Abstract
Introduction:
Many people with spasticity or pain managed with intrathecal drug delivery pumps have very impaired mobility and are at high risk of developing pressure sores, which may prevent them from travelling to a clinic in their wheelchair. Some patients may be transported by stretcher, but some will be bedbound at home. Routinely refilling all pumps at home is established practice in several parts of Europe and USA.
Method:
Freedom of information requests were emailed to trusts and health boards hosting intrathecal drug delivery services in the UK and Ireland, asking about frequency of home visits for pump refills, how many staff travelled, use of prefilled syringes and how they would manage a patient’s long-term inability to attend clinic.
Results:
Detailed data was received from all but 1 of 54 active clinics in the UK, but Irish legislation did not allow this data to be returned. Two thousand one hundred forty-five patients were managed with intrathecal pumps in the UK. Eleven services had less than 10 patients and 5 had more than 100. Twenty-one services did less than 1 home refill/year and 9 did more than 12/year. Twenty had travelled to patients more than 50 miles from the clinic, or to an island. Twenty-two services would send 2 clinicians and 10 would bring prefilled sterile syringes, rather than aspirate from bottles at the bedside.
Conclusion:
FoI requests can generate a very high response rate in the UK. Many services manage a small number of patients, which would make it difficult to provide cover when a single-handed clinician is unavailable or to train a second clinician.
Introduction
Following a reduction in available clinic space due to social distancing rules during the COVID pandemic and later relocation of the rehabilitation medicine clinic, we were no longer able to accommodate patients arriving by stretcher for intrathecal baclofen pump refills, and hence many more patients were seen at their homes instead. This prompted a local service evaluation, to identify adverse incidents and ways to improve the service. This in turn prompted a national survey of practice when patients with intrathecal drug delivery pumps cannot attend clinics. The focus of this report is on intrathecal drug delivery pump refills outside the usual clinic, and I did not explore differences in practice between people being treated with baclofen for spasticity or with morphine for pain. Freedom of information requests were used, to explore the efficiency of this method for gathering non-patient identifiable, routinely collected information from around the UK.
Method
Medtronic UK supplied a list of the trusts to whom they supplied pumps or refill kits in the UK and Ireland. A few more services were identified from a nationwide email forum (https://www.jiscmail.ac.uk/cgi-bin/webadmin?A0=INTRATHECAL-BACLOFEN). The Freedom of Information request email address was found on each trust or relevant Health Board’s website. The following email was sent to each, in December 2023:
“I am writing to all hospitals in UK and Ireland which provide intrathecal therapy services to enquire about their practice or policy for managing patients with pumps, when they cannot attend the clinic for a refill. This situation usually occurs because of intercurrent acute illness or pressure sores, but sometimes because of inadequate hospital transport services. I am using Freedom of Information Requests to ensure a high response rate, and because my hospital does not regard this as research and hence this method does not require any regulatory approval. I believe XXXXX in the YYY department is the most relevant contact. Alternatively you could ask your procurement team who has ordered Medtronic Synchromed intrathecal pumps or Medtronic 8551 refill kits. I propose to summarise these responses and write a report for publication in a medical journal. This information will inform service design and planning, particularly when key staff are retiring. It will also update a list of active services. What is the address of your base refill clinic, with postcode? How many How many How many How many Approximately how many pump refills does your service do each month at your usual locations ? Do you accept people attending on a stretcher for refills? How often do you refill pumps away from your base hospital clinic? Eg Once/month, once/year, once in 5 years What is the furthest you have travelled from your clinics to refill a pump, in miles or time, in the past 3 years? For the first time, a patient is unable to attend the clinic by wheelchair or stretcher for a planned refill, because of acute illness or transport failure. They are expected to recover and be fit to attend refill clinics in the future. They are a 90-minute drive from your usual refill service base and there is no more local service which could refill the pump before it runs out. Would you plan:
(a) no refill and when pump runs dry advise local doctors to manage withdrawal symptoms with medication by another route.
(b) staff from base clinic travels to refill pump at patient’s home or local hospital, before it runs dry.
Who would travel? Doctor who runs the service? doctor in training who is rotating through the service? Nurse who refills pumps in clinic? Do you arrange second person to travel to chaperone, check procedure or open vials to maintain sterility? No. /Yes, then who? Or a video call to check procedure/programming? When refilling away from usual clinic, would you ask pharmacy to prepare a sterile syringe with the total drug to be injected into the pump, or would you aspirate from separate vials at their home, as is usually done in clinics?
(c) patient transferred to your hospital for in-patient management and refill
(d) Other, please explain
If you have refilled it once while they are acutely unwell, but subsequently the patient is unable to attend the out-patient clinic by wheelchair or stretcher for any future refills, and resides 90 minutes’ drive from your usual refill service base, and there is no more local service which does home refills to take over long term management, would you plan:
(a) no visits for further refills or dose reduction
(b) no further refills but home visits to reduce ITB dose in stages before pump runs dry.
(c) regular home visits to refill pump until end of battery life, but pump not replaced.
(d) regular home visits to refill pump indefinitely and pump replacement at end of battery life.
(e) patient travels by stretcher ambulance, admitted overnight for regular refills
(f) Other, please explain.”
Where no response was received after further emails to the FoI address or it was suspected that the FoI response had omitted relevant services, individual clinicians were emailed for data.
The Cambridge intrathecal baclofen service data for home refills since 2006 (when letters were created and stored as easily searchable computer files), was examined in a service evaluation in 2022, Clinical Project ID4617 PRN10617.
Results
Response to FoI requests:
This method did not work in Ireland, as the FoI offices explained that Irish legislation required public hospitals to release Whereas individual hospital trusts are responsible for responding to FoI requests in England, FoI requests in Wales, Scotland and Northern Ireland are handled by Health Boards and, being more remote from the clinicians, some omitted relevant services and data. “We are not required to create information or to provide thoughts, opinions or professional judgements where these are not already held in recorded form.”
Time to respond:

Services:
Number of intrathecal pump services operating 54. Number closed 20
Not all questions were answered by every service, so the number of responses varies.
Number of patients in each service.

Total 2145 patients
Services accepting stretchers: all services except Cambridge would manage patients arriving by stretcher, and some commented on their process: “transfer them to a bed on the neuroscience day unit”; “we generally see these patients at home for refill”; “it is becoming a problem, as we cannot always accommodate transferring them to an alternative bed whilst waiting for return transport to arrive. The Trust has stopped paying for Wait and Return for stretcher patients”;
First inability to attend:
If a patient was unable to attend the refill clinic, 34 services would plan a home visit. Two would refill outside the clinic only if the patient was on another ward on campus. Four would plan admission to hospital for a refill.
Frequency of home visits:

In 8 services, a single clinician would visit the home, a nurse from 3 and a doctor from 5. Twenty-two services would send 2 clinicians, a doctor (11) or nurse, physio or clinical scientist (9), usually accompanied by a nurse or physio from the ITB service, but sometimes a local district or palliative care nurse. One service emphasised the need for staff to have motoring insurance which included cover for business driving.
Ten services would bring prefilled sterile syringes, prepared by their pharmacy. Almost all of these services routinely used prefilled syringes in their clinic. One service did not have access to pharmacy which could do this.
Distance:

When a patient was not going to be able to attend the clinic in the long term, 14 services would not refill, of whom some would visit to gradually reduce the dose; 5 would plan to admit overnight or to a day-case unit for refills; 3 would refill regularly at home but not replace the pump; 6 would refill regularly at home and replace the pump and 23 did not have a policy as the event was too rare to plan for.
Cambridge Service evaluation:
Number of home visits

Home visits are much less frequent now (13 in first 9 months of 2024-2025), as patients have died or been weaned off intrathecal baclofen as the pump reached its end of life.
Reasons for a home visit
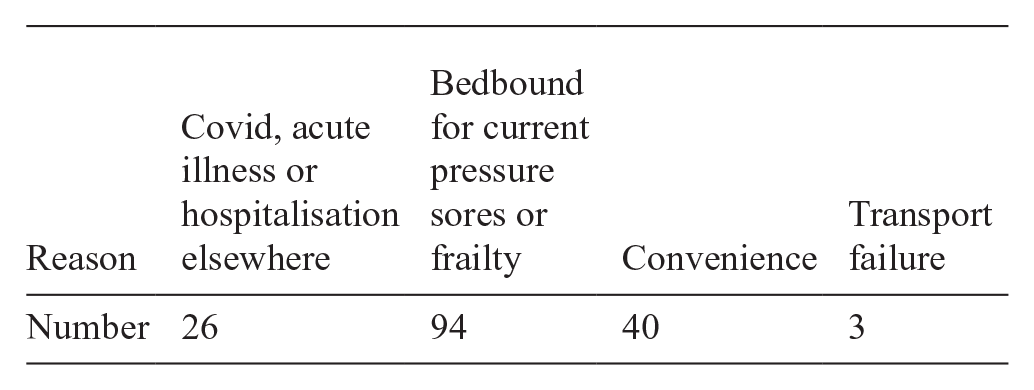
Patients refilled at home for “convenience” lived near the doctor’s usual route while driving from peripheral clinics and if they travelled to the base clinic the journey would threaten their previous pressure sores. Once patients had become accustomed to this during the Covid pandemic, several were very keen that refills at home continued after hospital appointment restrictions were lifted. Seeing them at home released an appointment slot in the base clinic. Many could have travelled to clinic by stretcher, but there is no space to accommodate them, and unpredictable transport failures are more disruptive than a home visit planned months in advance.
Distance
Mean 1 way distance from base clinic to site of refill: 30.6 miles
Mean 1 way detour from usual route for all visits: 8.9 miles
Adverse Incidents
Treating patients, by refilling their pump, in other hospitals without an honorary contract of employment with that hospital (4 occasions).
Before access to electronic medical records was possible remotely, the clinical note and letter were much delayed on 3 occasions, on 5 occasions an appointment for their next refill was not made. Because of this, pumps ran dry on 2 occasions, fortunately without ill effects.
The doctor was unable to get to see the patient at their home on 3 occasions: on one, she had gone to the hairdresser; once she was alone and couldn’t open the door and I did not have the key safe code, and on 1 occasion, the patient had died 2 days earlier.
Five drug related events were due to the drug getting hot in the boot of the car during very hot weather (1), wasting the drug which had been drawn up into a syringe, before discovering that the pump had flipped over and could not be accessed (1), forgetting to collect the drug from pharmacy before going to peripheral clinic (1), dispensing being delayed (1) and incorrect volume being prescribed (1).
Suboptimal conditions for the refill on 4 occasions were having to draw up and mix baclofen and morphine on the windowsill of the intensive care unit; rushing to refill pump in another hospital’s ITU while the patient was being prepared for urgent surgery; doing it in the patient’s toilet; with difficult access to the pump while patient sitting oddly in her wheelchair; and reversing the car into a low wall while leaving their driveway.
There were 5 equipment related events: the programmer not working because the batteries were exhausted (1); 10 mL Luer lock syringes being unavailable to drain the pump and subsequent failure to fit the filter when refilling(1); sterile gloves disintegrating (1); forgetting to bring alcohol swabs (1); forgetting to bring a sharps box or scissors to get into the bags in which pharmacy package their sterile filled syringes (multiple times).
Discussion
As information released by FoI requests is, by definition, publicly available and not identifiable, ethics approval is not necessary to seek and collate it. While being able to start a nationwide survey of NHS activity without waiting for R&D department or ethical committee approval saves time initially, this may be lost by delays in responses. Only half of trust FoI offices responded within 28 days, whereas direct emails to relevant clinicians produced complete information within a few days. This method has achieved a very high response rate, when supplemented by individual clinician contacts, but it is not possible to say if this would have been matched by just emailing all clinicians directly in the first place. Direct approaches to clinicians might be regarded as research, needing ethical committee approval, which would require a lot more preparatory work. The FoI method has achieved a much higher response rate than nationwide survey by email 1 to 4500 clinicians using intrathecal pumps for pain management in USA, which elicited 159 responses. A more personal approach to individual named clinicians has also been very successful and avoids limitations due to differences in legislation but does require an accurate, comprehensive address list to start. 2
It is easy to find a trust’s FoI email address, which is useful when the relevant clinician’s name or address is not known. This method did not work in Ireland, as Irish legislation requires public hospitals to release existing documents and records, but not to answer questions or create new reports. Also, many of the hospitals which have managed ITB pumps are private and are not required to respond to FoI requests. Scottish legislation did not require Health Boards to answer my “hypothetical” questions and responses from the FoI offices based in health boards, were less complete than from those based in individual trusts in England. This suggests surveys using FoI requests may work best in England alone.
Half of all reported patients were managed at 10 centres with more than 63 patients. At the other end of the range, 11 centres managed less than 10 patients. A similar pattern of wide variation in use of intrathecal drug delivery systems was reported in 2015 with pump implantation rates of 22/million to1/million in different parts of England. 3 It is likely that most of the services which are no longer operating, closed due to single consultants retiring, but some may have been due to consolidation of several services in a city onto 1 site. This raises the question of how big a service needs to be to be sustainable, and how many staff able to refill pumps should be involved in any service, to ensure that a service can continue when 1 person is unavailable. The number of refills performed/week will also influence how practical it will be to train someone to become competent to refill and reprogramme pumps independently.
All but 1 service accepted patients arriving by stretcher. Two explained that they would refill the pump while the patient stayed on the stretcher, sometimes in the ambulance, which requires the paramedic crew to wait. One service was reluctant to continue paying for this. A small number had access to a bed in their day-case unit.
On the first occasion that a patient could not attend, the great majority of services would go to the patient’s home. Among those who had not needed or been able to do this, some services would attend other wards within the hospital. Only 16 services did home visits more often than 2/year, which may not be sufficient to allow development of a standard routine or protocol. All but 8 services would send 2 staff.
Only those services whose pharmacies routinely prepare sterile syringes for every patient attending clinics would use a sterile syringe at home. A prefilled sterile syringe would make it easier to maintain sterility when only 1 person goes out to refill a pump, rather than aspirating from vials at the bedside.
A much smaller number of services would be able to keep going to a patient’s home, and most would plan to wean patients off the pump and convert to alternative methods of managing spasticity or pain. The Cambridge service was able to keep refilling pumps and weaning off as the battery life expired because the relevant consultant had regular clinical commitments at several sites each week, routinely did home visits to see bedbound patients, refilled pumps by himself and had access to a pharmacy who could make up sterile syringes for patients requiring bespoke concentrations.
Pump refills in patients’ homes are standard practice in some areas of Netherlands,4,5 Belgium, 6 Denmark7 -9 and USA, 10 where specialist agencies,11 -13 have been doing this for 30 years. Standards and guidelines for managing intrathecal drug delivery pumps14 -20 do not comment on home refills. A training curriculum for nurses has been developed. 21 Refilling the pump at home does not reduce the need for regular general reassessment of their condition by the doctor managing and prescribing their medication, which can be done at home if a doctor visits, but may still require the patient to travel to the hospital clinic in many services.
In Europe the higher density of pumps within a 50 km radius may make routine practice of all refills at home by a dedicated nursing team economically viable. Alternatively, the much greater distances some patients may have to travel to a clinic in USA may make it impractical for them to travel for many hours. The density and distances may not be enough to make such a service economically viable in the UK.
This information will inform debate about creating internal policies to avoid these adverse incidents and wider standards for home pump refills. While a standard requiring 2 staff or prefilled sterile syringes might further reduce some low frequency but dangerous risks, it probably would not have prevented the adverse incidents documented here. It may be impossible for some services to meet such a standard and hence they may be prevented from refilling a pump at home. This would increase the risk that patients’ pumps run dry before they can attend the clinic, which would carry a greater risk of morbidity than from a single person refilling from individual glass vials at the bedside. Recurrence of the adverse incidents we have experienced have largely been prevented by prompts from the departmental secretary and clinic nurse, remote access to electronic medical records and phoning the patient in advance.
Supplemental Material
sj-docx-1-rpo-10.1177_27536351251341016 – Supplemental material for Refilling Intrathecal Drug Delivery Pumps Outside the Clinic: Survey of Practice in the United Kingdom
Supplemental material, sj-docx-1-rpo-10.1177_27536351251341016 for Refilling Intrathecal Drug Delivery Pumps Outside the Clinic: Survey of Practice in the United Kingdom by Stephen G. B. Kirker in Advances in Rehabilitation Science and Practice
Footnotes
Acknowledgements
I acknowledge the help of Medtronic for identifying hospitals who have used their pumps and many clinicians who responded to the Freedom of Information requests. I am very grateful to all the clinicians who supplied data for this survey, who I hope will find this summary of interest.
Author Contributions
Stephen G. B. Kirker, conceived this study, collated and analysed the data returned by many hospitals and wrote the paper.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
A list of active intrathecal pump services in UK with addresses and types of patients they treat, is Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
