For those patients deemed suitable for weaning, the team established the baseline levels of respiratory support and set boundaries for cessation of spontaneous breathing trial (SBT). Patients were later commenced on twice daily SBT challenge with zero pressure support (PS). The duration of periods of zero PS was incrementally increased following each successful SBT. Trials of cuff deflation during periods of zero PS were initiated, with monitoring of voice, tracheobronchial secretions, and swallowing function. Prior to decannulation, the team ensured that the patient had a patent upper airway, an adequate cough to clear secretions, inspired oxygen requirements of less than 40%, absence of significant bronchopulmonary infection, minimal cardiovascular support, and an adequate level of consciousness. 9
Decannulation was attempted after the patient was able to tolerate continuous 30 minutes of zero PS with cuff deflation. A tracheostomy tube occlusion test was performed by briefly occluding the tube with a gloved finger during full cuff deflation. The process of decannulation was continued if the patient could breathe through their mouth and nose alongside intact phonation. Decannulation was deferred if airflow was absent during the tracheostomy occlusion test, or signs such as stridor, respiratory distress, laboured breathing, use of accessory muscles, or desaturation were observed. In such cases, the cuff was reinflated again. In the case of failed occlusion test, an ENT consultation was obtained for fibreoptic endoscopic examination of the upper airway to assess for tracheal stenosis, tracheomalacia, vocal cord palsy, or granuloma formation. Based on the status of decannulation at the time of discharge from our intensive care unit, we divided the patients into 2 groups: (1) those successfully decannulated at the time of discharge from ITU and (2) those who were discharged from the ITU with tracheostomy including 3 patients who continued to require mechanical ventilation.
Oral Secretion Management
In addition to speech and language therapy, we followed a stepwise approach to the pharmacological management of oral secretions. 10 For patients with delirium (Intensive Care Delirium Screening Checklist (ICDSC) score ≥ 4), 11 Glycopyrronium bromide was used as the initial option since it does not cross the blood-brain barrier. We used hyoscine hydrobromide transdermal patch for people who are not in delirium (ICDSC score ≤ 3). If first-line options were insufficient, we administered atropine 1% drops sublingually, starting with 1 drop (500 µg) every 12 hours and titrating up to 4 drops (2 mg) every 4 hours as needed. For refractory cases, not responding to the above, we injected botulinum toxin A (Xeomin) into the salivary glands.
Analysis
The information from the CRFs was extracted and collated into a single database. All identifiable information was anonymised. The data was coded and cross-checked by at least 2 investigators. The coded outcomes were then analysed using IBM SPSS Statistics (version 28.0)
12
and JASP (version 0.18.3).
13
Pairwise deletion was performed on variables with missing data. Distribution of the demographic parameters was assessed with Shapiro-Wilk test of normality (
Unpaired student’s
Degree of freedom (1): 0.10 (weak), 0.30 (moderate), and 0.50 (strong).
Degree of freedom (2): 0.07 (weak), 0.21 (moderate), and 0.35 (strong).
Concurrent link between sialorrhea and other parameters was calculated according to the specific nature of each variable. Longitudinal analysis exploring the temporal association between new onsets of pulmonary infection amongst patients with sialorrhea and their corresponding secretion management was performed. Frequency of onset of new pulmonary infections pre- and post-secretion management were documented, and their corresponding incidence rates were calculated.
Results
Clinical and Sociodemographic Information
Information sourced from the critical care unit registry showed 999 new admissions between July 2020 and December 2021. Of these admissions, 5.4% (n = 54) of patients with various neurological disorders required tracheostomy. Descriptive statistics summarising clinical and sociodemographic information of the cohort (N = 54), alongside their tracheostomy outcomes are shown in Table 2. Missing data was minimal and was only observed in the specific variables: GCS at admission (n = 2), admission weight (n = 2), and tracheostomy tube size (n = 1). Shapiro-Wilk’s test showed a significant departure from normality (
Demographics and care outcomes across different patient sub-cohorts.

Demographics and care outcomes across different patient sub-cohorts, stratified according to their Critical Care Unit (CCU) admission and decannulation outcomes. The second column represents patients who survived their CCU admission (n = 33), the third column represents those who died during their CCU admission (n = 6) and the fourth column depicts patients repatriated to their local CCU with their tracheostomy tube still in-situ (n = 15). The fifth and sixth columns depict patients who were decannulated (n = 32) and those who were not decannulated (n = 22) respectively. The final column represents the cumulative count of all tracheostomy patients in the study (N = 54), encompassing patients from the second, third, and fourth columns.
Primary Clinical Conditions
The most common diagnosis of people admitted to NCCU who had tracheostomy was neurovascular diseases (53.7%, n = 29), followed by traumatic brain injury (TBI; 13%, n = 7). Other pathologies include neuromuscular disorders (myasthenia gravis and Guillain-Barre syndrome), encephalopathy and malignancy (Table-3). Patients admitted to the NCCU due to vascular aetiologies had twice as many mortalities as those with other etiologies. Hydrocephalus interventions were more frequently performed in patients with vascular aetiologies (χ2 = 9.077,
Primary clinical conditions across different sub-cohorts of tracheostomy patients (N = 54).

The primary clinical conditions across different sub-cohorts of tracheostomy patients (N = 54), stratified into distinct categories: Vascular aetiologies including intracerebral haemorrhage (ICH), subarachnoid haemorrhage (SAH), and ischaemic stroke; Neuromuscular conditions; Traumatic brain injury (TBI); Encephalopathy; Malignancy; Disseminated brain abscess; and Cerebral palsy with bowel obstruction. Percentage within column (%) were reported alongside the frequency values.
Tracheostomy Care Outcomes
Tracheostomy care outcomes across different patient sub-cohorts are illustrated in Table-2. Thirty-three (61.1%) patients were discharged from NCCU. The tracheostomy tube could be removed in 32 (59.3%) with a median time of 23.5 days (IQR = 17-42 days) following tube insertion. Of the 33 patients discharged from NCCU, only 1 individual had a long-term tracheostomy tube inserted following numerous failed cuff deflation trials. A 11.1% (n = 6) died whilst the tracheostomy tubes were still in situ, with a median time to death of 45.5 days (IQR = 42-46 days) following tracheostomy. Fifteen patients (27.8%) with tracheostomy were transferred to their local hospital during the study period. Comparison between decannulation status across the tracheostomy outcomes in table-2 revealed significance of sample independence in 4 parameters: GCS at discharge (
Twenty-four (44.4%) patients required tube reinsertion, 17 of those were recannulated once, 6 were recannulated twice and 1 required 9 recannulations. Table-4 illustrates the specific indications for the replacement of tracheostomy tubes. Cuff leaks accounted for 18.4% (n = 7) of recannulations. One incident was attributed to a fractured flange which occurred in the absence of significant adverse events or tube displacements. Another instance involved a new clinical requirement for prolonged tracheostomy, prompting the transition from an LPC Shiley tube to a silver Negus tube.
Indication for tracheostomy tube replacement, (n = 38).

Specific indications accounting for the total documented cases of tracheostomy tube replacements, (n = 38).
Among the 54 patients, 22 underwent at least 1 neurosurgical procedure following the tracheostomy; 15 cerebrospinal fluid (CSF) diversion procedures including external ventricular drain (EVD) and ventriculoperitoneal shunt, and 7 other neurosurgical procedures comprising of decompressive craniectomy and craniotomy. Of the 14 patients who had seizures following tracheostomy, 7 developed seizures after CSF diversion procedures. This was significantly higher compared to patients who did not require such procedure [7/15 (46.7%) vs 7/39 (21.9%)] (χ2 = 4.65,
The most common medical complication after tracheostomy was infection (n = 49, 90.7%). Sixteen (29.6%) had at least 1 pulmonary infection episode. Presence of pulmonary infection after tracheostomy significantly delayed decannulation time from a median of 21 days (IQR = 12-39 days) to a median of 39 days (IQR = 20-61 days;
Oral Secretion Management
Table-5 depicts the comparison of patients with and without oral secretion management issues. Thirty (55.6%) patients had sialorrhea following tracheostomy. Median duration in which sialorrhea was identified as an issue following tracheostomy was 5 weeks (IQR = 2-8 weeks). Those with sialorrhea were more often referred for FEES, had longer length of stay in NCCU, higher GCS at admission to NCCU, longer interval between insertion and removal of tracheostomy tube, more often required reinsertion of tracheostomy tube, and more frequent chest infections and oral candidiasis (
Comparison of patients with and without secretion management issues (N = 54).
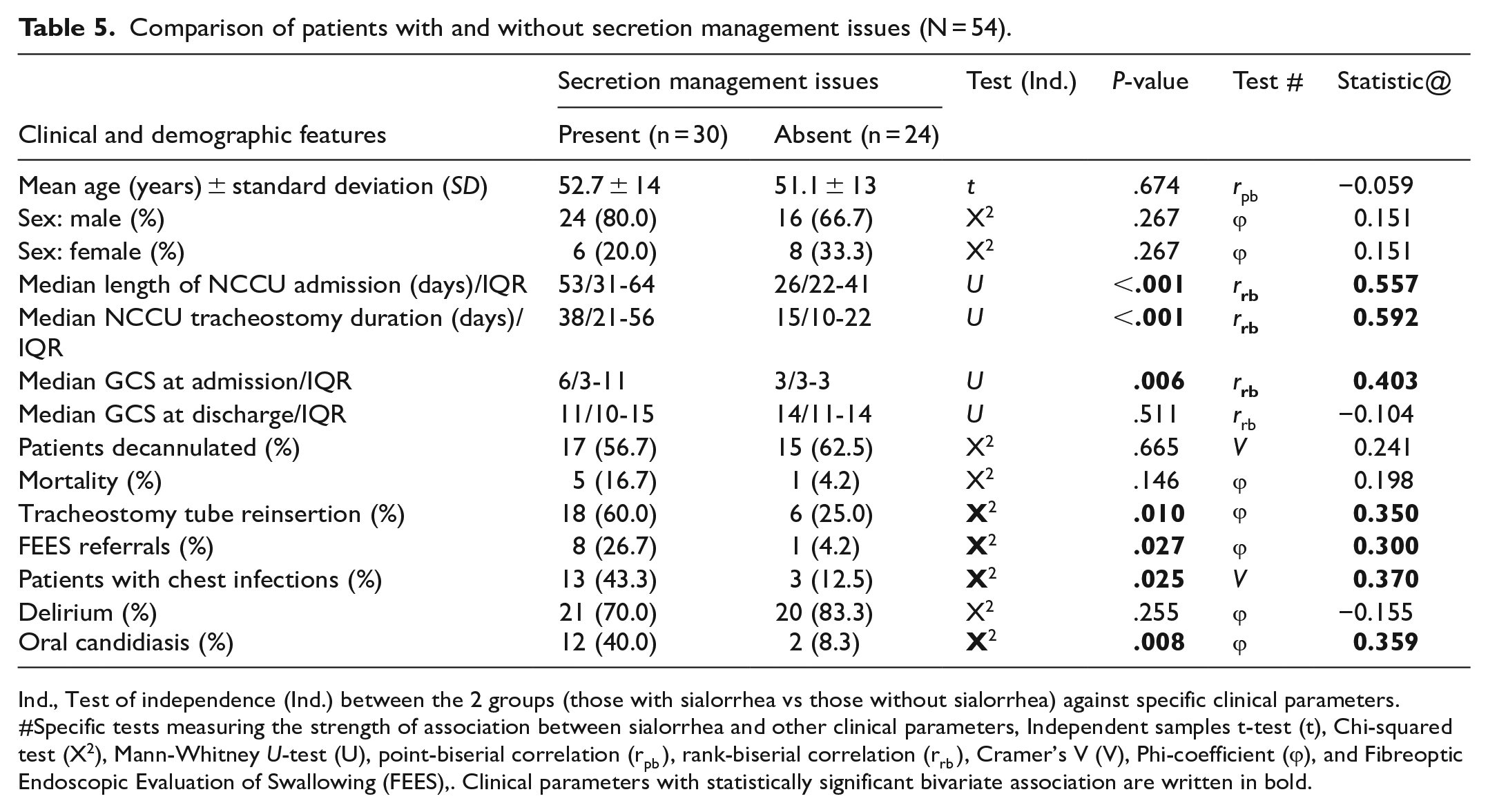
Ind., Test of independence (Ind.) between the 2 groups (those with sialorrhea vs those without sialorrhea) against specific clinical parameters. #Specific tests measuring the strength of association between sialorrhea and other clinical parameters, Independent samples t-test (t), Chi-squared test (X2), Mann-Whitney
Figure 1 illustrates the secretion management strategy employed in our cohort. Of the 30 patients experiencing issues with oral secretions, 29 received pharmacological therapies. Twenty-three (77%) of these patients, received pharmacological intervention within 1 week of onset of excessive oral secretions. Sixteen (55.2%) patients required only a single pharmacological agent. The remaining 44.8% (n = 13) received multiple agents of oral secretion management (Figure 1). Six were treated with Botulinum neurotoxin type A (Xeomin®).

Summary of the secretion management for the tracheostomy patients (N = 54), considering their decannulation status and level of pharmacotherapies administered.
Incidence of new chest infections before and after starting treatment for oral secretions was 304.3 per 1000 person-weeks (7 new cases over 23 person-weeks) and 70.4 per 1000 person-weeks (14 new cases over 199 person-weeks) respectively, with an incidence rate ratio (IRR) of 0.231 (95% CI 0.094, 0.567;
Discussion
Around 10% of patients requiring ventilation in NCCU undergo tracheostomy. 1 In our series, only 5.4% of all patients admitted to NCCU had tracheostomy. The difference in patient cohorts between ours and previous studies may account for the lower frequency of this procedure within our NCCU. In our cohort, survival following tracheostomy was dependent on age, sex, and aetiology of the neurological condition. The mean age of patients who died whilst tracheostomy tube was still in situ was higher (55.1 years) compared to those who survived. This observation is similar to findings from other studies, which reported an independent association between age and survival outcomes among mechanically ventilated patients.4,17 -20
Consistent with previous studies,21 -23 men in our cohort demonstrated a higher tracheostomy rate and mortality following tracheostomy, with all mortality cases being accounted for by them. Several studies have found that women with normal menstrual cycles exhibited significantly higher survival rates, better preservation of blood-brain barrier integrity, fewer sensorimotor impairments, reduced brain oedema, and smaller contusion volumes compared to age-matched males following TBI. 24 Murine models have also shown a dose-dependent neuroprotective response to progesterone. 25 Estrogen has an immunoprotective effect in the context of trauma and sepsis, while androgens have an immunosuppressive effect.26 -29 The higher mortality observed in men within our cohort may be partly explained by these variations in hormonal dynamics between genders. Participants who had neurovascular etiologies (predominantly haemorrhagic stroke) displayed higher overall mortality, further reaffirming the conclusion derived from a previous prospective multicentre study. 5
Like the observations in other studies, a significant proportion of patients were admitted with a low GCS of 3.0.4,17 Median duration for which the tracheostomy was in place was 22.5 days. Those with longer duration of tracheostomy also had a longer stay in NCCU. A significant increase in duration of tracheostomy was observed in those who did not survive their NCCU admission (45.5 days). Failure to decannulate after 3 weeks may prolong the length of stay in NCCU and increase the risk of death. Incidence of new pulmonary infection throughout the course of tracheostomy was also associated with a significant delay in decannulation (
Patients who underwent CSF diversion procedures had a higher prevalence of seizures. These findings were like that of another study which found that seizures post-CSF diversion procedures are common and were independent of their shunt location. 30
Explorative association analysis revealed a statistically significant link between sialorrhea and the length of NCCU admission (
Our longitudinal analysis suggested a notable difference in the incidence of chest infections before and after the initiation of oral secretion pharmacotherapies, indicating a potential therapeutic benefit of pharmacotherapies in preventing chest infections within this cohort. Due to the absence of a control group (comprising of tracheostomised patients with sialorrhea who did not receive any pharmacotherapeutic interventions for secretions), this study could not ascertain any definitive influence attributed to medical management of sialorrhea on the outcome of tracheostomy. However, incorporating such a control arm is not feasible due to ethical considerations.
Above-cuff suction ports were retrospectively added in some instances to manage the high subglottic secretion load in tracheostomised patients who lacked these ports initially. Due to the limited sample size, our study was unable to ascertain the beneficial evidence of subglottic secretion ports on the overall incidence rate of ventilator-associated pneumonia (VAP), as posited by another meta-analysis which looked at 20 different trials. 31 Mao et al 31 concluded a significant reduction in the rate of early-onset VAP following the introduction of subglottic secretion ports, with a number needed to treat (NNT) of 10.49, which meant that these ports effectively prevent 1 case of VAP for every 11 patients who were fitted with them. Results from this separate meta-analysis indirectly reinforced the idea of reduced incidence of pulmonary infections following mechanical optimisation of secretion load, hence externally validating the association seen in our study. Given these findings, an increased vigilance to secretion issues in patients with tracheostomy is crucial, as prompt initiation of secretion management may potentially mitigate pulmonary infections and other associated microbial colonisations.
We noted a statistically significant association, of moderate strength, between secretion issues and oral candidiasis from our exploratory association analysis (
Six patients were treated with Botulinum neurotoxin type A within our cohort. Several studies have established the efficacy of intraglandular botulinum neurotoxin type A injection in secretion management,32,36,37 that of which is greater than the anticholinergic effect exhibited by hyoscine hydrobromide patch. 32 Our patients treated with Xeomin® showed much greater clinical response compared to other agents, though the exact efficacy of this treatment could not be established due to the limited treatment sample size.
A considerable proportion of FEES referrals were made for patients identified with secretion issues (
A previous study demonstrated the utility of flexible bronchoscopy (FBS) in identifying and managing late tracheostomy complications, which were rarely the cause of decannulation failure. 40 Guided by FBS findings, targeted interventions such as selecting appropriate tracheostomy tubes and applying medical or laser therapies, successfully resolved tracheal lesions and enabled decannulation in a sizeable proportion of patients, including those who initially failed standard decannulation criteria. 40 Our study did not incorporate FBS as a routine protocol for decannulation. Instead, FEES was employed selectively in a subset of patients presenting with high secretion burden. Only 2 patients in our cohort underwent bronchoscopy to investigate potential tracheal alterations.
This study also examined the complications following tracheostomy, identifying infections as the most prevalent complication, occurring in 90.7% of cases. VAP was the leading cause of these infections, while line infections were the most frequent source of extrapulmonary infections, followed by oral candidiasis. There were 7 documented cases of central nervous system (CNS) infections (ventriculitis and CSF infection), all of which were associated with post-EVD procedures. In contrast, a separate retrospective study reported an infection incidence rate of 25.8% in their cohort of post-EVD patients. 41 Our findings not only confirmed the heightened risk of infection following EVD but also revealed a higher post-EVD infection rate of 46.7% in our patient cohort. Localised tracheostomy-related mucocutaneous infections were uncommon, with only 3 cases observed (5.6%).
This study’s prospective longitudinal design facilitated the systematic, real-time data collection over 18 months, offering practical insights into the progression and outcomes of patients requiring tracheostomy in NCCU. The design also captured a wide range of clinical scenarios and accounted for seasonal variations in admissions and care. As it was conducted at a single centre, the study benefited from consistent clinical practices and standardised data collection protocols, enhancing its internal validity. It is also the first study to explore sialorrhea in NCCU patients who require tracheostomy.
Limitations
Geographical homogeneity and small sample size inherent to this single-centre study limit generalisation of the findings. The small sample size and mortality events further limited us from adopting survival analyses including Cox regression model to explain the specific degree of variance contributed by each clinical predictor. 42 The clinical course and outcome of the people repatriated to local general intensive care units were also not collected. We did not collect details of location, dimensions, and other characteristics of the brain lesions.
We collected the data on complications including infections from the clinical records. We did not collect the data on the nature of the micro-organisms causing the infections. The diagnosis of infection was made by the treating team. We did not collect the data on bronchoalveolar lavage, bronchial aspiration procedure, and use of high flow nasal cannula by tracheostomy and their contribution to infection.
Bivariate association analysis between sialorrhea and other clinical parameters (Table-5) was exploratory in nature (no priori hypotheses were being tested), hence correction for multiple testing (Bonferroni Correction) was not done. Type I error rate would not be inflated since the objective of such analysis was to seek tangible associations rather than confirming/refuting priori hypotheses. All significant associations would need to be further validated in a larger multicentre study.
Tracheostomy-related interventions and other secretion management practices were difficult to standardise due to the observational nature of this study. We did not collect details like number and duration of the speech therapy sessions. Consequently, this diminished the statistical power of reported findings when compared against randomised controlled trials. This study was also conducted during the period of COVID-19 pandemic, which significantly delayed the operationalisation of this study.
Another limitation of this study is the lack of detailed and standardised indicators of the clinical state of patients. While GCS offers a general assessment of consciousness, it does not comprehensively capture motor, cognitive or functional disability, nor the severity of dysphagia. Established measures, such as the Functional Independence Measure, 43 Disability Rating Scale, 44 Barthel Index, 45 Dysphagia Outcome and Severity Scale, 46 or the Coma Recovery Scale-Revised (CRS-R), 47 could have provided more robust and multidimensional insights. Moreover, the absence of assessments like CRS-R is a significant limitation of our study. CRS-R is considered the gold standard for evaluating consciousness levels in patients with sABI. 48 Its inclusion would enhance the accuracy and depth of the findings, particularly in understanding the relationship between different states of consciousness and decannulation outcomes. 49
These limitations underscore the importance of future prospective studies that incorporate systematic multimodal assessments of patient functionality, cognitive state, dysphagia severity, and radiological features of TBI. Such approaches would provide a more nuanced understanding of the predictors of decannulation and their implications for patient outcomes, enabling more informed clinical decision-making.
Conclusion
This study informs clinicians on the risks associated with secretion issues and other tracheostomy-related clinical determinants, enabling them to anticipate future health events and proactively undertake early mitigative measures to minimise such risks. This study also highlights the potential role of pharmacotherapy in managing oral secretions and mitigating its complications, including chest infections. Oral secretion issues demonstrated significant associations with the length of stay in NCCU, time taken to decannulate after tracheostomy, need for recannulations, and pulmonary infections. Pharmacological treatment for oral secretions is associated with an increased prevalence of oral candidiasis. Treatment with pharmacological agents resulted in adequate secretion control. Further trials focussing on oral secretion management within the NCCU are required to establish treatment standards and to confirm the associations highlighted in this study.
Footnotes
Ethical Considerations
We confirm that this study has all the regulatory approvals of our institution. We confirm that this study adhered to ethical guidelines, was reviewed and approved by the Sheffield Teaching Hospitals NHS Foundation Trust clinical audit and effectiveness unit (STH 20830). The project was initially reviewed by the Clinical Research and Innovation Office (CRIO) of Sheffield Teaching Hospitals NHS Foundation Trust (STH 20830). The CRIO opined that it is a service evaluation and forwarded it to the Clinical Effectiveness Unit for approval. The project was approved by Clinical Effectiveness Unit of our institution as a service evaluation (STH 20830) which does not require informed consent. We confirm that the manuscript complies with all instructions to authors and all authors listed have met the authorship requirements. The final draft was reviewed and approved by all authors. This manuscript has not been published elsewhere and is not under consideration by another journal.
Author Contributions
Amin Mohamed Abu Baker: formal analysis, visualisation, methodology, writing – original draft, and writing – review and editing. Sachin Watve: funding acquisition, investigation, project administration (lead), supervision, and writing – review and editing. Sabrina Arabel Eltringham: data curation, investigation, and writing – review and editing. Amanda Forrester: data curation, investigation, and writing – review and editing. Vikrant Devgire: investigation, formal analysis, and writing – review and editing. Krishnan Padmakumari Sivaraman Nair: conceptualisation, methodology, project administration, supervision, formal analysis, and writing – review and editing.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Statistical support: Dr Kathleen Baster; Scientific support: Dr Karl Brennan. This study was supported by the NIHR Sheffield Biomedical Research Centre/NIHR Sheffield Clinical Research Facility. The views expressed are those of the authors and not necessarily those of the NHS, the NIHR, or the Department of Health and Social Care. This project was supported by a financial grant from Merz, manufacturer of Xeomin. The funder had no input into the design, data collection, or analysis of the results. This research was supported by the National Institute for Health and Care Research (NIHR) Sheffield Biomedical Research Centre (BRC) and NIHR Sheffield Clinical Research Facility (CRF). The views expressed are those of the author(s) and not necessarily those of the NHS, the NIHR, or the Department of Health and Social Care (DHSC).
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Amin Mohamed Abu Baker has no disclosures to make. Dr Sachin Watve has no disclosures to make. Dr Sabrina Arabel Eltringham has no disclosures to make. Amanda Forrester has no disclosures to make. Dr Vikrant Devgire has no disclosures to make. Prof. Krishnan Padmakumari Sivaraman Nair received a speaker honorarium from the Neurology Academy and has a leadership/membership role in the British Society of Physical & Rehabilitation Medicine and the Royal College of Physicians UK.
