Abstract
Introduction
The evolution of precision medicine and novel therapeutics has resulted in a current overall 5-year survival rate of 85% in children diagnosed with cancer (Surveillance Epidemiology and End Results Program, [SEER], 2018). Despite increases in survival rates, cancer remains the leading cause of disease-related death in children in the United States, with approximately 1,800 children and adolescents aged birth to 19 years dying a cancer-related death each year (SEER, 2018). The therapies used to treat various cancer types lead to poorly controlled physical and psychological symptoms that are often distressing and negatively impact health outcomes, including quality of life (QoL; Hooke et al., 2020; Linder & Hooke, 2019). Children with advanced cancer, defined as relapsed, refractory, or progressive disease, are especially vulnerable to treatment-related toxicities and symptoms, and poor health outcomes.
QoL is an important health outcome in pediatric oncology, with implications for research and clinical practice. The Pediatric Quality of Life Inventory (PedsQLTM) is the most common patient-reported measure for QoL in pediatric oncology research (Anthony et al., 2014). Most studies evaluate QoL as a symptom outcome in children undergoing curative treatment or among survivors and often exclude those with advanced cancer or at end-of-life (Momani et al., 2016). However, in the first multisite, prospective, longitudinal study of children with advanced cancer, or the PediQUEST study, researchers documented several distressing symptoms and their associations with poor patient and family outcomes, including child QoL (Rosenberg et al., 2016). Additional limitations of QoL research include the primary use of cross-sectional designs, homogenous samples (restricted to only certain diagnoses), and variation in symptom and QoL measurements. Together these limitations support the need for research to longitudinally evaluate QoL among children diagnosed with different types of cancer, including those with advanced disease (Momani et al., 2016; Steineck et al., 2022).
The evolution of patient-reported outcome (PRO) monitoring in symptom science has yielded several symptom measures to capture patient voices across developmental levels, including Memorial Symptom Assessment Scale (MSAS), SSPedi, and PediQUEST MSAS (Leahy et al., 2018). Recently, researchers developed and validated the Pediatric PRO version of the National Cancer Institute's (NCI's) Common Terminology Criteria for Adverse Events (Ped PRO-CTCAE®) to document symptom AEs from the child's perspective (Reeve et al., 2020). Because the Ped PRO-CTCAE® is aligned with CTCAE reporting requirements associated with clinical trials, it is an optimal symptom measure ready to integrate into clinical trials for patient-reported adverse event (AE) monitoring and reporting. However, the use of the new Ped PRO-CTCAE® in various pediatric oncology subpopulations has yet to emerge. Further, the relationship between symptom AEs and QoL in children with advanced cancer remains undocumented.
To address this gap in evaluating the relationship between reported symptom AEs and QoL in children with advanced cancer, we collected symptom AE and QoL data from children diagnosed with advanced cancer over 6 months. Our primary aim was to test the feasibility and acceptability of electronically administering the Ped PRO-CTCAE® and our secondary aim was to explore the relationship between symptom AEs and QoL over time. Here we report results from the secondary aim, while results from the primary aim are reported elsewhere (Montgomery et al., 2022). Based on recent findings validating the use of Ped PRO-CTCAE® to assess symptoms when compared to existing symptom measures (Reeve et al., 2020) and previously documented negative relationships between patient-reported symptoms and QoL (Rosenberg et al., 2016; Steineck et al., 2022), we expected to observe poorer QoL with higher reported symptom AEs over time.
Methods
This paper presents the analysis of symptom AE and QoL data generated from a longitudinal, prospective, multisite study.
Participants and Settings
Participants were recruited from one of three Children's Oncology Group-affiliated pediatric cancer centers (American Family Children's Hospital, Children's Hospital Colorado, and Children's Hospital Los Angeles) between January and October 2020. Children aged 2 to 18 with a diagnosis of advanced cancer defined as a 1-week or longer history of progressive, recurrent, or nonresponsive disease or a decision not to pursue curative-focused therapy and who were English or Spanish literate were eligible to participate. Caregivers of eligible children aged 2 to 7 were eligible if they were English or Spanish literate. Children and caregivers were excluded if they were unable to read, write, or understand English or Spanish or were otherwise unable to complete study procedures. Additionally, children who were referred to a participating center for a clinical trial with an expected treatment course of less than six months were also excluded. Once eligibility was confirmed by the research team, permission was sought from the primary treating team to approach the child and caregiver about study participation. Research personnel conducted consent (and assent when applicable) procedures for interested children and caregivers per institutional standards.
Ethics
Participating sites obtained approvals from the institutional review board of record.
Design
Symptom AE and QoL data were collected at enrollment and every 2 weeks for 6 months. Participants completed surveys on an electronic device after receiving a text or email notification from a Research Electronic Data Capture (REDCap) database hosted at the primary site. Electronic reminders were sent every 2 to 3 days with a final phone call reminder on the last day of the 7-day data collection window. Children aged 8 to 18 provided self-report data, while caregivers of children aged 2 to 7 provided proxy-report data. A 2-week interval was selected to capture potential changes in symptoms in response to disease status and new and ongoing cancer treatment regimens and informed by our team's previous work (Montgomery et al., 2020). Child demographics and clinical characteristics (e.g., disease status, cancer-directed treatment, etc.) were extracted from the medical record by research personnel. Participants received compensation in the form of a $10 gift card for each completed time point.
Measurement
Symptom AEs were evaluated using the Ped PRO-CTCAE®. The measure captures physical and psychological symptom AEs and the associated frequency, severity, or interference. Individual symptom AE items are scored on a 0 to 3 scale with higher scores indicating more frequent, severe, or bothersome AEs. Frequency scores were calculated to determine the presence of a symptom AE, with scores of 1 to 3 indicating the symptom AE was present and a score of 0 indicating the absence of a symptom AE. Mean scores were calculated to reflect an average individual symptom AE score (e.g., pain frequency, pain severity, and pain interference). To minimize participant burden and align with Food and Drug Administration recommendations to include patient-reported outcomes in pediatric cancer clinical trials (Murugappan et al., 2022), 24 items (15 core and nine additional) were selected from the 62-item Ped PRO-CTCAE® library to measure commonly experienced symptom AEs. Children aged 8 to 18 completed the self-reported Ped PRO-CTCAE® version, while caregivers of children aged 2 to 7 completed the caregiver version. The Ped PRO-CTCAE® has been validated in English-speaking children diagnosed with cancer and their caregivers (Reeve et al., 2020, 2021), but has not yet been formally validated among Spanish-speaking children and parents. Validity of the Ped PRO-CTCAE® self-report measures was demonstrated by acceptable correlations (0.62–0.80 for children aged 7–12, 0.44–0.94 for children aged 13–15, and 0.65–0.98 for adolescents aged 16–18) with the MSAS and PROMIS Pediatric measures (Reeve et al., 2020). Test–retest reliability demonstrated 54.3% to 93.5% agreement between two different time points (Reeve et al., 2020). The Caregiver Ped PRO-CTCAE® measure demonstrated similar acceptable validity and reliability when compared to Pediatric PROMIS parent-proxy measures (Reeve et al., 2021). Spanish versions of the measures were provided by the Ped PRO-CTCAE® research team upon request. In response to the absence of research validating Spanish translation of the Ped PRO-CTCAE® measures and the intention to be inclusive of Spanish literate children, we obtained a certificate of translation per institutional policy for the Ped PRO-CTCAE® self-report and parent-proxy measures.
QoL was evaluated using the PedsQLTM Cancer Module 3.0. The PedsQLTM Cancer Module 3.0 is comprised of eight different dimensions: pain and hurt, nausea, procedural anxiety, treatment anxiety, worry, cognitive problems, perceived physical appearance, and communication, and is scored on a 0 to 100 scale with lower scores indicating poorer QoL. Children aged 8 to 12 completed the 27-item Child Report and adolescents aged 13 to 18 completed the 27-item Teen Report by self-report. Caregivers of children aged 5 to 7 completed the 26-item Parent Report for Young Children and caregivers of children aged 2 to 4 completed the 25-item Parent Report for Toddlers by proxy. The PedsQLTM Cancer Module 3.0 has demonstrated reliability and validity in children diagnosed with cancer and their parents (Varni et al., 2002). Internal reliability coefficients were 0.72 for the child report, 0.87 for the parent report, and adequate construct validity was demonstrated based on the ability to distinguish between children on and off cancer treatment (Varni et al., 2002). Cross-culturally adapted and linguistically validated Spanish versions of the PedsQLTM Cancer Module 3.0 provided by Mapi Language Services were administered to Spanish-speaking participants (Fontibón et al., 2017).
Demographic variables, including child age, ethnicity, race, and sex were collected at baseline from the medical record. Clinical characteristics, including cancer type (hematologic malignancy, solid tumor, and central nervous system [CNS] tumor), specific diagnosis, cancer status (active disease or remission), cancer-directed treatment in the last 10 days, and cancer-directed treatment intensity were collected from the medical record at baseline and with each completed timepoint.
Analyses
Descriptive statistics were used to calculate frequencies for categorical data and means and standard deviations for continuous data. Pearson correlation coefficients were used to measure the strength of the relationship between mean individual symptom AE scores and mean total PedsQLTM scores. Effect sizes and confidence intervals were calculated to determine differences between PedsQLTM dimensions and total scores and the diagnostic groups of hematologic malignancy, non-CNS solid tumors, and CNS tumors. A general linear mixed model for repeated measures was used to evaluate the effects of a group of participants reporting symptom AEs (symptom present) compared to a group of participants who did not report symptom AEs (no symptom) on total PedsQLTM scores over time. The imbalance in select covariates (age, sex, cancer status, and cancer-directed treatment in the last 10 days) was assessed between the two groups of participants (symptom present vs. no symptom), and a propensity weight was established to adjust for differences in the covariates using the generalized boosted modeling approach, a multivariate nonparametric regression technique, to estimate the propensity weights (Zhu et al., 2015). Pairwise comparisons between groups were conducted to test for significance at individual time points. All analyses were conducted using Stata (17.0, StataCorp LLC, College Station, TX, USA; StataCorp, 2021).
Results
Demographics and Clinical Characteristics
The final sample included 49 children with a mean age of 11.1 years (2–18). The sample was heterogeneous: female (33%), non-White (21%), and Hispanic or Latino (35%). The sample included children diagnosed with a hematologic malignancy, including leukemia and lymphoma (n = 18), non-CNS solid tumor (n = 22), and CNS tumor (n = 9). Full sample characteristics are reported elsewhere (Montgomery et al., 2022). Most participants were in the child 8–12 (n = 18) and adolescent 13–18 (n = 20) age groups compared to the child 2 to 4 (n = 7) and child 5 to 7 (n = 4) age groups. Additionally, most participants preferred to receive study measures in English (n = 46) compared to Spanish (n = 3). A descriptive summary of disease and treatment characteristics is provided in Table 1. Most participants had active disease (67%–89%) at each time point. Few participants (3%–34%) had a disease progression in the previous 30 days. Most participants (34%–73%) received cancer-directed treatment in the previous 10 days that was categorized as mild (oral or outpatient intravenous chemotherapy only or minor procedures).
Disease and Treatment Characteristics for the Sample at Each Time Point
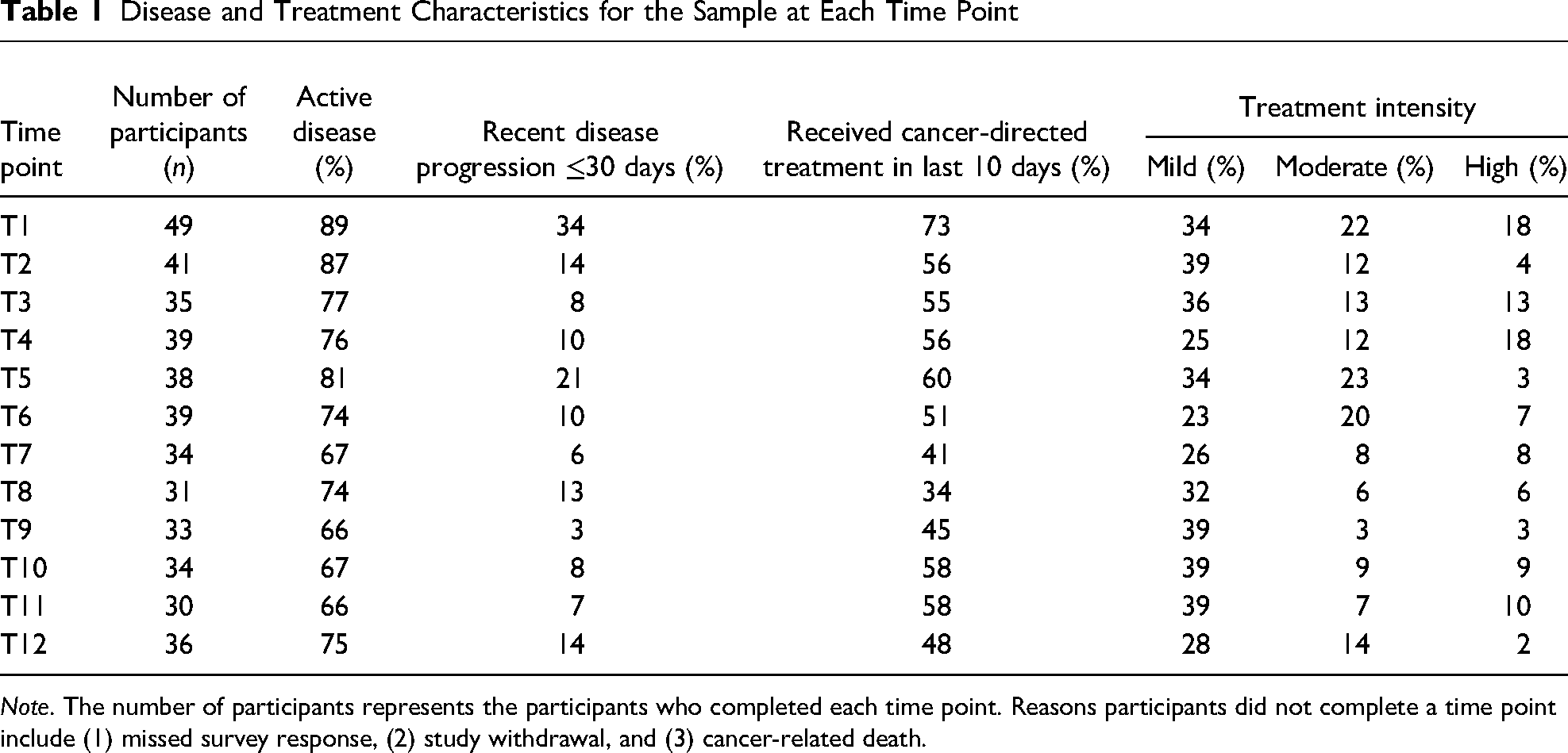
Note. The number of participants represents the participants who completed each time point. Reasons participants did not complete a time point include (1) missed survey response, (2) study withdrawal, and (3) cancer-related death.
Quality of Life
Participants completed 439 (85%) QoL measures over an average of 16 weeks (2–24 weeks). The PedsQLTM scores for the sample and by cancer type are reported in Table 2. The mean total PedsQLTM score was 73.68 (SD 17.83). The lowest scoring dimension was worry (66.43, SD 30.36), while the highest scoring dimension was appearance (78.57, SD 24.5). Comparisons of PedsQLTM dimension and total scores between cancer types demonstrated medium to large effect sizes for children diagnosed with a CNS tumor compared to children diagnosed with hematologic malignancies or non-CNS solid tumors. Children diagnosed with CNS tumors reported significantly lower scores on the worry dimension (0.84, p = .04) compared to children with non-CNS solid tumors (Table 3).
Mean PedsQLTM Scores
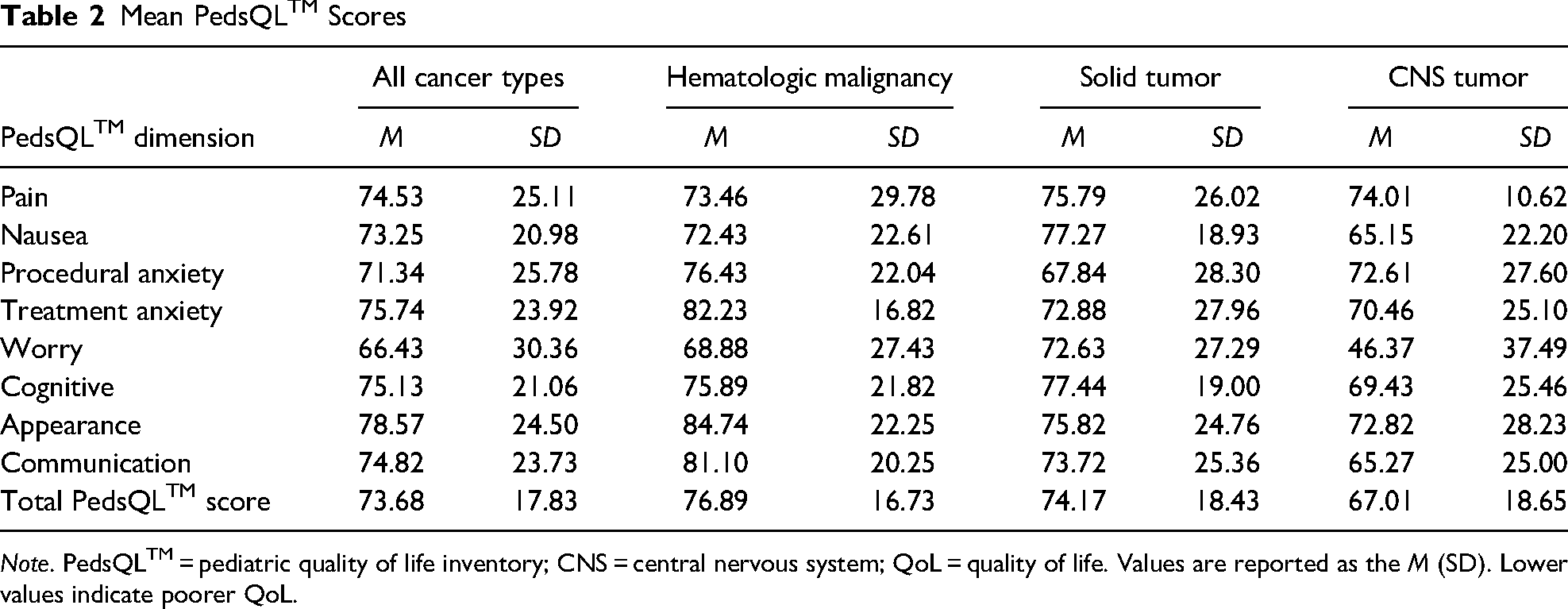
Note. PedsQLTM = pediatric quality of life inventory; CNS = central nervous system; QoL = quality of life. Values are reported as the M (SD). Lower values indicate poorer QoL.
Comparison of PedsQLTM Dimension and Total Scores Between Cancer Types
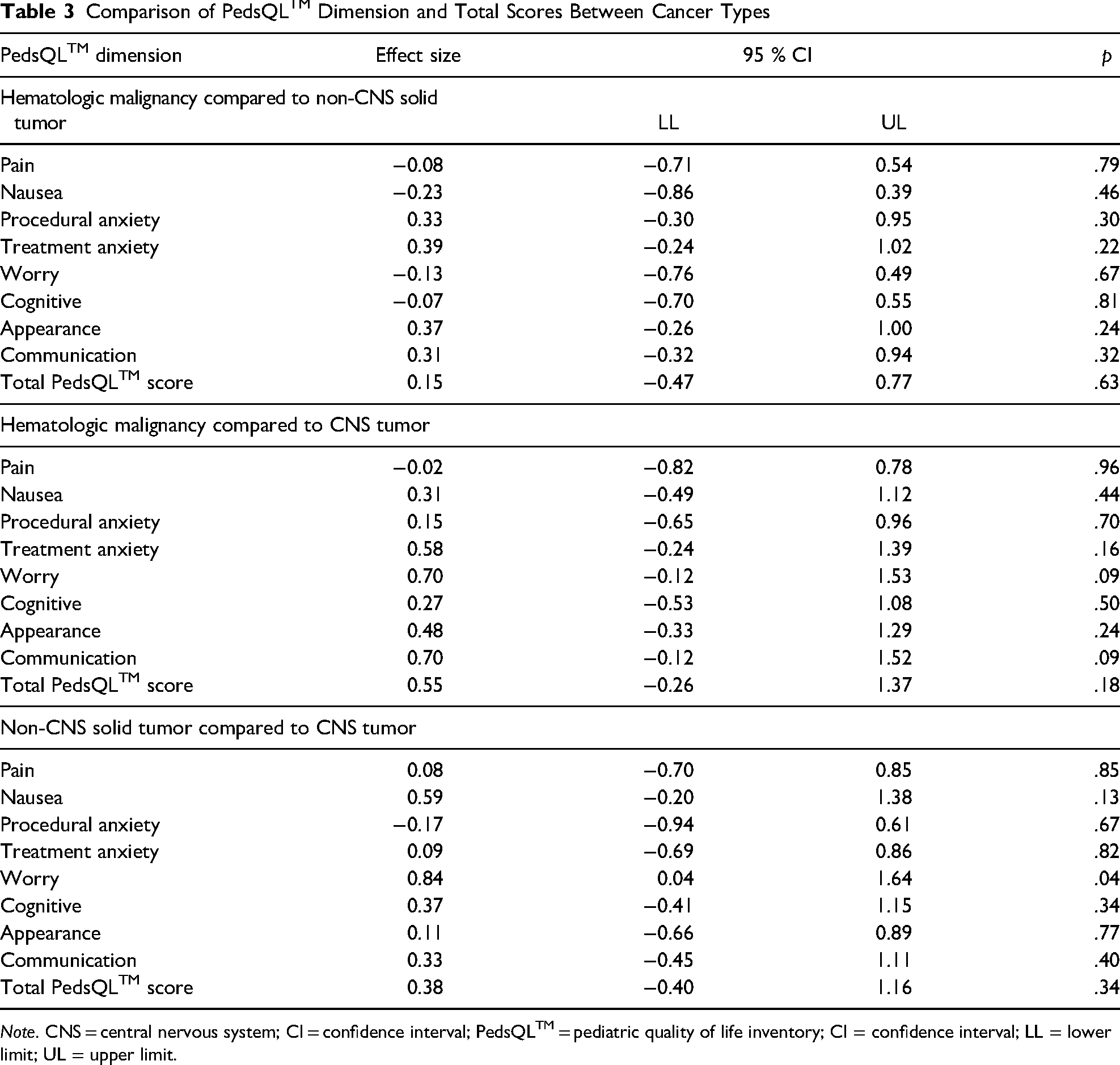
Note. CNS = central nervous system; CI = confidence interval; PedsQLTM = pediatric quality of life inventory; CI = confidence interval; LL = lower limit; UL = upper limit.
Symptom AEs and QoL
The results of correlational analyses between mean symptom AE scores and mean total PedsQLTM scores across all participants and time points are reported in Table 3. Most symptom AEs were negatively correlated with QoL, except for abdominal pain severity, concentration impairment interference, cough severity, oral pain severity, dyspnea severity, and dyspnea interference, which demonstrated insignificant weak positive correlation coefficients. Correlation coefficients > 0.4 and p < .01 were seen with frequency AEs of anxiety, pain, nausea, insomnia, and hot flashes, and severity AEs of fatigue and concentration impairment (Table 4).
Correlations Between Mean Symptom AE Score and Total PedsQLTM Score
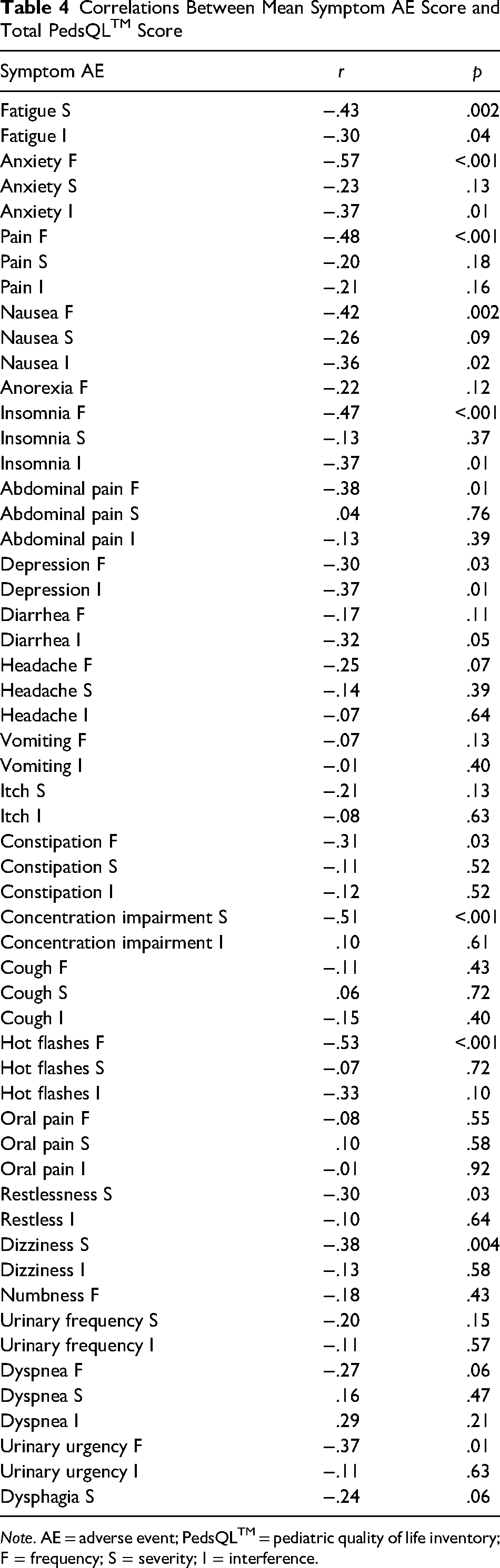
Note. AE = adverse event; PedsQLTM = pediatric quality of life inventory; F = frequency; S = severity; I = interference.
A general linear mixed effects model was used to evaluate the effect of the presence of the most frequently occurring symptom AEs with the strongest and most significant correlations in the sample (frequency AEs of anxiety, pain, nausea, insomnia, and fatigue severity) on the total PedsQLTM score, while adjusting for the covariates of age, sex, disease status, and recent cancer-directed treatment (Figure 1). Only data from the first six time points (12 weeks) were included in the model to closely align with the average length of study participation, maximize symptom AE and QoL data, and detect meaningful statistical signals. Isolated time-specific significant differences were noted for frequency AEs of nausea and anxiety at time point 4 (week 8), suggesting the presence of those symptom AEs further in the treatment trajectory may predict poorer QoL. A trend toward significance was noted for fatigue severity at time point 4. No significant differences in scores were observed for pain frequency or insomnia frequency.
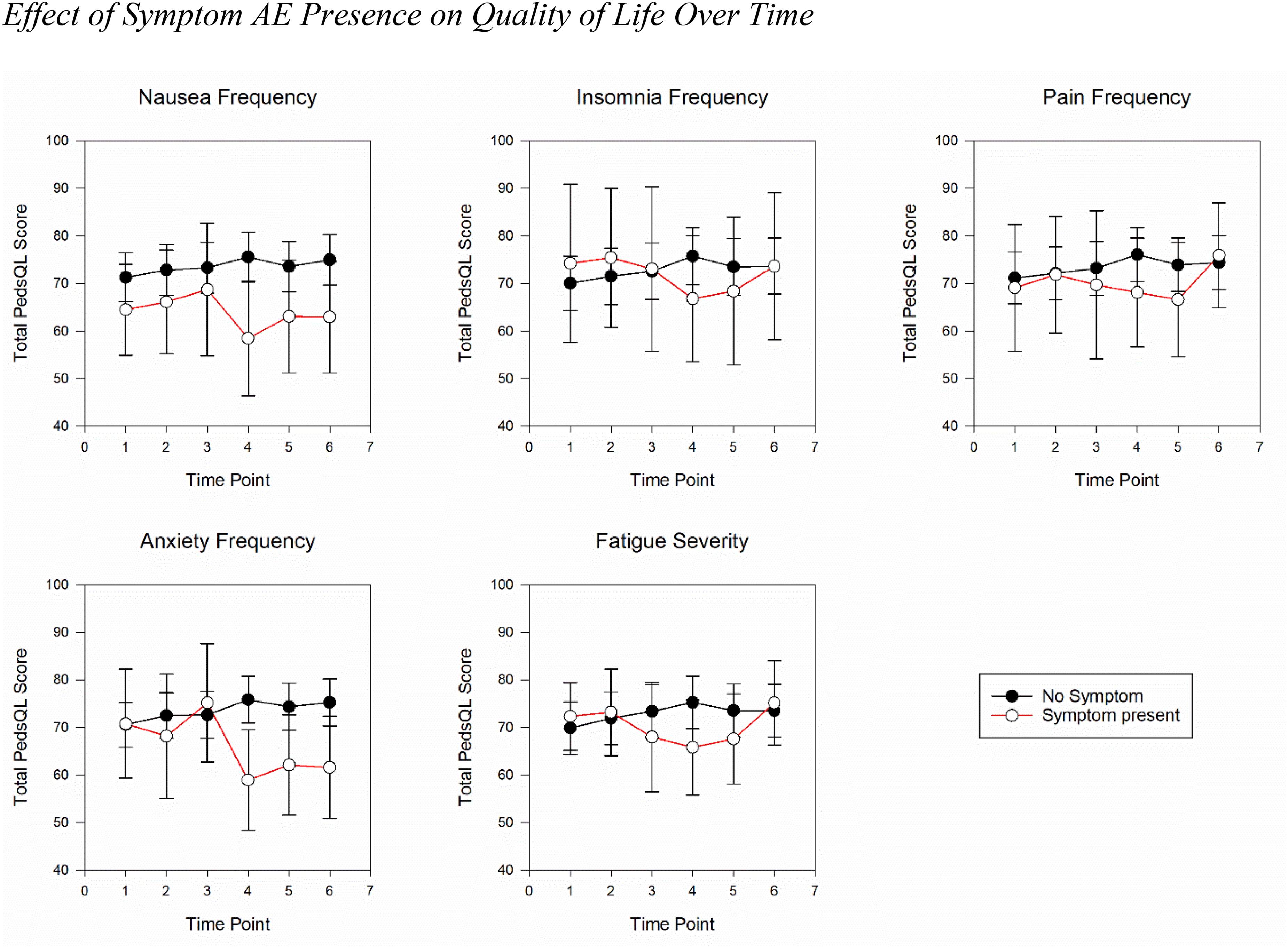
Effect of symptom adverse event (AE) presence on quality of life over time
Discussion
The effects of toxicities of cancer treatment and associated symptom AEs on QoL remain a priority for pediatric oncology. The average QoL score for all participants across time points in our study was comparable to a previous report of children with advanced cancer (Rosenberg et al., 2016) and to children without advanced disease (Rodgers et al., 2019). Notably, the average PedsQLTM scores for participants with a CNS tumor cancer type were nearly 10 points lower than that for participants with a hematologic malignancy, though this difference was not significant. This descriptive difference is consistent with previous reports describing lower QoL in children diagnosed with CNS tumors when compared to other cancer types (Eche et al., 2020; Momani et al., 2016) and suggests an opportunity for further investigation.
Results of this study indicate that researchers can use the recently validated Ped PRO-CTCAE® in conjunction with the PedsQLTM to evaluate the relationship between symptom AEs and QoL over time in children with advanced cancer. Further, the study added evidence of the negative relationship between the two constructs (symptoms and QoL), which has been previously described in this patient population and adolescents and young adults with cancer using a different symptom measure (Rosenberg et al., 2016; Steineck et al., 2022).
Fatigue severity and frequency AEs of anxiety, pain, and nausea were the most prevalent symptoms endorsed in this sample (Montgomery et al., 2022), and the presence of each resulted in a statistically significant association with poorer QoL. Given their prevalence and persistence in pediatric oncology literature, fatigue, anxiety, pain, and nausea are priority symptoms (Hooke & Linder, 2019; Linder & Hooke, 2019; Skeens et al., 2019). The negative relationship between these priority symptoms and QoL has been demonstrated by other researchers across the cancer continuum. The large prospective PediQUEST study led by Wolfe et al. (2014) found that pain, fatigue, and worry were associated with significant reductions in QoL in children with advanced cancer (Rosenberg et al., 2016). Nausea is known to be correlated with poor QoL in both acutely and chronically ill children with cancer (Dupuis et al., 2016; Hooke & Linder, 2019; Patel et al., 2017; Raybin et al., 2021). Pharmacologic interventions that are beneficial for nausea in late-stage cancer include benzodiazepines, often in conjunction with narcotics to provide multiple symptom relief (anxiety, pain, and nausea; Arland et al., 2013). However, few interventions have been developed and tested in children with advanced cancer who are not at end-of-life.
Anxiety has also been shown to be negatively associated with QoL in children undergoing cancer treatment (Martins et al., 2018). Further, there is evidence to support that emotional distress, which includes anxiety, mediates QoL among cancer survivors (Huang et al., 2017). Researchers have also demonstrated an association between worry about symptoms as signs of recurrence and fear of recurrence and poorer QoL in pediatric cancer survivors (Cunningham et al., 2021). Anxiety is a complex multidimensional symptom that may occur in response to or separate from disease and treatment. Anxiety is important to recognize as it may lead to other symptoms that can cumulatively affect QoL (Zernikow et al., 2019).
When interpreting the significant negative relationships between symptom AEs and QoL, researchers should be aware of the conceptual overlap between select items of the PedsQLTM Cancer Module 3.0 and the Ped PRO-CTCAE®. For example, the PedsQLTM Cancer Module 3.0 includes items assessing anxiety, pain, nausea, and concentration impairment in the context of cancer treatment. These items have considerable conceptual overlap with select Ped PRO-CTCAE® items, including frequency AEs of anxiety, pain, nausea, and the severity AE of concentration impairment, and may explain the significant negative relationships in this sample.
Our study also showed the presence of symptom frequency AEs of nausea and anxiety predicted poorer QoL later in the treatment trajectory. These findings also contrast research which showed decreases in nausea and increases in reported QoL over time during treatment (Hooke & Linder, 2019; Momani et al., 2016). Few studies have investigated a time effect between symptoms and QoL among children with heterogeneous disease and clinical characteristics, making it difficult to compare results. The proportions of our sample who had active disease, received cancer-directed treatment in the previous ten days, and received mild intensity treatment remained relatively stable across time points throughout the study period. Possible explanations for this finding in our sample include some children in the sample may have received treatment with increasing intensity and associated nausea over time or some children may have experienced increased anxiety with disease evaluations after initial treatment for advanced cancer. Additionally, whether children were receiving sufficient supportive care with antiemetic medications over time is unknown. More research is needed to fully examine the effect of time on changes in symptoms and QoL over time.
Strengths and Limitations
The multisite, prospective, and longitudinal design is the primary strength of this study. Further, the inclusivity of children who were Spanish literate, had diverse cancer types, and represented different developmental age groups is a strength. However, our study was limited by a small sample size and ability to test between groups with shared characteristics, and account for covariates that could affect the relationship between symptoms and QoL, such as active disease, recent disease progression, cancer-directed treatment, and treatment intensity. Additional limitations include the absence of or lack of robust psychometric testing for the Ped PRO-CTCAE® and PedsQLTM Cancer Module 3.0 measures among Spanish-speaking children diagnosed with cancer and their caregivers, and conceptual overlap between symptom and QoL measures.
Implications for Clinical Practice
Following the Children's Oncology Group Nursing Discipline's State of the Science Symposium on Symptom Assessment During Childhood Cancer Treatment in 2019, four key recommendations were published to inform generation of new knowledge about pediatric cancer symptoms and translate evidence into practice to improve quality care (Withycombe et al., 2019). This study's results support the recommendation to obtain a self-report of symptoms from the child whenever possible (Withycombe et al., 2019). Further, our biweekly data collection and study design supports the recommendation that symptom assessment of core or high-priority symptoms should occur at regular intervals as part of care delivery (Withycombe et al., 2019). However, guidance is still absent on how often to assess symptoms in the context of precision medicine and variable treatment regimens. Currently, integration of patient-reported symptom monitoring is scarce in pediatric oncology clinical trials (Riedl et al., 2021) and routine patient care (Leahy et al., 2018). To address the ongoing symptom suffering and promote QoL and other health outcomes among children with cancer, defining a feasible and ideal approach to symptom assessment that drives delivery of guideline-concordant care is needed (Dupuis et al., 2019).
Implications for Future Research
The development of the Ped PRO-CTCAE® provides researchers and clinicians with a new measure to assess symptom AEs. Symptoms and symptom burden continue to result in clinically meaningful reductions in QoL for children with advanced cancer (Steineck et al., 2022). Further, the influence of demographic factors and social determinants of health on QoL is only emerging (Robson et al., 2021). Leaders in symptom science research have emphasized the consistent use of symptom measures across populations for more effective comparisons (Miaskowski et al., 2007). Further, inconsistency in the measurement of QoL hinders researchers’ abilities to compare across studies (Momani et al., 2016). Beyond measurement inconsistency, conceptual overlap in items between measures compromises researchers’ abilities to interpret the strength of relationships between symptoms and QoL. Thus, researchers should consider QoL measures that exclude specific symptom questions when investigating symptoms and QoL. Future research must strive for consistency in measurement to better understand the relationship between symptoms and QoL and the biopsychosocial factors that predict symptom suffering and poor QoL to identify new targets for intervention development.
Footnotes
Acknowledgments
The authors would like to acknowledge members of the Ped PRO-CTCAE® Research Team, including Drs. Pamela Hinds and Janice Withycombe, and Ms. Molly McFatrich for their permission to use the Ped PRO-CTCAE® instrument.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was supported by the University of Wisconsin American Cancer Society Institutional Research Grant IRG-15-213-51. The REDCap database used for this project was supported by the UW Institute for Clinical and Translational Research and the Clinical and Translational Science Award (CTSA) program, through the NIH National Center for Advancing Translational Sciences (NCATS), grant UL1TR002373.
