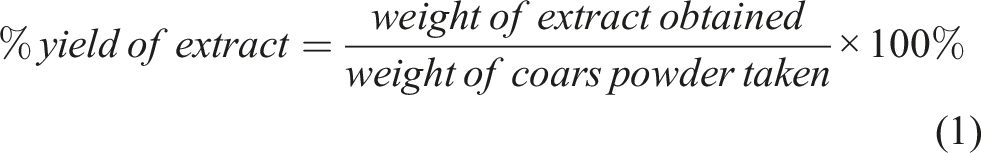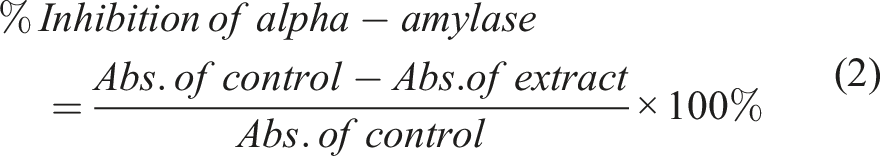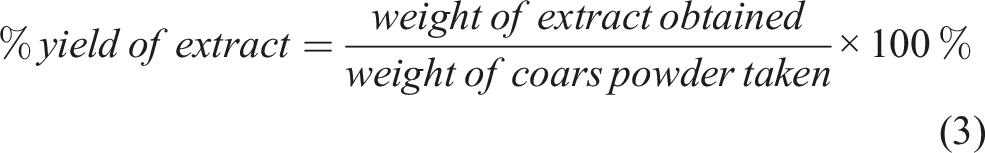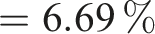Abstract
Objective
Diabetic individuals have a higher probability of suffering from illness and death due to small blood vessel-related problems such as retinopathy, neuropathy, nephropathy, and stroke than other complications. There are many synthetic anti-diabetic agents available, but these can be expensive and have undesirable pathological effects. The enzyme α-amylase (hydrolase), catalyzes the hydrolysis of starch to maltose and glucose via the cleavage of α-1,4-glucosidic linkages. Diabetes mellitus patients may benefit from a therapeutic strategy that involves slowing the hydrolysis of starch by inhibiting the activity of α-amylase. Thus, looking for cost-effective, natural, and safe antidiabetic agents is essential. This study aims to screen phytoconstituents and evaluate the in-vitro and in-silico α-amylase inhibitory activity of the ethanolic extract of Adhatoda vasica leaves.
Methods
The extraction of Adhatoda vasica leaves was performed using ethanol via the Soxhlet extraction process. Different concentrations (100 μg/mL to 1000 μg/mL) of ethanolic extract, Acarbose, and Sitagliptin, were prepared and evaluated for α-amylase inhibitory activity using the spectrophotometric method. Molecular docking (AutodockVina 1.2.0) and toxicity profiling (SToPToX web server) studies were performed.
Results
The ethanolic extract of Adhatoda vasica leaves showed the highest percentage inhibition against α-amylase (56.763 ± 0.0035) at a concentration of 1000 μg/mL. The in-silico study supported this inhibitory activity. Vasicoline (
Conclusion
It was concluded that Adhatoda vasica leaves possess some bioactive compounds that are responsible for controlling blood glucose levels, and their identification, purification, and isolation may lead to the development of new therapeutic agents with fewer side effects than the available drugs.
Introduction
Diabetes mellitus (DM) is a common metabolic endocrine disorder diagnosed via the presentation of hyperglycemia. This disorder is associated with many complications that may lead to the morbidity and death of a patient. 1 The burden of DM has increased globally, particularly in low-income and middle-income countries, including Nepal (8.5%, 2020). 2 The main clinical treatment strategy for DM is to control the blood glucose level using therapeutic agents (drugs). The most prevalent reason for an increase in one’s blood glucose level is the catalytic hydrolysis of carbohydrates by enzymes such as α-glucosidase and α-amylase. 3,4 α-amylase (hydrolase) is a prominent enzyme secreted by the pancreas and salivary gland, which are responsible for the catalytic hydrolysis of starch to maltose and glucose by cleaving α-1,4-glucosidic bonds. The inhibition of the activity of α-amylase delays the hydrolysis of polysaccharides, resulting in a reduction in the postprandial blood glucose level; that is believed to be an effective approach to the treatment of DM.5,6 The clinically approved inhibitors, ie, Acarbose, Voglibose, and Miglitol, have several side effects associated with gastrointestinal problems such as flatulence and diarrhea. Moreover, most of the approved antidiabetic drugs are expensive and have undesirable pathological effects like weight gain, diabetic ketoacidosis, fluid retention, and hypoglycemia. 7
Various traditional medicines and phytochemicals that exhibit α-amylase inhibitory activity are well known for their role in the prevention and treatment of diabetes. Some plant-derived constituents with antidiabetic properties have been isolated and shown to have a high potential and fewer side effects than clinically approved synthetic drugs.8–10 Therefore, further investigations into the development of more effective inhibitors for α-amylase are needed. Adhatoda vasica is 1 of the traditionally used medicinal plants. Furthermore, various studies in the literature have explained the anti-ulcer, antidiabetic, antioxidant, muscle relaxant, anti-allergic, hepatoprotective, and cardioprotective activities of different parts of Adhatoda vasica. 11 Although the in-vivo anti-diabetic activity of Adhatoda vasica leaves, roots had been thoroughly investigated, in-vitro and in-silico investigations have not yet been performed as per our best knowledge. The present study is designed to perform the phytochemical screening, and analysis of the in-vitro, in-silico α-amylase inhibitory effect of Adhatoda vasica leaves.
Methods
Reagents
All the chemicals of analytical grade were purchased from a commercial source.
Plant Materials Collection and Authentication
This study was carried out within 4 months of the proposal being approved by the Institutional Review Committee (UCMS/IRC/146/22) of the Universal College of Medical Science, Bhairahawa, Nepal at the Department of Pharmacy and Pharmacology. Leaves of Adhatoda vasica were collected from different areas of the Arghakhanchi district, Nepal (latitude = 27° 58′ 58.5372″, longitude = 83° 2′ 10.0968″ N). The herbarium was prepared and sent for identification to the Institute of Agriculture and Animal Science, Paklihawa campus, Rupandehi, Nepal. The plant identification report was validated by Dr Puspa Raj Poudel, Assi. Professor, Institute of Agriculture and Animal Science, Paklihawa campus, Rupandehi, Nepal.
Extraction
The freshly harvested leaves of Adhatoda vasica were collected and thoroughly rinsed with distilled water to eliminate any potential contaminants such as dust and foreign particles. The leaves underwent a process of air-drying under controlled laboratory conditions, ensuring adequate ventilation, and maintaining a constant room temperature. Subsequently, the leaves were subjected to additional drying in a hot-air oven at a temperature of 40°C for 24 hours to eliminate any residual moisture. The thoroughly desiccated leaves were pulverized using a grinder to acquire a granular powder, which was subsequently stored in a hermetically sealed container. A portion of 50 g of the powder was subjected to extraction with ethanol using a Soxhlet apparatus for 2 days each, at a temperature ranging from 40°C–50°C. Following the successful completion of the extraction process, the liquid extract was subjected to concentrate under vacuum. Subsequently, the resulting dry extract was carefully stored in a desiccator for future utilization.
12
The dried extract was weighed and the percent yield was calculated using the formula below:
13
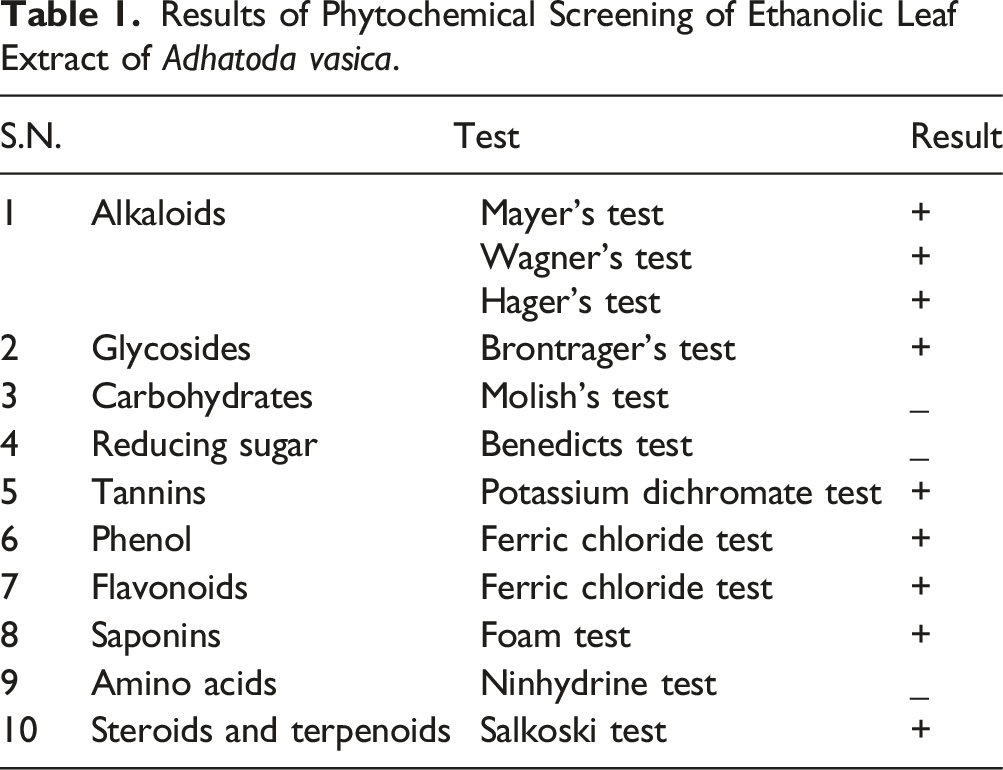
Phytochemical Screening
The phytochemical screening of the ethanolic extract of Adhatoda vasica leaves was performed to check the presence or absence of alkaloids, flavonoids, phenol, saponins, tannins, amino acids, cardiac glycoside, reducing sugar, carbohydrates, steroids, and terpenoid by slightly modifying a published methodology.14–16 (Supplemental file).
Determination of α-amylase Inhibition Activity
The α-amylase inhibition assay was performed using a published method with slight modification.17,18 About 250 μL of the test samples and standard drug (100-1000 μg/mL) were added to 250 μL of 0.20 mM phosphate buffer (pH 6.9) containing α-amylase (1 U/mL) solution and incubated at 25°C for 10 minutes. After incubation, 1 %, 200 μL of starch solution was added as a substrate to all the test samples and standard. Then, the solution was incubated at 25°C for 5 minutes for a second time. The reaction was stopped with 1.0 mL of 3,5 di-nitro salicylic acid color reagent. All test tubes were, then, incubated in a boiling water bath for 5 minutes, and cooled at room temperature. The reaction mixture was then diluted after adding 10 mL of distilled water and the absorbance (abs.) was measured at 545 nm using UV-visible spectroscopy. The absorbance of the control was measured accordingly without plant extract and labeled as the negative control. The experimental extract (test sample), Acarbose (standard drug), and Sitagliptin (standard drug) were tested with varying concentrations.19,20 The results were expressed as % inhibition (mean ± SD) of α-amylase activity and calculated according to the following equation:
21
Molecular Docking Procedure
Retrospective Spectral Profiling and Ligand Preparation
The genuine research articles were obtained from the indexed journal by using “GC-MS (gas chromatography-mass spectroscopy) profiling of Adhatoda Vasaka” as the search question. Approximately 8 appropriate articles were chosen, and the most significant, common, chemical constituent from each article was examined.22–26 We have selected 12 prevalent chemical constituents and subjected them to docking investigations.
The bioactive compounds from the Adhatoda Vasaka were identified from a previously published study and around 12 ligands (Figure 1) were selected through an extensive literature review as mentioned above. The 3 dimensional (3D) molecular coordinates were downloaded from PubChem database (https://pubchem.ncbi.nlm.nih.gov/) in sdf format. Next, energy minimization was performed using the Molecular Mechanics Force Field (MMFF94). This process optimized the ligand structures by minimizing their potential energy, resulting in stable conformations suitable for docking analysis. The ligand structures were then converted to pdbqt format using OpenBabel 3.1.1 to be compatible with AutoDock Vina 1.2.0, a widely used molecular docking software.
27
Chemical constitutes (C1-C7 alkaloid and C8-C12 flavonoids) of plant Adhatoda vasica and 3D structure of co-crystal native ligand (Acarbose C13) and reference compound Sitagliptin (C14).
Protein Selection and Preparation
Simultaneously, the 3D crystal structure of the α-amylase enzyme in complex with co-crystal Acarbose (PDBID: 3BAJ, X-ray crystallography resolution = 2.10 Å) 28 was downloaded from the Protein Data Bank (PDB, https://www.rcsb.org/). The protein structure underwent preparation steps, including the removal of water molecules, the addition of missing hydrogen atoms, and the assignment of proper charges. 29 The prepared protein structure was visualized using BIOVIA Discovery Visualizer Studio 2021, facilitating for a better understanding of its active site and potential binding pockets. 30 The binding site amino acids were determined using BIOVIA Discovery Visualizer Studio 2021 and validated according to a previously published original article about the protein (PDBID: 3BAJ). 31 Similarly, the grid parameters, to cover the active site, were determined as −9.632, 4.340, and −23.107 for the x, y, and z-axis respectively.
Docking Protocol
The molecular docking study provides the mechanistic view of the ligand-protein complex and determines the binding energy to explore the different poses of the ligand-receptor interactions.32,33 Molecular docking was performed in Linux (Ubuntu 20.04.6 LTS-GNU/Linux 4.4.0-19041-Microsoft x86_64) environment using AutoDock Vina 1.2.0 software (https://vina.scripps.edu/), 34 which predicted the binding modes and affinities of the selected ligands with the α-amylase enzyme. This analysis explored the various orientations and conformations of the ligands within the enzyme’s active site, providing insights into potential interactions and binding affinities. The ligand-target interaction and the nature of the interaction were visualized using PyMol 2.5 software (https://pymol.org/2/).35,36
To validate the docking results and assess their reliability, the Root-Mean-Square Deviation (RMSD) value was calculated using PyMol 2.5 software. For this, the native ligand was redocked to the active site and the surface of the ligand was taken as a reference for other ligands. Then, all docked ligands were superimposed with the native ligand surface in PyMol 2.5 software, and then the RMSD value was computed. This measure quantified the differences between the docked ligand conformations and the co-crystal structure of the native ligand-protein complex.37,38
Toxicity Study
Determining the molecular structure and its relationship with biological activity and toxicity is a very crucial step in drug design and development. The presence of certain functional groups, the rigidity of the structure, and the atomic composition and radii contribute to the development and occurrence of toxic properties in the molecule. The acute toxicity was predicted using a computational method. SToPToX (https://stoptox.mmL.unc.edu/), an atomic algorithmic online web server, was used to predict acute inhalation toxicity and acute oral toxicity. 39
Statistical Analysis
The in-vitro experiment was performed and the results were expressed in the mean ± SD. Descriptive analysis was performed using Microsoft Excel 2016.
Results
Percentage Yield
The extract was dried and then the percentage yield of plant extract was calculated; this was found to be 6.69 %.
The weight of the coarse powder of leaves was 50 g.
The weight of the extract obtained after extraction was 3.345 g.
Phytochemical Screening
Results of Phytochemical Screening of Ethanolic Leaf Extract of Adhatoda vasica.
α-amylase Inhibitory Activity
Percentage Inhibition of α-Amylase Activity (±SD) at Different Concentrations of Standard Drugs and Plant Extract and IC50 Value of Standard Drugs and Plant Extract.


Average percentage inhibition of α-amylase activity at different concentrations of the plant extract and standard drugs used and error bar diagram of % inhibition ±SD. This diagram represents the error bar diagram of the average percentage inhibition at a concentration of 100 to 1000 μg/mL.
Molecular Docking Results
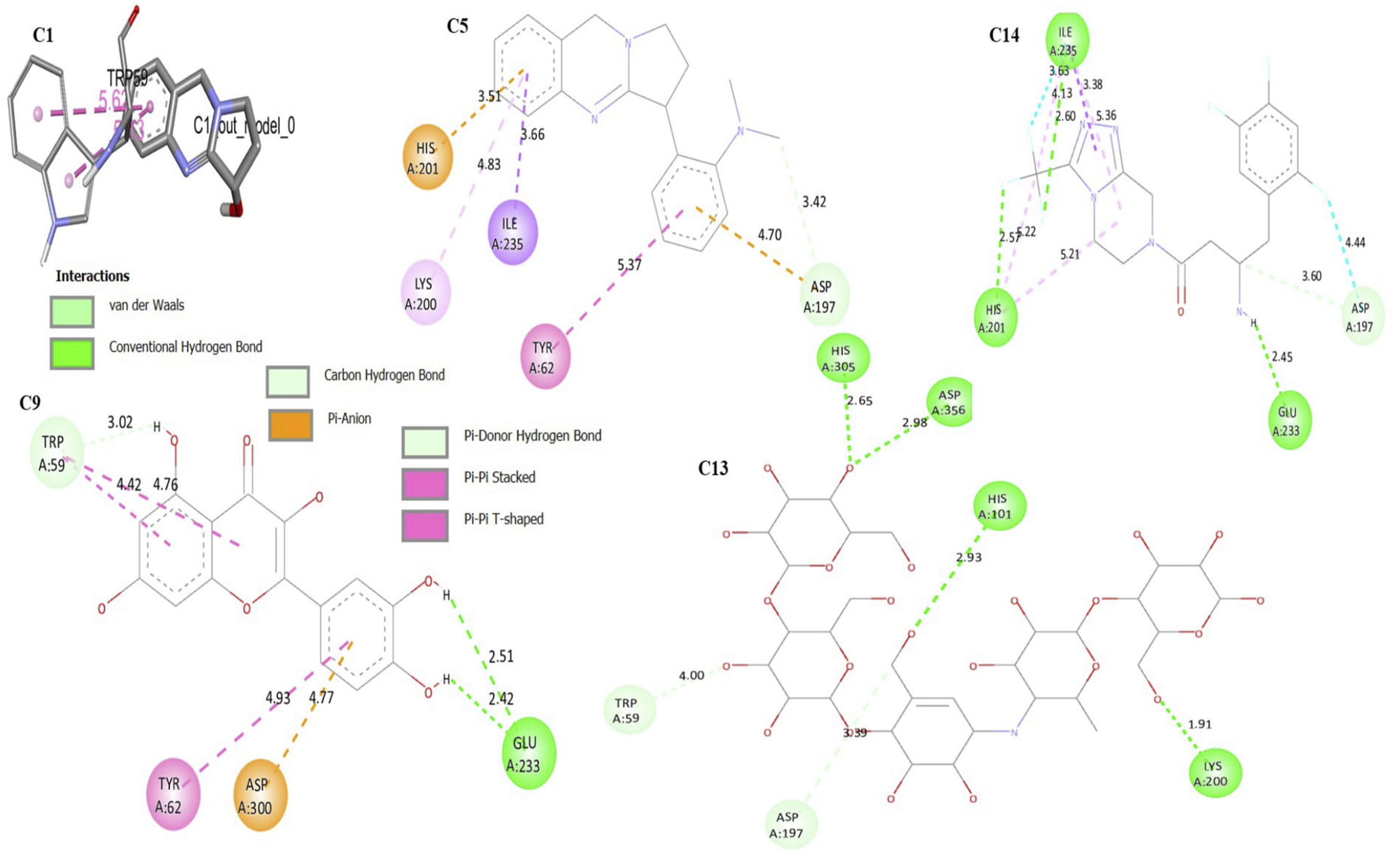
To generate the binding energy (ΔG, Kcal/mol), the phytoconstituents (C1 to C12) were docked into the active sites of α-amylase. A more negative binding energy (least binding energy) denotes a spontaneous connection and a stronger affinity between the ligand and protein, and consequently, a more powerful inhibition. All phytoconstituents showed a binding energy in the range of −5.9 Kcal/mol to −8.3 Kcal/mol. All the phytochemicals showed slightly higher binding energy (weak affinity) than the native co-crystalized ligand (Acarbose, C13, ΔG = −8.6 Kcal/mol) and standard drug Sitagliptin (C14, ΔG = −8.5 Kcal/mol). The binding energy, which measures the strength of the interaction between the compounds and the protein, was found to be −8.3 Kcal/mol for Vasicoline (C5) (Figure 4) and −8.0 Kcal/mol for Quercetin (C9). All the ligands showed a RMSD value less than 2 Å, which validates the docking results (Figure 3, Table 3). Table 3 provides details of the binding energy, amino acid required for interaction, and RMSD values Figure 4. The 3D structure of superimposed ligands (C1–C12) with co-crystal native ligand (C13) used to calculate the RMSD value. Binding Energy and Interactive Amino Acid. The 2D molecular interaction of Vasicoline (C5), Quercetin (C9), the co-crystal native ligand (Acarbose, C13), and the reference drugs Sitagliptin (C14), and Vasicin (Q1, 3D) against α-amylase.

Toxicity Profiling Analysis
The acute inhalation toxicity and acute oral toxicity of the active constituents were investigated using the SToPToX server. The predicted toxicities were indicated with a negative sign (−) for non-toxic compounds and with a positive sign (+) for toxic compounds. The contributing functional group in the toxicity properties was also predicted. The green color in the structure represents the functional group contributing to the non-toxic properties and the brown color represents the functional group contributing to an enhancement in the toxic character (Figure 5). Vasicoline (C5) was predicted toxic compound for inhalation. However, Quercetin (C9) showed a negative sign for both acute inhalation toxicity and acute oral toxicity. Therefore, it was a nontoxic compound. Details of the toxicity study are given in Table 4. ‘A’ and ‘C’ applicability domain (AD) for acute inhalation and acute oral toxicity of compound C5, respectively. ‘B’ and ‘C’ predicted fragment contribution for acute inhalation and acute oral toxicity of compound C5, respectively. The green color in the structure represents the functional group contributing to non-toxic properties and the brown color represents the functional group contributing to an enhancement in the toxic character. Acute Inhalation Toxicity and Acute Oral Toxicity of Phytoconstituents.

Retrospective Spectral Data Analysis
Discussion
The present study was carried out to investigate the potential α-amylase inhibitory activity of an ethanolic extract of Adhatoda vasica leaves by applying in-vitro and in-silico approaches. We also performed the phytochemical screening of Adhatoda vasica leaves. In diabetes, the critical mechanism involves the impairment of glucose regulation and metabolism. Symptomatic hyperglycemia, in which blood sugar levels are abnormally high, is attributed to an abnormally high glucose release. Understanding the regulation and inhibition of α-amylase activity is important for developing strategies to control postprandial glucose levels and manage diabetes effectively. Amylase inhibitors are regarded as starch blockers as they prevent the body from absorbing dietary starch and slow down digestion. This decreases the blood glucose level and thus has positive effects on insulin resistance and glycemic index control in diabetes.41,42
We investigated in which we measured the percentage inhibition of α-amylase activity using different concentrations (100-1000 μg/mL) of the ethanolic leave extract of Adhatoda vasica. The percentage inhibiting activity of the ethanolic extract at a concentration of 1000 μg/mL was found to be 56.763 ± 0.0035 which is less than the standard Acarbose (% inhibition = 74.879 ± 0065). The reason for this difference in activity could be the presence of a significantly low concentration of phytoconstituents (responsible for activity like Vasicine, Vasicinol, and Vasicinone) in the extract. It is worth mentioning that the phytochemical screening of the ethanolic extract of Adhatoda vasica leaves showed the presence of alkaloids, glycosides, phenol, flavonoids, saponin, tannin, steroids, and terpenoids. The extract did not contain the reduced sugar. The present study showed that the % inhibition was less than 50 % at a dose of 800 μg/mL. A similar study was conducted by Khadayat K. et al and evaluated the % inhibition of the α-amylase activity of Nepalese medicinal plants used in the treatment of diabetes mellitus. 43 The IC50 value (413.5 μg/mL) of their study was nearly half of our results (712.466 μg/mL). The reason behind this difference could be the extraction process and geographical difference which may have impacted the percentage composition of phytoconstituents responsible for anti-diabetic activity. However, we can rationalize that, the active constituents of the plant Adhatoda vasica (distributed in Nepal) exert significant α-amylase inhibitory activity.
The antidiabetic activity of the extract is due to the presence of alkaloids which was explained in a study conducted by Gao H. et al. They explained that alkaloids like Vasicine, Vasicinol, and Vasicinone are responsible for anti-diabetic activity via the inhibition of the α-amylase enzyme at more than 50% (the IC50 values were 125 μM to 250 μM). 44 This comparison highlights the consistency and agreement between our findings and the previous study regarding the presence and potential therapeutic effects of alkaloids in the plant extract. Graphical calculation revealed that the IC50 value of the ethanolic extract of Adhatoda vasica was 847.96 μg/mL, which indicates that it has an inhibitory effect against α-amylase. However, an earlier study conducted by Rudrapal M et al 45 reported a significantly lower IC50 value of 47.6 μg/mL for the Adhatoda vasica leaf extract. Their study involved the purification and isolation of specific constituents, particularly vasicine, using chromatographic techniques. These contrasting results suggest that the reported antidiabetic effect of Adhatoda vasica may not solely rely on the presence of alkaloids in general but rather on the specific constituents present, such as vasicine. The lower IC50 value observed in the study could indicate that vasicine, among other constituents plays a significant role in the anti-diabetic properties of Adhatoda vasica. Therefore, the effectiveness of the plant extract in managing diabetes may be attributed to a combination of alkaloids and specific constituents, emphasizing the importance of studying and isolating individual compounds for a comprehensive understanding of their therapeutic potential.
The results demonstrated a dose-dependent relationship, as higher concentrations of the extract led to greater percentage inhibition. This dose-dependent relationship was explained in a previous in-vivo study. The study explored the anti-diabetic activity of the ethanolic extract of the leaves and roots of Justicia adhatoda. This study examined the extract’s effect on the blood glucose levels of alloxan-induced diabetic rats. The results indicated that the ethanolic extract of Justicia adhatoda also demonstrated a dose-dependent reduction in blood glucose levels, meaning higher doses led to greater reductions in glucose levels. 46 We also observed the dose-dependent linear pattern in our study. The parallel dose-dependent patterns observed in both studies suggest a linear correlation between the inhibitory effect of the extract on α-amylase and (the ability to lower blood glucose levels) and the concentration of extract used. These findings support the notion that the ethanolic extracts of Adhatoda vasica and Justicia adhatoda hold promise for their anti-diabetic properties. This could be the result of alkaloidal binding to the (competitive or non-competitive) digestive enzymes (α-amylase), preventing the formation of an enzyme-substrate complex and ultimately reducing the activity of the enzymes.
To investigate the binding mode, binding energy, and ligand-protein interactions underlying the observed α-amylase selectivity, molecular docking studies have been performed. 47 The molecular docking method is virtual screening technique that helps to eliminate unsuitable ligands from the library. The docking result was validated by calculating RMSD value superimposed with the co-crystal native ligand.48,49
At locations close to the co-crystallized ligand (Acarbose), which is made up of the amino acids Trp59, Lys200, Asp356, His101, Ala169, His305, and Tyr159, it was discovered that all of the phytoconstituents (C1-C12) fit neatly into the active region of the enzyme. The docking interaction revealed that Vasicoline (C5, Figure 4) and Quercetin (C9, Figure 4) were well nested into the active site of α-amylase and presented a similar coordination with the active site of enzyme. The interactions mode obtained via molecular docking for Vasicoline, Quercetin, and the standard drug Sitagliptin (ΔG = -8.5 Kcal/mol) are illustrated in Figure 4. Quercetin (ΔG = -8.0 Kcal/mol) showed 2 hydrogen bonds with the amino acids Glu233 with bond distances of 2.51 Å and 2.42 Å each. Additionally, Quercetin had a shorter bond length, especially for hydrogen bond which explains its better stability. It is worth mentioning that Kaempferol (C8), Apigenin (C10), and Vitexin (C12) were among the top-scored phytoconstituents against α-amylase (Table 3).
Rudrapal M et al 45 conducted a molecular docking for vasicine against α-amylase enzyme and reported a binding energy of −6.7 Kcal/mol. However, we observed a slightly higher binding energy (weak affinity) for Vasicine (C1) against α-amylase enzyme (Table 3, ΔG = −5.9 Kcal/mol). Additionally, Vasicine (C1) showed a higher binding energy than the co-crystal ligand Acarbose (ΔG = −8.6 Kcal/mol). Intriguingly, Vasicine (C1) (Figure 4) lost its hydrophobic interaction with Trp59, Lys200, Asp356, His101, and His305. On the contrary, these amino acids were found to interact with the native co-crystal (Acarbose). The absence of these amino acids interacting with Vasicine (C1) could be the reason for its weak binding affinity with the α-amylase enzyme. The findings of the docking study revealed that the binding energies of Vasicoline (C5) and Quercetin (C9) were comparable to those of the standard drug (Sitagliptin and Acarbose, Table 3). Jhong C. et al 50 predicted the inhibitory activity of Quercetin against α-amylase. They also suggested the comparable binding energy of Quercetin (Docking score = −135.75 Kcal/mol) with the commercial α-amylase inhibitor Acarbose (Docking score = −166.28 Kcal/mol). The presence of 5 hydroxy groups (-OH) with Quercetin could be the reason for the similar binding energy because they formed a hydrophobic interaction with active site amino acid; this could have contributed to the binding energy. A similar result by Nabil A et al 51 suggested that Quercetin is the best α-amylase inhibitor, with a minimum binding energy of −8.8 Kcal/mol. They also revealed that the hydroxy group contributed to forming a conventional hydrogen interaction. Interestingly, the hydroxyl group acted as a hydrogen donor, like our findings. In Figure 4 (Quercetin, C9), Trp59 formed a carbon-hydrogen bond with the hydroxyl group of Quercetin at a bond distance of 3.02 Å. Meanwhile, in the interaction of Acarbose, a similar amino acid was seen but the bond distance of the pi-donor hydrogen interaction was 4 Å. The better stability of this contact can be explained by Quercetin’s shorter binding distance. Since Acarbose and Quercetin interact with the same amino acid, it can be said that Quercetin inhibits α-amylase via competitive inhibition. 52
Acute toxicity is the term used to describe the negative consequences that accompany the administration of a single dose or numerous doses of a chemical within a 24-hour period, or a 4-hour inhalation exposure. 53 In 1 study, when a group of 10 or more laboratory white rats inhaled a substance or a mixture of substances continuously for eight hours, 50% of the group died within 14 days; this is referred to as toxicity. 54 Most of the active constituents of the plant extract showed non-toxic characteristics when taken orally. For Vasicoline (C5), a three-nitrogen atom and the rigid benzene ring contributed to increase the in toxicity. The presence of the hydroxy, carbonyl, and oxy group, due to the saturated structure of the compound (Quercetin C9, Figure 1(s), supplemental file), increased its non-toxic characteristics. This indicates the safety profile of the extract. It can be taken orally for the treatment of DM when the allopathic medication is not effective or unavailable. 55
Limitations
The identification and isolation of the active phytoconstituent responsible for the anti-diabetic activity could not be performed due to resource limitations. The experimental in-vivo confirmation of the anti-diabetic effect of the plant’s active constituents could also not be added due to resource limitations.
Conclusion
The study revealed that, the ethanolic extract of Adhatoda vasica leaves is effective to inhibits α-amylase activity, which regulates the glucose level. The plant extract exhibited the greatest suppression of α-amylase activity (% inhibition = 56.763 ± 0.0035) when used at a concentration of 1000 μg/mL. The in-silico analysis supports the in-vitro results. The active components of the Adhatoda vasica leaves ie, Vasicoline and Quercetin exhibited the highest binding affinity of −8.3 Kcal/mol and −8.0 Kcal/mol, respectively against α-amylase enzyme. Molecular docking studies concluded that the presence of alkaloids and flavonoids support the α-amylase inhibitory activity. It was concluded that, that leaves possess certain α-amylase inhibitory activity which could be developed as an alternative medicine for DM.
Supplemental Material
Supplemental Material - In Vitro and in Silico Analysis of α -Amylase Inhibitory Activity of Ethanolic Extract of Adhatoda vasica Leaves
Supplemental Material for In Vitro and in Silico Analysis of α -Amylase Inhibitory Activity of Ethanolic Extract of Adhatoda vasica Leaves by Chandrajeet Kumar Yadav, Sandhya KC, and Shankar Thapa in Global Advances in Integrative Medicine and Health
Footnotes
Acknowledgements
The authors are thankful to the administration department of the Universal College of Medical Science, Bhairahawa, Nepal.
Authors’ Contribution
Chandrajeet Kumar Yadav, Sandhya KC, and Shankar Thapa have equal contribution to the research.
Preprint
Data Availability Statement
The data used to support the findings of this study are available from the corresponding author upon request.
Supplemental Material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.