Abstract
Background
Functional gastrointestinal disorders (FGIDs), a frequent cause of gastrointestinal (GI) symptomology, impact 40%–69% of the United States population. People who suffer from FGIDs often self-manage their symptoms using consumer healthcare products such as dietary supplements.
Objective
To investigate use of Digest-8 dietary supplement by people who purchased the product from pharmacies for their FGID symptoms.
Methods
This pharmacy-based observational study included a convenience sample recruited by flyers at 2 pharmacies upon purchase of Digest-8 dietary supplement to assess symptoms, satisfaction, tolerability, and side-effects through 7 weekly voluntary surveys completed by consenting participants.
Results
Twenty-two participants completed an initial survey and a collective total of 59 follow-up surveys. Analysis of follow-up surveys compared to baseline provided pilot data on symptom improvement, satisfaction, tolerability, and low side effect profile.
Conclusion
Preliminary findings of the study warrant a follow-up prospective randomized clinical trial to address the study limitations.
Keywords
Background and Objective
Functional gastrointestinal disorders (FGIDs) are a frequent cause of gastrointestinal (GI) cramps, pain, nausea, bloating, constipation and/or diarrhea, and flatulence. FGIDs are included in disorders of gut-brain interaction (DGBIs) and are described as a combination of motility disturbance, visceral hypersensitivity, altered mucosal and immune function, gut microbiota, and central nervous system processing. As such, FGIDs are cited to greatly impact patient quality of life 1 with 40%–69%% of the population in the United States meeting diagnostic criteria. 2 People frequently self-manage their symptoms with consumer healthcare products including over-the-counter medications and dietary supplements.3,4
Digest-8, a dietary supplement to support digestive health is an alcohol and water extract of 8 botanicals (Organic Dandelion, Organic Cinnamon, Organic Licorice, Organic Angelica, Organic Bitter Orange, Organic Fennel, Organic Ginger, Organic Yarrow) classified as Generally Regarded as Safe (GRAS) by the United States Food and Drug Administration (FDA).5–7 Similar to other non-prescription treatments for FGIDs, the botanicals in Digest-8 have varying degrees of evidence of promoting digestive secretions, soothing digestive discomfort, supporting gastric motility, and reducing mucosal inflammation and visceral hypersensitivity.8–10
With a paucity of information of Digest-8 botanical tincture in a consumer healthcare context, we collected and analyzed survey data from volunteer individuals with functional digestive symptoms who decided on their own or by recommendation of their doctors or pharmacists to take Digest-8. The suggested dose was 2.5 mL by mouth once a day at any time during the day, before, after, or between meals. Participants customized the duration of use of the product depending on their symptoms.
Methods
This observational study utilized Institutional Research Board (IRB) (Western Institutional Board approval #35294202) approved SurveyMonkey questionnaires. Participants who purchased Digest-8 at 2 designated pharmacies received a flyer describing the study and how to enroll if they fulfilled inclusion criteria (adults >18 years old) without any exclusion criteria (hypersensitive to ingredients and women who were pregnant or breast feeding). In accordance with the instructions on the flyer, participants sent their email address to the primary investigator and in return received the survey link that included a consent form and baseline questionnaire. Failure to consent to participate resulted in automatic exclusion.
Subjects received anonymous and voluntary baseline and 6 weekly follow-up questionnaires by email from September 2021 – September 2022. Baseline surveys included demographic information, prior usage of Digest-8 before the study began, description of symptoms (pain or cramps of upper and middle part of belly, heartburn; and pain or cramps of the lower belly, bloating, constipation), average daily severity of GI symptoms over previous 2 weeks (5-item Likert scale), symptom profile, and reason for using Digest-8 (medications did not work, medications had side effect, prefer not to use medication, other).
Weekly follow-up questionnaires asked participants how many days over last week Digest-8 was taken, average daily severity of GI symptoms over previous week (5-item Likert scale), overall improvement in symptoms since starting Digest-8 (5-item Likert scale), overall satisfaction with Digest-8 (5-item Likert scale), tolerability of Digest-8 over last week (5-time Likert scale), and side effects (no side effects, GI side effects, other).
Results
Participant Demographic Characteristics and Baseline Survey Responses.

Frequency of 6 Follow-up Weeks.
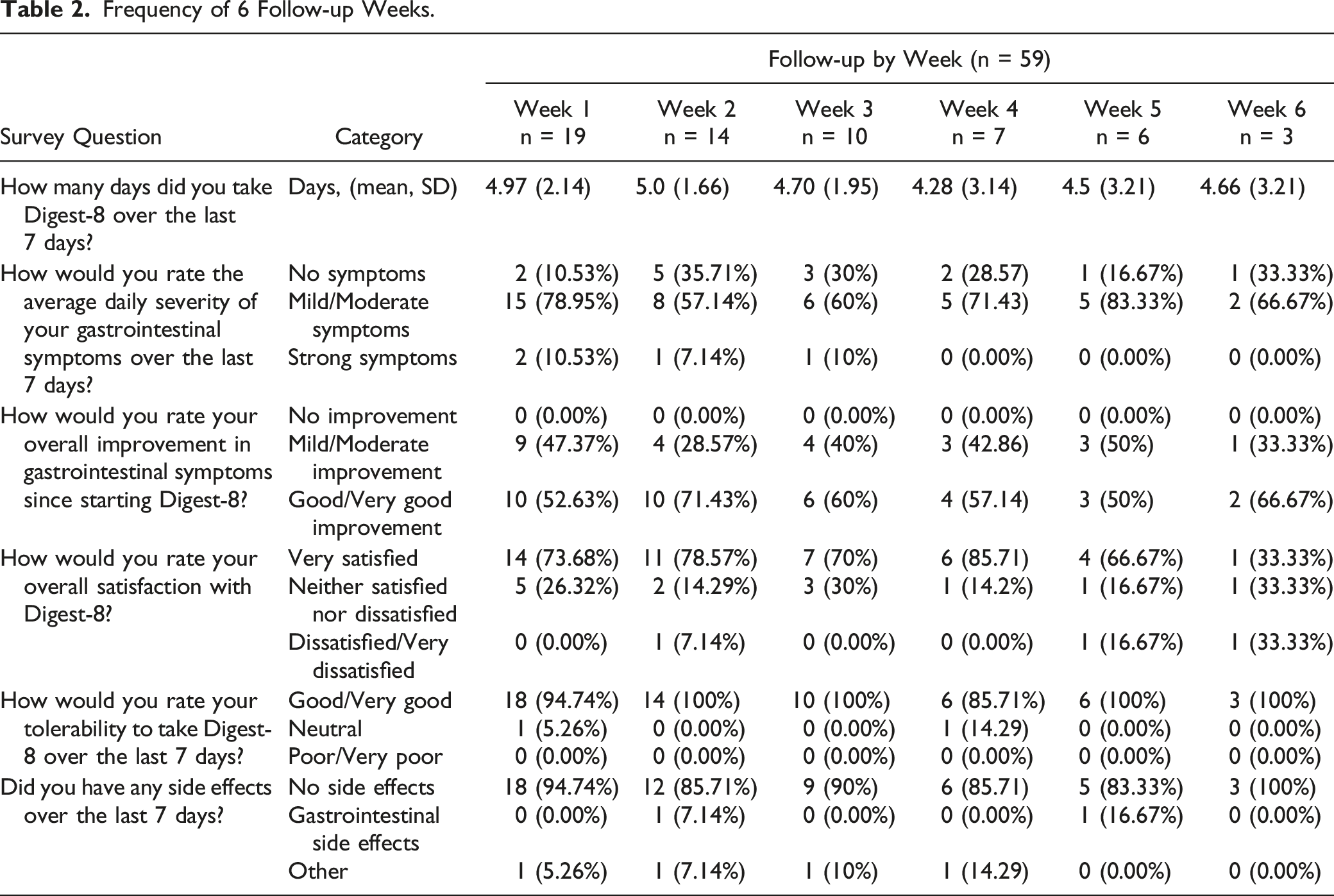
Three surveys were completed at 6-week follow-up which concluded the study. Evaluation of this data point is limited by the low response rate.
Discussion
As FGIDs are frequently self-managed, this study represents an innovative approach to investigate health consumer products that anecdotally help to relieve functional digestive symptoms.11,12 In addition, recent reports, including research conducted at the current institution, discovered associations between FGIDs and acute and long-term effects of Coronavirus disease 2019 (COVID-19) infection, further establishing the need for an increasing range of treatment options. 13
The value of consumer healthcare related data extends beyond FGIDs as more than a third to 73% of adults in the U.S. regularly consult the Internet 11 and social media for self-care of their ailments. 12 In response, various healthcare organizations and companies have developed symptom checker computerized algorithms. These programs ask users about themselves and their symptoms to provide potential diagnosis and triage to appropriate care such as emergent, non-emergent, or self-care. 2 Innovations in symptom checker algorithms may collect data of satisfaction of consumer healthcare treatments in cases of self-care triage. Smart-phone applications may also gather data on patient experience using these products 3 and potentially assess customer reports on self-diagnosed, self-treated, mild self-limited symptoms.
Despite the potential value of consumer healthcare related data, our study highlights potential limitations to the conclusions from results of data analysis. We did not calculate statistical significance because of the high potential for response bias with the small size (n = 22) and small number of follow-up survey responses (19/22 responses at week-1 follow-up and only 3/22 responses by 6-week follow-up) of the convenience sample.
Attrition bias limits data analysis to investigate if benefits were sustained during the duration of treatment. One hypothesis based on the experience of the author who provides patient care is that patients who improved were less likely to continue treatment and survey responses. Alternatively, those lost to follow-up may have improved to the same degree, improved to a lesser degree, or worsened relative to the participants who completed all follow-up surveys.
A follow-up prospective randomized clinical trial study using Health-Related Quality-of-Life (HRQOL) outcome measures such as Patient-Reported Outcomes Measurement Information System (PROMIS) could address the limitations of the study as well as the high placebo response rates of treatments for FGIDs. Since FGIDs are a diagnosis of exclusion, questions about diagnostic tests would rule our more serious conditions. In addition, the FGID patient population is heterogeneous which makes data collection challenging. Information technology tools may be useful for collecting data on this population to stratify FGID functional symptoms. Due to limited safety data on herbal supplements, a follow-up trial would also include detail on side effects.
Conclusion
Preliminary findings of this convenience sample of people who used Digest-8 dietary supplement for FGID symptoms represent low quality of evidence and warrant a follow-up prospective randomized controlled clinical trial to address limitations of interpretation of this data. Developing innovative tools of data collection and analysis for consumer healthcare practices may inform scientists what may be worthwhile to consider studying in more detail using more rigorous methods.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: MB is the inventor on United States Patent and Trademark Office (USPTO) number 10,993,979 for the Digest-8 dietary supplement formulation. MB has a financial interest/benefit. JH, KDH and SJR declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
