Abstract
Background
Children diagnosed with acute lymphoblastic leukemia (ALL) receive oral chemotherapies (OCs) for 2 years. The at-home self-management of OCs and their consequences on the child lead to multifaceted difficulties that can impact their optimal use and the family's well-being. We report the development process and the detailed plan of a program to support families throughout the treatment.
Method
The development combined a participatory approach with the three stages of Behavior Change Wheel (BCW) framework, namely understanding the behavior to change, identifying intervention options, and identifying content and implementation options. A working group (i.e., researchers, parents, and professionals involved in the care of children with ALL) participated at all development stages through online meetings and co-construction activities.
Results
The core objective targeted by the program was defined as: to enable families to face the difficulties met with OC by mobilizing tools and resources in collaboration with the healthcare team to support the proper use of OC and the family well-being. The program plan comprises three components with various interventions dispensed at key times during treatment: education and training for OC management and safe use, evaluation and counseling about OC side effects, and empowerment of families and normalization of their experience with OC.
Discussion
The combination of a participatory approach and a structured framework for the program development is likely to increase its relevance, acceptability, and efficacy. This program has the potential to support more effective and safe use of OC, as well as the well-being of the child and their family.
Introduction
Acute lymphoblastic leukemia (ALL) is the most frequently diagnosed cancer in children under 15 years (PDQ, 2024). The collaborative effort from pediatric research consortiums was instrumental in achieving an average 5-year survival rate of above 90% in these children (PDQ, 2024; Pui et al., 2015). This success relies on better supportive care (e.g., infection management) and tailored treatment based on clinical and genetic risk factors for treatment relapse (Pui et al., 2015; Pui & Evans, 2013; Raetz et al., 2023). Thus, most children with ALL are treated according to chemotherapy protocols from recognized childhood ALL consortiums (Pui et al., 2015), such as Dana-Farber Cancer Institute (DFCI; Vrooman et al., 2021) and Children's Oncology Group (Maloney et al., 2020). These protocols consist of different treatment phases with polychemotherapy administered by various routes, typically over a period of 2‒3 years (Maloney et al., 2020; Vrooman et al., 2021). Thiopurines (mercaptopurine [MP] and/or thioguanine) with corticosteroids (dexamethasone [DEX] and/or prednisone) are oral chemotherapy (OC) administered in the maintenance phase (last phase) of pediatric ALL treatment (Maloney et al., 2020; Toft et al., 2018; Vrooman et al., 2021). These OCs are taken at home for about 2 years. Methotrexate (MTX) is also given by mouth in certain protocols or situations (Maloney et al., 2020; Toft et al., 2018). The management of OC and its consequences for families can be complex in several respects. Among others, depending on the protocol and the drugs, OCs are administered intermittently or continuously and their daily dosage may differ between days or cycles (Maloney et al., 2020; Toft et al., 2018; Vrooman et al., 2021). Intake recommendations for drug efficacy and safety can also be cumbersome (Camiré-Bernier et al., 2021; Kahn et al., 2023; Tang et al., 2022). For example, depending on treatment protocols, MP is required to be taken on an empty stomach in the evening or at the same moment every day without regard to food (Kahn et al., 2023; Landier et al., 2017). MP and MTX should be handled with gloves due to their cytotoxicity properties (BC Cancer Drug Manual, 2018, 2023). An additional challenge encountered by families arises from recommendations to decrease the risk of infection such as limiting the child's group activities due to reduced immunity with chemotherapy (Camiré-Bernier et al., 2021; Dutta & Flores, 2018). While MP is generally well tolerated in terms of side effects, corticosteroids are associated with both physical and psychobehavioral effects on the child, such as food cravings, increased appetite, weight gain, hyperglycemia, hypertension, acne, insomnia, sadness, anger, and hyperactivity (Aljebab et al., 2017; McGrath & Rawson-Huff, 2010).
A few studies on families’ experiences and needs with OC in pediatric ALL highlight that these medications are associated with challenges affecting various aspects of the daily life over time and impacting the whole family (Camiré-Bernier et al., 2021; David et al., 2023; McGrath & Rawson-Huff, 2010; Tang et al., 2022). Our previous qualitative study with 13 parents of children aged under 15 years treated for ALL further emphasized that families would benefit from enhanced support throughout at-home OC treatment, including for its management and ensuing difficulties. Parents expressed a great desire to follow the OC intake recommendations, often prompting them to adopt a strict daily routine which was considered helpful but highly demanding, especially over such a long period. Moreover, respecting the infection control advice and dealing with the psychobehavioral effects of DEX on the child, and especially their unexpected intensity, can contribute to the child and family social isolation (Camiré-Bernier et al., 2021). Such difficulties are likely to have a negative effect on OC adherence as well as on the child and family well-being (Heneghan et al., 2020; Landier et al., 2011; McGrath & Pitcher, 2002).
Some interventions have been developed or evaluated with the aim of supporting families and the child diagnosed with ALL (Dragone et al., 2002; Tan et al., 2024; Wang et al., 2018; Williams et al., 2016; Zeng et al., 2023). Among these, some focused on treatment adherence (Zeng et al., 2023) while others provided general and essential knowledge to understand the treatment, ensure its proper management (e.g., drug name, preparation, storage), or deal with its common side effects (e.g., special precautions, classic adverse effects; Dragone et al., 2002; Tan et al., 2024; Wang et al., 2018). One intervention focused specifically on parenting skills during the maintenance phase (Williams et al., 2016). In these studies, participants reported high satisfaction with interventions that were developed with stakeholders (Dragone et al., 2002; Tan et al., 2024; Wang et al., 2018). Some interventions showed positive effects on MP adherence (Zeng et al., 2023), on the child's feeling of control over their health (Dragone et al., 2002), and on certain parental indicators related to psychosocial and knowledge domains (Wang et al., 2018).
Overall, none of these interventions specifically targeted the multifaceted difficulties met with combined at-home OC (thiopurines and corticosteroids). This prompted the development of such an intervention tailored to the unique context of families and health professionals involved in the care for children treated for an ALL with OC. Thus, to develop a program that can be implemented in a real-world clinical setting to support children with ALL and their families with the difficulties experienced with at-home OC, we paired the Behavior Change Wheel (BCW), a structured framework for designing interventions (Michie et al., 2014), with a participatory approach. BCW was chosen because it is a simple and practical tool which is suitable for a large range of clinical contexts. By offering a program that assembles tools and resources to help parents cope with the multiple difficulties associated with at-home OC, this could contribute to optimize OC use and family well-being. We report here the development process and the detailed plan of this program.
Method
Program Development Context
The program development process took place from 2021 to 2023 at a French-speaking university hospital center in the province of Québec (Canada) providing specialized and ultraspecialized care to the pediatric population and admitting ∼20 children newly diagnosed with ALL each year. Children receive medical, pharmaceutical, nursing, physical rehabilitation, nutritional, psychological, social, and educational care and services from a multidisciplinary team. At the time of this program development, most of the pediatric patients diagnosed with de novo ALL at the hospital site were treated according to the DFCI ALL Consortium Protocol 16-001 (ClinicalTrials.gov identifier: NCT03020030). This protocol includes intermittent oral DEX and MP on a 21-day cycle (first 5 and 14 days of cycle for DEX and MP, respectively; Vrooman et al., 2021). MTX is not routinely administered per oral route. This project was approved by the Institution's Ethics Board (no. 2021-5173) and a consent form was signed by stakeholders involved in the program development working group.
Development of the Program
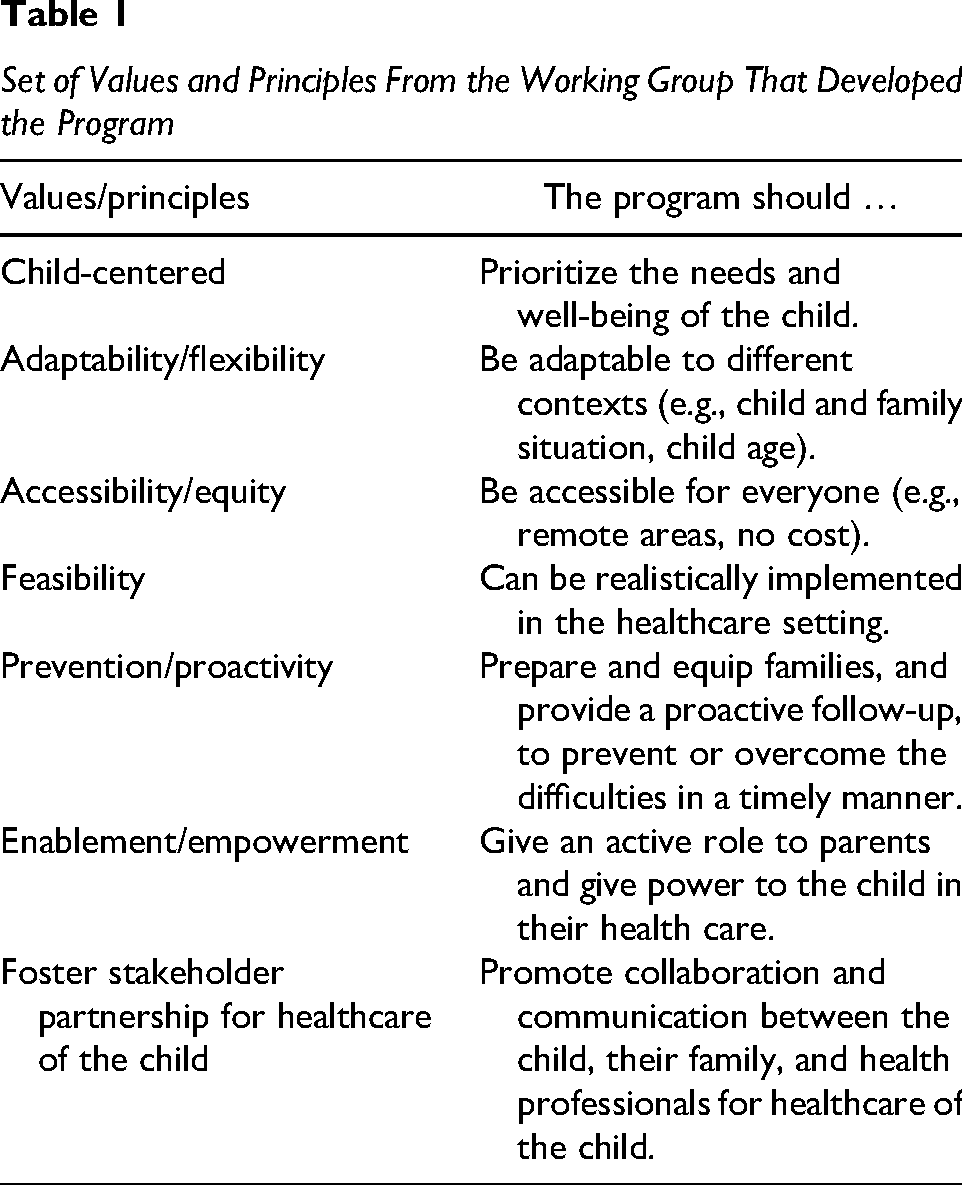
The program was developed using the BCW framework (Michie et al., 2014). As recommended by the BCW, decision-making throughout the process was guided by the APEASE criteria: affordability, practicability, effectiveness/cost-effectiveness, acceptability, side-effects/safety, and equity (Michie et al., 2014), as well as by a set of key values and principles that emerged from the discussions during the program development activities and deemed essential by all participants (Table 1).
Set of Values and Principles From the Working Group That Developed the Program
Participative Approach
The working group comprised of researchers as well as stakeholders directly involved in the care of children with ALL. Researchers had various backgrounds (pharmacy, pediatric oncology, social work, psychosocial oncology, patient education, epidemiology, anthropology, sociology). Stakeholders included four parents of children receiving OC as part of ALL treatment and six professionals of a multidisciplinary team providing care to patients with a pediatric cancer (Table 2). The roles of stakeholders were to (a) share experiential knowledge regarding OC as a parent of a child with ALL or as a professional providing health care and services to children with ALL, (b) comment on program proposals and prototypes prepared by the research team to achieve all steps of the BCW process, and (c) validate that the changes made were in accordance with their comments. The working group was actively involved at each stage of the program development through online meetings. A total of 10 two-hr meetings were held either with the parents (six meetings) or with the professionals (four meetings). The meetings were prepared and led by three to four members of the research team. Two parents along with three members of the research team also participated in two 3-hr in-person co-construction activities to develop a first prototype of the detailed program plan. For these co-construction activities, the parents individually completed a grid of relevant topics to include in the program (priority level, for whom, at what time of the care trajectory, using what types of intervention, and means of delivery). This grid was then used to fuel the in-person group discussion activity during which the most important items were chosen and placed on a timeline at points representing milestone of the 2-year treatment journey. The program plan was then refined and approved by all members of the working group during a regular online meeting. All online meetings and co-construction activities were audio-recorded and an anonymized summary of the discussions was compiled. Compensations were offered to stakeholders involved in the working group ($50 per online meeting, $75 plus reimbursement of the parking cost and a light meal for in-person co-construction activities).
Characteristics of the Parents (n = 4) and the Professionals (n = 6) in the Working Group
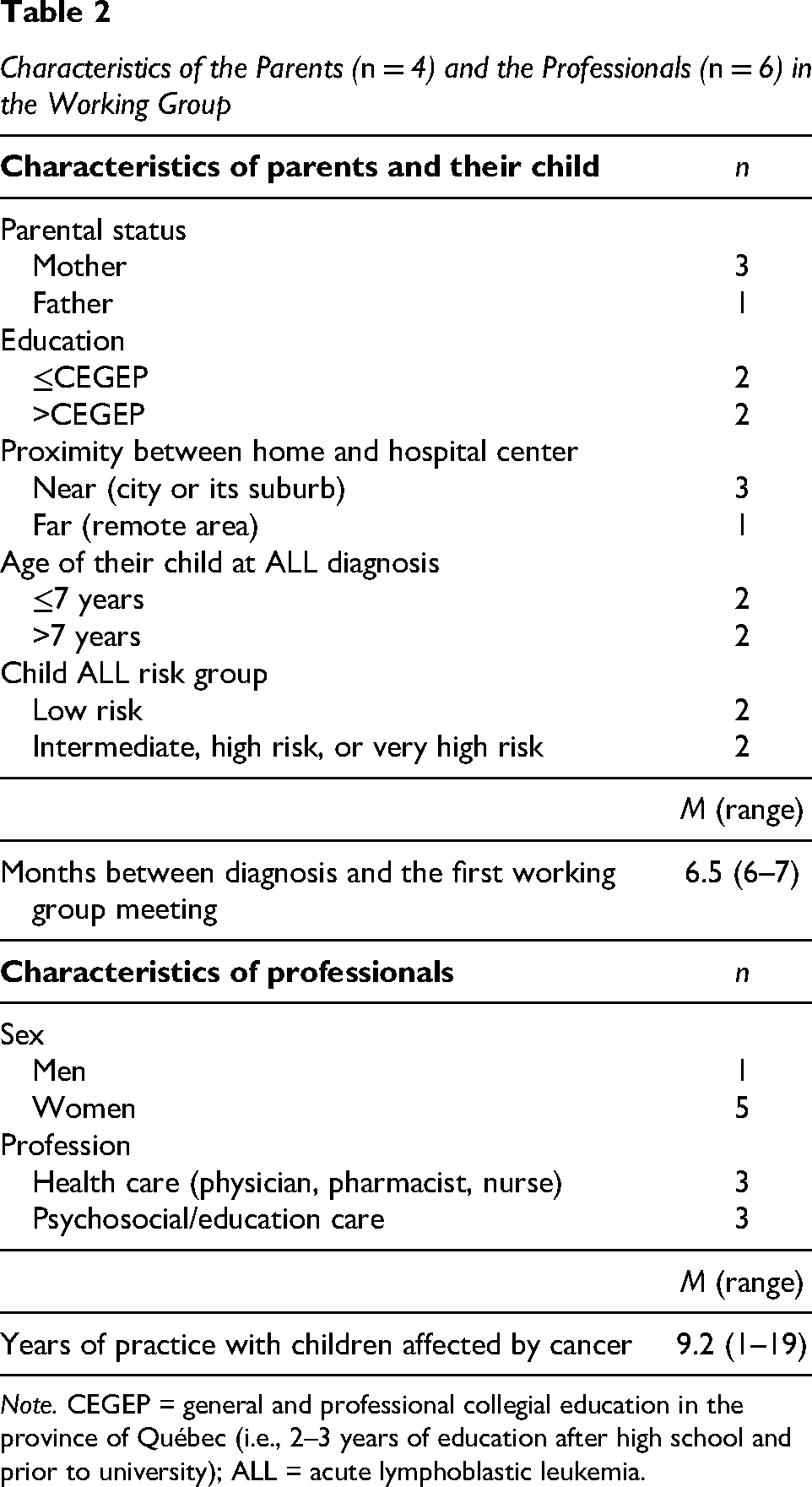
Note. CEGEP = general and professional collegial education in the province of Québec (i.e., 2–3 years of education after high school and prior to university); ALL = acute lymphoblastic leukemia.
Behavior Change Wheel
BCW is a comprehensive and systematic framework to guide the development of behavior change interventions (Michie et al., 2011, 2014). BCW has been extensively used to design interventions in health domains (Band et al., 2017; Gabriel et al., 2022; Los et al., 2021; MacPherson et al., 2021; Wray et al., 2021). It is anchored in the Capability, Opportunity, and Motivation-Behavior model (COM-B) in which each domain influences the adoption of a behavior and is potentially modifiable by an intervention (Michie et al., 2014). The capability (C) domain includes the physical and psychological capacities of the person to perform a behavior. The opportunity (O) refers to the physical and social opportunities in the person's environment that allow the behavior to occur. The motivation (M) can be reflective (e.g., a desire) or automatic (e.g., a need) and is what brings a person to want to do a behavior (B). COM-B regroups the 14 domains of the Theoretical Domains Framework (TDF; Figure 1; Atkins et al., 2017; Michie et al., 2014). BCW framework links COM-B and associated TDF domains to nine intervention functions, seven policy categories, and a taxonomy of 93 behavior change techniques (BCTs; Figure 1; Michie et al., 2013, 2014). BCW process is structured around three main stages subdivided into eight steps, defined as: Stage 1—“understand the behavior” to change, Stage 2—“identify intervention options,” and Stage 3—“identify content and implementation options” (Michie et al., 2014). Evidence-based knowledge from relevant scientific literature and experiential knowledge from the working group stakeholders were carefully evaluated and integrated throughout the developmental process. This included findings from studies on families’ experiences with OC (Aburn & Gott, 2014; Akkawi El Edelbi et al., 2023; David et al., 2023; Heneghan et al., 2020; Kahn et al., 2023; Kremeike et al., 2015; McGrath & Pitcher, 2002; McGrath & Rawson-Huff, 2010; Tang et al., 2022; Williams & McCarthy, 2015) and from our previous studies with 13 parents responsible for at-home OC (Camiré-Bernier et al., 2021) and 19 healthcare professionals of an interdisciplinary team in pediatric oncology (unpublished data).
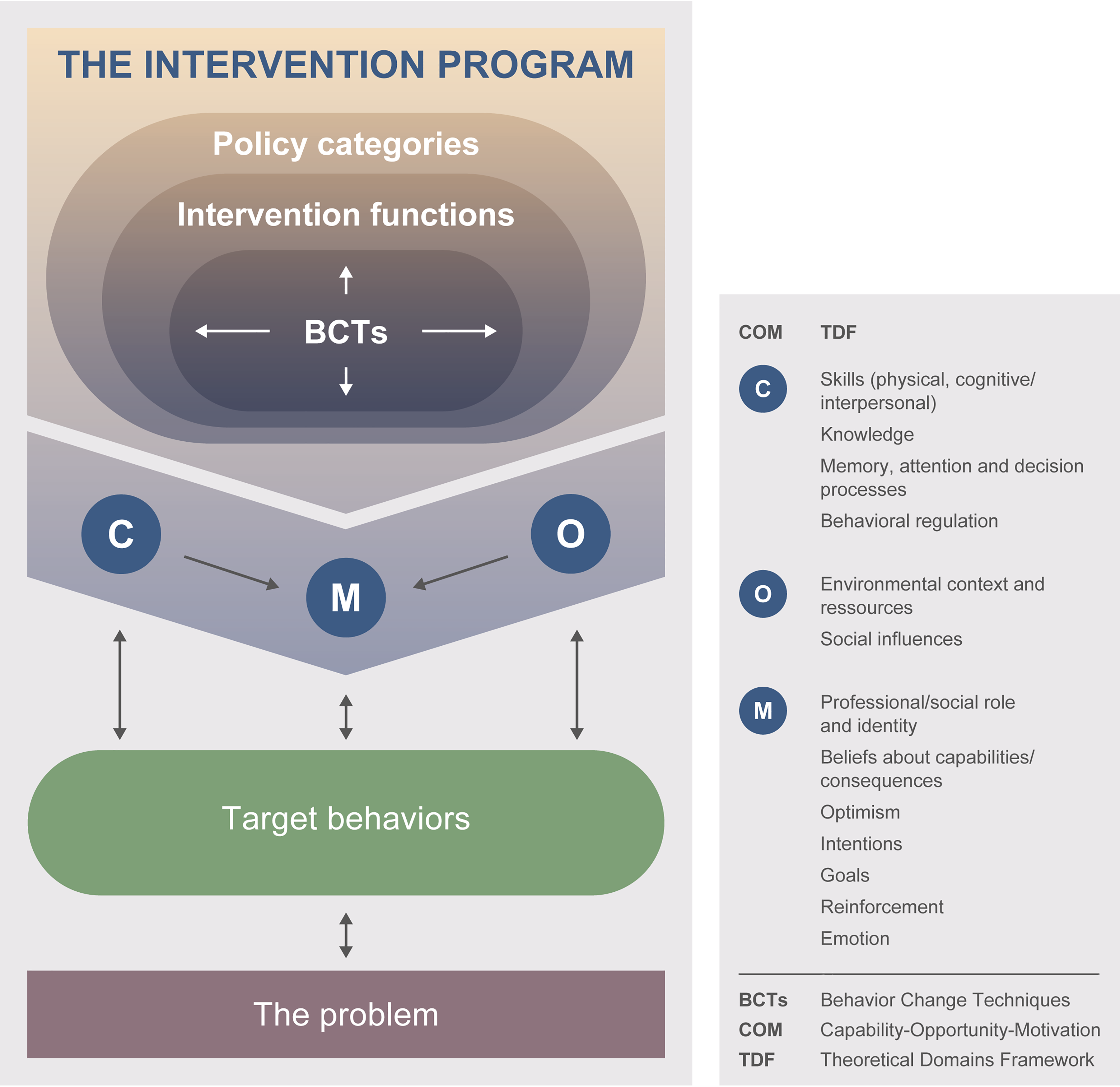
Simplified Schematic of the BCW Framework Used to Develop the Program.
Stage 1 (“understand the behavior to change”) aims to fully understand the problem to be solved, to then clearly define the main objective to be achieved with the program in the target population. This stage comprises four steps. Following BCW framework (Michie et al., 2014), the working group identified the behavior at the core of the problem (what), the individuals/groups involved in the behavior (who), and where it takes place (where) (Step 1). A list of all potential behaviors that could be adopted by the family to reach the main objective was then retained. Each behavior was evaluated for (a) its likely impact on the problem, (b) how easily it could be adopted, (c) its anticipated repercussions on other behaviors, and (d) its measurability (Step 2). The selected behaviors were further specified (Step 3) and the group identified what needed to be changed for the families to be able to adopt these targeted behaviors (Step 4), that is the factors that could influence their adoption. Those factors were classified into the most relevant COM-B and TDF domains (Atkins et al., 2017; Michie et al., 2014).
Stage 2 (“identify intervention options”) consists of two steps that are the identification of intervention functions (Step 5) and policy categories (Step 6). Intervention functions are categories of means used to bring a person or a group to change factors likely to promote the adoption of the targeted behaviors. BCW provides nine intervention functions linked with the COM-B and TDF domains: education, training, environmental restructuring, enablement, modeling, restriction, persuasion, incentivization, and coercion. The relevant authorities can also support the desired changes by implementing measures which fall into seven policy categories: guidelines, fiscal measures, environmental and social planning, communication and marketing, service provision, legislation, and regulation. Thus, the intervention functions most relevant to the COM-B and TDF domains determined at Stage 1 were identified (Step 5). The choice of policy categories was based on their good fit with the intervention functions and their accessibility in the context of the hospital center where the program was developed (Step 6).
At Stage 3 (“identify content and implementation options”), the BCTs from the BCT taxonomy (V1) that were most likely to lead to the desired targeted behaviors were identified (Step 7) (Michie et al., 2013, 2014). BCTs are methods used in interventions to act on factors poised to effect behavior change. We then developed the final program plan by choosing the components that translated and anchored the chosen intervention functions, policy categories, and BCTs into a coherent whole. We thus planned the method of delivery of the interventions (e.g., face-to-face/remote, individual/group session, written material/audio-visual material, etc.), the actors involved (providers, receivers), timing over the 2 years of treatment, and their specific content (Step 8). The final program prototype and its components was discussed with and approved by all members of the working group.
Results
BCW Stage 1: Understand the Behavior to Change
The problem to be solved with the program was summarized as: “Families of children with ALL can meet difficulties with at-home OC. These difficulties can impact various aspects of the family life, change over time and according to the family context, as well as affect the use of OC and the well-being of the whole family.” (Figure 2)

Problem Addressed by the Program and the Targeted Behaviors and Subbehaviors to Achieve Its Main Goal.
Therefore, the main objective to be achieved by the program was formulated as: “To enable families to face the difficulties met with OC by mobilizing tools and resources in collaboration with the healthcare team to support the proper use of OC and the family well-being.” From the list of behaviors that could help solve this problem, four key themes emerged: Theme 1—OC's safe and effective intake, Theme 2—the impact of OC on the child, Theme 3—the impact of OC on parents, and Theme 4—the impact of OC on the family unit. The first two themes were prioritized by the working group since the first one addressed the basics for safe at-home OC management and the second one addressed the most pressing unmet needs of families related to OC. These themes further aligned with the working group's core values, in particular “child-centeredness.” These themes should also have a positive spill-over impact on Themes 3 and 4 related to parents and family unit. For each of the first two themes, the main behavior to be targeted by the program was formulated and defined by subbehaviors necessary to achieve the targeted behaviors (Figure 2). Factors influencing their adoption were identified and categorized according to the most appropriate COM-B components and their associated TDF domains (indicated in brackets), namely physical capability (skills-physical), psychological capability (knowledge, skills-cognitive and interpersonal), reflective motivation (beliefs about capabilities), physical opportunity (environmental context and resources), and social opportunity (social influences). These factors are also supported by examples from the literature (Supplemental Table 1).
BCW Stage 2: Identify Intervention Options
Based on the COM-B and TDF domains identified above, the chosen intervention functions to be included in the program plan were education, training, enablement, modeling, and persuasion (Table 3). The policy category, service provision, was deemed appropriate and accessible in the context of our study and the care environment where we planned to first deploy the program (Table 3). Indeed, it was decided that the program would be developed with the perspective of being an added feature “bonus” to the standard care already offered to families to enhance support with at-home OC.
Summary of the Applicable BCW Concepts Based on Program Components

Note. BCW = Behavior Change Wheel; COM-B = Capability, Opportunity, and Motivation-Behavior model; TDF = Theoretical Domains Framework; BCT = Behavior Change Techniques; OC = oral chemotherapy.
The policy category that applies in order to support the chosen intervention functions is “service provision.”
BCT groups and the specific components of the BCT taxonomy (V1) applying (Michie et al., 2013).
BCW Stage 3: Identify Content and Implementation Options
This stage led to the development of the intervention plan by concretely translating the selected intervention functions, policy category, and BCTs. The identified BCTs belonged to seven of the 16 major BCT taxonomy groupings: goals and planning, social support, shaping knowledge, comparison of behavior, associations, comparison of outcomes, and antecedents (Table 3). The intervention strategies translated into three program components: Component 1—education and training on OC management and safe use, Component 2—evaluation and counseling for OC side effects, and Component 3—empowerment of families and normalization of their experience with OC (Table 3). The program was built to be compatible with the current care offered at our institution and to be delivered by professionals from the regular healthcare team, primarily the nurses and pharmacists. Thus, all its components were designed to integrate with regular treatment and follow-up appointments. The program materials were created with the intention of supporting the professionals already in place for program delivery, as well as allowing families to use them autonomously. The final program plan approved by all of the working group members is outlined in Figure 3 and described below.
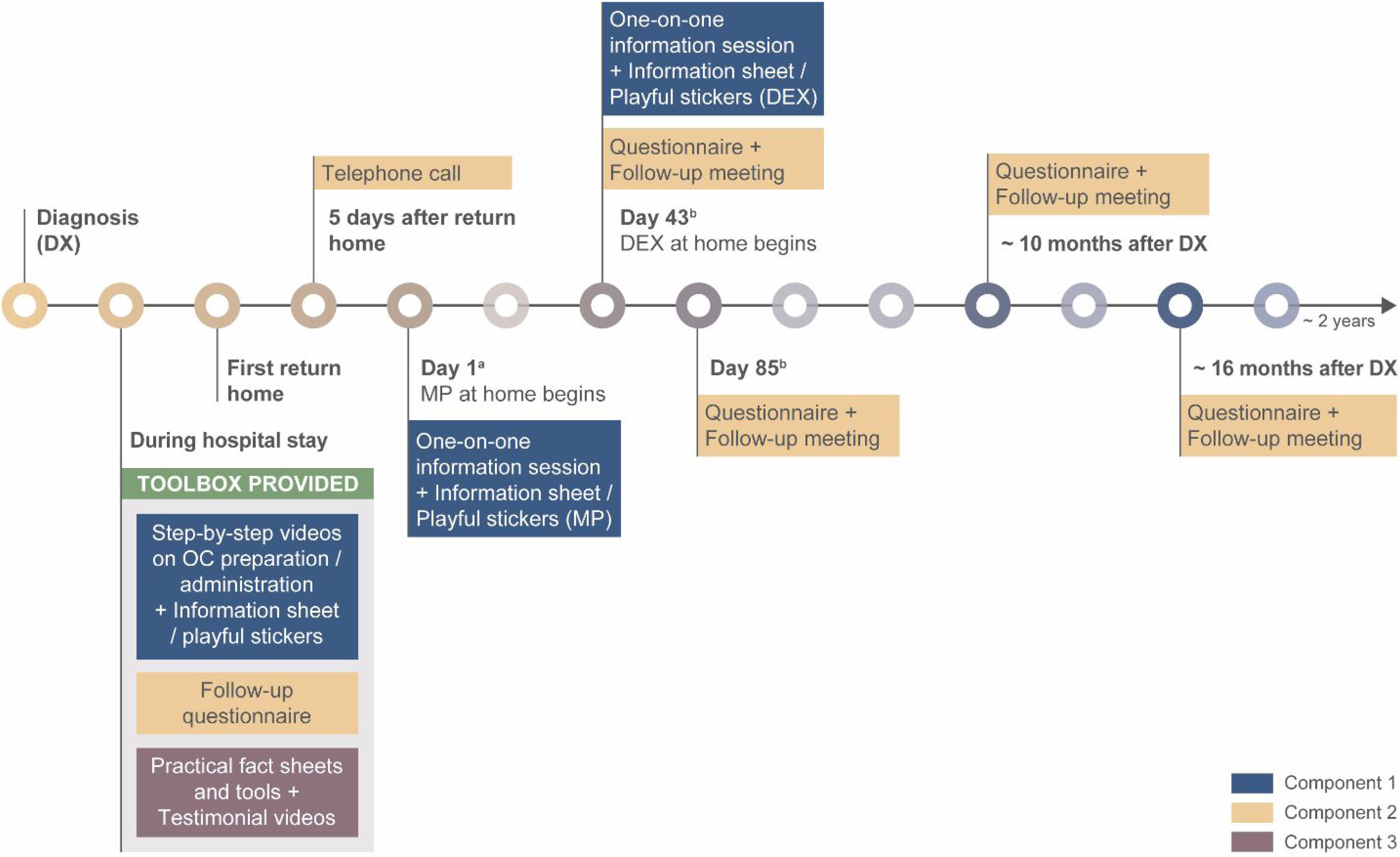
Overview of the Program Plan and Its Components Delivered Throughout the Treatment.
Program Components
The program components are presented in Table 3 as well as the COM-B/TDF domains, intervention functions, and the BCT group/specific BCTs supporting each of them. These are detailed below.
Component 1 (education and training) aims at preparing parents to administer OC at home, ensuring they gain the essential knowledge and skills to feel confident about their capability to efficiently and safely take over their child's OC treatment. Through diverse modes, parents will be provided with information adapted to the context of ALL therapy regarding the thiopurines (MP or thioguanine) and corticosteroids (prednisone or DEX). Two short standardized one-on-one information sessions with a health professional will be held before initiating each drug at home (before hospital discharge and/or during a follow-up visit, as applicable; targeted professionals: pharmacists). Parents will also be provided with take-home information sheets summarizing the key points. Specifically, the information will cover OC intake (dose, frequency, duration, with or without food, etc.), handling and storage safety recommendations, as well as the common side effects to monitor and those requiring rapid assistance from the medical team (intervention function/BCTs: education/instruction on how to perform the behavior). Playful stickers will be affixed on these information sheets to inform and prompt parents to use the other tools of the program according to their needs (intervention function/BCTs: education/prompts and cues). Short step-by-step videos (under 5 min each), accessible online at all times, will demonstrate how to handle, prepare, and administer cytotoxic drugs in tablets (including water dilution of MP tablets) or in liquid form, and inform about the option to use empty pill capsules to limit the number of pills to swallow (intervention function/BCTs: training/instruction on how to perform the behavior, demonstration of the behavior). We further expect that the information sheets and videos will help the healthcare team in their teaching of patients and serve as an at-home reminder to parents.
Component 2 (evaluation and counseling) aims to proactively identify difficulties that may arise with OC, and when necessary, to provide advices to help families cope with these situations. This will be achieved through systematically planned discussion periods throughout treatment between a member of the healthcare team and families, so that they can share their difficulties with OC, ask questions, and have easy access to resources (targeted professionals: oncology pivot nurses). Thus, a brief telephone call will take place 5 days after the first discharge from the hospital to proactively find out how the return home is going with the OC, answer questions, and advise or redirect to the appropriate tools or resources as necessary (intervention function/BCTs: enablement/social support). Four standardized follow-up meetings will also be held at key time points of the 2-year treatment trajectory when some support with OC may be needed, based on the literature and the working group. These time points include when difficulties are experienced for the first time shortly after the return home (e.g., establishing a new family routine to implement each OC at home, dealing with OC administration issues and adverse effects; Meetings 1 and 2); when novel difficulties occur later, toward the end of the first year of treatment (e.g., returning to school with OC; Meeting 3); and near the end of the 2-year treatment period, when the fatigue of dealing with OC and their side effects sets in (Meeting 4). In the context of the treatment protocol on which the program development is based, the first and second follow-up meetings will occur respectively after two consecutive at-home cycles with MP and DEX (i.e., after the second block of 14 days of MP and of 5 days of DEX on a 21-day cycle basis). This period of exposure with OC at home provides sufficient time for families to experience the difficulties most likely to be encountered at the start of at-home OC use, while allowing early management of these difficulties. The third follow-up meeting corresponds to the moment when the treatment becomes less immunosuppressive, making possible the return to normal activities. The fourth one is planned when families are most likely to need support to see the treatment through to completion. Follow-up meetings for a low-risk ALL treated with DFCI protocol (16-001) are exemplified in Figure 3. These meetings are planned at the regular appointment closest to the program target time to avoid extra hospital visits. The discussion during these follow-up meetings will be guided by a self-administered questionnaire completed by parents prior to the meeting to help them assess the presence and the degree of severity of the physical, psychological, and social difficulties most likely related to OC. With the help of the medical team, families will identify and try strategies that are best suited for them to overcome the most worrisome or disturbing difficulties they encountered with OC. This may include tips and tools offered in the program or any other interventions or resources proposed by the healthcare team. While offering support, the professional involved in those meetings will also help families to set up an action plan for the use of chosen tools, including determining objectively achievable goals, the trial period, and the efficacy evaluation criteria (intervention function/BCTs: enablement/social support, adding objects to the environment, action planning, goal setting). In the subsequent meetings, a review of the employed strategies and their results will be completed. If needed, the professional will help families adjust or change the intervention strategy (intervention function/BCTs: enablement/review goals).
Component 3 (empowerment and normalization) aims to enable parents to autonomously manage certain difficulties encountered with OC, at the very moment they arise at home, by implementing safe advice that has been successful for other families. By giving an active role to parents and their child in the management of difficulties experienced with OC, we also wish to foster a sense of empowerment toward their child's care. Moreover, it aims to provide a frame of reference more aligned with the new reality experienced due to ALL and its treatment. To this end, practical fact sheets will be provided to allow parents and the child to act on difficulties often met with OC and whose nature and intensity are not judged dangerous for the child. The topics covered are: anger/aggressivity, sadness, food cravings/polyphagia, insomnia, integration of OC in family routine, and tips/collaboration for taking OC. Each fact sheet will include, in lay terms, a definition of the difficulty, how to recognize it, what it can hide or express, a list of activities and tips to try out, as well as the signs and symptoms indicating a need for medical assistance. Some of the fact sheets proposed activities will be demonstrated in video capsules (intervention function/BCTs: training/instruction on how to perform the behavior, demonstration of the behavior; enablement/adding to objects to the environment, action planning). In addition, testimonial videos of parents and former patients (intervention function/BCTs: persuasion/credible source, social comparison), sharing the difficulties they faced during the treatment and what they put in place to face them (intervention function/BCTs: modeling/demonstration of the behavior), will be used to help families normalize their new reality with OC (e.g., psychobehavioral side effects, changes in daily life), encourage them, and provide additional tips or resources to cope with OC difficulties. These videos could also provide families with an aspirational model (intervention function/BCTs: persuasion/social comparison) and so, could help continue with the treatment until its completion and feel less alone in their situation. This material, and that for the other components (hard copies of information sheets, practical fact sheets, and questionnaire, as well as barcodes to access the online video capsules), will be placed in a toolbox given to each family during the first month of treatment, before hospital discharge. In accordance with the core value “accessibility/equity” that guided the program's working group, arrangements will be made for families who do not have access to the internet or electronic devices (e.g., provide a tablet during hospital visits for viewing video capsules, hard copy summary of video capsule content, etc.).
Discussion
By following the structured framework of BCW (Michie et al., 2014) and employing a participatory approach, we developed the detailed plan of the first multifaceted program specifically dedicated to support families of children diagnosed with ALL with combined at-home OC (thiopurines and corticosteroids). This three-component program provides clinical tools and services to better prepare and support families through the challenges most likely to occur during the 2-year treatment period with OC as documented by the literature (Aburn & Gott, 2014; Akkawi El Edelbi et al., 2023; Camiré-Bernier et al., 2021; David et al., 2023; Heneghan et al., 2020; Kahn et al., 2017, 2023; Kremeike et al., 2015; McGrath & Pitcher, 2002; McGrath & Rawson-Huff, 2010; Tang et al., 2022; Williams & McCarthy, 2015) and validated by stakeholders (parents and professionals) involved in our working group. This development process resulted in a program that is likely to (a) address pressing unmet needs of families with at-home OC, (b) be delivered in a manner adapted to the families and clinical practices, and (c) optimize the use and utility of the program for as many families as possible. We believe that these features are likely to increase the chance of the program's success, both in efficacy and implementation, but this will need to be formally evaluated by other studies. These crucial program features are further discussed below.
The program's content is designed to meet the essential and most pressing needs expressed by families and healthcare professionals regarding at-home OC. The literature and our previous work underlined that the intensity of psychological side effects of DEX are unexpected and worrisome for families and are aspects that would definitively benefit from more support (Camiré-Bernier et al., 2021; David et al., 2023; McGrath & Pitcher, 2002; McGrath & Rawson-Huff, 2010; Williams & McCarthy, 2015). Among the published interventions studied in pediatric ALL, one specifically focuses on strategies to help parents manage the emotions and behavior of their child during the maintenance phase of treatment, without being specific to DEX (Williams et al., 2016). A distinctive and innovative aspect of our program is the practical and easily accessible tools to help parents and the child manage the psychobehavioral consequences of DEX on the child. By informing families about these expected side effects and the intensity they can reach, as well as by providing resources to acquire the knowledge and skills to identify and manage the ensuing difficulties, the program has the potential to lessen the families’ burden and improve the well-being of all family members. Indeed, timely management of difficulties could limit their worsening or avoid long-term family fatigue. Such an approach also has the potential to prevent children and their families from developing unhealthy or ineffective strategies that may lead to negative impacts in the longer term (Andrés-Jensen et al., 2020; Arpaci et al., 2022; Williams et al., 2014).
Particular attention was paid to developing a program that is uniquely adapted to the reality and the burden of families and healthcare professionals caring for a child with ALL. The program is designed to be delivered in a timely manner over the entire treatment period to help families cope with the various difficulties likely to be met over the 2 years of OC. The program is also suitable for various clinical situations or family contexts. Thus, it has the potential to better respond to the family's needs and difficulties as they arise during the care trajectory. Among others, the families will have repeated opportunities to (a) be reminded of essential information and resources available to help them face OC difficulties; and (b) have follow-up meetings with a professional to detect, discuss, and be counseled on difficulties they experienced at home with OC. The opportunity to ask questions or to be assisted by a qualified person with childcare was also offered in some interventions studied in pediatric ALL through systematic telephone calls or social networking tools (Wang et al., 2018; Williams et al., 2016). Unlike the systematic meetings offered with our program, these opportunities were not particularly designed to assist parents in the screening of the difficulties encountered by their children with at-home OC nor to advise them on tools and resources personalized to their needs. Given the emotional burden and workload that childhood ALL diagnosis and care can inflict on parents (Aburn & Gott, 2014; Camiré-Bernier et al., 2021; McGrath, 2001; Tang et al., 2022), keeping them engaged with the program for 2 years could be challenging, and potentially even more so for families living in remote regions. For instance, previous feasibility studies in pediatric cancer showed that time-demanding programs, those requiring extra visits at the hospital, or whose content was not specific to cancer, were less attractive for families as highlighted by difficulties in recruiting and retaining participants or low participation rate in the program components (Beaulieu-Gagnon et al., 2019; Williams et al., 2016). This was the case for a program tested in pediatric ALL that was developed for another clinical context (Williams et al., 2016). The main criticisms were the time commitment and contents not adapted or relevant to ALL. This emphasized the importance of programs tailored to the unique needs and considerations of families in pediatric ALL. To limit such foreseeable issues, our program comprises a limited number of standardized follow-up meetings at critical time points of the care trajectory. They are short and scheduled to coincide with standard care, thus adding no extra visits to the hospital. We also provide tools that can be used independently by parents at home if difficulties arise. By limiting the workload associated with the program and by tailoring it to the child's needs, we expect to increase the chances that families have the time and interest to use the program and benefit from it throughout its duration.
The interventions and material provided with the program will be delivered in various modes suitable to different learning or user preferences. As highlighted by us and others, being provided repeatedly with accurate and consistent information using diverse modes supports the understanding and retention of important information by parents, and the feeling they can properly take charge of their child's at-home OC (Akkawi El Edelbi et al., 2023; Camiré-Bernier et al., 2021). Thus, the program contains practical fact sheets, videos, families’ testimonies, a screening questionnaire, and one-on-one meetings. These interventions and tools, and the way they are delivered, are based on the rigorous evaluation and selection of the most appropriate evidence-based intervention functions and BCTs, guided by BCW (Michie et al., 2014). Moreover, several of the tools offered with the program are made to be used autonomously by families. These choices are also aligned with the key values and principles of the working group and are likely to resonate with parents of children with ALL. For instance, through education and training, the program may help equip and empower families to prevent or overcome difficulties from OC. By taking all these considerations into account, our goal is to optimize the use and utility of the program for families.
The approach chosen to develop this program has provided a structure for building a complex and coherent theory- and evidence-driven program that is also grounded in field-based needs and realities. Addressing such multifaceted problems required a multicomponent program that would have been difficult to conceive without a systematic and practical framework (Skivington et al., 2024), such as BCW (Michie et al., 2014). We further adopted a participative approach with stakeholders concerned by the problem to gain experiential and practical knowledge on the problem and the reality of caring for a child diagnosed with ALL (Pomey et al., 2015; Skivington et al., 2024). In summary, the BCW framework has structured the working group activities, while the stakeholders’ participation has been instrumental in defining and respecting a set of values and principles that guided our choices throughout the program development journey. Although the methodology used increases the likelihood that the program will be effective, relevant, and achievable in a real-life context, challenges were encountered from combining these two approaches. BCW is practical and suitable for various clinical contexts. However, it does not provide explicit guidance to pursue it as part of a participatory approach with stakeholders. The most important challenge we faced was determining how to concretely involve stakeholders of the working group in BCW stages involving fewer intangible notions, such as in Stages 2 and 3 regarding intervention functions and BCTs. Indeed, it was hardly conceivable to perform them during regular online meetings. As reported by other groups, the strategy we adopted was to translate the tasks to be performed into concrete co-construction activities and then retrospectively code the work produced in intervention functions and BCTs (Hall et al., 2020; Wray et al., 2021). By doing so, we strived to enhance the understanding, participation, and creativity of parents participating in the working group.
Strengths and Limitations
The working group was composed of stakeholders with diverse backgrounds (e.g., family context, education, expertise, etc.), with the exception of sex where men representativity was low. This may have limited our ability to capture father-specific concerns with OC. All involved parents had extensive experience with and showed a high level of health literacy on ALL and its treatment. They also had sufficient perspective, which allowed them to adopt a more objective look on their past and current experience with OC, on what was helpful or lacking in coping with OC, and what were the critical moments of the families’ journey with ALL treatment. However, parents often mentioned that their own experience might not always be identical to other families starting the treatment. Moreover, we did not consult children with ALL about their experience with OC, although it would have been highly relevant. To address this limit and ensure the acceptability and feasibility of the program and its material for a more diverse population, we plan to obtain feedback from children and parents, including fathers, with different realities and health professionals that did not participate in its development. Finally, the decision to meet parents and professionals of the working group separately was made to ensure participants were comfortable to freely express their opinions since the parents’ children were still receiving care and services at the cancer center. However, having parents and health professionals in the same meeting could have allowed for discussion on divergent perspectives and led to the emergence of new ideas. To mitigate this, we kept each group informed of the work being done with the other group. We also recognize the fact that clinical practices and settings can differ between hospitals and ALL-treatment protocols, and this must be considered when preparing for program implementation. Thus, certain adaptations of program components may be required for its implementation on a larger scale to accommodate local realities of the practice environments. For instance, this may include rethinking which professionals will deliver program components depending on the available resources and to adjust the predetermined times of the systematic one-to-one meetings consistently with the variable OC administration schedule between treatment protocols. Since other drugs such as MTX are given orally in certain protocols, modifying existing program material or developing new material specific to these drugs might also be considered. Nevertheless, the behaviors and factors targeted by our program are not limited to the specific context of our hospital center as the literature that guided those decisions included diverse treatment protocols, countries, ethnicities, and so on. Moreover, the program was designed to make these adaptations feasible. Another important aspect that can limit program implementation in a clinical setting is the incremental human and financial resource burden associated with the program. Indeed, it is likely that professionals will need additional time to deliver the program as well as to be trained on the program. To address such issues, we plan as next steps to implement and evaluate the program acceptability, feasibility, and preliminary effects in a single-center pilot study with the collaboration of healthcare and management teams in place. The pilot study will provide valuable insights into how the program can be sustained by the regular healthcare team and how much additional professional time may be needed.
Implications for Pediatric Care and Research in Oncology
We have demonstrated that it is feasible to apply a participative research approach in pediatric oncology to develop a program following a structured framework, such as BCW. This was possible by making some adjustments, including translating technical and abstract concepts into lay terms and concrete activities. As stated by many organizations and authors, the involvement of healthcare users’ in health research is expected to support a better alignment of research efforts with their needs and realities. Even though involving community stakeholders in the research process is more demanding on time, it has the potential to help develop more accountable health services—for example by being more aligned with the field problems or by promoting health equity and empowerment of underrepresented populations by giving them a voice (Brett et al., 2014; Haldane et al., 2019; Harting et al., 2022; Kuchler et al., 2022). In future evaluation and implementation stages of our program, the principles defined with the working group will continue to guide our choices. These have been key in keeping the focus on important elements that will likely resonate with target users. We encourage clinical and research teams to determine with stakeholders such guiding values and principles in the early development process.
Conclusion
The approach used in this project led to the development of a flexible and adaptable three-component program plan to support families with a child having ALL in coping with the multifaceted needs arising from at-home 2-year OC treatment. By facilitating earlier management of difficulties, or by reducing their intensity or negative consequences, the program is expected to promote the safest and most effective use of OC as well as the well-being of affected children and their families during treatment. It could also potentially lead to long-term benefits in young survivors of ALL, such as improved psychosocial health. The program may enrich clinical practices of health professionals in pediatric oncology involved in its delivery.
Supplemental Material
sj-docx-1-jpo-10.1177_27527530251337037 - Supplemental material for Development of a Program to Optimize the Experience and the Use of At-Home Oral Chemotherapies in Pediatric Acute Lymphoblastic Leukemia: A Participatory Approach Using the Behavior Change Wheel
Supplemental material, sj-docx-1-jpo-10.1177_27527530251337037 for Development of a Program to Optimize the Experience and the Use of At-Home Oral Chemotherapies in Pediatric Acute Lymphoblastic Leukemia: A Participatory Approach Using the Behavior Change Wheel by Angéline Labbé, Laurence Guillaumie, Marianne Olivier D’Avignon, Pierre-Marie David, Sophie Lauzier and Isabelle Laverdière in Journal of Pediatric Hematology/Oncology Nursing
Footnotes
Acknowledgments
We would like to thank the parents and health professional partners who were instrumental to the development of the program, and Andrea Neumann, who reviewed the manuscript for English.
Declaration of Conflicting Interests
S.L. has received unrestricted research grants from Eli Lilly Canada and Pfizer Canada for other studies not related to the one presented in this manuscript.
Funding
Authors disclose the following financial support: the Fondation Charles-Bruneau (to I.L.) and the Réseau Québécois de Recherche sur les Médicaments (to I.L. and S.L.), S.L. is a research scholar and I.L. is a clinical research scholar with funding from the Fonds de Recherche du Québec-Santé (Québec Health Research Fund; grant number FQ122109 to S.L., grant number FQ122099 to I.L.), and A.L. was awarded master's scholarships by Fonds d'enseignement et de recherche de la Faculté de pharmacie de l'Université Laval, Centre de Recherche sur le Cancer de l'Université Laval, and Chaire de recherche en soins palliatifs de l'Université Laval.
Ethical Approval and Informed Consent
This study was approved by the Institution's Ethics Board (no. 2021-5173) and a consent form was signed by stakeholders involved in the program development working group.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
