Abstract
Mandibular reconstruction following Odontogenic Keratocyst (OKC) resection poses significant challenges in restoring both structural integrity and functional dentition. Traditional approaches require multiple surgeries and extended rehabilitation periods. This case report presents a 70-year-old male requiring anterior mandibular reconstruction following OKC resection from teeth #31 to 46. Using an innovative fully digital workflow, a patient-specific titanium implant (PSI) with pre-integrated dental implants via Direct Metal Laser Sintering (DMLS) was designed and fabricated. The PSI featured a gradient mesh architecture optimized through finite element analysis, with dental implants precisely positioned and laser-welded during fabrication. Single-stage surgical placement was followed by prosthetic rehabilitation at 3 months using a digitally designed DMLS-fabricated acrylic-metal hybrid prosthesis. At 18-month follow-up, the patient demonstrated complete functional restoration with optimal mastication, speech articulation, and facial symmetry. Radiographic evaluation confirmed excellent osseointegration without complications. The integrated digital workflow reduced treatment time by 40% compared to conventional protocols. This case represents the first reported use of pre-mounted dental implants within a PSI for mandibular reconstruction, demonstrating how advanced digital technologies can streamline complex rehabilitative procedures while achieving superior functional outcomes.
Keywords
Introduction
Odontogenic Keratocyst (OKC) is an aggressive benign lesion with recurrence rates approaching 30% following conservative treatment. 1 Surgical resection, while curative, creates significant mandibular defects that compromise facial esthetics, occlusion, and oral function. 2 Traditional reconstruction of these defects were multistage, the first stage included the resection of lesion and graft placement using autogenous grafts or stock reconstruction plates followed by placement of endosseous dental implants and a third stage of prosthetic rehabilitation.3,4 Recent advances in digital technology have enabled the development of patient-specific implants (PSIs)-custom titanium frameworks manufactured through computer-aided design/manufacturing (CAD/CAM) that precisely match individual anatomy, hence reducing treatment duration, improving accuracy of implant positioning, and minimizing overall surgical burden. 5 While PSIs have demonstrated superior outcomes in mandibular reconstruction, conventional protocols still require secondary surgeries for dental implant placement, prolonging rehabilitation. 6
We present an innovative approach integrating pre-mounted dental implants within a PSI framework, enabling single-stage reconstruction with immediate foundation for prosthetic rehabilitation. This technique, which was the first reported, exemplifies how digital workflows can transform complex reconstructive procedures. This case report has been reported in line with the SCARE checklist.7,8
Case Presentation
Clinical History
A 70-year-old male reported to the Department of Prosthodontics with a progressive swelling lingual to teeth #41 to 44 (Figure 1a-c: preoperative photographs). A detailed medical history was taken, following which radiographic examination was carried out. Radiographic examination revealed a well-defined radiolucent lesion extending from the left mandibular central incisor to the right first molar. Incisional biopsy confirmed the OKC diagnosis. Given the lesion’s size and aggressive nature, segmental mandibular resection with immediate reconstruction was planned. The decision for immediate reconstruction was based on intraoperative and histopathological confirmation of complete resection with clear margins. Patient-specific factors, including advanced age and desire to avoid multiple surgeries, also supported the single-stage approach. Should recurrence occur, the PSI’s modular design allows for surgical access or partial explanation.
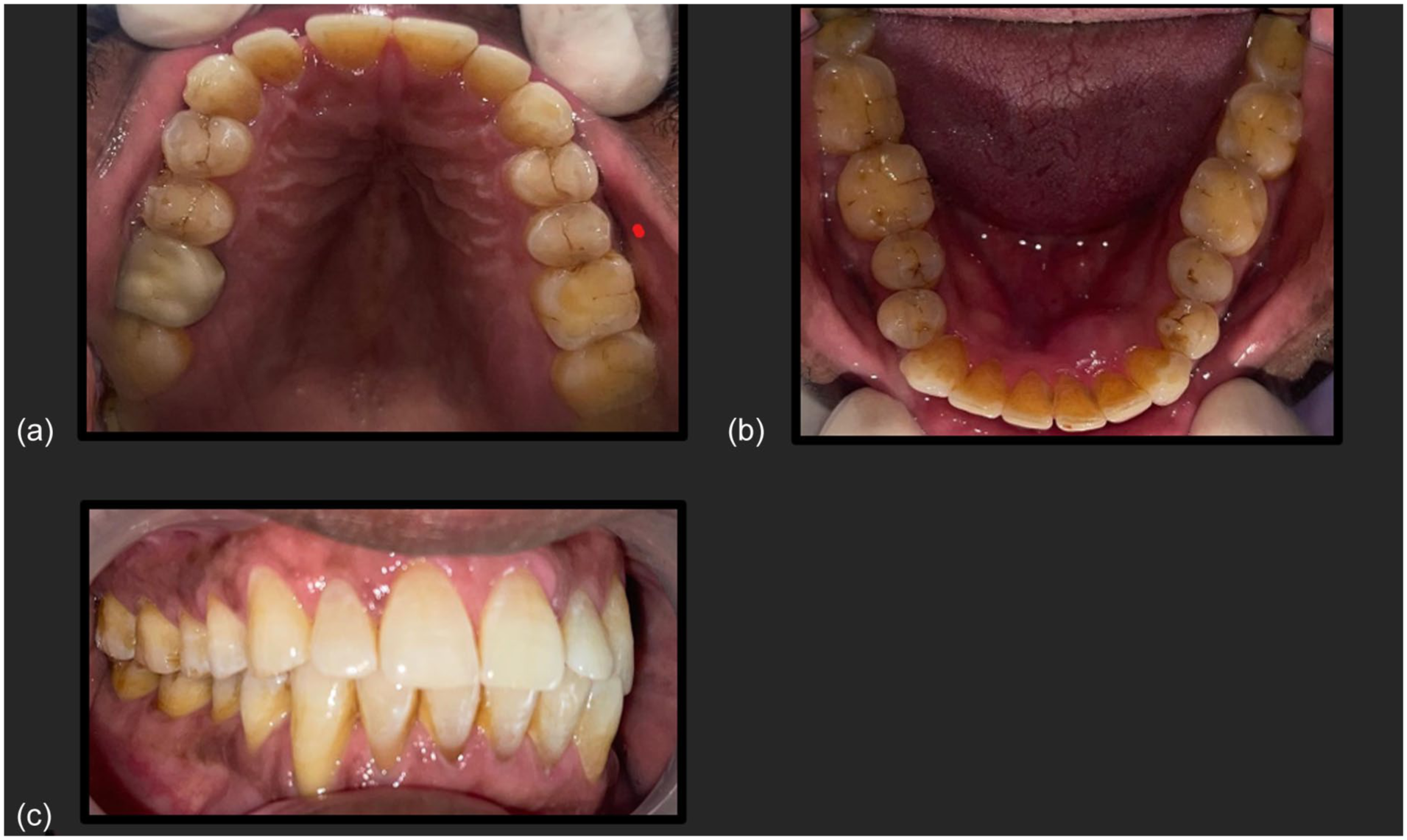
Preoperative intraoral presentation of the intact maxilla and mandibular lesion depicting buccal and lingual cortical expansion from the region of teeth #41 to #44, with intact mucosal covering: (a) Maxillary occlusal view, (b) Mandibular occlusal view, and (c) Lateral view illustrating occlusion.
Digital Planning and PSI Design
High-resolution cone-beam computed tomography (CBCT) data were acquired and converted to 3D models using specialized medical imaging software (Materialize Mimics, Belgium; Figure 2a-c: CBCT scan). Clinical and radiographic examinations confirmed the presence of a large anterior mandibular defect with no signs of infection or pathological fracture. Virtual resection margins were established with 5 mm safety borders. The PSI framework was designed using Geomagic Freeform (3D Systems, USA) with precise contouring of residual mandibular segments and strategic fixation point placement. The structural design incorporated a gradient mesh architecture transitioning from solid titanium (Young’s modulus: 110 GPa) in load-bearing regions to a 65% porous structure for tissue integration. Six platforms for 4.0 mm diameter dental implants were strategically positioned based on prosthetic requirements, with textured surface regions to promote soft tissue adaptation. Finite element analysis validated the design’s biomechanical performance under physiological loading conditions (500 N vertical, 200 N lateral forces)
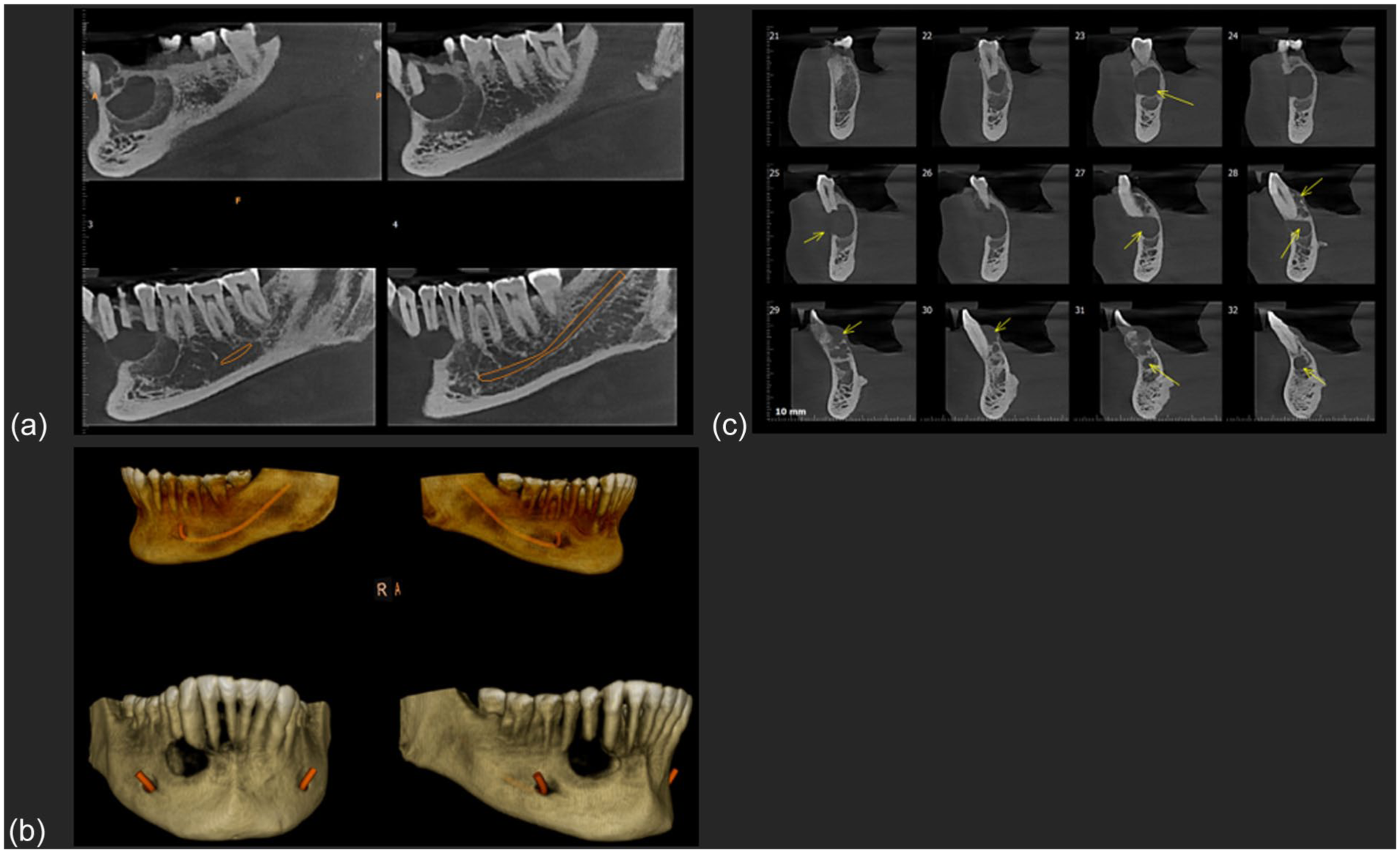
Preoperative radiographic assessment using cone-beam computed tomography (CBCT): (a) Multiplanar reformatted CBCT images showing a well-demarcated unilocular radiolucency extending across the anterior mandible, with cortical thinning and expansion, but no signs of cortical perforation, (b) three-dimensional volume-rendered reconstructions of the mandible illustrate the spatial relationship between the lesion and vital structures, including the inferior alveolar nerve canal, and (c) serial CBCT cross-sections (coronal slices) identifying internal septations and the full extent of the lesion from the lingual to buccal cortices, aiding in defining the resection margins.
Manufacturing Protocol
The patient-specific implant (PSI) design process began with the acquisition of high-resolution CBCT data, which was converted to a 3D model using medical imaging software. The PSI was designed using CAD software optimized explicitly for medical applications (Figure 3a and b: Patient Specific Implant designed on CAD software, Figure 3c and b: Construction of patient-specific implant). The titanium framework was manufactured using Direct Metal Laser Sintering (DMLS) technology, specifically employing a Grade 5 Titanium alloy. Manufacturing parameters included a layer thickness of 30 μm, laser power of 280 W, scanning speed of 1200 mm/s, and hatch spacing of 140 μm. post-processing involved stress-relief heat treatment at 650°C for 3 hours in an argon atmosphere. Surface modifications were performed sequentially, beginning with mechanical polishing of external surfaces to achieve a surface roughness (Ra) less than 0.8 μm. Tissue-contact regions underwent aluminum oxide sandblasting using 250 to 500 μm particles, followed by acid etching in HCl/H₂SO₄ solution at 60°C for 30 minutes. Final passivation was achieved using 30% HNO₃ at room temperature for 60 minutes.
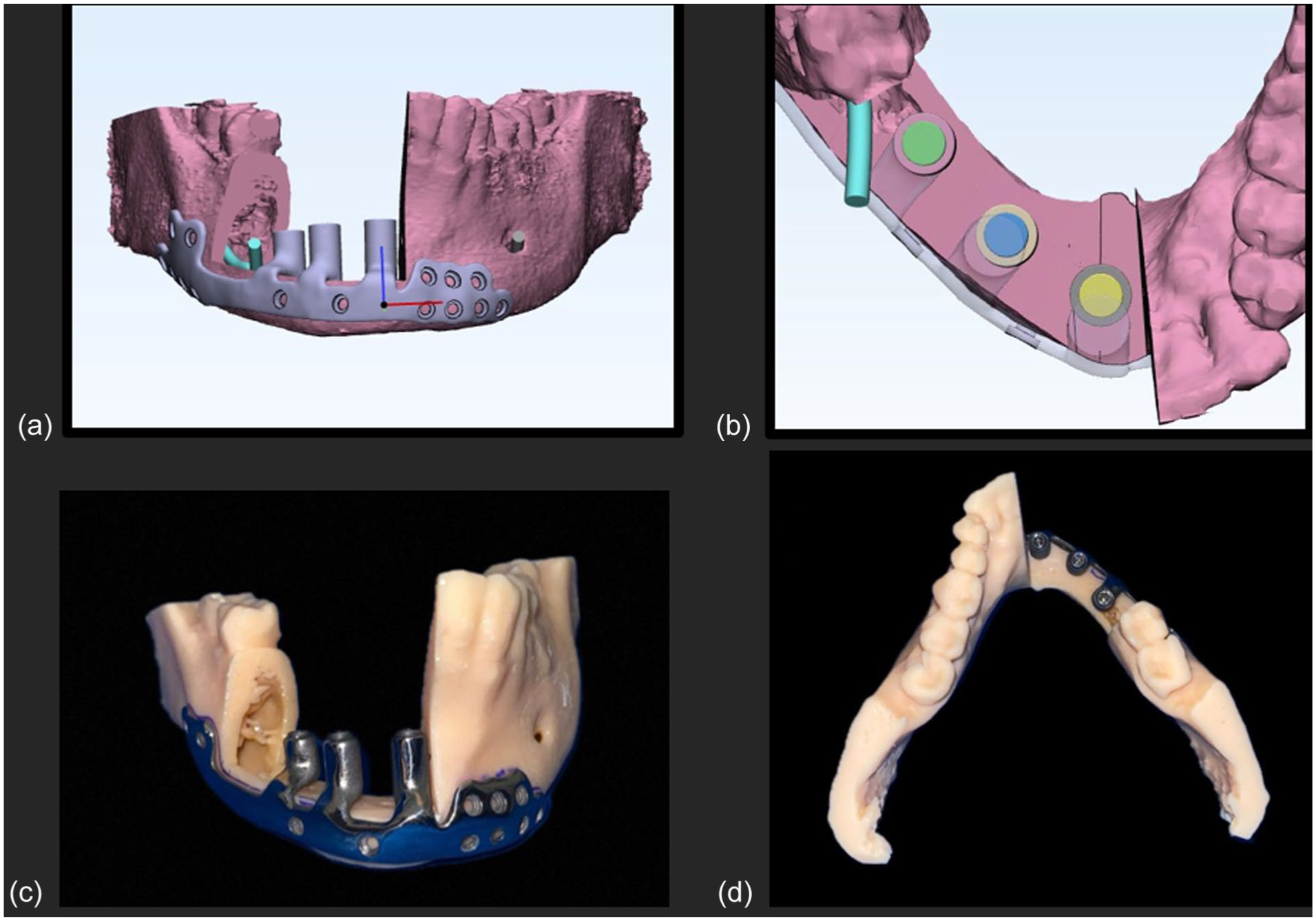
Design and fabrication process of the patient-specific titanium implant (PSI) with integrated dental implants: (a) virtual planning of the mandibular resection and PSI design using CAD software, showing resection margins and implant channels, (b) top-down view of implant positions integrated into the PSI framework, aligned to the prosthetic envelope, (c) fabricated PSI prototype fitted onto a stereolithographic mandibular model, demonstrating anatomic conformity and implant orientation, and (d) occlusal view of the 3D-printed mandibular model with the PSI in situ, highlighting prosthetic space planning and implant emergence profile.
Dental Implant Integration
Six dental implants (Straumann BLT, 4.0 × 10 mm) were precisely positioned using a digitally fabricated positioning guide. Integration was achieved through a systematic protocol from CNC machining interface surfaces to a tolerance of ±0.05 mm. Multi-point tack welding provided initial stabilization, and circumferential laser welding using an Nd: YAG laser system operating at 1064 nm wavelength, 20 Hz pulse frequency, and 300 W peak power. Radiographic verification confirmed weld integrity throughout the integration process. This framework served as the load-bearing structure, and the implant support was achieved mechanically. Quality control includes coordinate measuring machine verification with tolerances of ±0.1 mm for critical features and ±0.2 mm for non-critical surfaces, followed by terminal gamma sterilization at 25 kGy.
Surgical Procedure
Under general anesthesia, the surgical procedure utilized a submandibular approach with anterior release incisions to provide adequate visualization and access for precise PSI placement. Mental nerve preservation was prioritized. The PSI was stabilized with 2.0 mm titanium screws, guided by 3D-printed anatomical models. Soft tissue closure was achieved using native mucosa, and no vascularized soft tissue flap was used in this case. However, postoperative healing was closely monitored, and meticulous hygiene maintenance instructions were provided to prevent exposure or infection.
Prosthetic Rehabilitation
At 3 months post-surgery, digital impressions were captured using an intraoral scanner (3Shape TRIOS 4). The prosthetic workflow began with the fabrication of a digital verification jig to confirm implant positions, followed by virtual articulation and occlusal design. A DMLS framework with acrylic veneering was fabricated and delivered at 6 months with screw retention, completing the rehabilitation process. The scan data was processed to generate a 3D-printed working model with integrated analog implant positions. Following verification, a bite registration was made to verify occlusion, followed by a wax try-in. The final prosthesis was designed using CAD software, incorporating the verified measurements and esthetic parameters from the digital wax-up.
DMLS Acrylic Plus prosthesis was fabricated, combining the durability of a metal framework with the esthetic advantages of acrylic. This advanced manufacturing technique ensured precise fit, optimal strength, and superior esthetics. The prosthesis was delivered 6 months post-surgery. Clinical evaluations and digital occlusal analysis confirmed the successful integration of the implant and restoration of optimal occlusal function
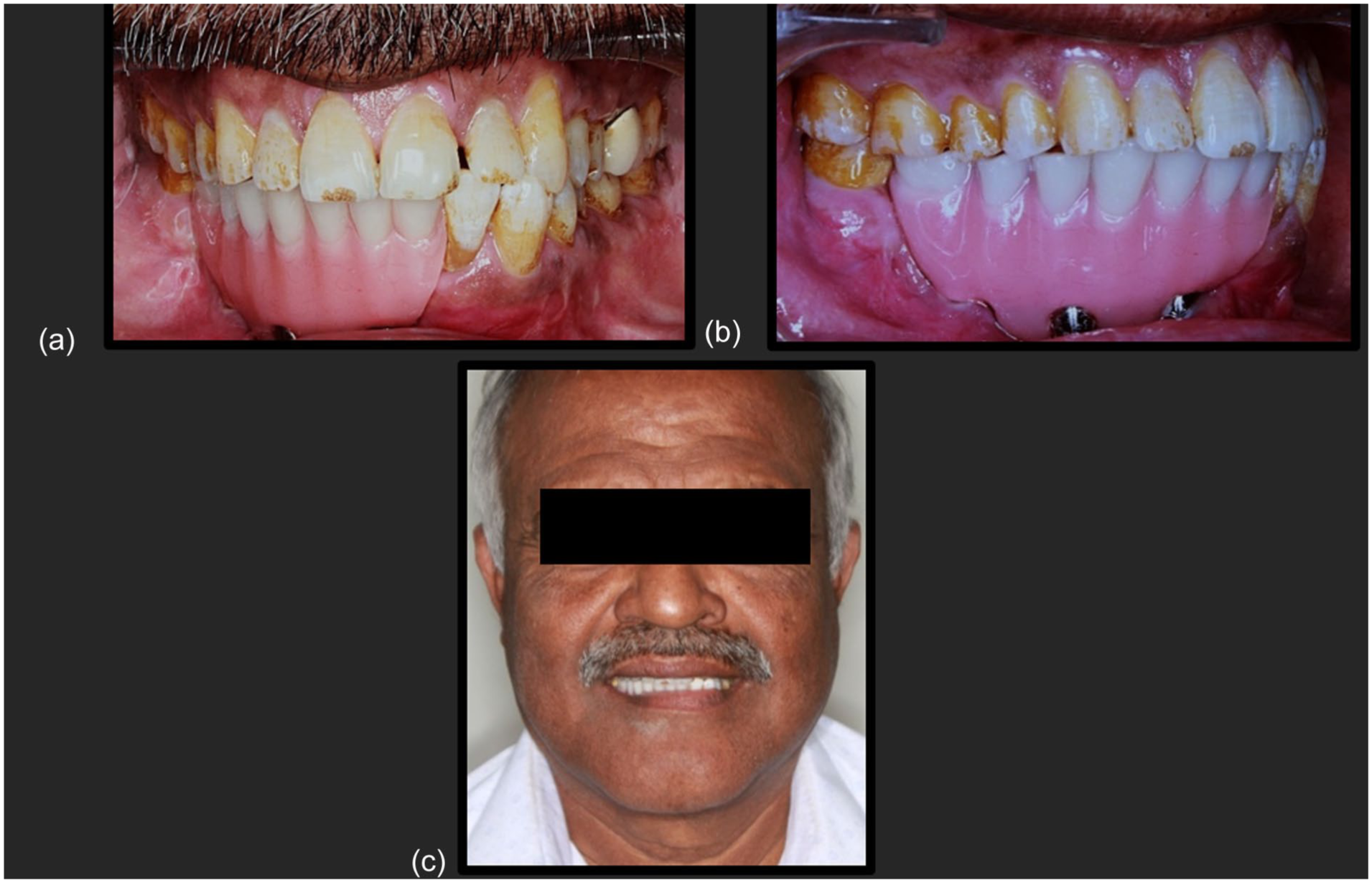
(a-b) Post-operative intraoral views following prosthetic rehabilitation, and (c) Extraoral frontal view showing the patient’s postoperative smile and overall facial harmony.
Outcome
A structured follow-up protocol was implemented with assessments at 3 days, 1 month, 3 months, and 6 months post-insertion. The patient demonstrated complete restoration of mastication and speech function. Facial symmetry and lip support were successfully maintained, addressing the initial esthetic concerns. Patient satisfaction was exceptionally high, with an Oral Health Impact Profile (OHIP-14) score of 4/56, indicating minimal impact on quality of life.
Discussion
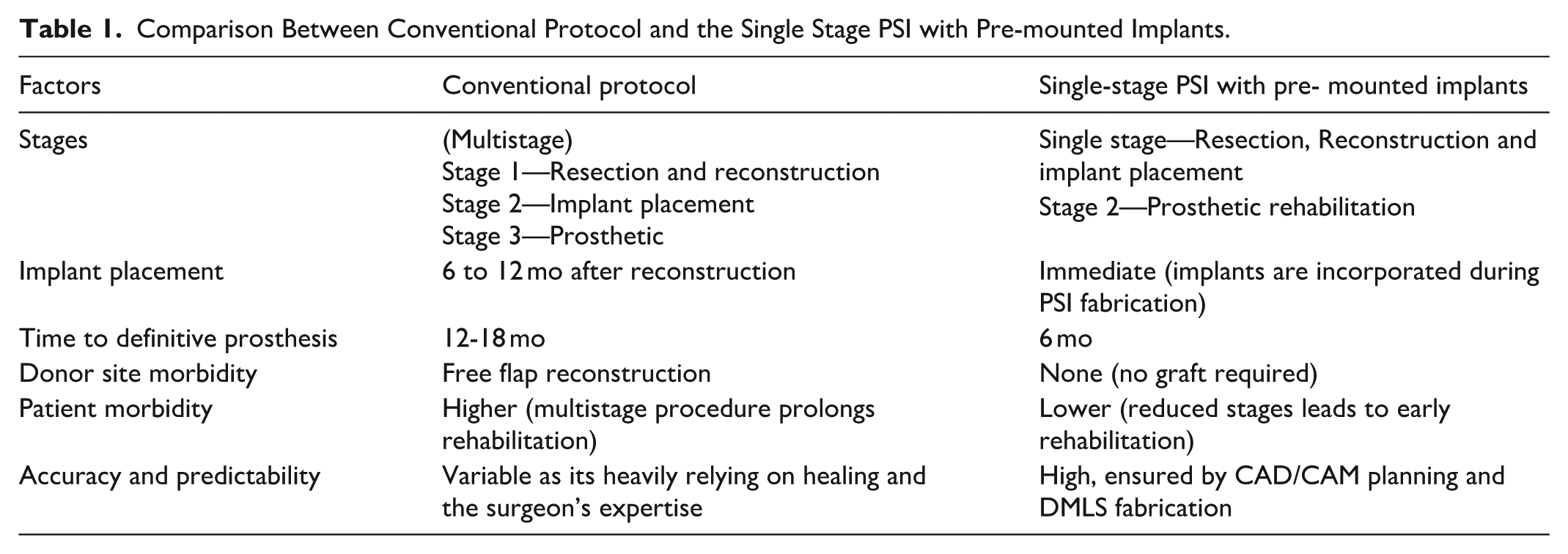
This case highlights the transformative potential of integrating dental implants within patient-specific implant (PSI) frameworks for mandibular reconstruction. The conventional workflow typically involves 2 to 3 surgical stages, with dental implant placement delayed by several months and prosthetic rehabilitation extending up to 18 months (Table 1). However, the adoption of a single-stage approach eliminated the need for secondary surgeries, while the fully digital workflow ensured prosthetically driven outcomes with high precision and efficiency. Patient selection and oncologic safety are critical to the success of this protocol. Ideal candidates include individuals presenting with extensive anterior mandibular defects, adequate residual bone height, and favorable soft tissue conditions. While this technique does not replace the gold standard of vascularized free fibula flap reconstruction, it provides a viable alternative in cases where flap surgery is contraindicated due to systemic comorbidities, patient preference, or logistical constraints. 9
Comparison Between Conventional Protocol and the Single Stage PSI with Pre-mounted Implants.
Oncologic safety was addressed through en bloc resection with a 5 mm safety margin beyond the radiographic extent of the lesion, as guided by cone-beam computed tomography (CBCT) and clinical correlation. Given the benign nature of odontogenic keratocyst (OKC) and its low malignant potential, this approach allowed for immediate reconstruction without compromising oncologic principles. The absence of soft tissue or perineural involvement further supported the decision. This strategy enabled optimization of functional recovery, reduction in treatment duration, and improvement in overall patient quality of life, while maintaining vigilant postoperative surveillance.
From a technical perspective, the PSI framework incorporated a gradient mesh architecture, enhancing stress distribution while facilitating osseointegration. The pre-mounted dental implants, laser-welded during the fabrication phase, provided enhanced mechanical stability compared to conventional intraoperative placement. This innovation eliminated the need for delayed implant insertion and shortened the rehabilitation timeline. The reported 40% reduction in treatment time, attributable to integrated digital planning, reduced operative time, and immediate prosthetic workflow, represents a significant improvement over the traditional multistage protocol. 10 Clinically, the integration of pre-mounted implants enabled early prosthetic planning and delivery, reducing errors in implant positioning and improving functional predictability. The seamless digital workflow, from surgical planning to prosthetic fabrication, minimized patient morbidity, and streamlined interdisciplinary collaboration. 11
Given the known recurrence potential of OKC, a structured follow-up protocol was established. Serial CBCT imaging was scheduled every 6 months during the first 2 years, followed by annual assessments. The PSI design featuring porosity and modularity not only facilitated soft tissue integration but also permitted radiologic evaluation of underlying structures. In the event of recurrence, conservative re-intervention using enucleation and peripheral ostectomy may be feasible, potentially through selective removal of PSI segments. The screw-retained fixation system allows for partial or complete framework explantation when surgical re-entry is required. Continuous monitoring of peri-implant mucosal health further enhances early detection of pathologic changes, ensuring recurrence can be effectively managed without compromising structural or functional outcomes.
Despite its promising results, this case report has several limitations. First, the follow-up duration of 18 months is insufficient to assess long-term outcomes, including risks of mechanical fatigue, peri-implant disease, or structural degradation. Second, the absence of autogenous or regenerative bone beneath the PSI means that prosthetic success is entirely dependent on the durability and integration of the titanium framework. While no complications were observed during the current follow-up period, extended longitudinal studies are necessary to validate the stability of the system under continuous occlusal load. Additionally, the high cost and need for specialized digital planning and manufacturing may limit the adoption of this technique in resource-constrained settings. Future advancements in bioactive coatings, regenerative scaffolds, and smart materials could restore biological continuity alongside mechanical support, improving long-term outcomes and expanding the applicability of this approach.
Conclusion
This case represents a paradigm shift in mandibular reconstruction, demonstrating how digital technologies can integrate surgical and prosthetic rehabilitation into a streamlined protocol. The successful outcome validates the concept of pre-mounted dental implants within PSI frameworks, offering a patient-specific solution that addresses structural and functional rehabilitation in a single stage. As digital technologies continue advancing, such approaches may become the standard of care for complex maxillofacial reconstruction.
Footnotes
Acknowledgements
The authors thank the biomedical engineering team for technical PSI design and manufacturing support.
Ethical Considerations
Every patient undergoing a basic oral examination is required to provide consent for their clinical information to be used for academic and research purposes. This consent ensures that anonymized data may be utilized to advance dental education, enhance research, and improve patient care practices, while maintaining strict confidentiality and adherence to ethical standards.
Consent to Participate
Written informed consent was obtained from the patient for publication of this case report and accompanying images. A copy of the written consent is available for review by the Editor-in-Chief of this journal on request.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Anonymized data supporting this case report, including selected CBCT images and clinical findings, are available upon request.
