Abstract
Background
Medicines constitute the fulcrum of therapy across diseases, with patients needing to follow prescribed regimens (practice medication adherence (MA)) to derive optimal clinical benefits from medications. Poor MA is linked with increased mortality, morbidity and health costs, worse disease outcomes and reduced quality of life. Aotearoa New Zealand (NZ) operates a publicly funded health/medicines system, but available MA supports are unclear.
Methods
Using document analysis, this narrative review collated MA interventions and strategies recommended in several broad NZ health and medicine -specific policies to outline the NZ MA policy landscape for the first time. Eight policy documents underwent document analysis to identify proposed MA-related interventions and the responsible bodies.
Results
The identified interventions targeted health system, socioeconomic, disease, therapy and patient-specific factors. Incorporating MA promotion across the health system (in IT platforms, MA services and others) by all healthcare professionals, pharmacists especially, was central to the interventions, as was including patients in medicines therapy decisions via robust communication. The interventions aligned with the MA needs of NZ chronic disease patients, indicating that implementing them could improve patients’ MA. The lack of a national MA policy or responsible entity and other policy gaps, however, appeared to hinder implementation, ownership and accountability.
Conclusion
The wide-ranging MA promotion interventions recommended across NZ health and medicines policies need implementation which would require multipronged efforts of stakeholders across government and the pharmacy/health sectors.
Introduction
Medicines are an essential part of therapy across diseases, and “the process by which patients take their medication as prescribed” is described as medication adherence (MA). 1 MA is important for therapy effectiveness but, in developed countries, it has been reported that only half of chronic disease patients are adherent to long-term treatment regimens. 2 Poor MA is a public health challenge as it contributes to poorer health outcomes and quality of life in the population, as well as an increased burden on health services and (avoidable) economic costs to society. For instance, it factors in around 200,000 untimely deaths and a financial burden of €125 billion in Europe annually. 3
MA is not solely impacted by patients’ behaviours but by numerous other elements, with the WHO outlining five interconnected dimensions of MA encompassing socioeconomic-, health system/health team-, therapy-, condition- and patient- related factors. 2 Across Europe, multi-factorial policy-based solutions have been advanced based on expert consensus, with practical initiatives for implementation in different countries. 4 Likewise, public MA programmes such as Medicines Use Reviews are available in countries including Australia and Canada.5,6 MA challenges, nonetheless, remain a significant barrier to successful medication therapy.
Consistent with the international literature, poor MA has been reported in over a third of patients across several chronic conditions in Aotearoa New Zealand (NZ).7–13 Although research on the costs of poor MA in NZ is scarce, the public burden is thought to be substantial. 14 Given the significant financial resources invested in pharmaceuticals in NZ, the reduced health system burden associated with better adherence to medications, and the importance of medicines access to facilitate MA, suitable adherence policies, articulating MA support strategies, are important in the healthcare system.
Policies for medication access, usage and adherence are important to determine what interventions are resourced (by funding and workforce engagement), but policy omissions of necessary services or strategies would mean that these are not prioritized, and may reflect a lost opportunity.
For context, Aotearoa New Zealand has multiple government entities that have oversight on the use and funding of medicines. These include: Manatū Hauora-Ministry of Health (MoH), which reports directly to the Minister of Health on all areas of health including medications. Medsafe New Zealand (New Zealand Medicines and Medical Devices Safety Authority) is the regulator of medicines. PHARMAC (Pharmaceutical Management Agency) is the funding agency that determines which medications are financed on the public health system schedule. Provision of funding for medication supply (i.e., community pharmacy contracts) are held within Te Whatu Ora Health NZ, the national health agency. These government agencies all play a role in medication access and adherence. Further, professional bodies such as the Pharmaceutical Society of New Zealand (PSNZ) and the former New Zealand Medical Association (NZMA) alongside Medical Colleges for different specialisations including the 17 colleges represented by the Council of Medical Colleges (CMC), 15 and several other organisations, have interest and expertise in medications (alongside healthcare delivery) and these organizations support their professions to deliver services that meet patient medicines needs.
While many comparable health systems, such as in England, 16 have Chief Pharmacists or Chief Pharmaceutical Officers who hold oversight for “best use of medicines” including appropriateness, access and adherence, Aotearoa New Zealand (NZ) does not. Therefore, in Aotearoa New Zealand, multiple national agencies and professional associations have independently created policy documents touching on medicines access/appropriateness/adherence.
Aotearoa New Zealand has a predominantly public health system, with small out of pocket costs compared to other similar health systems. All hospital and specialist treatments are funded by the government and the government contributes significantly to general practices (GPs); however, most practices have an additional co-payment requirement for consultations/prescription issuance/medicines administration. This amount is fixed by individual GPs; so, it can vary widely and constitute a substantial cost. Effective July 1, 2024, for all medications listed on the publicly-funded schedule, a flat patient co-payment of $5 NZD is standard per prescription; additionally, once a patient/family has a total of 20 items dispensed in a year, the $5 co-payment is waived as they get a Prescription Subsidy card. 17 This co-payment is also waived for patients <14 and ≥65 years old and some special groups. This represents a policy change due to a change in government as from July 1, 2023 until June 30, 2024 this co-payment was eliminated and publicly covered for all patients, with few exceptions. Notwithstanding, private health insurance and hospitals are available. It is noteworthy that inequities in obtaining healthcare and medications continue; these arise throughout the process of patients reaching a prescriber, obtaining a prescription, reaching a pharmacy, collecting the medications and then taking them as prescribed. 18 These challenges require resolution.
Given its flexibility in data gathering and interpretation, allowing for expert recommendation, and, thus, its utility for knowledge synthesis especially in developing fields with limited data/literature, 19 a narrative review was adopted for this study. The purpose of this narrative review policy analysis is to collate the disparate policy documents that pertain to medications access and adherence within Aotearoa NZ, and to analyse the recommendations for MA specific interventions therein. These recommendations are then mapped across the WHO domains for MA, and priority actions and policy gaps identified. This analysis could inform future policy development, implementation strategies and priorities, and be used as the basis for an overarching consensus on MA policy for Aotearoa NZ.
Methods
This study assessed references to action points/strategies/interventions regarding MA and/or its definition as “the process by which patients take their medication as prescribed”,
1
in NZ health/medicines policy documents. The specific objectives were to: (1) identify NZ policy documents addressing MA; (2) characterize key features of the policies including: (i) the policy holders (authors); (ii) what action points the policies articulate; (a) map these to WHO identified MA barriers via an evidence and gap map; (iii) how the action points are/were to be implemented; (3) underline what policy gaps exist.
Literature Search
Policy Documents Analysed.

Document Analysis
This study employed document analysis (DA) which is “a systematic procedure for reviewing or evaluating documents” entailing a quick read through and then an in-depth perusal of the document(s) followed by interpretation of the data. 27 A unique advantage of DA is that data in documents are already collated/available and are thus not influenced by the researcher; this allows for the sidestepping of social desirability bias in data collection. 27 Also sidestepped is recall bias as documents preserve historical information. Hence, as opposed to data generated from interviews or surveys (e.g., of policy stakeholders) which might be impacted by personal opinions, evaluation of policy documents gives an insight into the formal viewpoints of the publishing entity, for example, government departments.
The DA was conducted using the READ approach: “(1) ready your materials, (2) extract data, (3) analyse data and (4) distil your findings”. 28 First, the relevant documents were identified, then the strategies outlined for helping patients take their medications as prescribed (i.e., MA) were curated. Next, the interventions were categorized based on the WHO MA dimensions. 2 Finally, the data was analysed vis-à-vis the self-reported MA needs of NZ chronic disease patients in the literature. The first step was conducted by Amiesimaka O, who conducted the next two steps through an iterative process involving the review of Braund R. All four authors contributed variously to the drafting, composition and editing of this article. All steps were manually conducted. To conduct a narrative review by way of document analysis, the data was analysed from a “direct realist” epistemological framework. The documentary data were therefore considered at “face value”, taking the perspective of the data wholly reflecting a specific reality. 29 This paper would be of value to researchers, academics, clinicians and policy-makers in MA policy.
Reflexivity
Acknowledging the impact of researcher subjectivity in qualitative analysis, exploring the researchers’ “construction of meanings” through reflexivity is essential.29,30
Amiesimaka O holds a PhD with a public health (policy) focus, alongside prior qualifications and experience in biomedical sciences, global/public health and academia. Braund R is a professor of pharmacy with decades long experience in pharmacovigilance and public health, and top-level engagement with NZ professional bodies in pharmacy alongside advisory roles to the government on pharmacy. Both their research covers MA policy, calculations, data-based monitoring and general advancement. Hence, they are strong proponents of evidence-based pharmacy/medicine and the importance of MA for health and wellbeing. These shaped their data interpretation as they took the perspective that patients should be robustly supported in attaining the best possible levels of MA. They also both brough a wealth of MA lived experience from distinct health settings, as Amiesimaka O is from Sub-Saharan Africa and Braund R is from Aotearoa New Zealand.
Ethical Approval
Ethical approval for this type of study is not required by our university.
Results
Analysis
Across all the documents (Table 1), the concept of patients taking their medications as prescribed was referred to directly, described as medicines/medication adherence (MA) or contained within references to medicines access, medicines optimization, responsible use of medicines and/or medicines management services (MMS). There was a consensus that patients following their prescription is important for achieving the best health outcomes and that multiple factors, “financial, organisational and social or cultural” (Policy E), 18 impact on patients’ MA. All the policies supported interventions in assisting patients to practice good MA.
For concision, this study focussed on proposed action points that were closely targeted at aiding patients’ MA from the point of prescription onwards. The strategies advanced were analysed in line with the five interconnected WHO domains of MA. 2 As the domains are interlinked, the proposed solutions are often applicable across the dimensions.
Socio-Economic Factors
Several socio-economic factors can impact MA including: race/ethnicity, racism/ethnic bias, medication unaffordability, lack of nearby health/pharmacy facilities, poverty, transportation unaffordability, certain cultural/general beliefs, poor education, age and more besides. 2 The policy documents propose strategies which address these factors.
Inequities in health outcomes/medicines access experienced particularly by Māori were acknowledged in the documents (Policies E, D, C, H).18,22,23,26 Consequently, integrating the principles of Te Tiriti o Waitangi (The Treaty of Waitangi – the founding treaty of NZ between the British Crown and Māori signed in 1840), 31 across the health system was recommended (Policies A, H, E).18,20,26 Multiple approaches were advanced in line with this and to attain equity in health outcomes for Māori, for example, “Initial priority will be given to our Te Tiriti partner, Māori, who are well evidenced to experience health inequities” (Policy E). 18 Additionally, the formation of the Māori Health Authority (MHA), with system-wide responsibility for impacting Māori health outcomes, was endorsed (Policies A, H).20,26
To aid timeous physical access to prescriptions/community pharmacies/testing, service combination and flexibility in opening hours was recommended (Policies A, E).18,20 “Some or all services being open earlier or later in the day, providing clinics at the weekend, or participating in outreach. It could mean aligning services’ opening hours and workforce in similar locations and times, so a person or whānau can conveniently meet all their needs at the same time” (Policy A). 20 Also, PHARMAC should consider ways schedule guidelines on “supply orders for rural areas”, and “alternative mechanisms of medicine distribution and supply” (Policy E) 18 can be employed in improving physical access.
Strategies were also proposed for reducing the out-of-pocket costs of accessing medications. These costs include general practice (GP) fees for consultations, obtaining (refill) prescriptions and medication administration, besides transportation expenses and medicine co-payments. To help with affordability, existing subsidy schemes were to be re-assessed to limit the impact of access fees (Policy B). 21 However, the complete removal of co-payments was not endorsed in Policy A as, given existing subsidies, it “would disproportionately benefit higher-income households”. 20 Instead, Policy A recommends investments in “prevention, population health management, more outreach services and better care coordination”. Outreach services involve providing health services (e.g., medicines optimization), normally done in health centres, elsewhere including in patients’ homes, marae (Māori meeting place), and religious centres. Also proposed was funding the administration of more therapeutics nearer patients, for example, at home/GPs and “funding medicines with shorter infusion times, or moving infusions to oral therapy” (Policy E). 18 Moreover, relevant stakeholders should collaborate on ways to limit costs of medicines access via various pathways.
Health System/Healthcare Team-Related Factors
The nature of the health system considerably impacts on MA with barriers including: inadequately trained/skilled workforce in MA; insufficient resources to monitor and support (behavioural changes for) patients’ MA; limitations in managing long-term diseases; and low awareness/understanding of MA and its associated factors. 2 The policies advance interventions across the health system which could address these.
Strategies for leveraging digital tools in managing the range of services needed to facilitate patients’ MA were broadly recommended. Central to this was developing an integrated (nationwide) platform particularly for electronic prescription and sharing electronic health records (EHRs), including dispensing/prescription, allergies, adverse events, and diagnosis data, to facilitate comprehensive medicines management (Policies A, E, B, C).18,20–22 This platform would be available to both patients and healthcare professionals (HCPs) across the whole health system. An “Information Use Framework and Health Information Governance” was to be developed “to implement standard operating procedures and rules for access, and… audit processes for information transfer and management” (Policy B). 21 Several platforms including the National Health Investment Platform (nHIP), My List of Medicines, NZ ePrescription Service (NZePS), eMedicine systems for hospitals and “a shared care platform to support …pharmacy medicines adherence plans” were also to be developed (Policies A, B).20,21 The NZePS would also provide a platform for indicating patients’ eligibility for the prescription subsidy which could help ensure those qualified actually receive the subsidy. Digital tools would also facilitate communication between patients and HCPs (e.g., via telehealth) as well as prescribers and pharmacists, and help overcome challenges of distance and transport to service centres (Policy B). 21
The National Pharmacist Services Framework details medicines management services (MMS) comprising MA and other medicines optimization services which pharmacists could provide; so (primary) health sector stakeholders/funders can choose which to implement (Policy D). 23 Patients’ understanding their prescribed medications and using them safely and efficaciously are central to MMS. Thus, MMS “involve a discussion with the patient about their medicines and delivering … activities to improve utilisation and manage … harm from their use or misuse” (Policy D). 23 The services are delineated into those on medicines adherence namely the Long-Term Conditions (LTC) service and Medicines Use Review (MUR), as well as on medicines optimization, namely Medicines Therapy Assessment (MTA), Comprehensive Medicines Management (CMM) besides the Community Pharmacy Anticoagulation Management Service (CPAMS) (Policy D). 23 Different tiers of support for patients and their whānau (family) are available via these services depending on need and each service has specific eligibility criteria. The LTC and MUR allow the pharmacist reconcile and synchronize the patients’ medicines, issue MA advice and aids, for example, blister packs, provide educational interventions, as well as develop a medicines management plan to share with patients and HCPs, besides others (Policies D, G).23,25 In line with increasing clinical complexity, the level of expertise/certification needed by pharmacists to deliver these services increases from the LTC to MUR, MTA and CMM, with the CPAMS also needing formal training of providers (Policy D). 23
It was recommended that these services be provided “across primary, secondary and aged care settings” (Policy B), 21 and in hospitals as part of Hospital Clinical Pharmacy Services (Policy D) 23 in line with an integrated health system approach.
Overall, integrated care provision involving all HCPs across the health system was advocated in the policies. HCPs, especially in primary care and pharmacies, were encouraged to communicate better, work collaboratively, co-locate service provision, use shared digital tools and seize innovative opportunities to support patients’ MA (Policies F, B, C).21,22,24 Although “all of the health workforce is responsible for medicines optimisation, … pharmacists play a key role as medicines experts” (Policy A). 20 Pharmacists were to lead robust MMS/personalized MA services, including monitoring medication utilization, in convenient locations for patients and their whānau, and making recommendations to other HCPs, for example, prescribers (Policies A, C).20,22 Moreover, Policy A proposed that MA services be part of a range of integrated health services (e.g., GP, community pharmacy services etc.) to be made mandatorily available across all regions so patients access them as a “single service” (Policy A). 20 This would achieve “a seamless continuum of care …that involves diagnosis, prescribing and dispensing of medicines, medicine therapy optimisation, monitoring and patient adherence support…” (Policy F). 24 Such MA services, included in medicine optimization, were to be targeted to priority groups including persons with chronic conditions and polypharmacy (Policies A, C).20,22 Also advocated was training pharmacists in specialist skills, and all HCPs in relevant MA skills; alongside making needed policy changes to advance MA (Policies A, C).20,22
Further, the Manatū Hauora-Ministry of Health and pharmacy stakeholders were to advance and manage the framework for prescribing by pharmacists (Policy B). 21 The policies further recognize that prescriptions are issued by other non-medical doctor HCPs including midwives, registered nurse prescribers, dietitians and optometrists (Policy C). 22 The PHARMAC special authority rules were spotlighted for updating (Policies E, C),18,22 to permit pharmacist prescribers “to apply for special authority for some medicines” (Policy E). 18 Although not making primary diagnoses, pharmacy prescribers may issue initial prescriptions and “modify therapy (including discontinuation or maintenance of therapy) originally initiated by another prescriber” (Policy D). 23 Expanding the prescriber workforce this way could increase the number of prescribers available to patients, thus increasing access to medicines and MMS in furtherance of patients’ MA. It would also allow pharmacists use their expertise to play a more integral role in the patients’ overall medicines management including their MA. However, as at 2016, there were only 15 pharmacist prescribers in NZ (Policy C) 22 and this number had risen to only 37 by 2021. 32
Structurally, funding of MA services was also highlighted as important alongside adopting models of care that advanced patients’ MA across the regions (Policy C). 22 The importance of continued MA research was also underscored, with only PHARMAC mentioned as funding MA studies (Policies H, A).20,26 Also, recommended was the reconsideration of which entity was best placed to oversee “responsible use of medicines” (Policy H), 26 highlighting the difficulty in multiple agencies working independently. More so, crafting a new medicines strategy with a focus on medicines optimization and equity in line with Te Tiriti O Waitangi principles was endorsed (Policy H). 26
Therapy-Related Factors
Medication adherence can be impacted by elements of the treatment/therapy employed. Some of these include: length of therapy, presence of side-effects, variations in the therapy, how complicated the medication regimen is, the nature of support available to address adverse-effects, and more besides. 2 The policies propose some action points relating to these factors.
Prescribers need to be up to date with the funded-medicines available to inform the most appropriate therapy to adopt considering the disease, medication side effects, alternative medicines, drug interaction profiles for patients with polypharmacy, among others. Such information can be provided by PHARMAC (via its Implemention Team), MedSafe (MoH), professional regulatory bodies, and DHBs (now Te Whatu Ora) (Policy E). 18 Although no single organization can fulfil all medicines information responsibilities, it was proposed that PHARMAC continue making such information available to HCPs to achieve better medicines access equity (Policy E). 18
The nature of the medication regimen is influenced by the PHARMAC Schedule rules which determine the period within which the PHARMAC subsidy applies – typically within 3 months from the date of prescription. Moreover, some pharmaceuticals may only be dispensed in monthly tranches. These might create access issues for managing stable long-term diseases by requiring repeated patient visits to obtain refill prescriptions and dispensings at GPs/specialists and pharmacies (Policy E). 18 It was proposed that such schedule rules be reappraised in furtherance of access equity (Policy E). 18 PHARMAC’s Schedule rules also permit pharmacists, including when providing MMS services for example, LTC and MUR, to optimize the frequency of patients’ dispensing collection visits in line with their MA support needs (Policies G, H).25,26 Additionally, under the MTA, pharmacists could help address the challenges of patients facing adverse effects and/or with wories about medication effectiveness (Policy H). 26 An appropriate care plan could be drafted, collaboratively with the prescriber, to better achieve the therapy goals. Further, patients with complex clinical needs could receive MA support (e.g., medicines monitoring, regimen review etc.) from a pharmacist embedded within the healthcare team via the CMM service (Policy H). 26
Condition-Related Factors
The nature of the disease can influence MA based on factors such as: disease progression/severity, related disability, co-morbidities, symptom severity, and others. These could all affect patients’ views of the therapy and the need for MA. 2 The policies outlay some strategies to handle such issues.
The policies advocate for greater integration across the health system, with all HCPs working together via shared platforms throughout the patients’ healthcare journey (Policies B, C, A, D, F).20–24 As all HCPs are tasked with medicines optimization (Policy A), 20 both prescribers and pharmacists should ensure that prescribed medications are appropriate to the disease conditions. The MMS provide avenues for patients to raise concerns about the therapy and medicines effectiveness, and for pharmacists to address such concerns collaboratively with prescribers, for example via the MTA (Policy D). 23
Patient-Related Factors
Patient’s motivation, expectations, beliefs and knowledge about the disease, treatment and MA, all influence their adherence behaviours. 2 The policies propose action points related to these factors.
Effective communication within a trusting, respectful and culturally competent prescriber/pharmacist-patient relationship is essential to MA (Policy E). 18 Centering Te Tiriti o Waitangi principles would facilitate this (Policies A, H, E).18,20,26 Such communication is necessary for noting what beliefs/expectations/motivations patients have, addressing any misperceptions and equipping patients with information/knowledge/skills to self-manage their medicines and practice good MA. Policy B recommended that all HCPs “demonstrate improved communication with consumers [on how medicines work, MA, self-monitoring effectiveness/side effects] at every point of care”. It also advocates that all health entities empower patients and their whānau for medicines self-management through culturally-suitable health literacy/education interventions. Consequently, socio-linguistic and cultural competence should be an intrinsic part of all HCP training for example, within a ‘single framework for prescribers’ to be outlined by the MoH (Policy B). 21 Multiple avenues for communication were also proposed via online/written/multimedia applications besides in-person meetings. All these would serve to treat patients/whānau as ‘active partners’ in medicine-related decision making, increase accessibility for people living with disabilities, tackle cultural bias, and enhance patients’ understanding of the prescribed regimen and avenues for accessing needed information, while being “responsive to [their] needs, values, beliefs and knowledge” (Policy B). 21
As all five dimensions of MA are interlinked, individual strategies would impact one another and fit in more than one dimension. Consequently, multiple synergistic system-wide interventions would be needed for maximum impact on patients’ MA.
Relation to Chronic Disease Patients’ MA Needs
As life-long medication use is central to managing chronic diseases, MA is important for achieving best health outcomes. Chronic diseases hamper the performance of activities of daily living and often lead to disability and increased mortality. Poor MA has been reported for 46%, 22% and 37% of people with chronic diseases namely inflammatory bowel disease (IBD), gout and diabetes, respectively.8,33,34 Moreover, chronic diseases, including cardiovascular diseases and cancers, are the major causes of death in NZ. 35
In NZ, people over 65-years of age generally, besides people with diabetes and others with IBD,7,36–38 have all reported that adverse effects as well as regimen characteristics, for example, large pill sizes, high numbers of medicines/dose frequency, and prescription unaffordability hindered their MA. Obtaining refill prescriptions, mainly from GPs, every 3 months was considered a particularly needless obstacle to MA for people with IBD as patients are sometimes on unchanging regimens for extended periods. 38 Obtaining refills entails both the time and financial cost of GP appointments and collecting/co-paying for dispensed medicines at pharmacies. The patients highlighted the unavailability of GP appointments as a significant bottleneck and would prefer for pharmacies to dispense refills for longer periods and/or until otherwise informed by prescribers. They appreciated the prescription subsidy scheme but also wanted all out-of-pocket costs (for GP prescriptions/consultations, medicine co-pays) eliminated. 38
Family and friends, besides HCPs, were identified as major supporters of MA in patients with IBD, and diabetes, although some whānau were unsupportive.37,38 Proactivity was also framed as central to MA self-management and a trusting/respectful patient-HCP relationship was important for MA. 38 This trust, alongside cultural competence, aided MA for bipolar disorder patients of Chinese background and other diabetes patients in NZ, and differences in perceptions of medicines-taking between people of different ethnicities have also been reported in NZ.37,39,40
Evidence and Gap Map on the Alignment of Medication Adherence (MA) Interventions Recommended in NZ policies with Factors Impacting Chronic Disease Patients’ MA.
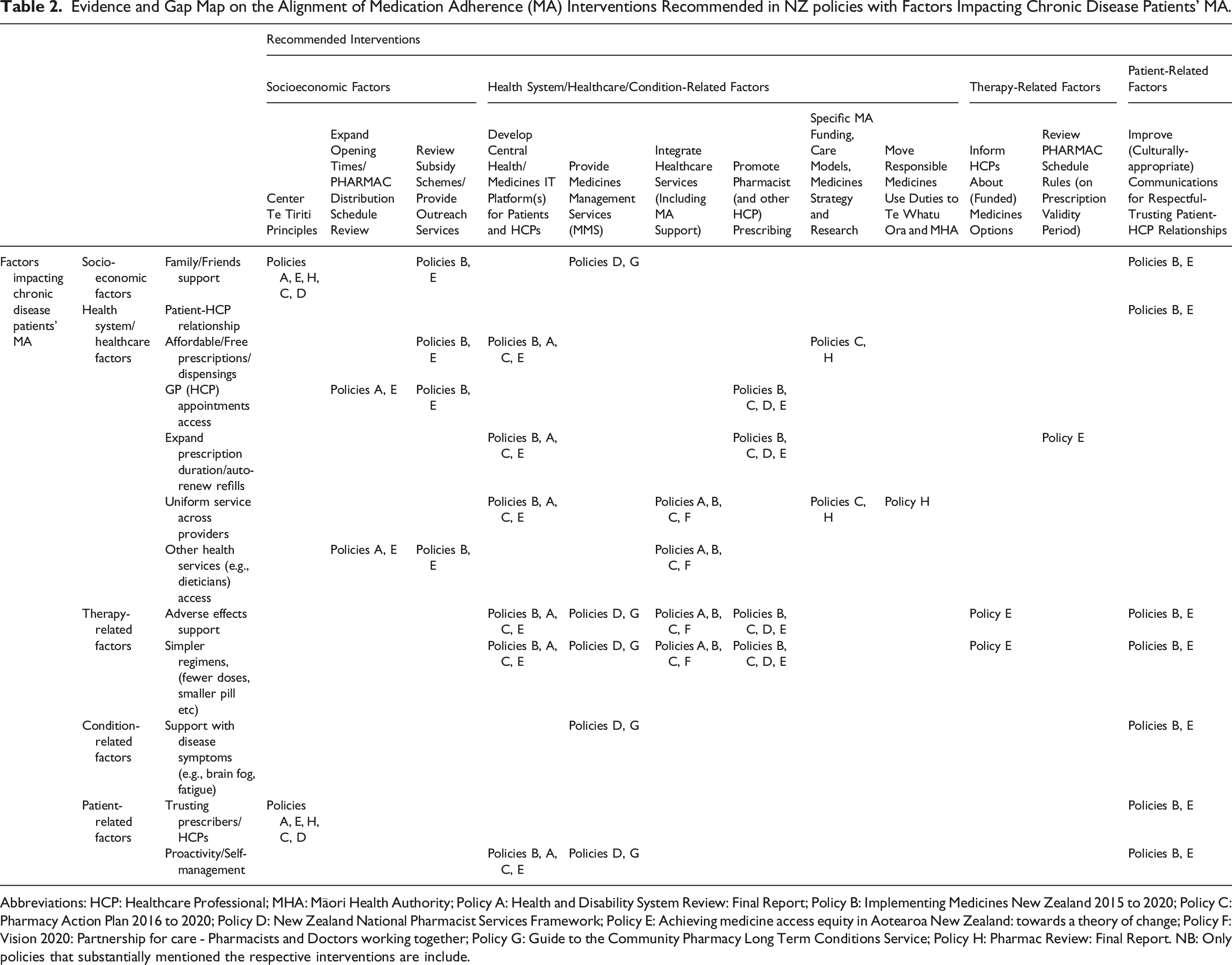
Abbreviations: HCP: Healthcare Professional; MHA: Māori Health Authority; Policy A: Health and Disability System Review: Final Report; Policy B: Implementing Medicines New Zealand 2015 to 2020; Policy C: Pharmacy Action Plan 2016 to 2020; Policy D: New Zealand National Pharmacist Services Framework; Policy E: Achieving medicine access equity in Aotearoa New Zealand: towards a theory of change; Policy F: Vision 2020: Partnership for care - Pharmacists and Doctors working together; Policy G: Guide to the Community Pharmacy Long Term Conditions Service; Policy H: Pharmac Review: Final Report. NB: Only policies that substantially mentioned the respective interventions are include.
Discussion
This analysis found that a broad range of strategies, interventions and action points to support medication adherence (MA) in NZ have been proposed in policy documents. The policies were authored by the Manatū Hauora-Ministry of Health (MoH) or its agencies, for example, PHARMAC, and professional pharmacy and medical organizations. Suggested interventions span the five WHO dimensions of MA 2 and encompass socioeconomic factors, for example, improving health facility access via expanded opening times/outreach services; health system elements, for example, providing medicines management services (MMS); therapy factors, for example, reviewing PHARMAC Schedule rules on prescription validity; condition factors, for example, providing multi-disciplinary MA support; and patient factors, for example, fostering trusting patient-HCP relationships. These recommendations address many of the self-reported MA needs of chronic disease patients in NZ, with individual interventions having the potential to help resolve multiple challenges. This indicates that investments in the action points could prove valuable and useful in attaining the WHO recommendation for the universal accessibility of essential medicines to all patients. 42 However, there is no nationally consistent MA support provided in Aotearoa NZ, indicating that while the consensus is that MA is important and strategies have been proposed, no entity has been specifically mandated to actually implement the strategies recommended. This challenge persists as the New Zealand Health Strategy 2023 makes limited reference to medicines as it only mentions “a stronger role for pharmacists and nurses in providing health advice and prescribing medication”, 43 in alignment with the documents analysed in this review. Unfortunately, it does not mention MA, an MA policy or a responsible entity. This decentralisation also represents a fragmentation of resources (e.g., funding) specifically for MA, as the different health bodies receive funding for the sum of their functions, with little clarity on which funds are earmarked for MA advancement. Reasons for this lack of foregrounding of MA despite consensus about its importance requires research attention but may be due to a lack of knowledge of decision-makers about the intricacies of MA – from equity to health systems - demanding specific resourcing.
The NZ interventions align with consensus MA improvement proposals of cross-European policy stakeholders. 4 These European proposals targeted patients, healthcare professionals (HCPs), patient-HCP interactions, medicine regimens, health systems and governments. Also, the NZ interventions are congruent with the Organization for Economic Co-operation and Development (OECD) system-level enablers of MA “acknowledge, inform, incentivise, and steer and support”. 3 Stakeholders must acknowledge the importance of MA; information/data on patients’ MA behaviours and MA-associated indicators should be routinely collected (the OECD could facilitate this); incentives should be provided for MA prioritization; and system-wide support (IT resources, education/training etc.) should be given to patients and HCPs to aid MA. Aotearoa NZ is an OECD member state. 44
The NZ policies referenced the concept of patients following prescriptions, that is MA, using various terms including medicines adherence/optimization/management services, and these were often combined with other issues, for example, meeting health needs. This pattern of limited MA ‘visibility’ was also found in European policies 45 and could limit the prioritization/implementation of MA services. Further challenges to implementing MA proposals in Europe included deciding which MA proposals to adopt, limited funding/resources/staffing, difficulties in determining the effectiveness of MA initiatives, and poor synergy of MA interventions across the health system. 45 The implementation also focussed on patient-level changes and less on government/systemic ones. Whether these apply to the NZ context needs to be explored in future studies alongside the selection of the NZ recommendations that were implemented and funded, the duration of implementation, and the impact on patients’ MA. Again, the lack of a national MA policy and an accountable entity for MA promotion in NZ constitute a major hurdle to the provision of MA supports as well as conducting needed research. Exploring these would also be beneficial for MA policy development across health systems globally, with the chief point being that responsibility/accountability for a function (MA) must be clearly situated as shared responsibility often connotes a lack of ownership and accountability; hence, the function is prone to being inadequately executed. This may be especially instructive in countries with decentralised governance systems like the USA, operating a three-tiered federal system (federal, state, local) each with (some) responsibility for health and medicines.
Theoretical frameworks facilitate the articulation of concepts and the wider translation of research such as this. By way of illustration, the Consolidated Framework for Implementation Research (CFIR) allows dominant domains from an intervention perspective to be identified. 46 Although the present study was not premised on such a theoretical framework but rather the alignment of the policies’ action points with the WHO dimensions of MA, 2 the CFIR is instructive. The findings herein align, in particular, with the CFIR’s ‘Outer Setting’ domain’s ‘Policies and Laws’ construct and the ‘Inner Setting’ domain’s ‘Culture: Recipient-Centredness’ construct. The former encompasses the policy and regulatory landscape, whilst the latter evaluates how patients’ needs, alongside barriers/facilitators for meeting same, are reflected, valued and prioritised.
Policy Gaps
Notwithstanding the breadth of the proposed interventions, some policy gaps exist which should be considered.
NZ chronic disease patients found MA when travelling to be very challenging, due to issues with (cold-)storage and advance stock-ups of medications, timings of refill appointments and others, but the policies did not speak specifically to this issue. Chronic disease patients found the cost of GP consultations and medicines to be burdensome.37,38 10.2% and 3.1% of New Zealanders reported not seeing a GP or not obtaining prescribed medications, respectively, in 2020/2021, due to such fees. 47 Although some patients desired that those fees be eliminated, 38 Policy A opposed the elimination of publicly-funded medicines co-payments as the impact on access might be limited, but advocated other strategies; for example, more outreach services to improve medication affordability. 20 However, eliminating the NZ$5 medicines co-payment especially has been linked with fewer hospitalizations for patients resident in regions of high socioeconomic deprivation. 48 In response to advocacy efforts, the NZ government eliminated this co-payment from July 1, 2023, with few exceptions, but it was reinstated from July 1, 2024 after a change in government. 17 Attention should still be paid to other financial factors impacting patients’ MA including the GP costs. A new medicines policy which considers patient contribution to their care, balanced with access and equity, would provide support for the consideration and development of the most appropriate payment strategy.
Further, although medicines management services (MMS) were proposed, referral by patients or HCPs is needed to access the services. Thus, patients need to be aware of the services and their eligibility, while HCPs (pharmacists, GPs, nurses etc.) need to monitor patients’ MA to identify those eligible. However, despite the policies recommending the development of centralized digital platforms including prescription and dispensing records, they do not mention real-time or periodic evaluations of MA levels using these data. Such evaluations would require centralised pooling of pharmacy data in real-time, which could highlight patients with low MA levels needing intervention. 49 It is however noteworthy that the MMS must be aimed not just at identifying individual patients’ MA needs, but must proffer solutions through the use of needed MA improvement approaches to be effective at bettering the patients’ MA.
Recognising the current lack of unified digital infrastructure in the health and medicines system, the policies also recommended the development of system-wide IT platforms for health/medicines data, but different approaches and platforms were suggested. These should be synergized to avoid duplicated efforts, further fragmentation and wasted resources. 49
The policies did not propose a specific entity to ensure that MA is prioritized and resourced, nor one for monitoring, coordinating, and evaluating the effectiveness of MA initiatives across the health system. NZ only has a medicines regulator, MedSafe, and a funder, PHARMAC. A lack of coordination has been underlined as a challenge to implementing MA policy proposals in Europe. 45 This highlights the need for a specific coordinating entity with responsibility for developing and maintaining a new medicines policy and ownership of/accountability for MA support provision.
While promoting MA was couched as the responsibility of all HCPs, the vast majority of interventions were expected to be delivered by pharmacists. This reveals perceptions of pharmacists as medicines experts and that support for patients literacy and practical solutions are often believed to be best delivered by pharmacists. Care should be taken to ensure that the roles of other HCPs in MA are not overlooked in implementation stages particularly as medication adherence does not occur exclusively in the realm of pharmacy as the different WHO dimensions (e.g., socio-economic factors) bear out. Further, pharmacist prescribing was endorsed to leverage pharmacists’ expertise and reduce the GP appointment bottleneck by expanding the prescriber options. Besides NZ, pharmacist prescribers also exist in the UK, USA and Canada. 50 As NZ GPs were resistant to the idea of pharmacists making diagnoses, 50 it should be highlighted that Policy D proposes that pharmacists not be the primary diagnosticians but make ongoing/refill prescriptions. 23 Nevertheless, the financial costs of expanding (pharmacist) prescriber roles to the health system need to be evaluated vis-à-vis the benefits to the patients’ MA and health outcomes.
Overall, there was no MA policy outlaying multi-level system-wide MA interventions and a strategy for implementing them. Such a document would need to be drafted, after comprehensive stakeholder engagement, if MA support is to be prioritized in the NZ health system. Our paper constitutes a first step in exploring the policy landscape and pooling pre-existing proposals which could guide future research/actions. The aforementioned gaps/misalignments should also be considered in developing an MA policy and implementing the programmes.
This study only included select publicly available policies; hence, any internal documents of relevant organizations would have been excluded. For concision, this study’s analysis was limited to only policy action points which impact MA from the point of prescription onwards. Thus, higher-level strategies, for example, on organizational structure/governance, notwithstanding their impact on MA, were excluded. These should be the focus of future research as part of a further in-depth policy analysis adopting an appropriate theoretical or conceptual lens such as implementation science, systems governance or equity frameworks. Moreover, as underlined in the ‘Reflexivity’ section, the authors’ perspectives shaped the data interpretation and conclusions in ways which might differ from other authors. Further, although a comprehensive breadth of policies were included in this study, sufficient for providing an foundation of the policy landscape, the possibility remains that some relevant policies might have been inadvertently omitted as this is a non-systematic narrative review. 51 This limitation applies also to the NZ patient MA needs considered.
Conclusion
Wide-ranging interventions to promote medication adherence in NZ have been recommended in several broad health and medicines -specific policy documents. This exploratory study takes the first step in collating the proposed strategies to advise future actions. Policy gaps are also spotlighted for future resolution. Evidence was scarce about whether the recommendations have been implemented and actioning them may have been hindered by the lack of a designated responsible office(r) with ownership of medication adherence support and development of a national MA policy.
Footnotes
Ethical Considerations
Ethical approval for this type of study is not required by our university.
Author Contributions
Amiesimaka: Conceptualization; Data curation; Formal analysis; Investigation; Methodology; Project administration; Resources; Software; Roles/Writing - original draft; and Writing - review & editing. Aluzaite: Conceptualization; Methodology; Supervision; and Writing - review & editing. Schultz: Conceptualization; Methodology; Supervision; and Writing - review & editing. Braund: Conceptualization; Data curation; Investigation; Methodology; Project administration; Resources; Supervision; Roles/Writing - original draft; and Writing - review & editing.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Dr. Obreniokibo I. Amiesimaka received a PhD scholarship from the Department of Medicine, Dunedin School of Medicine, University of Otago.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
All policy documents cited in this manuscript are publicly available.
