Abstract
Background:
Alloplastic cranioplasty is a common procedure in cranial reconstruction, yet factors related to success and failure have been incompletely characterized.
Methods:
The authors conducted a large-scale retrospective review of patients who underwent alloplastic cranioplasty between 2014 and 2021 at a single institution. Information was collected regarding demographics, wound healing comorbidities, indications for surgery, and outcomes. A multivariable regression analysis was used to determine variables associated with operative complications, implant explantation, and contour defects.
Results:
One hundred and one patients underwent alloplastic cranioplasty. Fifty-seven percent of patients had at least 1 wound healing comorbidity. The most frequent indications for surgery were trauma (44%), cerebrovascular accident (18%), and cancer (18%). The operative complication rate was 24%. Thirty-six percent of patients had a postoperative contour deformity, and 16% underwent additional surgeries related to cosmesis. At a median follow-up of 1.5 years, 99% of patients maintained either a primary (84%) or secondarily placed (15%) implant. On multivariable analysis, level 4 ASA classification (P = .05) and heavy alcohol use (P = .01) were significantly associated with complications. Cerebrovascular accident (CVA) was associated with contour defects (P = .03). The only factor associated with explantation was heavy alcohol use (P = .05).
Conclusion:
This study reviews predictive factors for complications, implant explantation, and poor contour outcomes after alloplastic cranioplasty in a large cohort. Results indicate that alloplastic cranioplasty can have a high success rate with reasonable aesthetic outcomes.
Keywords
Introduction
Cranioplasty is an operation which restores cranial form and the soft tissue envelope in a craniectomy defect. Goals of surgery are to restore protection to the cerebrum, restore normal pressure and cerebrospinal fluid (CSF) dynamics, and improve soft tissue anatomical structure and appearance. 1 Common indications for cranioplasty in large scale craniectomy defects include: traumatic injury, intracranial hemorrhage, cancer, and bone resorption. Alloplastic cranioplasty was developed as an alternative in cases of insufficient patient craniectomy bone segment for autologous reconstruction, such as trauma, resorption, or oncologic involvement. In recent years, titanium mesh, hydroxyapatite (HA), polyetheretherketone (PEEK), porous polyethylene (Medpor), and polymethylmethacrylate (PMMA) have been become widely used with no clear consensus as to the best material. 2 Benefits of alloplastic materials include custom shaping using computer aided design/manufacturing (CAD/CAM), non-resorbability, bone-like hardness, and decreased operative time compared to autologous methods.1,3
However, complications after cranioplasty remain high, with overall complication rates cited to be as high as 40%.3,4 These often include infection leading to implant explantation, hematoma, seroma, CSF leak, and wound breakdown. While alloplastic materials can provide sufficient brain protection, many patients can experience temporal hollowing after reconstruction. Soft tissue loss with temporalis muscle malposition and or atrophy are perceived putative factors in this temporal deformity.
With the aforementioned challenges in alloplastic cranioplasty, much work remains to optimize form and function in the reconstruction of large-scale cranial defects. Prior studies have highlighted the risk of complications by material type and compared the risk of complications between alloplastic and autogenous cranioplasty.5 -8 However, few robust studies exist specifically for alloplastic cranioplasty, and literature is scant regarding aesthetic outcomes. 9 Herein we present a retrospective review of 101 cases of alloplastic cranioplasty and investigate predictive factors for complications, implant explantation, and postoperative aesthetics.
Methods
Patient Selection
We performed an institutional review board-approved (#00000311) retrospective cohort study between 2014 and 2021 at a single institution. Eligible patients had a cranial defect larger than 5 cm in diameter. All patients underwent decompressive craniectomy, followed by alloplastic cranioplasty by either a plastic surgeon or neurosurgeon. No patients were included who required additional flaps or grafts for soft tissue augmentation.
Data Extraction
Clinical data was obtained through the electronic medical record. Information was gathered regarding patient demographics, wound healing comorbidities, indications for surgery, operative information, and outcomes. Patients with missing operative data or follow-up were excluded.
Indications for surgery were categorized as trauma, neoplasm, cerebrovascular accident (CVA), osteomyelitis, resorption of autologous cranioplasty, or other. Demographic information collected included patient age, sex, and race. Wound healing comorbidities reported as a dichotomous variable were defined as the presence of hypertension, diabetes (type I or II), active tobacco smoking, steroid use, hemodialysis, or heavy alcohol use. Heavy alcohol use was defined as an alcohol use disorder diagnosis, hospitalization for alcohol withdrawal, or individuals who consumed greater than an average of 14 alcoholic beverages weekly for men and 7 beverages weekly for women. Patients who had impaired pre-operative neurological status or obstructive sleep apnea (OSA) were also identified.
Operative information included time to surgery from craniectomy, American Society of Anesthesiologist physical classification (ASA), cranioplasty location (hemispheric or bifrontal), type of implant (titanium, PEEK, or PEEK/Medpor composite), and CSF manipulation (ventricular catheter, extra ventricular drain, or lumbar drain). Three categories of surgical outcomes were collected, including complications, implant explantation, and contour defects. Complications were divided into operative and non-operative complications. Operative complications included all cases of hematoma, seroma, infection, implant exposure, and all other concerns that required an operative intervention. Non-operative complications included wound healing issues that were managed only with local wound care and fluid collections that resolved without intervention or with only office-based aspiration. Only operative complications were included in statistical analysis. Poor contour was defined as the presence of a temporal hollow, palpable implant edges, or paralysis of the frontal branch of the facial nerve. This information was ascertained by chart review of surgeon documentation during post-operative visits.
Statistical Analysis
Continuous variables were described as means with standard deviations and categorical variables as percentages. In bivariate analysis, factors associated with operative complications, implant explantation, and poor contour were identified using Mann–Whitney U and Chi-squared tests, where appropriate. All variables with P < .10 were moved to multivariable regression analysis. Logistic regression analysis was then used to understand the relationship between the factors identified on bivariate analysis and their respective outcomes. Results were expressed as odds ratios with a 95% confidence interval and standard error. P-values less than .05 were considered statistically significant. Analyses were performed using Stata 13.0 (StataCorp, College Station, TX, USA).
Results
A total of 101 patients underwent alloplastic cranioplasty, 72% (73/101) were men and 28% (28/101) were women (Table 1). Eighty-two percent (82/101) of patients self-identified as Caucasian. Mean patient age was 42 ± 18 years. All but 3 patient were >18 years of age (one 13-year-old and two 16-year-old patients were included). Fifty-seven percent (58/101) of patients had at least 1 wound healing comorbidity. Six patients had obstructive sleep apnea, 1 patient had a bleeding disorder, 1 patient had chronic kidney disease and underwent regular hemodialysis, and no patients took chronic steroids. Fifty-one percent (51/101) of patients had some form of neurological impairment pre-operatively.
Study Cohort Patient Characteristics and Perioperative Information.

Continuous variables as mean ± standard deviation or median (interquartile range); categorical variables as percentage (number).
Indications for cranioplasty included trauma 44% (44/101), CVA 18% (18/101), cancer 18% (18/101), osteomyelitis 9% (9/101), resorption of prior autologous cranioplasty 4% (4/101), and others 8% (8/101). Other indications included an epidermoid cyst, cranial fibrous dysplasia (2 cases), recurrent epilepsy, a prominent glabella leading to vision distortion, an unruptured aneurysm, bone resorption surrounding a prior alloplastic cranioplasty performed at an outside institution, and patient preference for improved cosmesis after a prior autologous implant. Implant material types included PEEK 64% (65/101), titanium 24% (24/101), and PEEK-Medpor composite 12% (12/101). Eighty-three percent (84/101) of craniectomy defects were hemispheric and 17% (17/101) were bifrontal. Of the patients who underwent bifrontal craniectomies, 76% (13/17) also underwent frontal sinus cranialization prior to cranioplasty. Craniofacial plastic surgeons were the primary surgeon in 90% (91/101) cases, and 10% (10/101) of cases were performed by neurosurgeons alone. Median time to surgery from craniectomy was 5 months.
Intraoperative CSF manipulation occurred in 10% (10/101) of patients. This included placement of a ventricular catheter (6%, 6/101), extra-ventricular drain (3%, 3/101), and lumbar drain (1%, 1/101). Of the 10 patients who had CSF manipulation, 3 went on to receive a lumboperitoneal or ventriculoperitoneal shunt.
Complications and Implant Explantation
Operative complications occurred in 24% (24/101) of patients, see Figure 1. The rate of postoperative infection was 7% (7/101). Operative hematomas and seromas each accounted for 6% (6/101) of complications. CSF leak accounted for only 3% (3/101) of complications. Three miscellaneous complications included an adhered drain, exposed implant over the brow, and implant detachment. One patient experienced both a hematoma and infection. There were no in-hospital postoperative deaths.
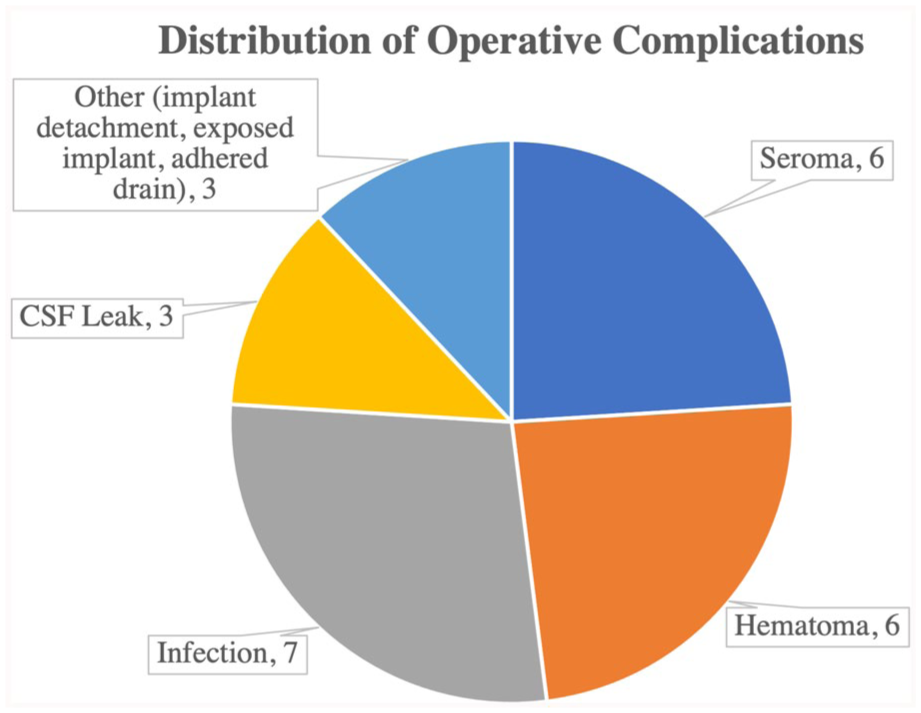
Distribution of operative complications.
The incidence of nonoperative complications was 13%. All cases were small fluid collections that resolved without any intervention or with office-based drainage procedures.
Implant removal occurred in 16% (16/101) of patients. The most common reason for explantation was infection (44%, 7/16). Other reasons included wound breakdown after radiation therapy (12.5%, 2/16), hardware malfunction or shifting (12.5%, 2/16), epidural hematoma (12.5%, 2/16), patient preference (12.5%, 2/16), and persistent seroma (6%, 1/16).
Of the 16 patients who underwent explantation of their primary implant, 94% (15/16) went on to have a successful secondarily placed implant. The patient who did not have an implant replaced had severe medical co-morbidities and resided in a long-term care facility. At a median time to follow-up of 1.5 years, 99% (100/101) of all patients successfully maintained either a primary or secondarily placed implant.
Postoperative Contour
Contour abnormalities occurred in 36% (36/101) of patients (Figure 2). Eighty-nine percent (32/36) of these patients had undergone temporoparietal cranioplasties. Of the temporoparietal group with contour defects, 91% (29/32) had PEEK implants, 6% (2/32) had PEEK/Medpor implants, 10 and 3% (1/32) had titanium implants. In the bifrontal group, 75% (3/4) had titanium implants and 25% (1/4) had PEEK implants. Further, 44% (16/36) of all patients with a contour defect underwent subsequent operations related to cosmesis. Indications for revision surgery were timing at least 6 months after the index operation and the presence of an obvious physical deformity for which the patient desired additional intervention. For temporal hollow specifically, this included medpor implants (2/16), fat grafting (5/16), acellular dermal matrix (ADM) plus fat grafting (1/16), and ADM plus medpor implant (5/16). Other secondary surgeries related cosmesis included scar revisions (2/16) and implant replacement due to prominence (1/16). See Figure 3 as an example of a patient with good post-operative contour after a composite PEEK-Medpor cranioplasty.
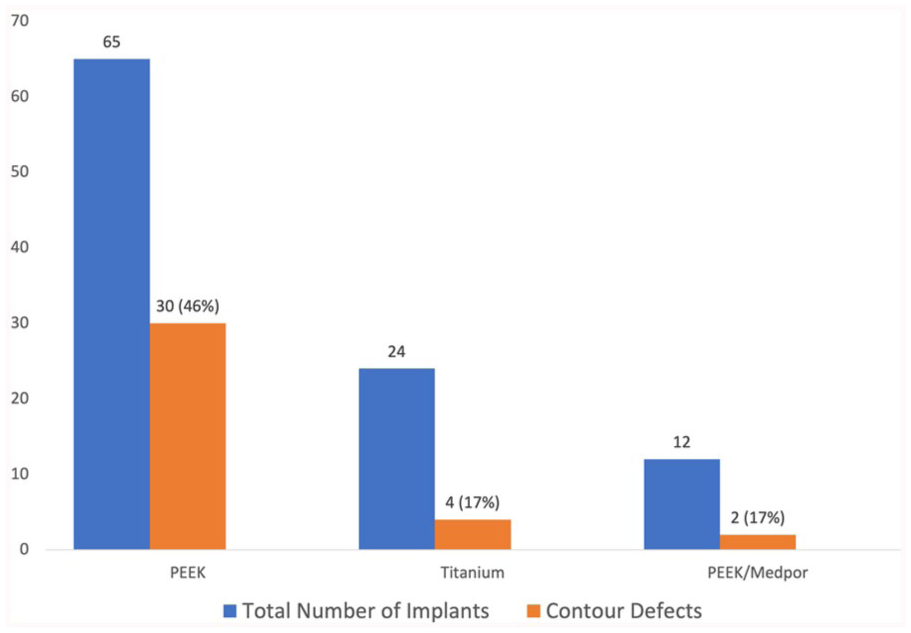
Distribution of postoperative contour defects by implant type.
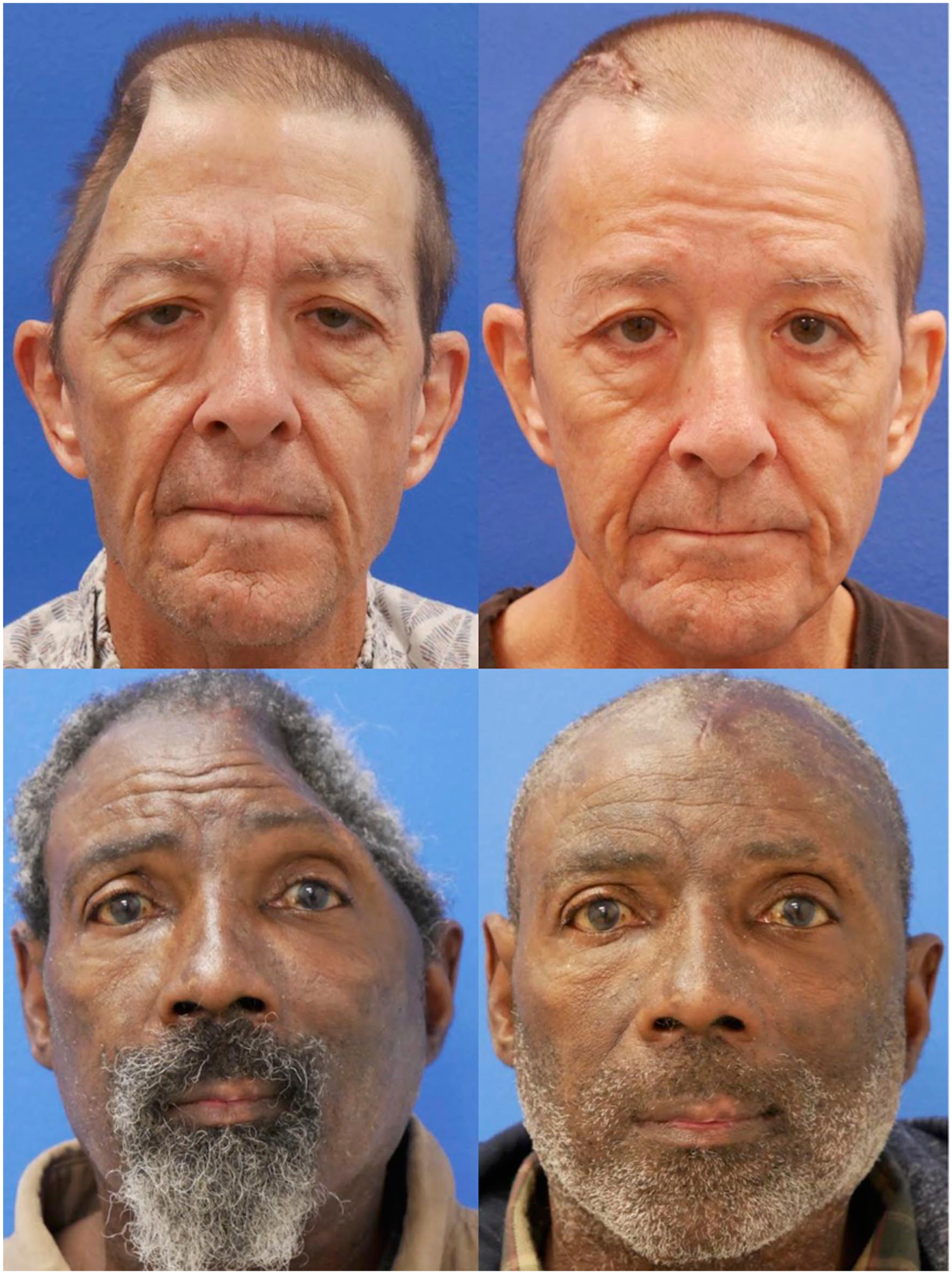
Pre-operative (left) and post-operative (right) images of patients undergoing alloplastic cranioplasty.
Logistic Regression Analysis
Bivariate analysis identified several variables associated with the occurrence of a perioperative complication, contour abnormalities, and implant explantation (Table 2). Factors associated with operative complications included ASA classification (P = .10), heavy alcohol use (P = .001), and craniectomy location (P = .01). Factors associated with contour abnormalities included indication for surgery (P = .001), type of implant (P = .01), impaired pre-operative neurological status (P = .08), age (P = .02), and time to surgery (P = .02). The only factor significantly associated with implant explantation was heavy alcohol use (P = .05).
Bivariate Analysis of Factors Associated with Operative Complications, Contour Irregularities, and Implant Explantation.
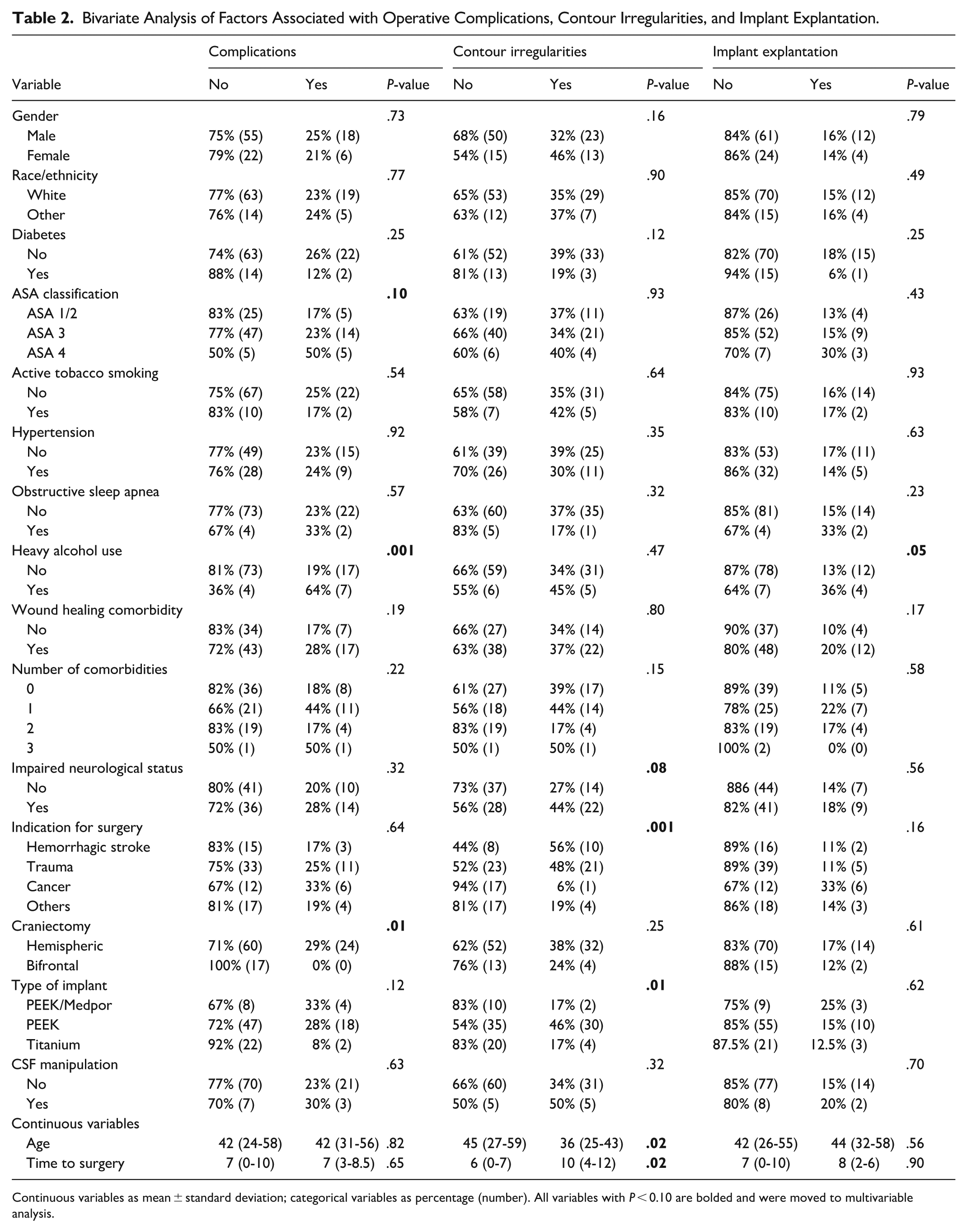
Continuous variables as mean ± standard deviation; categorical variables as percentage (number). All variables with P < 0.10 are bolded and were moved to multivariable analysis.
Candidate predictors from the bivariate analysis were assessed further in a multivariable logistic regression model (Tables 3 and 4). Only outcomes for which predictors were significant on bivariate analysis were included in the regression analysis. ASA classification of level 4 (OR: 5.9, 95% CI: 1.03, 33.1, P = .05) and heavy alcohol use (OR: 6.2, 95% CI: 1.5, 25.5, P = .01) were associated with operative complications. Craniectomy location was not included in multivariable analysis, because there were no complications in the bifrontal group.
Logistic Regression Analysis of Factors Associated with Operative Complications.
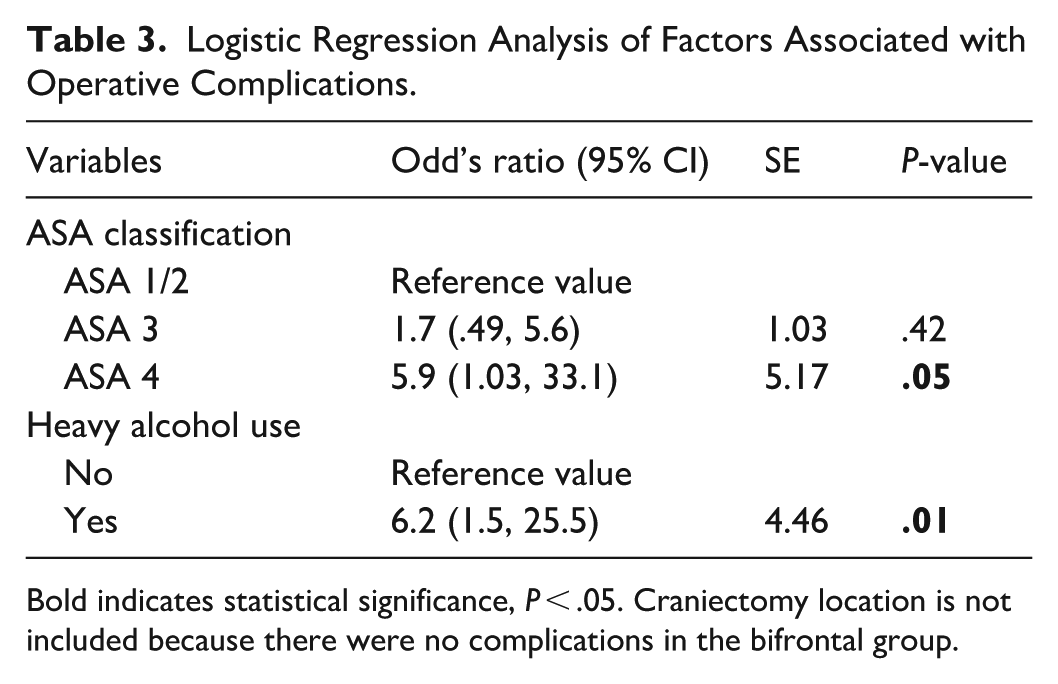
Bold indicates statistical significance, P < .05. Craniectomy location is not included because there were no complications in the bifrontal group.
Logistic Regression Analysis of Factors Associated with Contour Irregularities.
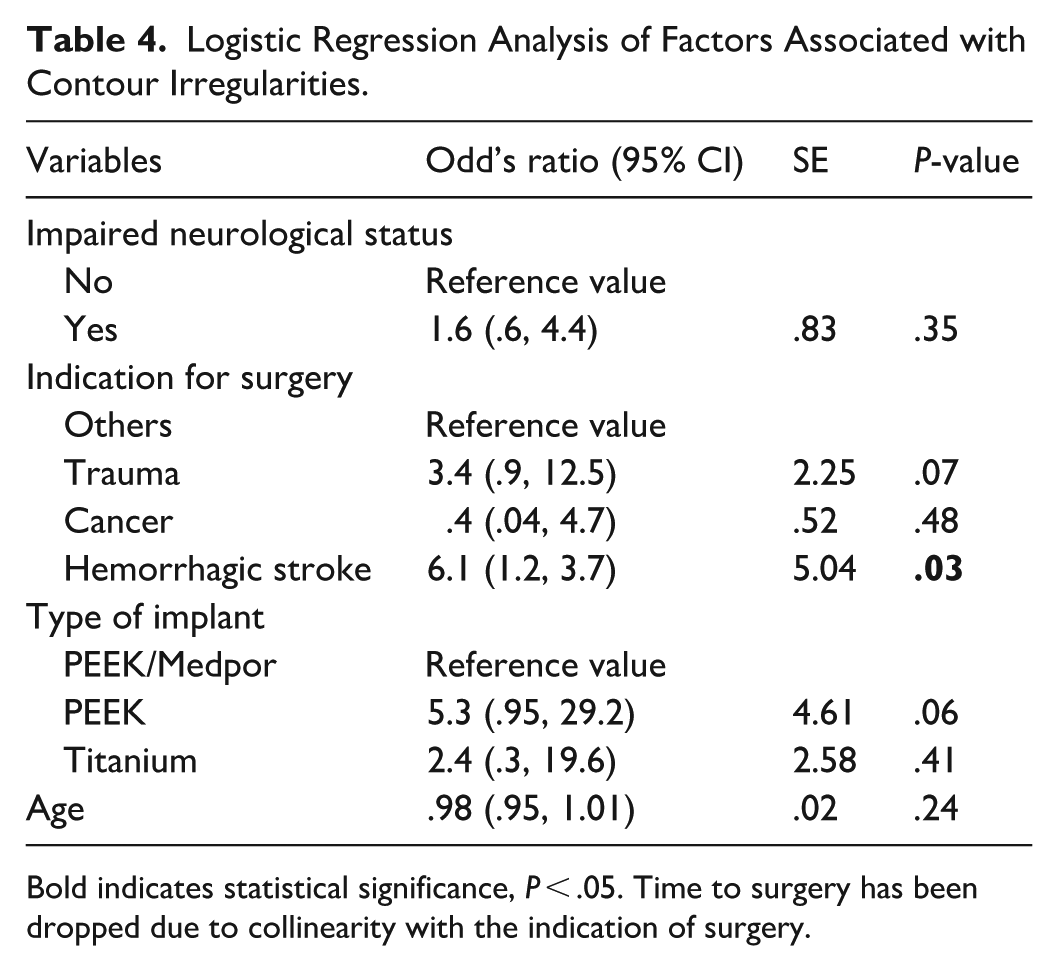
Bold indicates statistical significance, P < .05. Time to surgery has been dropped due to collinearity with the indication of surgery.
Indication of CVA for surgery was a predictor of contour irregularities (OR: 6.1, 95% CI: 1.2, 30.7, P = .03). PEEK implant type and trauma as an indication for surgery approached significance as a factor associated with contour irregularities. Time to surgery was not included in this analysis due to collinearity with indication for surgery.
Discussion
Cranioplasty, though a common and established procedure for both craniofacial and neurological surgeons, is known for the difficulty of achieving optimal results. Autologous reconstruction benefits from cost effectiveness, high accessibility, and use of native tissue, but is susceptible to resorption in as high as 22% of adult patients in addition to the complications seen in alloplastic cranioplasty.1,2 Additionally, use of a patient’s native autologous bone may not be feasible due to trauma or oncologic involvement. For this reason, alloplastic implants have become more widely used in recent years and are associated with shorter operative times, decreased blood loss, good cosmetic outcomes, and high customizability with CAD/CAM technology, but are relatively expensive and time-intensive.1,3,11 With the increased use of alloplastic materials for cranial reconstruction, we sought to review our experience with alloplastic cranioplasty, with retrospective analysis of 101 cases taking place from January of 2014 to January of 2021. To our knowledge, this is the largest review of predictive factors of outcomes in alloplastic cranioplasty in a civilian cohort with these materials to date.
Brain Protection
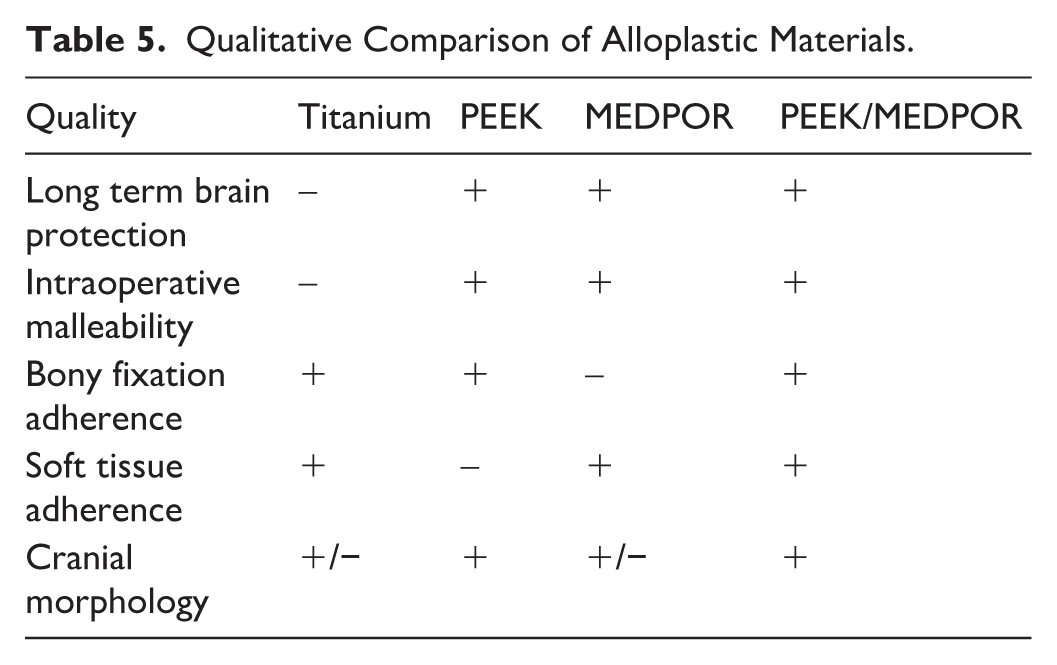
The primary goal of cranioplasty is to achieve brain protection. Thus, implant material selection is of the utmost importance to ensure adequate strength and durability of the implant. Materials available for alloplastic implants include hydroxyapatite, polyetheretherketone (PEEK), combined PEEK/porous polyethylene (Medpor), polymethylmethacrylate (PMMA), and titanium. However, there is no consensus on the best material. 12 When compared to titanium, PEEK can withstand more force without as much surrounding damage in mechanical stress models. 13 The senior author previously reported on 99 PMMA or titanium alloplastic cranioplasties conducted in a military population that resulted in a complication rate of 27%, primary implant retention rate of 95%, and contour abnormalities in 10% of the cohort. 14 It is possible that Medpor is a weaker material independently than when combined with other alloplastic materials, and strength testing is needed. Table 5 is a summative, qualitative comparison of several popular alloplastic materials used in craniofacial surgery today. In our study, 100/101 patients have maintained either their primary or secondary implant at follow up, indicating a high degree of success using alloplastic materials in cranioplasty.
Qualitative Comparison of Alloplastic Materials.
Aesthetic Considerations
In the limited number of studies that do report aesthetic outcomes after cranioplasty, 1.5% to 64% of cases have been reported to be unsatisfactory.4,7,9,15 -17 The wide range of poor cosmetic outcomes could be due to the lack of a standardized, scientifically validated method of cosmetic evaluation for cranioplasty, and/or the inability of soft tissues, including the temporalis muscle to adequately adhere to the alloplastic material.
Temporal hollowing is a frequent adverse outcome and can be attributed to numerous factors, such as temporalis atrophy or soft tissue loss from repeated operations or trauma. 17 Work has been done to quantify this soft tissue loss by Thiensri et al, 18 which found statistically significant volume differences in muscle, soft tissue, and bony areas. Secondary methods to correct temporal hollowing have been described such as injectable fillers, fat grafting, botox, muscle suspension, or solid implant sup plementation.18 -25
In this cohort, 36% (36/101) of patients had a cosmetic abnormality, with the most frequent by far being temporal hollowing. 44% (16/36) of the patients with a contour abnormality underwent an intervention to improve cosmesis, and 13 of these surgeries were specifically to address temporal contour. The most significant predictor for postoperative temporal form irregularity was CVA. The authors expected patients with a history of trauma to have a high rate of contour defects following surgery due to extensive soft tissue damage. However, trauma was found to be a predictor of poor contour of bivariate analysis, but was not statistically significant on multivariable analysis, possibly due to a small sample size of 18. No implant type was found to be a predictor for poor cosmesis.
PEEK implants were a significant predictor for contour irregularities in bivariate analysis, possibly due to poor soft tissue adherence to the smooth surface of the PEEK. PEEK/Medpor composite implant use led to fewer aesthetic defects than PEEK alone (17% vs 46%) due to better soft tissue adherence to the porous surface of Medpor, which is consistent with previous work reported by the primary author. 10 While titanium implants also had a low rate of contour defects (17%), tensile strength is suspect over the long term, therefore a PEEK/Medpor implant may better address both brain protection and enhanced soft tissue adherence. However, only 12/101 implants were the combination PEEK-Medpor design. The authors are working to publish a larger cohort specifically looking at outcomes with this technique.
Complications
Complications after cranioplasty are relatively common with overall rates between 11% and 40.4% for varying implant materials.4,6,26 Bone resorption and infection are cited as the most common complications when autologous materials are included.6,27,28 Further, many studies reviewing cranioplasty outcomes include both autologous and alloplastic implants, which limits the broad applicability of the results.29,30
This study found an operative complication rate of 24% and an implant replacement rate of 16%. These findings are consistent with other larger retrospective reviews, one of which found a reoperation rate of 23% in 195 cranioplasties and another a reoperation rate of 15.2% in 500 cranioplasties. However, both studies included both alloplastic and autologous implants together,29,31 while this study involves a pure cohort of alloplastic cranioplasty patients, making our results particularly clinically relevant. The most common cause for operative complication and replacement was infection. After multivariable analysis, ASA Classification of 4 and heavy alcohol use were significant predictors for postoperative complications. The American Society of Anesthesiologists defines ASA Class 4 as, “a patient with severe systemic disease that is a constant threat to life.” 32 This is an expected finding as more ill patients would have a higher propensity for complications postoperatively, however, this has not been confirmed as an association with complications in alloplastic cranioplasty in previous studies. In cases where alloplastic cranioplasty timing is flexible, it may be beneficial to optimize medical co-morbidities contributing to ASA class prior to surgery.
In addition to being a predictor for operative complications, heavy alcohol use was the only predictor of implant explantation (P = .05). Of the 11 patients with heavy alcohol use, 4 underwent implant explantation. Chronic alcohol use is proposed to impair wound healing by limiting the inflammatory response to tissue injury, thus causing increased rates of hospital acquired infections and surgical site dehiscence. 33 These findings suggest that a robust social history should not be overlooked as part of the pre-operative assessment, and attempts to treat heavy alcohol use preoperatively may be beneficial.
Other Considerations
Most of the literature on cranioplasty emanates from the neurosurgical community. At our institution, the vast majority of our cranioplasties are performed in a multidisciplinary fashion by a craniofacial trained plastic surgeon along with a neurosurgeon both intra and post-operatively. Therefore, the results presented in this article should be placed in this unique context compared to the existing cranioplasty literature. Future work should include the collection of patient reported outcomes, evaluating outcomes across institutions, further research on aesthetic results, and analyzing the effects of CAD/CAM technology for implant design.
Limitations
Limitations include operative variability, as multiple craniofacial and neurosurgeons performed these procedures. This study is also a retrospective design and conducted at a single institution, limiting the patient cohort size and demographics. Median time to follow up was 1.5 years, which may not encompass the entire cohort of patients who develop complications or poor cosmesis, as implant extrusion and thinning of the overlying soft tissue may not manifest until several years following surgery. Development of an implant registry, similar to the breast implant population, may be helpful in monitoring long-term complications on a nationwide scale. Three pediatric patients were included in this study, future work with larger cohorts could look at this population separately as they may confound results. Despite these limitations, we believe these findings will help craniofacial and neurosurgeons in understanding the complications and cosmetic outcomes of alloplastic cranioplasty on a broad scale.
Conclusion
We present a series of alloplastic cranioplasty in 101 patients with both aesthetic and functional outcomes. At a median follow-up of 1.5 years, 99% (100/101) of patients retained a primary or secondarily placed implant, and 36% of patients had postoperative contour defects. Preoperative ASA class 4 status and heavy alcohol use were predictive for operative complications, CVA was predictive for contour defects, and heavy alcohol use was predictive for implant explantation. These findings indicate a high success rate is possible with alloplastic cranioplasty and that much work remains to optimize aesthetic patient outcomes.
Footnotes
Acknowledgements
David Ring, MD, PhD.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Raymond Harshbarger is a member of the Editorial Board of FACE and did not take part in the peer review or decisionmaking process for this submission.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical Considerations
This study was approved by the Institutional Review Board (IRB) at the University of Texas (Approval #00000311). All data was collected retrospectively and kept confidential and anonymous.
Consent to Participate
This was a retrospective study, thus consent to participate was not necessary. All data collected was de-identified.
