Abstract
Emphasis in ASB reconstruction is placed on achieving a watertight dural closure, providing structure for cranial and orbital contents, obliterating dead space, and separation between cranial and sinonasal contents in order to maintain a functional sinonasal system. Pediatric ASB reconstruction also requires consideration for future craniofacial skeletal growth, donor-site morbidity, and psychosocial impact when selecting a reconstructive donor site. Large defects (>3 cm) have traditionally been repaired with free flaps. We describe the successful use of bilateral temporoparietal fascia flaps for a large anterior skull base defect in a pediatric patient after tumor resection. Utilizing bilateral, stacked temporoparietal fascia flaps, which are regionally close to the ASB, avoids morbidity from both extraneous incisions and free flap transfer. The authors show that temporoparietal fascia flaps provide enough tissue bulk and vascularity for safe and effective use in pediatric ASB reconstruction.
Introduction
Pediatric skull base tumors are overall rare and vary widely in pathology, with approximately half of cases being malignant.1-7 Surgical resection of these tumors leaves variable ablative defects. 1 Goals of reconstruction include providing structural support for the cranial and orbital contents, obliterating dead space, providing a watertight dural seal, separation between cranial and sinonasal contents, and maintaining a functional sinonasal system. Failure to do so may result in consequences such as CSF leak, infection, or functional deficit.
Oncoplastic reconstruction of these defects is particularly challenging in a pediatric population, given their overall small anatomical structure size and potential disruption in sinonasal and skull base development.2,7-9
Anterior skull base defects can be accessed either through a traditional open approach or endoscopically through an endonasal approach (EEA). EEA has been shown to be a safe and efficacious option for complex skull base tumors in both adults and children, with a lower rate of morbidity compared to the open approach. 10 The endoscopic endonasal approach utilizes local intranasal flaps for reconstruction, with the nasoseptal flap being the most common.9,11-13 Open approach is indicated for large central anterior skull base ablative defects, and in irradiated or compromised nasopharyngeal donor sites which may render local nasoseptal flaps unreliable.9,14,15 In such cases, open approach with extranasal flaps remain the standard. 8
Extranasal flaps may include either locoregional flaps or microvascular free tissue flaps. Traditional regional flaps for anterior skull base reconstruction include the pericranial flap for central defects, and temporoparietal flaps for more lateral defects.6,9,16,17 The use of a TPF flap has also been described in anterior skull base reconstruction as both a tunneled flap in endoscopic skull base reconstruction and in the form of a free flap. 1 To our knowledge, this paper reports the first pediatric case of open anterior skull base reconstruction utilizing bilateral pedicled temporoparietal fascial flaps for large defect anterior skull base reconstruction.
Case Presentation
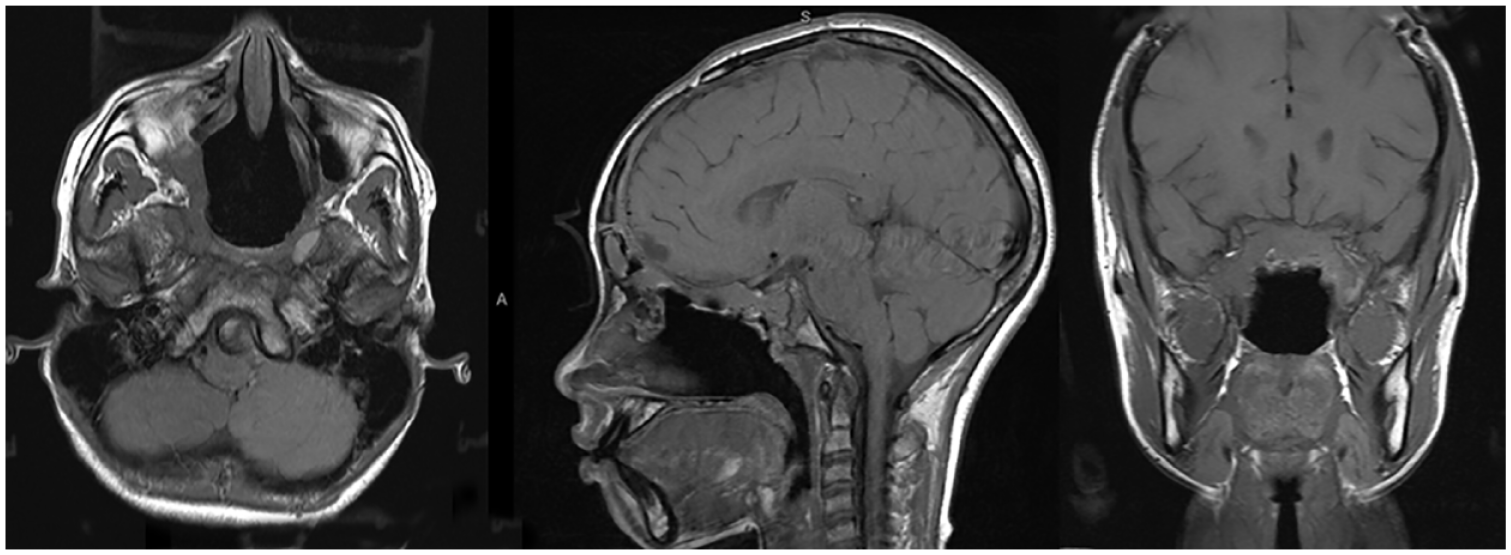
A 12-year-old male presented with complaints of progressive vision loss, bilateral proptosis, ocular motility deficits, and congestion. Magnetic resonance imaging (MRI) revealed a large anterior skull base heterogeneous mass (5.4 × 6.9 × 5.8 cm) arising from the sphenoid and central skull base (Figure 1). The mass displayed suprasellar extension and involved the ethmoid air cells and nasal cavity. The midline retrobulbar tumor resulted in bilateral orbital compression and optic neuropathy. The patient was taken to the operating room by otolaryngology for biopsy and nasal endoscopy which revealed a large mass medial to the right middle turbinate, with left septal deviation that caused complete obstruction of the left nasal cavity. Biopsy confirmed the diagnosis of a juvenile ossifying fibroma.
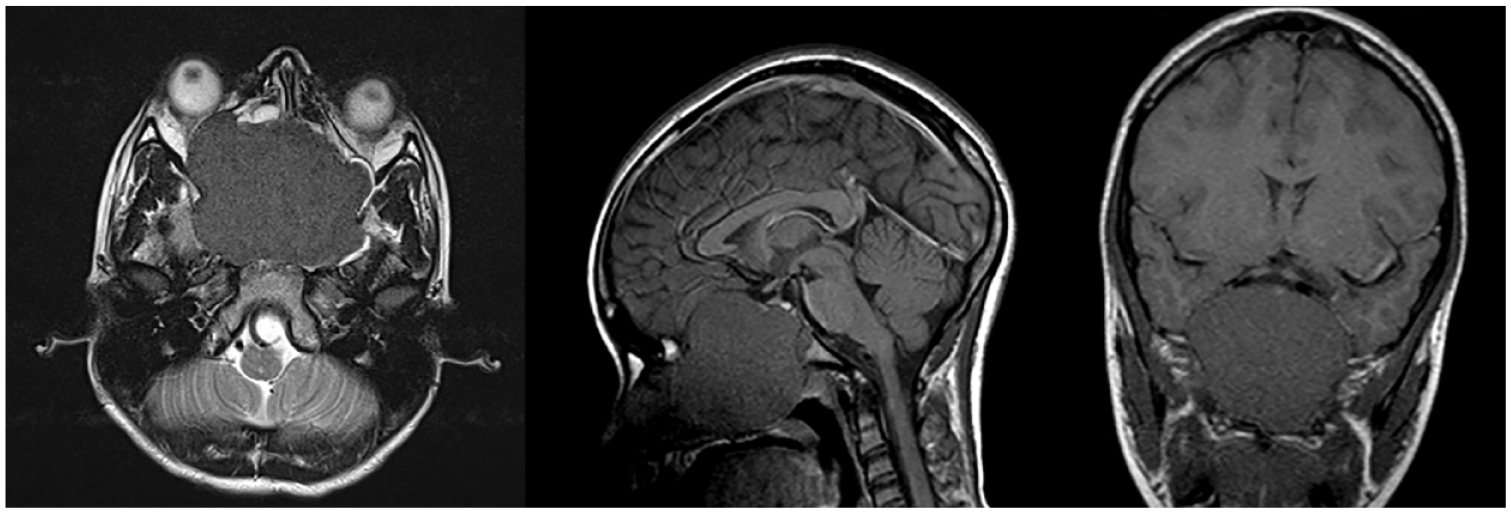
MRI revealing 6.4 × 6.7 × 6.8 cm mass at midline and paramedian sinonasal/frontal skull base, involving the ethmoid air cells and nasal cavity.
A multidisciplinary team from neurosurgery, otolaryngology, oncology, and plastic surgery services was involved in the surgical management of the case. The patient underwent a 3-staged surgical approach for tumor resection and reconstruction.
Stage I Resection
Stage I resection was performed in piecemeal fashion by otolaryngology via a combined endoscopic endonasal and Caldwell-Luc approach. Post-stage I MRI revealed a residual intracranial tumor measuring 5.4 cm AP by 6.2 cm transverse by 3.9 cm craniocaudal dimension. The residual tumor remained in the central skull base and involved the cribriform plate, fovea ethmoidalis, planum sphenoidale, basisphenoid, and the floor of the sella turcica (Figure 2).
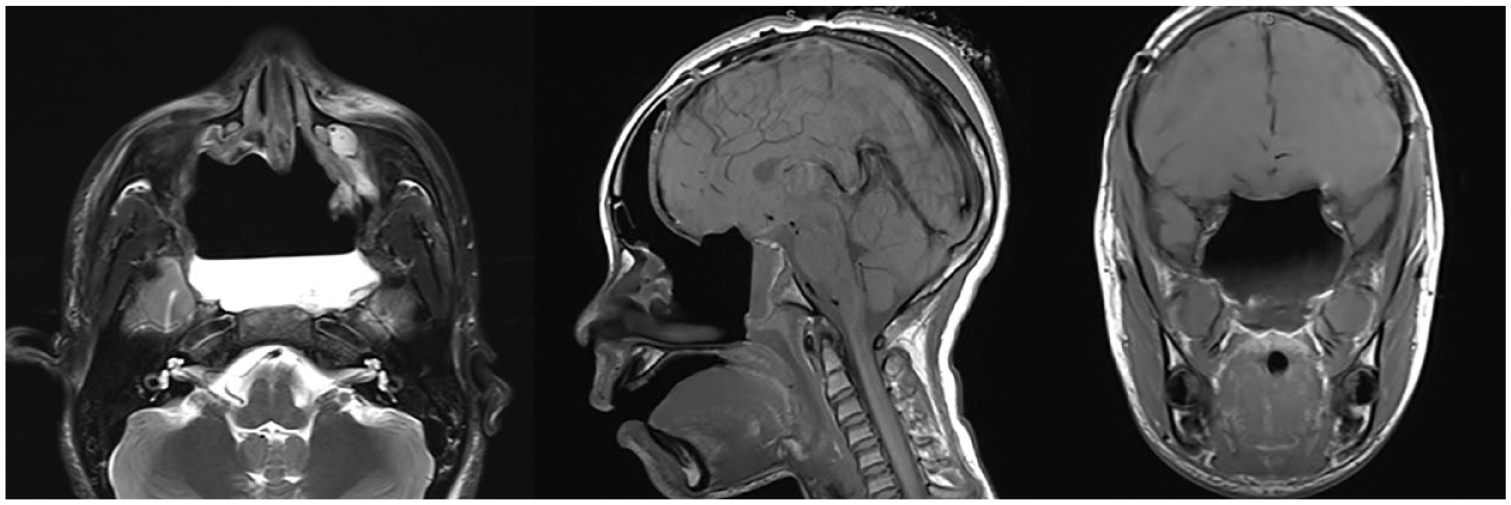
MRI brain revealing complete resection of the central/anterior skull base lesion. There is postoperative epidural pneumocephalus along the anterior cranial fossa with small edema/contusion in the right frontal lobe.
Stage II Resection
Stage II intracranial resection via bifrontal craniotomy by neurosurgery was performed 8 days following stage I of the resection. Prior to resection, a lumbar drain was placed. Final reconstruction was not planned during this stage in order to first confirm gross total resection on MRI (Figure 2). However, plastic surgery was present during this stage to assist in neurosurgical exposure while preserving the bilateral temporoparietal flaps (TPFF) for final reconstruction. A bicoronal incision was created and dissection was carried out just deep to the subcutaneous tissue—different from the more typical subgaleal plane—leaving down the galea and temporoparietal fascia for later elevation as TPFFs. One large TPFF was raised by careful dissection of the loose areolar tissue between the pericranium and galea centrally, and superficial and deep temporal fascia laterally. Dissection in this plane was carried anteriorly, careful to avoid injury to the superficial temporal pedicle and frontal branch of the facial nerve bilaterally. After elevation, the large flap was split at the midline, yielding 2 TPFFs (Figure 3).
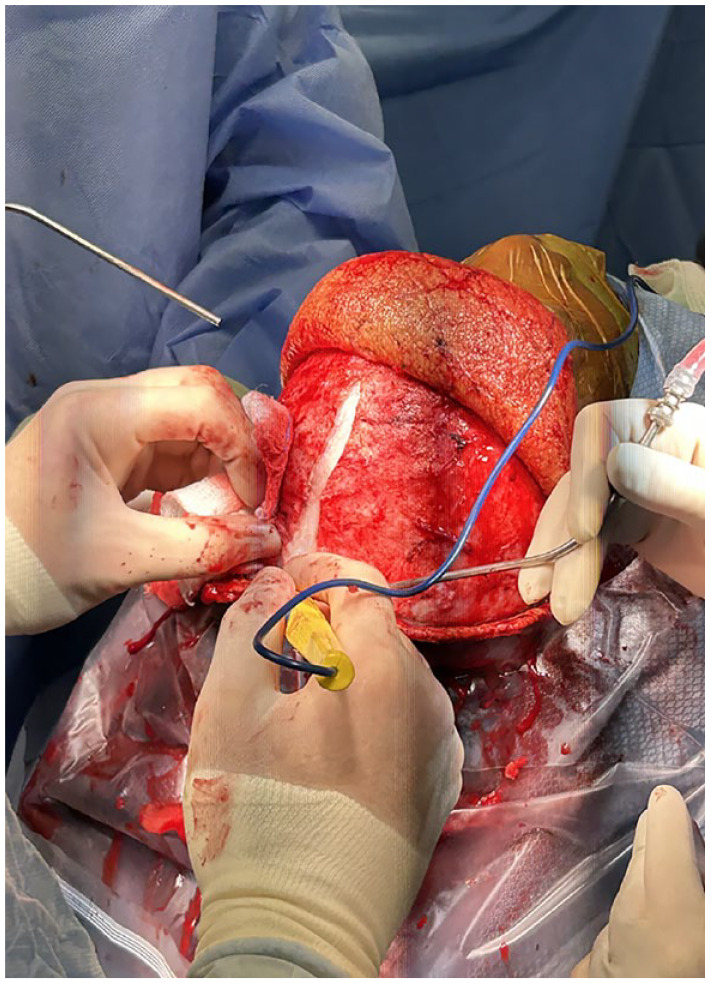
TPFF divided at the midline to create 2 temporoparietal fascia flaps.
Neurosurgery then proceeded with surgical resection of the remaining mass. Bifrontal craniotomy was performed and the orbital bar was removed. Dura was dissected from the skull base, resulting in a small, low flow dural tear which was repaired primarily. At the level of the clivus, the dura was found to be invaded, which required excision. This resulted in an additional low-flow CSF leak, which was repaired utilizing DuraGen. Finally, the remaining portion of the tumor was dissected off the nasopharynx to achieve complete macroscopic resection of the tumor. Temporary closure was performed with the intent of confirming gross total resection (GTR) on postoperative MRI. The TPFFs were then laid back into their anatomical position for later use during final reconstruction, and the skin was closed with placement of a 7F size Jackson-Pratt (JP) drain.
Stage III Reconstruction
MRI confirmed GTR of the lesion, and reconstruction of the anterior skull base defect commenced the following day. The bicoronal incision was reopened, flaps re-elevated, and orbital bar removed to expose the dura and skull base defects. The cavity was inspected under a surgical microscope and the DuraGen patch was confirmed to be intact. For further dural reinforcement, the posterior edges of both TPFFs were cut as free grafts and laid over the 2 dural defects at the clivus and cresta galli. The TPFFs were then further released to allow additional advancement and rotation into the defect (Figure 4). The flaps were then inset to cover the exposed dura at the tuberculum sella, down to the nasopharynx. The inferior tip of a single TPFF was sutured to the posterior pharynx to achieve full coverage of the exposed dura at the skull base. The remaining TPF flap was secured on top of the first TPF flap (Figure 5). Two lateral openings were burred in the craniotomy bone flap to allow for tunneling of the flaps into the intracranial space without compression of the pedicle. The bone flaps were then fixed using plates. Finally, nasal packing with Merocel was performed endoscopically after confirmation of complete separation of sinus and intracranial space by the TPF flaps. A 7F drain was placed beneath the scalp. The scalp was then closed with 3-0 vicryl and 3-0 plain gut.
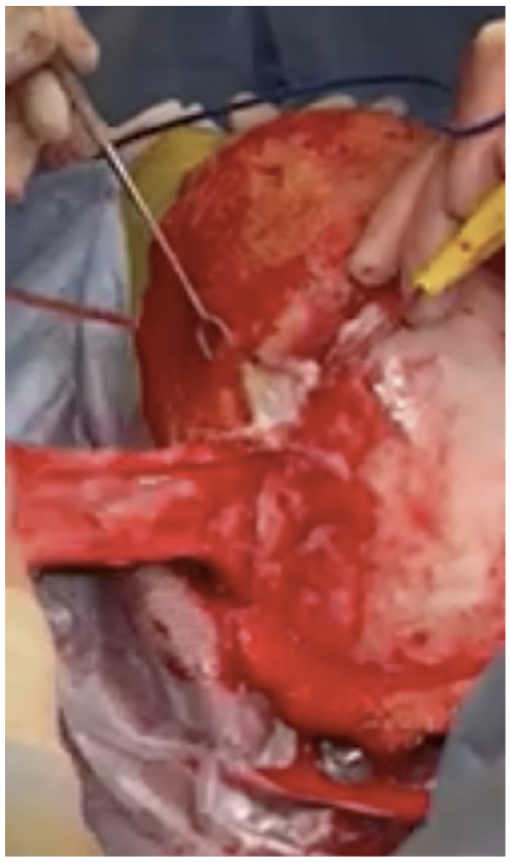
Further elevation and advancement of the left TPFF.
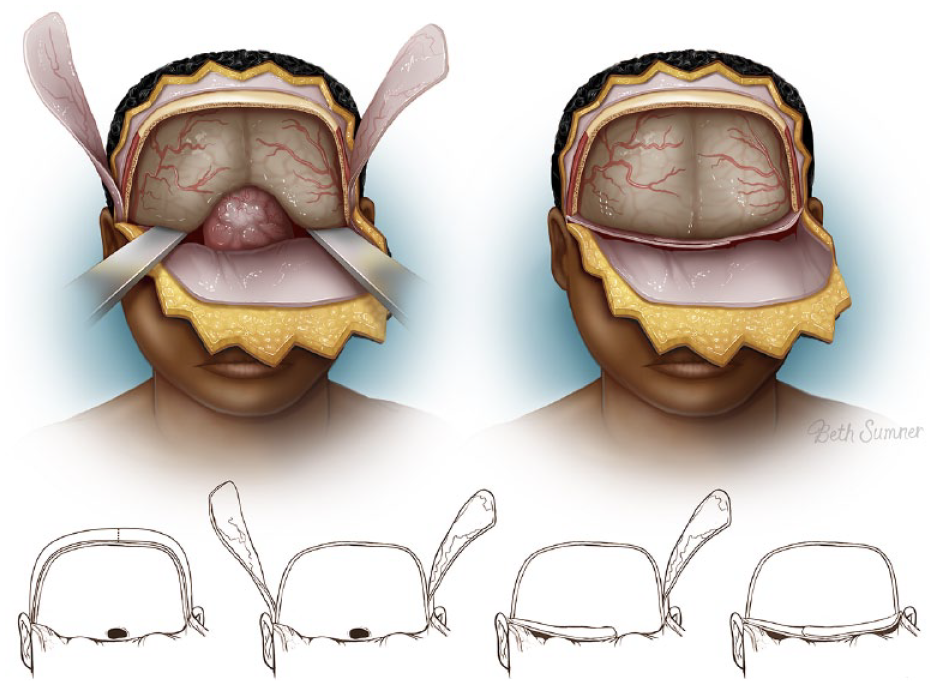
Bilateral temporoparietal fascia flap inset to the anterior skull base defect. Printed with permission from Texas Children’s Hospital.
Written informed consent for use of patient information, imaging, and pictures was provided by the patient’s legally authorized representative.
Outcome
The postoperative course was uncomplicated. The lumbar drain (LD) was removed 6 days after final reconstruction, and nasal packing was removed on the seventh day without evidence of CSF leak. The patient was discharged 8 days after final reconstruction. Otolaryngology nasal endoscopy follow-up 4 weeks postoperatively revealed a well healing surgical site without evidence of leak. MRI brain was repeated 2 months post-operatively (Figure 6).
MRI 2 months postoperatively.
Discussion
Juvenile ossifying fibroma (JOF) is an aggressive, non-malignant tumor that affects the anterior skull base and surrounding structures such as the maxilla, sinonasal cavity, and orbit, with a mean age of onset of 11 years old. 18 JOF enacts painless, rapid growth in a short amount of time which may lead to disruption of vital surrounding structures such as the globe. 18 In addition to aggressive tumor behavior, JOF has a high rate of recurrence, necessitating aggressive radical resection.18,19 Anterior skull base tumor resection has been successfully completed with both open and endoscopic approaches. Approach of resection and reconstruction should be tailored to the size and structures involved.20-22
The primary goals of ASB reconstruction include providing structural support for the cranial and orbital contents, obliterating dead space, providing a watertight dural seal, separation between cranial and sinonasal contents, and maintaining a functional sinonasal system.1,5,16 Bony reconstruction has historically been debated in skull base reconstruction. 16 Proponents for bony reconstruction cite the need of structural support for the cranial contents to prevent herniation. However, many studies have shown that the bony skull base defect has no functional or esthetic consequence for the patient, and that the introduction of free bony grafts or synthetic material has the potential to interfere with healing. 16
In ASB, reconstructive emphasis should be placed on a water-tight dural closure, as this region is much more likely to leak than other intracranial areas postoperatively.5,16,23 As such, ineffective reconstruction can result in serious complications such as cerebrospinal fluid (CSF) leak, meningitis, brain herniation, and tension pneumocephalus.1,24 ASB in a pediatric population also requires special considerations for future craniofacial skeletal growth, donor-site morbidity, and psychosocial impact. 1
Traditional means of ASB repair are generally based on defect size. 21 Small ASB defects (<1 cm) are repaired with a multilayer onlay graft utilizing non-vascularized tissue. Autologous graft sources used for small defects include fascia lata, temporalis fascia, and autologous fat. 21 Non-autologous grafts can also be used such as cadaveric fascia lata, lypophilized dura, and cadaveric acellular dermis (Alloderm; LifeCell Corp. Woodlands, Texas, USA).2,21,25,26 Medium defects (1-3 cm) are typically repaired with locoregional pedicled flaps, with the specific flap choice depending on location of the ASB defect and approach utilized. If resection is performed solely by endoscopy, medium defects are repaired with nasoseptal or middle turbinate flaps. In an open approach, central defects are traditionally repaired with pericranial flaps, while lateral defects are repaired with temporoparietal fascia and temporalis muscle flaps. Large defects (>3 cm) reconstructed with free flaps or regional myocutaneous flaps. 21
While the temporoparietal fascia flap (TPFF) has been traditionally described for use in smaller lateral ASB defect reconstruction, our case demonstrates the successful use of bilateral TPFFs for a large central ASB defect in a pediatric patient undergoing open ASB reconstruction. By utilizing 2 temporoparietal pedicled flaps, there is enough reach and arc of rotation to both cover the large ASB defect and afford a stacked, multilayer closure for additional protection against CSF leak. Use of the TPFF does not require overlying skin grafts for coverage of the nasopharyngeal space and will mucosalize within days of surgery. 1 The TPFF also carries the advantage of residing distant from the defect site and is therefore reliably available following resection. 8 Compared to the traditional pericranial flap, bilateral TPFFs provides superior bulk with a robust axial blood supply. Compared to a free flap, it provides the same utility without the increased operative time, blood loss, and postoperative hospital stay. 5 Further, free muscle flaps are often bulky and can inadvertently mask local tumor recurrence on radiology, which is not seen with low-bulk fascia-only flaps. 2 Although it did not reach statistical significance, Yano et al reported that using free flaps for pediatric patients tended to result in a higher postoperative complication rate than locoregional flaps, and therefore advocated for locoregional flap use first, if large enough to reach, in pediatric skull base reconstruction. 5
Conclusion
In this case, the authors demonstrate that bilateral temporoparietal fascia flaps are a safe and efficacious option for large anterior skull base defects in the pediatric population.
Supplemental Material
sj-docx-1-fac-10.1177_27325016231220621 – Supplemental material for Pediatric Anterior Skull Base Reconstruction With Bilateral Temporoparietal Fascial Flap
Supplemental material, sj-docx-1-fac-10.1177_27325016231220621 for Pediatric Anterior Skull Base Reconstruction With Bilateral Temporoparietal Fascial Flap by Anna J. Skochdopole, Heather R. Burns, Ammar Hashemi, Nicholas H. Yim and Edward P. Buchanan in FACE
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Informed written consent was obtained by the patient.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
