Abstract
Keywords
Introduction
Carotid body tumors (CBTs), or paragangliomas, are rare, slow-growing neuroendocrine tumors that typically grow near major blood vessels and nerves. This neoplasm arises from neural crest-derived ectoderm and can either be non-functioning or hormone-secreting due to chromaffin cells that produce adrenaline and noradrenaline. Most commonly, they are located in the adventitia of the carotid bifurcation and receive sympathetic innervation (ie, stimulus for secretion) from the glossopharyngeal nerve. 1 Carotid body tumors are rare in the general population, with a reported incidence of 1 to 2 cases per 100 000 people, and make up approximately 0.6% of all head and neck tumors. They are even rarer in the pediatric population (Supplemental Table 1). A 2024 systematic literature review identified only 29 published cases of pediatric carotid body tumors since 1968. 2 Although rare, they are still among the most common paragangliomas in the head and neck and should be included in the differential in a young patient presenting with a neck mass. 3 Primary treatment for carotid body tumors is subadventitial resection with or without prior embolization. Surgical resection, however, carries high risk, including the potential for vessel or nerve injury, uncontrollable bleeding, or stroke, all of which could lead to brain injury and other major complications.
We describe a case of a 14-year-old unimmunized boy presenting with a right-sided neck mass diagnosed as a carotid body tumor and treated with en bloc surgical resection without prior embolization. We aim to highlight the importance of both early recognition and diagnosis, as well as the crucial role of thorough preoperative surgical planning.
Case
A 14-year-old boy with a recently diagnosed right carotid body tumor at an outside hospital presented to our institution for a second opinion, further evaluation, and treatment. Beginning 2 years prior, he first noticed a small right-sided neck mass following a 1-week history of illness he characterized as “cold symptoms,” which included mainly coughing and congestion. At the time, the mass was thought initially to be a swollen lymph node. However, the mass persisted, and 3 months prior to his initial presentation and workup he noted the swelling began to worsen progressively. He was taken to a local hospital in Hawaii, where his family was living at the time. A thorough workup was performed, including Complete Blood Cell count (CBC), Purified Protein Derivative (PPD), Bartonella henselae testing (cat scratch disease), ultrasound, Magnetic Resonance Imaging (MRI), and Fine Needle Aspiration (FNA) of the mass. Ultrasound showed lymphadenopathy. The FNA, although limited by poor cell preservation, demonstrated a polymorphous lymphoid infiltrate. On MRI, a possible right carotid body paraganglioma was noted. No significant lab testing abnormalities were noted.
Shortly after the biopsy, he developed “cold symptoms” once again: sinus headaches, congestion, malaise, lightheadedness, and feeling warm with no fevers measured at home. Notably, his neck mass dramatically increased in size in the 2 weeks after his illness. He was then taken to Los Angeles due to a lack of specialists in their region of Hawaii, and per the mother’s report, the diagnosis of a carotid body tumor was confirmed. However, the mother sought a second opinion and further evaluation and care at our institution, where they planned to move and would have a support system in place, ultimately bringing him to our hospital.
On presentation to our facility, he had about a 2-week history of neck stiffness, fatigue, intermittent chills, and fever. He also complained of lightheadedness, dizziness, occasional headaches, congestion, and snoring due to his recent illness. The generalized fatigue had been ongoing for about a year, which he felt was likely due to the “volcanic smog” in the area in which he lived. A few days after these symptoms began, his mother noted diffuse swelling on both sides of his neck and inguinal regions, and 2 days before presentation, sinus congestion, and sneezing started. Pertinent history included no personal history of hospitalizations or significant illness; however, he was unimmunized, having only received 1 tetanus shot in his lifetime. His mother reported a Mumps diagnosis 1 month prior that she said was currently resolving. Surgical history included bilateral strabismus surgery at age 6 with no complications. He had a full-term birth with no complications or developmental delays. Family history included Hashimoto’s Thyroiditis in the mother, as well as hypothyroidism in several other family members. Of note, his brothers also had neck and inguinal lymphadenopathy similar to the patient.
Physical examination showed mild tachycardia at 105 beats per minute. He appeared pale, with bilateral diffuse swelling of the anterior cervical regions, more prominent on the right than the left. In addition, there was a pulsating mass in his right cervical region. He had full range of motion of his neck with no signs of meningismus. The only other physical exam findings were enlarged lymph nodes in his inguinal region bilaterally. Based on his history and physical exam, initial differential diagnoses were carotid body tumor versus diffuse lymphadenitis.
Workup included a CBC, lactate dehydrogenase (LDH), Uric Acid, C-reactive protein (CRP) level, Cytomegalovirus (CMV), Epstein-Barr Virus (EBV), Bartonella, and blood culture. Imaging consisted of MRI, magnetic resonance angiography (MRA), magnetic resonance venography (MRV), and computed tomography angiography (CTA). LDH and uric acid levels were normal, suggesting no evidence of tumor lysis. CRP was elevated. CBC showed low hemoglobin and mean corpuscular volume (MCV) levels demonstrating iron deficiency anemia, as well as high white blood cells, neutrophils, and monocytes. CMV and Bartonella were negative. While there was evidence of previous EBV infection, nothing was consistent with an acute process. Blood culture showed no growth. MRI/MRA/MRV scans demonstrated a hypervascular mass splaying the right carotid bifurcation, consistent with a carotid body tumor. No tumor was seen in the left bifurcation. No stenosis was present, and the right jugular vein was dominant. CT angiography showed the same result, measuring the tumor as at least 6 cm in the cephalocaudal dimension and 5 cm transverse. The right jugular vein was slightly impinged upon and displaced laterally. Prominent cervical adenopathy was seen, and the right sphenoid sinus was completely opacified (Figure 1).
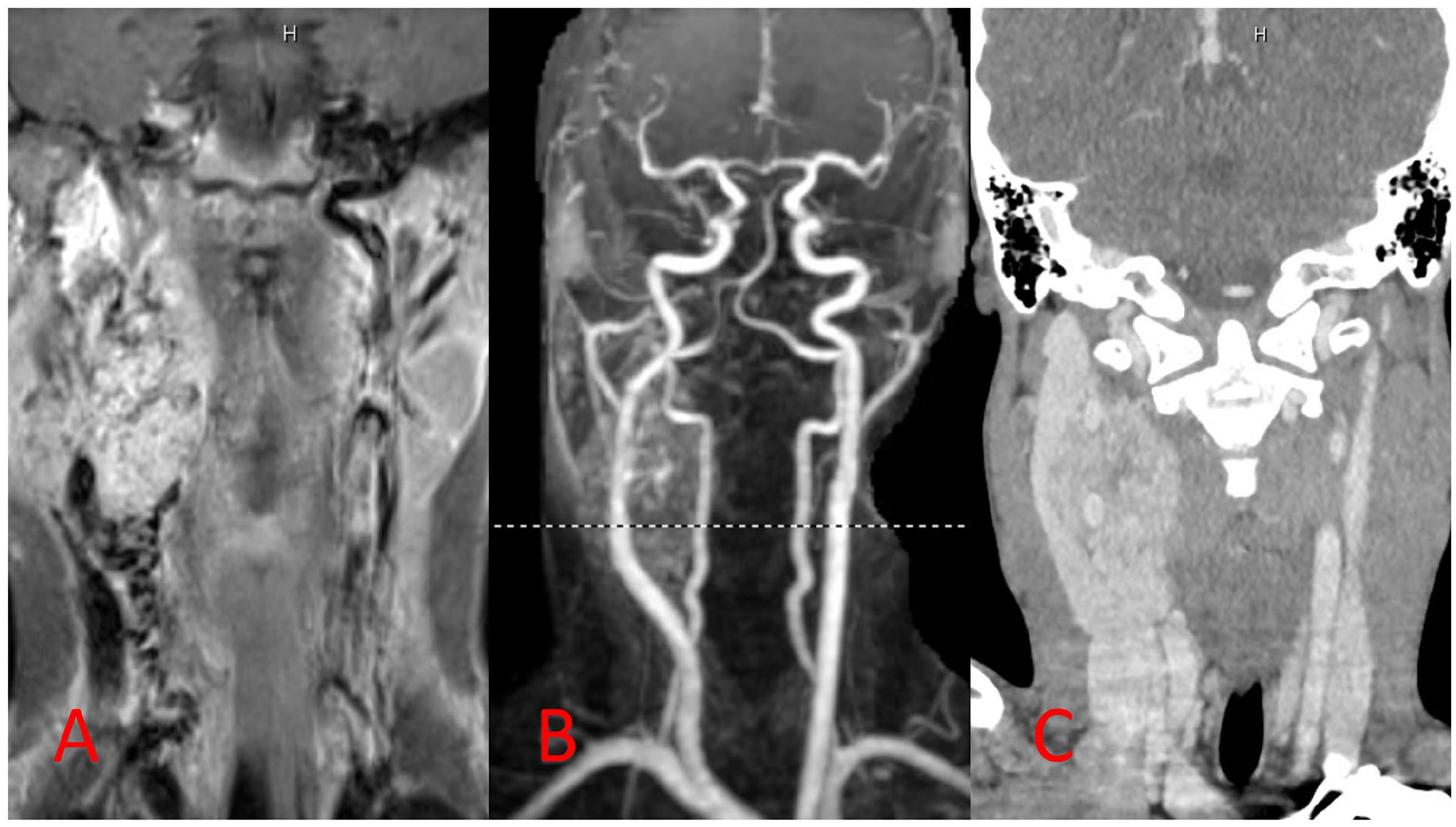
(A) T1-weighted preoperative MRI scan demonstrating a mass splaying the right carotid bifurcation. (B) Preoperative MRA scan demonstrating the same hypervascular mass. (C) Preoperative CTA (AP view) demonstrating jugular impingement by the mass.
Plastic surgery, hematology oncology, infectious disease, and neurosurgery were consulted. At this point, surgical removal and biopsy were recommended as the treatment plan, once his acute viral illness subsided.
Preoperative evaluation involved a carotid balloon occlusion test to assess perfusion from his left carotid artery, helping determine whether the right carotid artery could be safely sacrificed in case it could not be salvaged during surgery, or if shunting would be necessary. Intraoperatively, this test was performed by gaining access to the internal carotid through the Seldinger technique through the left common femoral artery. A 5-French sheath was placed, and then a multi-purpose catheter was guided through the sheath into the internal carotid artery. Once there, the patient’s sedation was lightened to obtain the baseline exam. Electroencephalogram (EEG) leads were also placed to monitor brain activity during vessel occlusion. The balloon was inflated for 15 minutes. During these 15 minutes, arterial blood pressure decreased by 20% from baseline. The patient remained neurologically stable and maintained excellent movement of the left upper and lower extremities, as well as visual fields and speech function. No facial weakness was observed. EEG also remained symmetric without evidence of a reduction in voltage or frequency. Based on these results, it was determined that the right internal carotid could be taken if needed during the carotid body tumor resection, and shunting was not necessary. The rest of the angiogram proceeded to demonstrate normal collateral flow in both the left internal carotid and vertebral arteries.
Initial plan for the resection was to attempt to dissect the tumor off the carotid while preserving it; however, if not feasible, an en bloc resection with immediate carotid reconstruction with reverse saphenous vein graft (RSVG) would be carried out. The patient also had braces placed in case partial mandibulectomy was necessary to access the internal carotid. Embolization of the tumor was not performed.
Surgical Resection
Surgical resection was carried out by the neurosurgery and plastic surgery teams. EEG leads were placed for intraoperative monitoring. A plan with anesthesia was developed to address potential rapid and large-volume blood loss. A preauricular facelift incision was made, extending curvilinearly down the neck. Initial dissection was performed anterior to the facial nerve to facilitate skin mobilization. The platysma was vertically split to preserve the marginal mandibular branch of the facial nerve.
The carotid sheath was opened, and the distended venous system was observed surrounding the vagus nerve and extending to the base of the common carotid artery, clearly identifying the tumor localization. The tumor was extremely hypervascular, and it was noted that venous structures were abnormally dilated and appeared arterialized. Dissection of the tumor away from the carotid artery revealed several areas where the tumor infiltrated the adventitia and penetrated the lateral wall of the carotid artery, consistent with Shamblin Type 3. As expected, during dissection, there was significant bleeding encountered, requiring the surgical team to gain systematic control quickly. At this point, it was clear that en bloc resection with RSVG would be necessary. Ligation of the distal external and internal carotids, along with the common carotid just 2 cm below the tumor, was performed, followed by an en bloc tumor removal. Tissue margins were sent for fresh frozen pathology; margins and 2 resected lymph nodes were noted to be clear of malignancy (Figure 2).
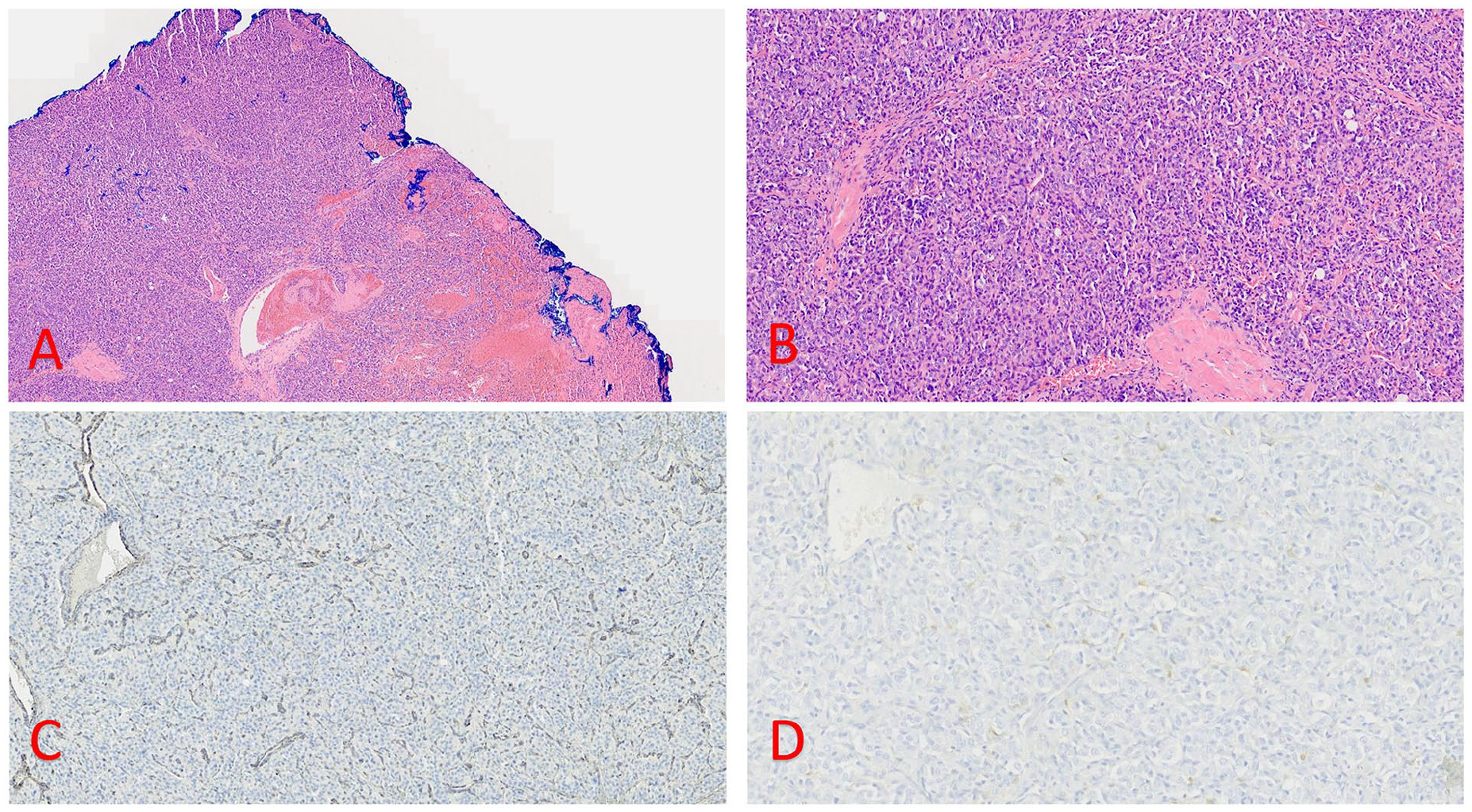
(A) 50× magnification hematoxylin and eosin (H&E) stain of the carotid body tumor. (B) 100× magnification of carotid body tumor. Note nested arrangement of epithelioid tumor cells, interrupted by pink sclerosis. (C) 200× magnification, S100 immunohistochemistry. Note staining of substentacular cells surrounding tumor nests (arrows) (D) 200×, Succinate Dehydrogenase (SDHB) immunostain. Note absence of cytoplasmic granular staining in tumor cells. Background vessels are internal positive controls.
Concurrently, the left saphenous vein was harvested for the reconstruction of the right common internal carotid artery. The RSVG was anastomosed to the distal and proximal internal carotid artery with 8-0 nylon. Sufficient flow was demonstrated on clip removal with no leaks between the anastomoses. The length of surgery was 10 hours and 2 minutes. Estimated blood loss (EBL) was 1000 mL. He was given 4400 mL of crystalloid, 3 units of packed red blood cells (PRBC), and 2 units of fresh frozen plasma (FFP). No EEG changes were noted during surgery.
Postoperative Course
The patient was stable postoperatively; however, he was kept intubated and sedated in the ICU for resuscitation due to high intraoperative blood loss. There were also concerns of nerve injury and laryngeal paralysis, as well as some lip and tongue swelling. He received neurologic examinations along with Doppler vascular checks every hour.
He was successfully extubated in the operating room on the first day after his surgery. Immediately following, however, the patient struggled with anxiety, insomnia, and refusal to eat due to fear of choking, and experienced gagging with any oral intake. He also developed right-sided Horner’s syndrome.
He had a hoarse voice and stridor consistent with vocal cord paralysis and had difficulty swallowing, requiring continuous suctioning. A nasojejunal (NJ) tube was placed to permit feeding. An otolaryngologist performed a swallow study, endoscopy, and right vocal cord injection to help reapproximate the paralyzed vocal cord for improved speech and swallowing function. Speech therapists were consulted to further assist with these difficulties. After 12 days post-op, the NJ tube was removed, and the patient progressed to more viscous oral intake. The patient was discharged home on post-op day 13.
The margins on final pathology were negative for malignancy. Genetic testing revealed that the patient had a homozygous deletion in the 11q23.1q23.2 region, which included the SDHD (succinate dehydrogenase complex subunit D) gene. Further testing was conducted on the patient’s blood, confirming a constitutional abnormality, indicating that the mutation arose within the patient’s cells rather than being inherited.
The patient followed up for 2 years with our clinic before relocating to another state. During this time, he received biannual MRIs of the neck, which showed no recurrence. (Figure 3) The patient did state some neurological symptoms, including an improving right-sided Horner’s Syndrome, occasional orthostatic hypotension, and right-sided numbness and tingling on the ear and face. The patient was managed by a multidisciplinary medical team ranging from ENT, speech therapy, ophthalmology, hematology/oncology, infectious disease, neurosurgery, and plastic surgery.
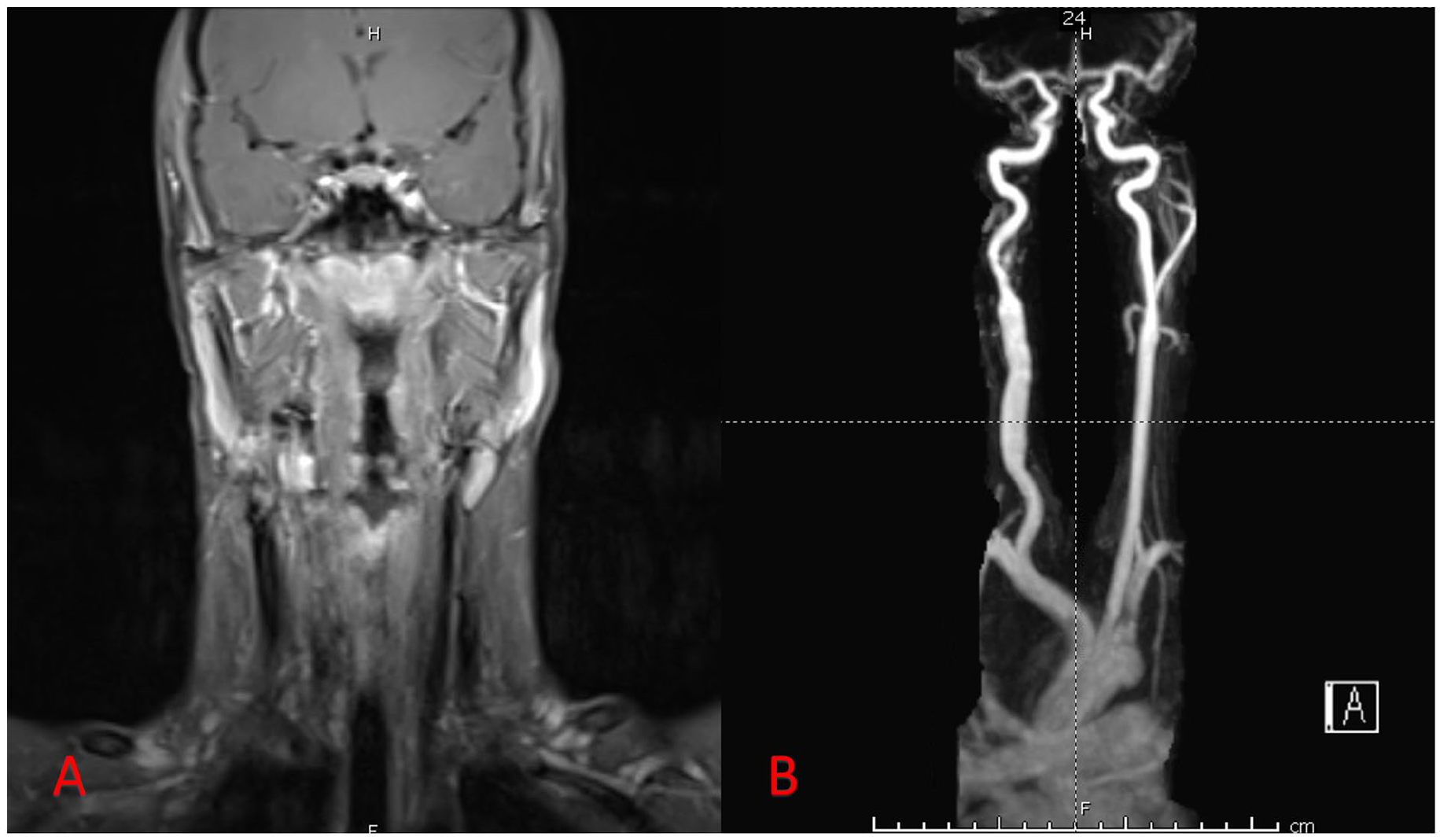
(A) T1-weighted MRI obtained 1 year after surgery. (B) Postoperative (9 months) MRA reveals adequate flow in the carotid artery reconstructed with a right saphenous vein graft.
Discussion
Carotid body tumors are uncommon in both the adult and pediatric populations; however, although rare, extra-adrenal paragangliomas are most commonly seen in the head and neck. 2 These tumors often present silently, but can be extremely dangerous due to high vascularity and proximity to critical neurovascular structures in the head and neck. 4 If not promptly treated, as might have occurred in our patient, these tumors can lead to severe complications, including vascular blowout, neurological damage, local invasion, and possibly metastatic disease requiring radiation and systemic therapy. Therefore, early surgical recognition and intervention are essential to prevent such life-threatening outcomes. 2
The etiology of CBTs is still largely unclear and is sometimes linked to genetic mutations. Some specific mutations that have been described include alterations in the succinate dehydrogenase (SDH) complex, the Von Hippel-Lindau gene (pVHL), neurofibromin-1 (NF-1), and myc-associated factor X (MAX). 4 Our patient demonstrated a homozygous deletion in the 11q23.1q23.2 region, which included the SDHD gene. He also demonstrated the absence of SDHB (succinate dehydrogenase complex iron-sulfur subunit B) staining on pathology. Both SDHD and SDHB are part of the succinate dehydrogenase complex, and loss of SDHD expression has been linked to sporadic paragangliomas. 5
Due to their rarity and silent growth, these masses are often misdiagnosed as branchial cleft cysts or lymphadenopathy, as was the case for our patient for almost 2 years.1,2,6,7 It is important to note as well that our patient was diagnosed with Mumps on initial presentation, a condition characterized by parotid gland swelling. When considering the differential diagnosis for unimmunized patients, it is crucial to include acute viral illnesses that may cause lymphadenopathy and head and neck symptoms. Additionally, there are also environmental exposures to consider. Our patient grew up in Hawaii and was continuously exposed to what they described as “volcanic smog,” which likely contributed to his symptoms of cough, congestion, and lymphadenopathy. A comprehensive review of the patient’s medical and exposure history is essential, but it is also important not to let these factors overshadow a more serious final diagnosis. Therefore, when presented with a pediatric patient with a head and neck mass, carotid body tumors should be included in the provider’s broad differential.
Clinically, carotid body tumors most commonly present as a gradually enlarging painless submandibular neck mass, typically located anterior to the sternocleidomastoid muscle at the level of the hyoid bone.1,3,4 The mass can usually be manipulated horizontally, but not craniocaudal, a phenomenon known as Fontaine’s sign. 4 In some cases, patients may complain of sensory changes such as facial numbness, pain, tingling, or tinnitus. 2 On examination, a bruit or thrill may be present, transmitting the carotid pulse, and rarely, individuals may experience flushing and palpitation indicative of a functional tumor.1,4
The most common treatment for CBTs is a total surgical resection.3,4 However, for a successful operation with minimal complications intraoperatively and postoperatively, a thorough preoperative workup and surgical plan are vital. A thorough approach includes multiple forms of imaging: ultrasound examination, CTA, and MRI. Ultrasound examination can visualize blood flow to the tumor, however, CTAs and MRI, including arterial and venous scans, provide a clearer picture of the tumor’s mass effect on surrounding vessels and size.1,4 In addition, with children, it is important to consider the potential for radiation-induced malignancies as a result of CTA, so MRI may be the safest and best option for visualization in the pediatric population. 8 Blood workup and urinary metanephrines can also help confirm the diagnosis if the tumor is functional. 2 Other preoperative tests include an angiogram and balloon occlusion test. An angiogram can help assess collateral circulation, and a balloon occlusion test is crucial for evaluating the safety of total en bloc resection. This test is particularly important in cases where intraoperative findings suggest that the tumor may be inseparable from the blood vessels. In the present study, obtaining thorough imaging helped characterize the tumor and plan for resection and potential revascularization, however, the tumor infiltration into the adventitia could not be adequately visualized until the carotid sheath was opened intraoperatively.
Surgical resection remains the gold-standard treatment for CBTs, though it can be more challenging in children due to the thinner intima and media of their blood vessels. 1 The goal is subadventitial resection of the tumor to minimize unnecessary vascular disruption and avoid the need for revascularization. However, in cases where the tumor completely encases the artery, classified as Shamblin Type III, a total en bloc resection may be necessary, potentially requiring revascularization of the external or internal carotid artery, or both. In our patient’s case, reconstruction of the internal carotid artery was required, and we used the left saphenous vein. This approach is common for vascular reconstruction and offers the added benefit of growth potential in children.1,8 Alternatively, polytetrafluoroethylene (PTFE) grafts have been employed in adults.
Vascular reconstruction after CBT resection has been associated with increased operative blood loss. Preoperative embolization, a procedure that reduces blood supply to the tumor, has been widely debated in the literature for both adult and pediatric populations. Some authors have described it to be safe in children and successful in decreasing EBL and postoperative mortality.1,4 However, the same authors cautioned its use as embolization may make the surrounding tissues friable and inflamed, making the resection more difficult or may increase the risk of cerebral embolization and stroke. We elected not to proceed with embolization. Intraoperatively, we encountered increased bleeding initially as we attempted to preserve the carotid artery. However, given the tumor’s vascularity, associated blood loss, and the elevated risk of recurrence, we ultimately opted to convert to an en bloc resection. En bloc resection without embolization was deemed preferable, provided that the balloon occlusion study was favorable for prolonged carotid cross-clamping without need for shunting.
Several complications frequently arise after CBT resection, including vocal cord paralysis, Horner’s syndrome, orthostatic hypotension, pain, tongue paresis, hearing loss, hoarseness, and dysphagia. These complications are typically caused by mass effect on cranial nerves, cranial nerve injury during surgery, or carotid body autoregulation. They can be temporary, though some cases have reported permanent cranial nerve palsy.1,2,7 In our case, the patient experienced vocal cord paralysis and dysphagia after surgery. To address his dysphagia and nutritional deficits, an NJ tube was placed, which allowed him to gradually progress to more viscous oral intake after 2 weeks. Additionally, he underwent a right vocal cord injection, resulting in improved speech and swallowing function. The patient also developed right-sided Horner’s syndrome, likely due to mass effect, which showed improvement at his last visit. A multidisciplinary team, including ENT, speech therapy, ophthalmology, neurosurgery, anesthesia, hematology/oncology, and plastic surgery, was essential to take optimal care of our patient preoperatively, intraoperatively, and postoperatively.
Conclusion
Carotid body tumors should be considered in the differential diagnosis when evaluating a pediatric patient with a slow-growing neck mass. Effective management starts with early identification and involves a comprehensive assessment, including a detailed physical exam, various imaging modalities, and a well-prepared surgical plan with a multidisciplinary team.
Supplemental Material
sj-docx-1-fac-10.1177_27325016251352084 – Supplemental material for Carotid Body Tumor in a 14-Year-Old Unimmunized Patient
Supplemental material, sj-docx-1-fac-10.1177_27325016251352084 for Carotid Body Tumor in a 14-Year-Old Unimmunized Patient by Yasmine Soubra, Hung B. Le, Sarah A. Frommer, Steven L. Henry, Patrick D. Combs and Patrick K. Kelley in FACE
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Considerations
Ethical approval was not required for this study.
Informed Consent
Consent to participate in research was obtained from the patient and his family. Consent for publication was obtained from the patient and his family.
Data Availability Statement
Data files are stored in a password-protected cloud source.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
