Abstract
Background
Romdoul (Sphaerocoryne affinis) is a flowering plant of the Annonaceae family and has been used customarily in folk medicine. The bioactivities of this plant, especially the anti-cancer effect, however, remain surprisingly few.
Objective
this study aimed to elucidate the anti-leukemic effect of romdoul fruit extracts and their underlining mechanisms.
Methods
The extracts were prepared from fresh fruits and the phytochemical contents were evaluated by biochemical assays and HPLC method. The promising extract was identified via the inhibition of HL60 as well as normal NIH-3T3 cell densities utilizing MTT assay. The underline mechanism of the extract’s effect was studied by accessing the treated HL60 cell population overtime (via MTT assay). The morphology of abnormal cells was examined by bright-field microscopic imaging. Hallmarks of apoptosis including nucleus characteristics and caspase 3 activation were analyzed by fluorescence imaging. The underline mechanisms of apoptosis and proliferation inhibition were accessed via RT-qPCR examination of involved genes.
Results
Our findings showed that the ethyl acetate extract of romdoul fruit (SA-EA) was found to be an exceptional anti-leukemic candidate (IC50 was as low as 4.11 μg/mL). More interestingly, the treated HL60 cells expressed nuclear fragmentation and caspase 3 activation, indicating the effect could follow an apoptotic mechanism. Importantly, the transcription assessment of apoptotic and proliferative genes suggested that SA-EA might suppress the growth of HL60 cells and induce p21-dependent apoptotic pathway.
Conclusion
This study demonstrated one of the first scientific evidence for the anti-cancer activity of Sphaerocoryne affinis fruit-derived extract. Thus, our findings exhibited a novel and promising anti-leukemic candidate for future studies.
Introduction
Leukemia, a malignant transformation of blood-forming cells, was the 11th most lethal cancer globally accounting for more than three hundred thousand deaths in 2020 according to Global Cancer Observatory. 1 Moreover, the economic burden that involves leukemic diagnosis, treatment, and related toxicity management is dauntingly increasing.2,3 In addition, common leukemia treatments (i.e., chemotherapy, radiation, and stem cell transplantation) may pose numerous downsides which seriously deteriorate various organs and tissues of cardiovascular, skeletal, neuronal, endocrine, immune, and gastrointestinal systems.4-9 Hence, there is an urgent need for developing novel, safe, and cost-effective cancer therapeutic regiments. Among reasonable candidates, medicinal plants have emerged as a potential source for anticancer screening. Several renowned plant-derived compounds have been extensively prescribed (e.g. vincristine, paclitaxel, vinblastine, etc.) or studied in late-phase clinical trials (e.g. quercetin, betulinic acid, curcumin, etc.).10-12
Romdoul (Sphaerocoryne affinis (Teijsm. & Binn.) Ridl) is a flowering plant in the Annonaceae family which has prevalently occurrence in Southeast Asia areas such as Borneo, Cambodia, Java, Malaysia, Philippines, Thailand, and Vietnam. On the one hand, the flowers, and fruits of S. affinis have been used traditionally as cardiac stimulant, anti-ulcer, dizziness, and digestive problem treatments as well as reducing cancer progression; on the other hand, its bark has been harnessed to relieve fever and insect’s bite.13,14 More importantly, several scientific research strongly indicated the anticancer activity of extracts and compounds from parts of S. affinis toward cervical and breast carcinoma cells.14–16 In addition to the anticancer activity, the water fraction of the S. affinis fruits was proved to be a robust antioxidant regimen in the Drosophila model. 17 The bioactivities of S. affinis even extended toward anti-plasmodial trait as demonstrated by Abdul-Wahab Salae et al. 18 It is reasonable that these bioactivities, especially anticancer features, might come from the contained phytochemical constituents of S. affinis. Benzylated flavonoids as well as phenolic compounds were isolated from leaves, stems, bark, fruits, and flowers of S. affinis.19,20 It is worth noting that plant-derived flavonoid and phenolic compounds have been reported to possess effective anticancer activity, especially inducing apoptosis in cancer cells.19–22 Moreover, many species belonging to the Annonaceae family were identified as potential sources for antineoplastic agents. 13 Compounds isolated from Melodorum fruticosum, a member of Annonaceae family and a close relative of S. affinis, demonstrated the effective inhibitory activity toward many cancer cell lines.23–28 Recently, Le-Trung et al. reported the substantial toxicity against cervical cancer HeLa cells of ethyl acetate and hexane extracts of S. affinis fruits as well as acetylmelodorinol isolated from the ethyl acetate extract. These extract and compound provoked apoptosis and arrest cell cycle of HeLa cells via various mechanisms including DNA damage, increased Bax/Bcl-2 expression, and AKT/mTOR pathway activation.14,15 Works of Le-Trung et al. significantly stress the promising anti-cancer activity of substances from S. affinis fruits. Surprisingly, there has been no report about anti-leukemic activity of S. affinis.
Therefore, in this study, the anticancer effects of extracts from S. affinis fruits cultivated in Vietnam were examined on HL60 human leukemia cell line. We also analyzed the anticancer mechanism of potential extract, focused on the apoptosis induction and growth inhibition.
Methods
Plant Material
S. affinis (local name: “chùm đuông”) fruits were collected from Tay Ninh province, Vietnam. Binomial nomenclature and voucher specimen (PHH0004912) were identified and preserved by PhD. Dang Le Anh Tuan, Laboratory of Ecology, University of Science, Vietnam National University Ho Chi Minh City, Vietnam.
Preparation of Fractional Extracts
Fresh fruits of S. affinis were collected and freeze-dried until the flesh of fruits became fine powder. The dried material was then extracted using 70% ethanol (Xilong, China) (ratio of 10% w/v) at room temperature for 24 hours. Obtained solution was filtered and condensed using a rotary evaporator (Hahnvapor, Korea) at 40°C. Half of the solution was dried using a freeze-drier (Benchtop K Virtis, USA) to obtain the ethanol extract (SA-EtOH), while the remaining half was consecutively mixed with petroleum ether (Xilong, China) and ethyl acetate (Xilong, China) solvents (ratio 1:1) using separatory funnel. The ethyl acetate (SA-EA) and petroleum ether (SA-PE) fractions were filtered and concentrated using the rotary evaporator and freeze drier. The remained water fraction (SA-H2O) after organic solvent separation was also freeze-dried. All dried extracts were weighed using analytical balances scales (Sartorius, Germany) and reconstituted with absolute DMSO (Merck, Germany) to prepare a stock solution. The stock solutions were stored at −30°C until being diluted in phosphate-buffered saline (PBS, Merck, Germany) to experimental concentration, then sterilized by 0.22 μm filter and stored at 4°C for cytotoxicity assay. The main steps for preparing the extracts used in this study were presented in Supplemental Figure 1.
Qualitative Phytochemicals Analysis
Phytochemical constituents in fractional extracts were detected using screening methods referred to previous reports,29-31 including Molisch’s test for carbohydrates, Mayer’s and Wagner’s tests for alkaloids, foam test for saponins, alkaline reagent test for flavonoids, lead acetate test for phenolics, ferric chloride, lead acetate and HCl hydrolysis tests for tannins, Salkowski’s test for steroids, Trim-Hill test for terpenoids. All of the chemicals used for these assays were purchased from Xilong Chemical (China). The flavonoids and phenolics contents of SA-EA were also examined via HPLC method following the procedure from previous report. 32
Cell Lines and Culture Media
The HL60 human leukemia cell line was grown in RPMI 1640 medium (HiMedia, India) supplemented with 10% fetal bovine serum (Merck & Co., USA). The murine fibroblast cell line NIH-3T3 was grown in DMEM high glucose medium (Merck & Co., USA) supplemented with 10% bovine calf serum (Merck & Co., USA). Cells were maintained in humidified incubator (Esco, Singapore) with 5% CO2 at 37°C.
In Vitro Cytotoxicity Screening Assay
Suspension HL60 cells were seeded into each well of a 96-well plate (Corning, the USA) at density of 1 × 104 cells/95 μl/well in medium containing 5% serum and incubated for 1 hour. For fractional extract screening, 5 μl of SA-EtOH, SA-PE, SA-EA and SA-H2O extracts were added into each well to the final concentration of 40 μg/mL. For cytotoxicity assay of potential extracts, an optimized MTT assay was utilized.
33
Briefly, the serial diluted concentrations of extract solution in medium containing 5% serum were added to each well to the final concentrations ranging from 0.8 to 200 μg/mL. The same DMSO concentration in extract solution (0.05%) was used as negative control. After 48 hours of treatment, cells were treated with 0.25 mg/mL MTT (3-(4,5-Dimethylthiazol-2-yl)-2,5-Diphenyltetrazolium Bromide) (Sigma-Aldrich, Germany) for 4 hours and the formazan crystals was dissolved by adding 60 μl SDS lysis buffer (0.3 g/mL SDS, 0.03 N HCl) and 90 μl DMSO (Merck, Germany) into each well. The plates were shaken at 700 rpm for 10 minutes and measured absorbances at the wavelength 550 nm (OD550nm) by Multiskan plate reader (Thermo, the USA). Optical density values were converted to inhibition percentage according to the formula:
The half maximal inhibitory concentration (IC50) value was interpolated from nonlinear sigmoidal dose-response curves by GraphPad Prism. Selectivity index (SI) was obtained by calculating the ratio of IC50 on normal fibroblasts to that on cancer cells. Potential extracts from screening and cytotoxicity assay were then subjected to further analysis.
Evaluation of Cell Proliferation Rate and Cell Morphology
After being seeded into each well of 96-well plate in a similar way to cytotoxicity assay, HL60 cells were then treated with SA-EA extract at IC50. In addition, HL60 cells were also treated with the chemotherapy drug doxorubicin at 0.1 μg/mL for 24 hours. The cell density was determined using MTT assay and the cell morphology was observed by Nikon Eclipse TiU microscope system (Nikon, Japan) at different incubation times (0, 24, 48, 72 and 96-hour time points).
Detection of Nuclear Fragmentation and Caspase 3 Activation
Nuclear fragmentation was detected by DAPI staining. The level of activated caspase 3 was investigated by cleaved caspase 3 staining using anti-cleaved caspase 3 antibody (product number: 9661, Cell Signaling Technology). HL60 cells were seeded into each well of a 96-well plate in a similar way to cytotoxicity assay and treated with SA-EA extract at IC50. After treatment time, cells were fixed in 4% paraformaldehyde (Scharlau, Spain) for 10 minutes and permeabilized with 0.05% Triton X100 (Pancreac AppliChem, Spain-Germany-Italia) for 5 minutes in a humid chamber at 37°C. Fixed cells were washed gently with PBS and incubated with 10% normal goat serum (NGS) (Vector Laboratory, Japan) for 30 minutes, stained with anti-cleaved caspase 3 antibodies (product number: 9661, Cell Signaling Technology) (1:200) v/v for 1 hour, washed and co-stained with secondary antibodies (1:500, product number: A-11008 Invitrogen, USA) and DAPI solution (product number: 62 248, Thermo, the USA) for 1h. The staining sample was washed with PBS and immediately observed under Nikon Eclipse TiU fluorescence microscope system (Nikon, Japan).
Determination of Apoptosis-Related and Proliferation-Related Genes Expression
To investigate the mRNA expression level of apoptosis-related genes BAX, BAK, BID, BCL-2, APAF-1 (encoding BAK, BAX, TBID, BCL-2, and APAF-1 protein), proliferation-related gene CDKN1A (encoding p21) and PCNA (encoding PCNA protein), the quantitative reverse transcription polymerase chain reaction (qRT-PCR) was carried out.
HL60 cells were seeded into each well of a 24-well plate (Corning, the USA) in a similar way to cytotoxicity assay and treated with SA-EA extract at IC50. After 24-hour treatment, total RNA was isolated using Trisure solution (Bioline, UK) according to the instruction of the manufacturer. Five hundred nanogram of total RNA sample was reverse transcribed into cDNA using Tetro cDNA Synthesis Kit and oligo-dT primers (Bioline, UK). One microgram of cDNA templates was subjected to realtime PCR amplification using SensiFASTTM HRM Kit (Bioline, UK) and primers (Phu Sa, Vietnam) in LightCycler96 instrument (Roche Diagnostics, Germany). The qPCR program comprised an initial denaturation step at 95°C for 10 minutes, followed by 45 cycles of denaturation step at 95°C for 15 seconds, annealing step at annealing temperature for 15s, and extension step at 72°C for 20s. The sequences of qPCR primers for analysis were referred to previous studies34-38 as follows: BAX-F 5′-CAAACTGGTGCTCAAGGCCC-3’; BAX-R 5′-GGGCGTCCCAAAGTAGGAGA-3’; BAK-F 5′-TTTTCCGCAGCTACGTTTTT-3’; BAK-R 5′-TGGTGGCAATCTTGGTGAAGT-3’; BID-F 5′-TGGACTGTGAGGTCAACAACG-3’; BID-R 5′-AGTCTGCAGCTCATCGTAGCC-3’; BCL-2-F 5′-CTGGTGGACAACATCGCCCT-3’; BCL-2-R 5′-TCTTCAGAGACAGCCAGGAGAAAT-3’; CDKN1A-F 5′-GTCACTGTCTTGTACCCTTGTG-3’; CDKN1A-R 5′-CGGCGTTTGGAGTGGTAGAAA-3’; PCNA-F 5′-TTTGGTGCAGCTCACCCTG-3’; PCNA-R 5′-CGCGTTATCTTCGGCCCTTA-3’; ACTB-F 5′-TCCTGTGGCATCCACGAACT-3’; ACTB-R 5′-GAAGCATTTGCGGTGGACGAT-3’. Gene expression levels were analyzed using LightCycler96 software. The expression levels of above genes were normalized to the expression level of housekeeping gene ACTB (β-actin) in each sample by the formula 2−ΔCt.
Data Analysis
Data was analyzed by GraphPad Prism 8 software. Cytotoxicity experiments were performed in three replicates and the results were expressed as growth inhibition percentage (I%). IC50 values were interpolated from sigmoidal dose-response curves with nonlinear regression model using GraphPad Prism 8 software. The results were described as mean ± standard deviation (SD). The differences between mean values were analyzed by unpaired Student’s t-test. Differences were statistically significant if P < 0.05.
Institutional Approval
The contents of this research (project number: SH-CNSH 2022-07) were approved by University of Science, Vietnam National University Ho Chi Minh City, Viet Nam.
Results
Cytotoxic Activities of S. affinis Extracts on Leukemia HL60 Cell Line
To identify the potential extracts against tested cancer cell line we evaluated the effect of the four fractions at the concentration of 40 μg/mL on cell growth. The screening results indicated that SA-EA, which strongly inhibited more than 90% of cell growth, was the most lethal extract toward HL60 cell line (Figure 1(a)). Further experiments revealed the IC50 value of SA-EA on HL60 cells was 4.11 μg/mL (Figure 1(b)). These results were in line with the guideline of National Cancer Institute (NCI), in which a crude extract, whose IC50 value on a cell line lower than 20 μg/mL, is considered as a highly cytotoxic agents to that cell line.
30
Therefore, the anticancer activity of SA-EA extract on HL60 cancer cells was investigated in subsequent experiments. Cytotoxic activities of S. affinis extracts and ethyl acetate fraction (SA-EA). A: Cytotoxicity screening of four fractional extracts on HL60 cell line at the concentration of 40 μg/ml. B: Dose-response curves of inhibitory activity of SA-EA extract on HL60, and NIH 3T3 cell lines. C: Inhibitory effects of SA-EA on HL60 cell line over time. D: Percentage of inhibition of SA-EA (4.1 μg/ml) on HL60 cells over time. Each value represents mean ± SD of three independent experiments. The statistical differences were analyzed by two-tailed Student’s t-tests (ns P > 0.05, *P ≤ 0.05, **P ≤ 0.01, ***P ≤ 0.001).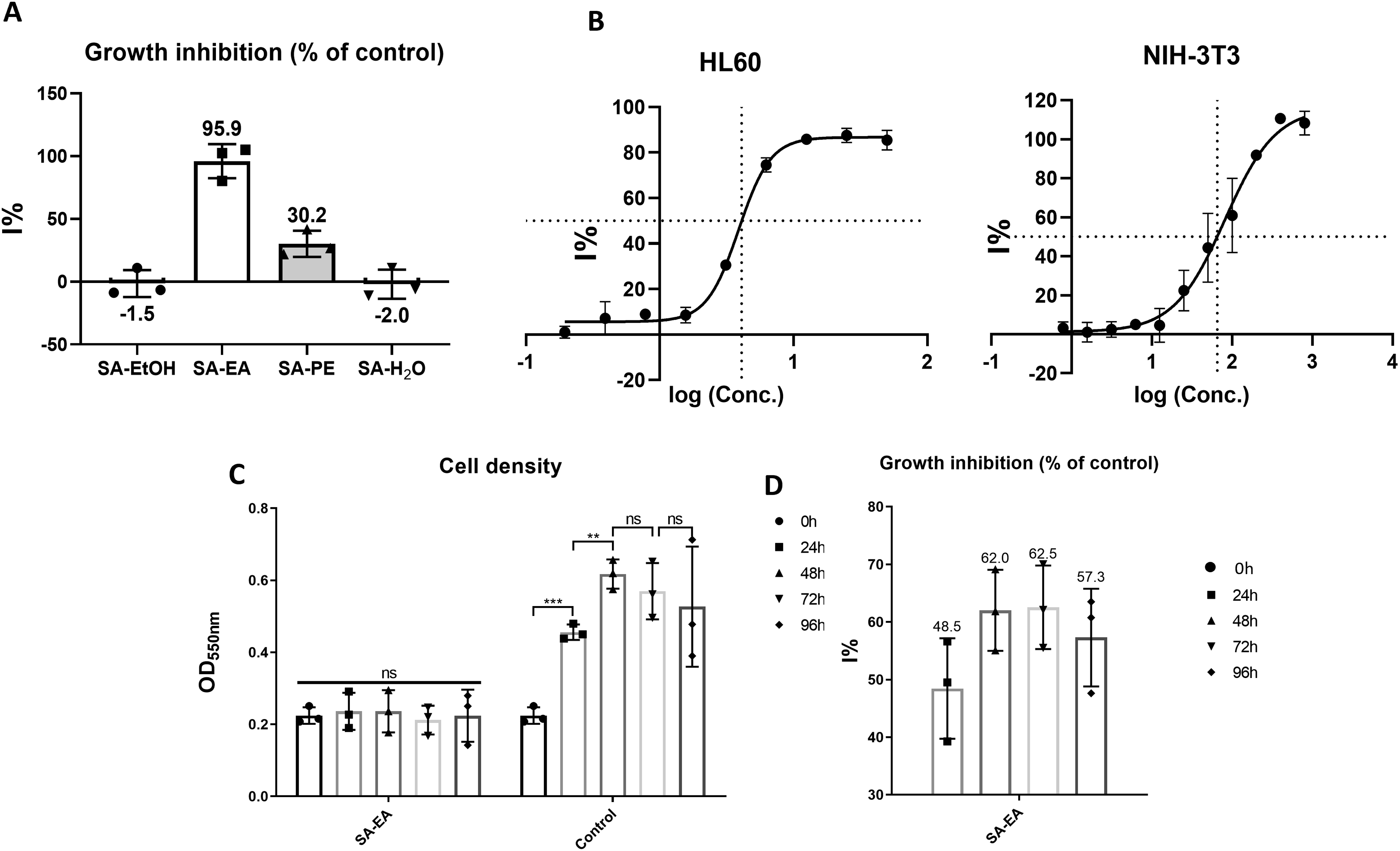
Besides being highly lethal against cancer cells, a potential extract must demonstrate relatively low cytotoxicity toward non-cancer cells. This characteristic is demonstrated via the selectivity index (SI). Briefly, the SI value >2 indicates the anticancer activity of the given agent is highly selective against a particular cancer cell line. 39 Hence, we also examined the cytotoxic specificity of SA-EA on NIH-3T3 normal fibroblast cell line with this extract (Figure 1(b)). The relatively high IC50 value (65.12 μg/mL) of SA-EA indicated the specificity (i.e., selectivity index (SI) = 15.89) of this extract against cancer cells.
For further investigation on the time-dependent effects of SA-EA on HL60 cells, we treated the cells with SA-EA at the concentration of 4.1 μg/mL and evaluated the changes in cell density for 96 hours. SA-EA-exposed HL60 cell density showed no significant change at all time points as opposed to the augmented population of the control group, especially in the first 48 hours (Figure 1(c)). Hence, the inhibitory activity of SA-EA on HL60 population increased from 48.46% to 62.54% throughout the course of the experiment (Figure 1(d)). This result indicated that SA-EA could inhibit HL60 growth in a time-independent manner. All these results strongly suggested the anticancer property of SA-EA. In addition, the unchanged cell population might be originated from the hindered proliferation and/or the balance between induced cell death and cell replication. Therefore, we parallelly qualified the potent phytochemical groups in SA-EA and accessed the morphology of treated HL60 cells.
Qualitative Phytochemical Analysis of S. affinis Fruits
Qualitative Analysis of Common Phytochemical Groups in S. affinis Fruit Extracts.
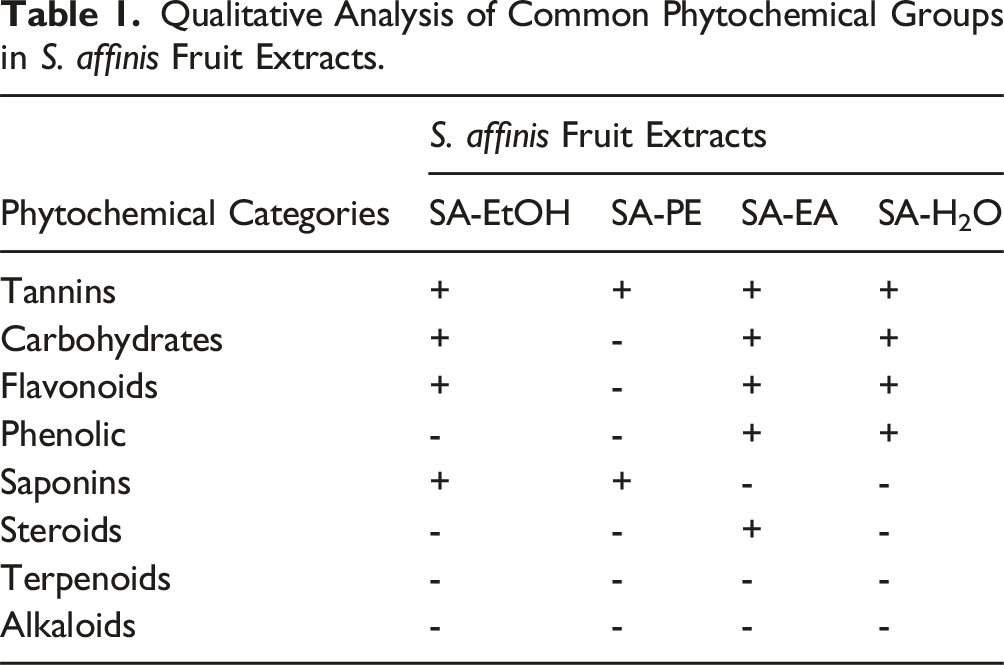
Since various parts of S. affinis have been reported to contain flavonoid and phenolic compounds19,20 together with the fact that SA-EA was shown to be the most potent extract against HL60 cells (Figure 1), we also performed the HPLC analysis of these chemical groups in SA-EA (Supplemental Figure 2). The presence of quercetin and kaempferol (concentrations of 0.32 and 0.19 mg/mL respectively) was confirmed by two signal peaks whose retention time was the same with reference standards (Supplemental Figure 2A, B). Other flavonoid and poly phenolic-related compounds were also analytically found in SA-EA via three peaks with retention time in the range of 3.6 to 18.1 minutes, which are correlated with gallic acid (1.48 mg/mL), epicatechin gallate (92.45 mg/mL), and epigallocatechin gallate (0.23 mg/mL) respectively (Supplemental Figure 2C, D). Interestingly, these compounds have long been shown to exert potential anticancer properties via numerous mechanisms.44-46 Thus, the HPLC analysis results not only proved the phytochemical consistency of our S. affinis fruits in comparison to previous studies but also further consolidated the anticancer potential of S. affinis fruit extracts.
SA-EA Induced Morphological Alterations of HL60 Cells
To study the underlining mechanism of SA-EA on HL60 cells, we first examined the morphological changes of HL60 cell line under SA-EA treatment at IC50 via bright-field microscopic images captured at different time points (from 24 to 96 hours).
The featured morphological characteristics and size homogeneity of non-treated group remained unchanged till the end of the experiment (Figure 2). Specifically, HL60 cells appeared as spherical shape with bright and intact membrane. Morphology of HL60 cells under SA-EA treatment. A: HL60 cells treated with SA-EA (4.1 μg/ml) and 0.05% DMSO (control) for 96 hours. B: HL60 cells treated with doxorubicin (0.1 μg/ml) for 24 hours. Red arrows indicate spindle-shaped cells, blue arrows indicate shrunk cells, and black arrows indicate blebbing/fragmented cells. The scale bars represent 50 µm.
In SA-EA-treated HL60 sample, there are three types of abnormal morphology emerged at different time points. The first unusual morphology is spindle-shape with long tails at both opposite ends which appeared as early as 24-hour time point and diminished after 72 hours of SA-EA treatment (Figure 2). This abnormal morphology bears similar trait with the mitotic catastrophe wherein the cell replication is halted. The incomplete cell division might subsequently lead to programed cell deaths, most commonly apoptosis. 47 This explained, to some extent, the proliferative inhibition effect (Figure 1) and apoptotic-liked morphology observed in later time points. Shrunk cells and cells with blebbing membrane, surrounded by small bodies were the other two abnormal shapes, which emerged at 24-hour time point and gradually augmented till the end of the experiment (i.e., 96 hours) (Figure 2). These shrunk and fragmented cell morphologies were similar to the morphology of doxorubicin treated HL60 cells, which were shown to be the apoptotic traits of this cell line under doxorubicin effects. 47
Moreover, the shrinking, blebbing and fragmented cells observed in this study were similar to apoptosis cells described in previous publications. 48 Hence, the inhibition on cell growth rate caused by SA-EA could involve cell death. Since apoptosis is a common and important mode of action of many chemotherapies and the activation of apoptotic pathway is the major target of clinical drugs, 49 we hypothesized that SA-EA might induce apoptosis in HL60 cells. Therefore, we further examined other hall marks of apoptosis such as nuclear fragmentation, caspase 3 activation, and apoptosis-related gene expression.
SA-EA Induced Typical Traits of Apoptosis in HL60 Cells
Caspase 3 Activation and Nuclear Fragmentation
The activation of cysteine-aspartic acid protease 3 (caspase 3) is an important marker of apoptosis. 50 In normal cells, caspase 3 presents as inactive proenzyme. The apoptotic process is initiated by different molecules in intrinsic or extrinsic pathway which then regulate downstream caspase through the cleave process, especially caspase 3. Activated caspase 3 further activates a cascade of enzyme responsible for cytoskeleton destruction, membrane blebbing, DNA fragmentation, cell-cell, and cell-matrix adhesion deformation. 48 Thus, DNA fragmentation and activated caspase-3 are typical characteristics of apoptosis. 48
Since the treatment of SA-EA on HL60 cells resulted in membrane blebbing and cell partition, we aimed to assess caspase 3 activation and nuclear fragmentation by immunostaining using anti-cleaved caspase 3 antibody and DAPI staining, respectively.
In SA-EA exposed HL60 samples, after 24 hours of treatment, caspase 3 activation and abnormal nuclei (i.e., condensed, or fragmented) were observed in shrunk and blebbing cells whilst the spindle-shaped cells showed neither activation of caspase 3 nor unusual nuclei. At the 36-hour time point, however, spindle-shaped HL60 cells exhibited both hall marks of apoptosis. At the end of the experiment (i.e., 48-hour time point), shrunk and blebbing cells which had atypical nuclei and activated caspase 3 predominated the population (Figure 3(a)). In comparison with the non-treated controls, no sign of activated caspase 3 and unusual nucleus were observed (Figure 3(b)). This observation strongly suggested that the apoptotic cell death together with the cell division inhibition in lesser extend could be the mechanism of SA-EA effect on HL60 cells. The caspase 3 activation and nuclear morphological changes for 48 hours in HL60 cells treated with (A) SA-EA (4.1 μg/ml) and (B) 0.05% DMSO (control). Red arrows indicate spindle-shaped cells, blue arrows indicate shrunk cells, black arrows indicate blebbing/fragmented cells, yellow arrows indicate cells with fragmented nuclei, white arrows indicate cells with condensed nucleus. The scale bars represent 50 µm.
SA-EA Induced Transcriptional Alterations of Apoptosis-regulating Genes
As mentioned earlier, apoptosis is the preferred mode of action of anticancer candidates. With evidence from the former experiments, we firmly conclude the apoptosis characteristics of SA-EA treated HL60 cells. Subsequently, to further understand the apoptosis signaling pathway induced by SA-EA, we evaluated the relatively transcriptional levels of several apoptosis-related genes like pro-apoptotic genes BAX, BAK, BID, anti-apoptotic gene BCL-2, and APAF1 gene which involves in pro-caspase activation (Figure 4). Apoptosis and proliferation–related gene expression at 24-hour time point of treatment. (A) Relative mRNA levels of apoptosis–related genes in HL60 cells treated with SA-EA (4.1 μg/ml) in comparison with control cells (0.05% DMSO). (B) Relative mRNA levels of proliferation–related genes in HL60 cells treated with SA-EA (4.1 μg/ml) in comparison with control cells treated with 0.05% DMSO. Each value represents mean ± SD of at least three independent experiments. The statistical differences were analyzed by two-tailed Student’s t tests (*P ≤ 0.05, **P ≤ 0.01, ns P > 0.05).
The treatment with SA-EA, on the one hand, gave rise to a two-fold surge in BAK transcriptional expression in HL60 cells. On the other hand, other genes showed no significant alteration.
Among apoptosis regulators, BAK plays an important role in the intrinsic pathway, which auto-oligomerizes or co-oligomerizes with BAX to form pores on mitochondrial membrane leading to the decrease of mitochondrial membrane integrity. Hence, many components of the mitochondria like APAF1, cytochrome c and Ca++, etc., are released. In the cytoplasm, the formation of APAF1 complex with other proteins resulted in apoptosome that activates pro-caspase 9. This, in turn, activates other effector caspases (caspase 3/7). 51 In addition, BID is a signaling factor which directly involves in the crosstalk between extrinsic and intrinsic pathway, so BID expression increase might be the consequence of extrinsic pathway induction. 52 Previous studies showed that tBID (truncated BID by extrinsic pathway) could insert itself into mitochondrial membrane, induce the transformation and oligomerization of BAK. 53 On the one hand, the unchanged mRNA level of BID indicated that the apoptosis observed in SA-EA treated HL60 cells might not be involved, transcriptionally, in the extrinsic pathway. On the other hand, our results showed that SA-EA might induce apoptosis in HL60 cells via upregulation of BAK mRNA level. Hence, this finding suggested the involvement of apoptotic intrinsic cascade of SA-EA treated HL60 cells.
SA-EA Inhibited Cell Proliferation in HL60 Cell Lines
The lower SA-EA-treated HL60 populations in comparation to the non-treated groups (Figure 1) suggested the involvement of inhibited proliferation beside apoptotic cell death. Thus, to understand the effects of SA-EA on HL60 cell growth, we investigated the relative transcription level of CDKN1A and PCNA genes which are important in cell cycle arrest and DNA replication respectively.
PCNA gene encodes DNA replication related protein PCNA in S phase of cell cycle. PCNA also interacts with cyclin A-CDK2 or p21 to control cell cycle. 54 P21, encoded by the CDKN1A gene, block cyclin B-CDK1 complex to suppress the transition from G2 to M phase and block cyclin E− CDK2 to suppress the G1/S transition. When interacting with PCNA, p21 blocks its activity, therefore inhibiting DNA replication. 55 Moreover, p21 is also involved in initiating apoptosis. 56
The treatment of SA-EA resulted in a 10.04-fold increase of CDKN1A expression in HL60 cell line. The upregulation of p21, therefore, helps explain the growth inhibition, and to some extent the apoptotic phenomena, of SA-EA treated cells. In contrast, SA-EA induced decreased PCNA expression by 1.70 times in these cells (Figure 4). The lower level of PCNA might reduce the rate of DNA duplication, thus led to significantly slower cell density observed in SA-EA treated cells (Figure 1). Therefore, our finding suggested that SA-EA might, beside apoptotic death, hinder HL60 cells proliferation via p21 signaling pathway as well as decrease PCNA-dependent DNA replication.
Discussion
Because of the everlasting threat of leukemia globally, there is an urgent need for identifying and developing novel and effective anti-leukemic agents. Various compounds obtained from plants of Annonaceae family have demonstrated potential anticancer activities.13,28 A member of this family, Sphaerocoryne affinis, colloquially known as romduol, is commonly used in folk medicine of several Southeast Asian nations. Surprisingly, the anti-cancer activity of this plant was only reported on cervical cancer HeLa cells.14,15 This study, therefore, aimed to elucidate the effects of S. affinis fruit extracts, namely, SA-EtOH, SA-PE, SA-EA, and SA-H2O on promyelocytic leukemia HL60 cell line.
Among four extracts, SA-EA showed the most promising effects toward HL60 cells with low IC50 value (4.11 μg/mL) together with high selectivity against this cell line (i.e., SI = 15.86), which agreed with the guideline of NCI. 24 This activity might be attributed to the effects of tannin, flavonoid, phenolic and steroid phytochemical groups identified in SA-EA. HPLC analysis further confirmed the presence of several flavonoids and phenolics including quercetin, kaempferol, gallic acid, epicatechin gallate, and epigallocatechin gallate. Encouragingly, the cumulative body of knowledge has firmly demonstrated the anticancer effects as well as the underline mechanisms of these class of compounds.22,57-59 The other phytochemical group, steroids, are also potential anticancer candidates due to their specific affinity toward various receptors and enzymes.40,43 Importantly, corticosteroids have been widely used as supportive treatment for cancer patients to reduce the related complications and side effects of cancer treatments.60-62 However, the presence of steroids might also raise caution due to their adverse effects. Therefore, further tests must be performed to verify the effective and safe dosage range of S. affinis extract for anticancer applications. Interestingly, the ethyl acetate and hexane extracts of S. affinins whole-fruits and seeds also demonstrated potent cytotoxicity toward HeLa cells in previous reports.14,15 Furthermore, the IC50 value of our SA-EA (4.11 μg/mL) is relatively similar with such values of the S. affinis extracts in Le-Trung’s study (from 2.76 to 8.15 μg/mL),14,15 highlighting the consistent cytotoxicity of S. affinis fruit extracts toward sensitive cancers.
Further evaluation of the effects of SA-EA at IC50 demonstrated time-independent inhibition on proliferation of HL60 cells. SA-EA-exposed cells exhibited early noticeable morphological changes (i.e., at 24-hour time point) including elongation, shrinkage, and blebbing. These are distinct morphological features of apoptotic cells. 51 This finding prompted us to further examine the apoptotic traits in HL60 cell line under SA-EA treatment.
As induced apoptosis is a prevalent pharmacological effect of many anticancer medicines, deficiency, or suppression of proapoptotic genes, or overexpression of antiapoptotic regulatory factors might play important roles in the progression of cancers and treatment resistance.51,63 Selective induction of apoptosis is an efficient way to eliminate cancer cells with limited inflammation damage to surrounding normal cells and tissue as opposed to necrosis. 64 Necrotic cell death might release pro-inflammatory mediators which increase angiogenesis, cancer cell proliferation, metastasis and immune cell infiltration, all of which can promote cancer progression. 64 Hence, apoptosis induction is a desirable target of drug discovery.
Treatment with SA-EA at IC50 value resulted in apoptotic specific morphologies, nuclear fragmentation, and caspase 3 activation in HL60 cells. Activated caspase 3 is required to activate the caspase-activated DNase (CAD) responsible for cleaving DNA, cytoskeleton proteins and adhesion complexes. Subsequently, this cascade of signal leads to typical apoptotic morphological changes together with the detachment of cells from basement matrix, the fragmentation of DNA, and finally the formation of apoptotic bodies.48,51 Because caspase 3 is required in both intrinsic and extrinsic (death receptor) pathways of apoptosis,
53
we supposed SA-EA could induce apoptosis in HL60 cell line. Interestingly, Le-Trung et al. recently isolated acetylmelodorinol (
In intrinsic (mitochondria-mediated) apoptotic pathway, BAK (Bcl-2 homologous antagonist killer) and BAX (Bcl-2-associated X protein) play critical roles in apoptosis as proapoptotic proteins. Normally, they remain inactivated in cytoplasm by binding with pro-survival protein Bcl-2. After being activated, they indirectly stimulate the initial caspases (i.e., caspase 9), which, in turn, cleave effector procaspases 3/7. At this stage, the apoptosis is irreversible. 51 Our results showed that the transcriptionally increasing BAK levels together with unchanged BCL-2 mRNA correlated with apoptotic morphologies in SA-EA treated HL60 cells. This outcome suggested the BAK oligomerization might occur owing to the relative excess amount of BAK over Bcl-2. Interestingly, activated caspase 3 signals were observed in treated HL60 cells since 24-hour time point (Figure 3(a)) whilst the transcriptional expression of APAF-1 remained unchanged. Some studies reported that apoptosis may occur through alternative pathways which are independent of APAF-1 and activated caspase 9.65-67 These results suggested the activation of apoptotic process might take place in an intrinsic APAF-1 independent manner in SA-EA-treated HL60 cells. In the already-mentioned study of Le-Trung et al., AM from S. affinis also induced the intrinsic apoptotic pathway in HeLa cells via activation of both caspase-3 and -9. 15 Hence, this comparison further indicates the potent and consistent anticancer activities of S. affinis fruits’ compounds.
To further examine the possible involvement of extrinsic pathway (death receptor-initiated pathway), we evaluated the transcriptional level of BID gene in SA-EA treated cells. Extrinsic pathway is initiated by the binding of compatible ligands to the tumor necrosis factor receptors (i.e., CD95/FAS, DR3, CD266, CD120a, CD120b among others) followed by the activation of initial caspases 8 and 10. 53 Activated caspases 8/10, in turn, cleave effector caspases 3/7 and Bid (BH3-interacting domain death agonist), an interconnector between intrinsic and extrinsic pathway via activation of BAX.51,53 Here, we showed that the treatment of SA-EA led to no change in BID expression in HL60 cells. This transcriptional evidence suggested that apoptotic process might not occur by extrinsic pathway in HL60 cell line. In contrast, the AM compound, which is also found in S. affinis whole fruit, prompted the extrinsic apoptotic pathway in MDA-MB-231 cells in Chutamas Thepmalee et al.’s research. Briefly, AM treated breast cancer cells exhibited the up-regulation of caspase-8, death receptors, and their respective ligands, all of which are crucial markers of the extrinsic cascade. 16 Thus, our findings further provide evidence for the diverse underline mechanisms of S. affinis extracts and compounds, which is of important for multi-target anticancer strategies.
Beside apoptotic cell death, we also elucidated the involvement of cell cycle arrest in SA-EA treated cells by evaluating the expression of CDKN1A (encoding p21 protein) and PCNA genes. P21 is a cyclin-dependent kinase inhibitor which universally inhibits cyclin-CDK complexes, especially CDK2. 63 Moreover, this protein also has a pro-apoptotic role independent of the presence of functional p53. 56 Thus, p21 has been regarded as an important factor in cell cycle arrest and apoptosis.68,69 The mentioned knowledge strongly supports the increase in CDKN1A expression in p53-defective HL60 cell line used in this study. CDKN1A expression elevated dramatically at 24-hour time point suggested that SA-EA might induce the CDKN1A expression in a p53-independent manner. Thus, cell cycle arrest, and apoptosis in some extend, might occur in HL60 cells in accordance with the upregulation of CDKN1A.
In addition, we found that the SA-EA treament decreased the PCNA mRNA expression (Figure 4) and inhibited the growth of HL60 population (Figure 1). PCNA plays an important role in DNA replication through interacting with DNA and serving as a processing factor for DNA polymerase δ. 54 In addition, the high level of p21 in treated HL60 cells might interact with PCNA to magnify the DNA synthesis inhibition and cell proliferation. 54 Therefore, this finding indicated that SA-EA also restricted cell proliferation through lowering PCNA expression. Interestingly, the HL60 cell-growth inhibition acknowledged in our study is also in line with the same reported effects of S. affinis derived extract and compound on HeLa cells. In those study, the agents from S. affinis also significantly lower the levels of PCNA and other proteins of the p21-involved pathway (i.e., cyclin E, CDK1, CDK2).14,15 Therefore, the collective results of our research and others suggest the common proliferative inhibition of S. affinis fruit on sensitive cancer cell lines.
Conclusion
In our study, S. affinis fruit extracts, especially ethyl acetate fraction, with content of tannin, flavonoid, phenolic and steroid constituents, had selective anticancer activity against HL60 cells. This anticancer activity was the combination of apoptosis activation (intrinsic apoptosis via Apaf-1 independent manner) and antiproliferation (CDKN1A upregulation and PCNA downregulation). This study, for the first time, provided initial results of anti-leukemia potential of S. affinis fruits, and suggested further studies on their specific bioactive compounds as well as on the underlying mechanisms.
Supplemental Material
Supplemental Material - Ethyl Acetate Extract from Romdoul (Sphaerocoryne affinis) Fruit Induced Apoptosis in Human Promyelocytic Leukemia Cells
Supplemental Material for Ethyl Acetate Extract from Romdoul (Sphaerocoryne affinis) Fruit Induced Apoptosis in Human Promyelocytic Leukemia Cells by Nguyen Thi Hoai Nga, Tran Thanh Long, Truong Thi Bich Ngoc, Nguyen Hoang Khoi Nguyen, Dang Thi Phuong Thao, and Nguyen Thi My Trinh in Global Advances in Integrative Medicine and Health
Footnotes
Authors’ Contributions
NTHN, DTPT, and NTTM designed research. NTHN did most of the experiments. TTL, TTBN, and NHKN did several experiments. NTHN and TTL wrote the paper. TTL revised the paper. NTMT had primary responsibility for final content. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Faculty of Biology and Biotechnology, VNU-HCMUS, University of Science, under grant number: SH-CNSH 2022-07.
Ethical Statement
Data Availability Statement
All data of this study is available from the corresponding author on reasonable request.
Supplemental Material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

