Abstract
Objective:
We present a case series of 25 pediatric patients who underwent maxillary and mandibular oncoplastic reconstruction at a single institution. Further, we discussed the present review of the literature and underscored the unique considerations for the pediatric population.
Methods:
A retrospective chart review of 25 pediatric oncoplastic patients undergoing maxillary and/or mandibular reconstruction was performed from March 2015 to January 2022.
Results:
Thirteen patients underwent maxillary reconstruction, and 12 patients underwent mandibular reconstruction. The mean age of the maxillary cohort was 10.2 years, and the mean age of the mandibular cohort was 9.8 years. The age range of patients followed was from 6 months to 18.8 years. All patients displayed satisfactory postoperative swallowing and facial symmetry at subsequent follow-up visits.
Conclusion:
Maxillary and mandibular reconstruction following oncologic resection requires special attention in the pediatric population because of the difficulty to predict bony growth of the mid and lower face, and the resultant effects on dentition and symmetry. Our case series demonstrates successful pediatric maxillomandibular reconstruction utilizing microvascular free tissue transfer and 3D virtual surgical planning.
Introduction
While pediatric craniofacial tumors are less common than in their adult counterparts, the tumors that do manifest are often destructive to surrounding hard and soft tissues of the face, carrying the potential to create significant deformity in the child.1,2 Oncologic reconstruction in children is particularly challenging due to the consideration of future skeletal growth, smaller donor sites, and concern for donor site morbidity in a developing child.1-3 Maxillary and mandibular involvement portends further challenges, as these structures are vital supports for dentition, oral competence, and facial symmetry.1-3 The maxilla undergoes anteroposterior and vertical expansion up until skeletal facial maturity, which takes place between ages 15 and 17.1,4 Full mandibular length is achieved between 10 and 12 years old in girls and 12 to 14 years old in boys. 5 The maxilla, mandible, and basal skull develop synergistically, so disruption of any of these structures influences the development of the other 2, making operative precision paramount in these patients.1,6
Different reconstructive approaches have been proposed, such as primary reconstruction, where the defect is repaired immediately following tumor resection, versus delayed secondary reconstruction, in which reconstruction is staged at a later date from resection.7-9 The use of neoadjuvant chemotherapy and/or radiotherapy may pose additional risks to the reconstructive wound bed. Our center has developed simple algorithmic approaches to maxillary and mandibular reconstruction based on the size of the defect and age of the patient, which will be described in detail. This study presents a large case series and discusses important reconstructive considerations when treating children with maxillary and mandibular extirpative defects.
Methods
A retrospective chart review of patients who underwent maxillary or mandibular reconstruction following extirpative surgery from March 2015 to January 2022 was performed. Patients with post-ablative defects that spared the palate were excluded. During the allotted period, a total of 25 patients were identified, with 12 mandibulectomy and 13 maxillectomy reconstruction patients. The average age at time of reconstructive surgery was 9.6 (range 6 months-18.8 years) years old.
Data collected included patient demographics, tumor etiology, use of neoadjuvant/adjuvant chemotherapy and/or radiotherapy, defect characteristics, reconstructive modalities used, postoperative complications, and surgical outcomes. Maxillectomy procedures were characterized based upon resection of the hard palate and surrounding structures such as the zygoma, orbital floor, and maxillary alveolus. Mandibulectomy procedures were classified as complete if the condyle was removed, and subtotal if the condyle was spared. All the data collected is shown in Tables 1 and 2.
Clinical Characteristics, Interventions, and Reconstructive Outcomes of Patients Undergoing Oncoplastic Palatal Reconstruction.
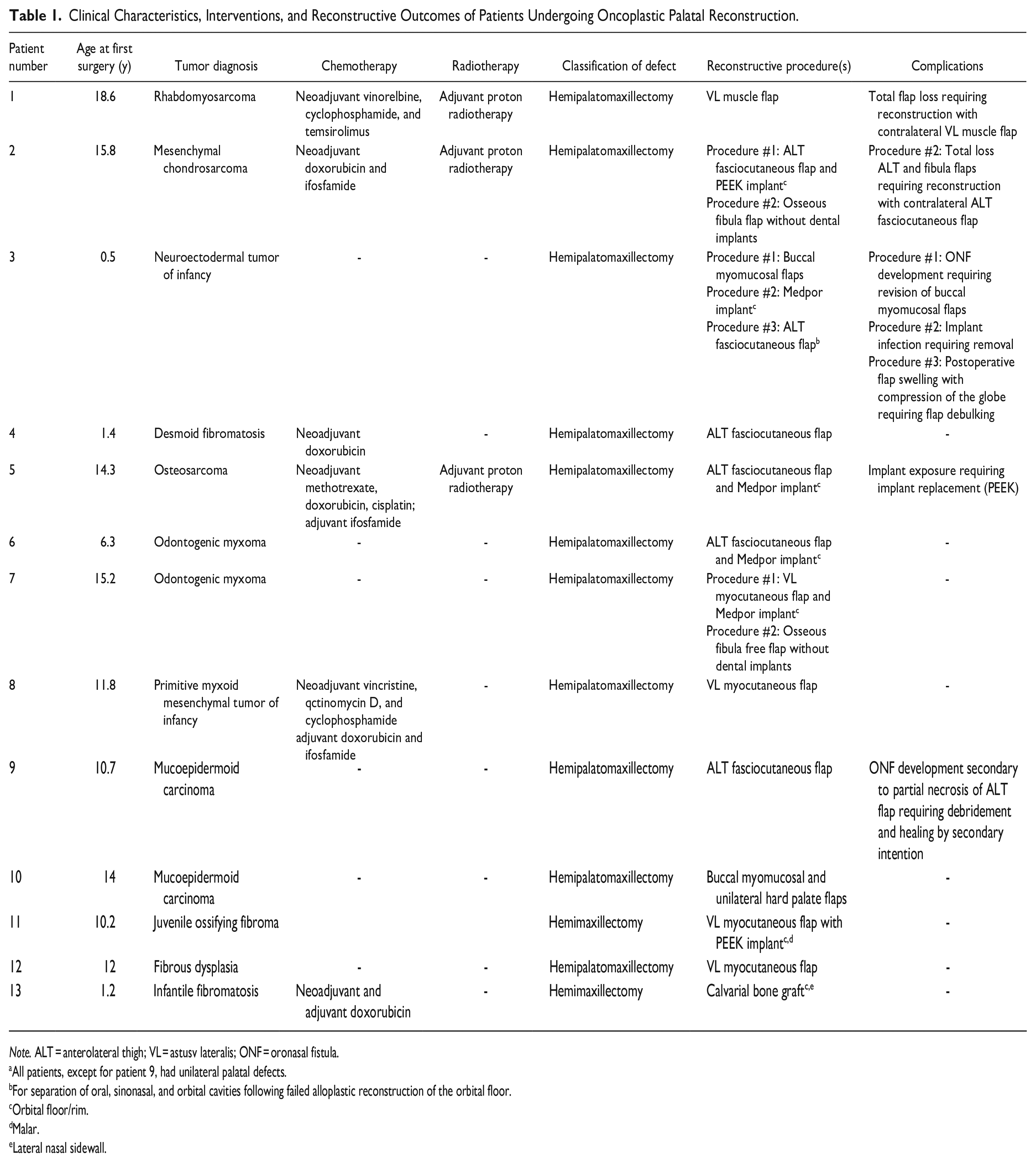
Note. ALT = anterolateral thigh; VL = astusv lateralis; ONF = oronasal fistula.
All patients, except for patient 9, had unilateral palatal defects.
For separation of oral, sinonasal, and orbital cavities following failed alloplastic reconstruction of the orbital floor.
Orbital floor/rim.
Malar.
Lateral nasal sidewall.
Clinical Characteristics, Interventions, and Reconstructive Outcomes of Patients Undergoing Oncoplastic Mandibular Reconstruction a .
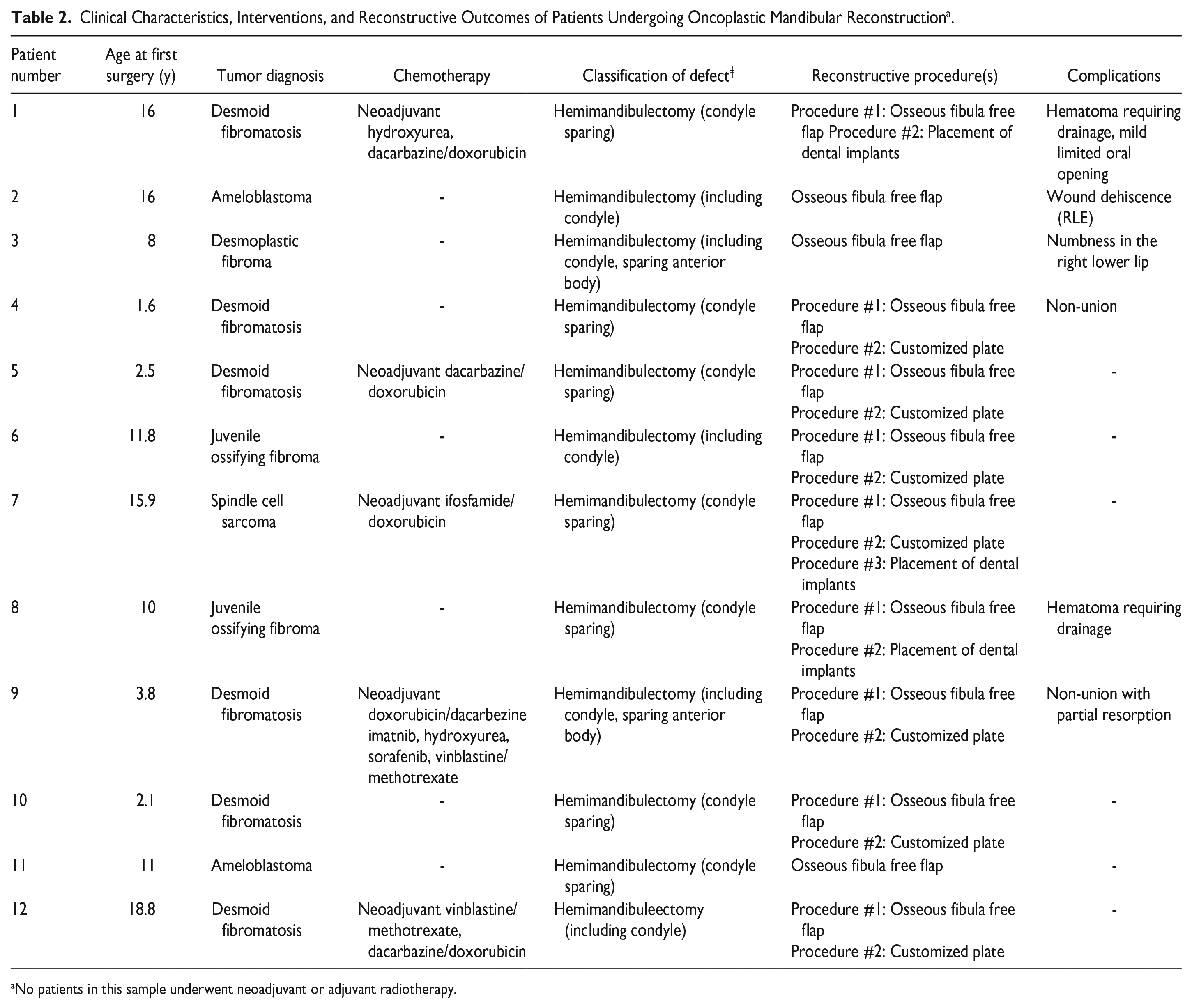
No patients in this sample underwent neoadjuvant or adjuvant radiotherapy.
Virtual Surgical Planning (VSP)
Craniofacial surgeons and representatives from Depuy Synthes (Johnson & Johnson, Warsaw, IN) collaborated to formulate a virtual surgical plan for all patients undergoing mandibular reconstruction. In these planning sessions, CT scans are sent for rendering using 3D, virtual surgical planning software. Critical nerves and blood vessels are identified at both the recipient and donor sites and a three-dimensional reconstruction of the patients’ fibula is superimposed onto the mandibular defect. Fibular osteotomies are planned to best recreate the shape and angles of the patient’s native mandible. The goal is to optimize the number and cutting plane of the osteotomies, bone-to-bone contact, and fibular segment lengths. Mandibular models, cutting guides for the mandible and fibula, and plate-bending templates are manufactured according to the specifications defined by each planning session.
Results
Patient demographics—The mean follow-up period was 28.5 months. A total of 13 maxillary reconstruction patients with a mean age of 10.2 years were included in the study. A total of 12 mandibular reconstruction patients with a mean age of 9.7 years were included in the study. Underlying maxillary pathologies included fibrous dysplasia, mucoepidermoid carcinoma, juvenile ossifying fibroma, desmoid fibromatosis, osteosarcoma, rhabdomyosarcoma, odontogenic myxoma, mesenchymal chondrosarcoma, neuroectodermal tumor of infancy, and primitive myxoid mesenchymal tumor of infancy. Mandibular pathologies included desmoid fibromatosis, ameloblastoma, juvenile ossifying fibroma, and spindle cell sarcoma.
Tumor Diagnosis and Neoadjuvant/Adjuvant Chemotherapy and Radiotherapy
Of the maxillary patients (n = 13), 5 patients (38.5%) had a tumor of fibrous etiology (desmoid fibromatosis, infantile fibromatosis, juvenile ossifying fibroma). Two patients (15.4%) had mucoepidermoid carcinomas, 2 patients (15.4%) had odontogenic myxomas, 2 patients (15.4%) had tumors of mesenchymal etiology (primitive myxoid mesenchymal tumor of infancy, osteosarcoma), 1 patient (7.7%) had a neuroectodermal tumor, and 1 patient (7.7%) had a recurrent rhabdomyosarcoma. Six patients received neoadjuvant chemotherapy (Table 1). Three patients underwent adjuvant chemotherapy, and 3 patients received proton radiotherapy. Of the 13 maxillary reconstructions, 3 (25%) had defects isolated to the hard palate. Five (41.7%) had defects involving the maxillary arch and dentition. The average maxillary post-extirpative defect size was 5.7 cm (range 12-2.5 cm).
Of the mandibular patients (n = 12), 7 patients (58.3%) were diagnosed with a desmoid fibroma or desmoplastic fibromatosis, 2 patients (16.7%) had a juvenile ossifying fibroma, 2 patients (16.7%) had an ameloblastoma, and 1 patient (8.3%) had a spindle cell carcinoma tumor. Four patients received neoadjuvant chemotherapy (Table 2). None of the mandibular patients received radiotherapy. The average mandibular post-extirpative defect size was 6.8 cm (range 9.6-3.1 cm).
Types of Reconstruction
Maxillary—Primary separation of the oral and sinonasal cavities was achieved using free tissue transfer (FTT) in 10 patients (76.9%) with the remainder undergoing buccal myomucosal flaps in 2 patients (15.4%) or calvarial bone graft reconstruction in 1 patient (7.7%) (Figure 1). For FTT, a vastus lateralis myocutaneous flap was used in 4 patients, anterolateral thigh (ALT) flap in 6 patients, and a vastus lateralis (VL) muscle only flap in 1 patient. Two patients underwent staged reconstruction (one ALT flap, 1 VL myocutaneous flap) with an osseous fibula free flap without dental implants. Six patients (46.2%) underwent additional orbital floor and rim reconstruction, using a Medpor (Stryker Corporation, Kalamazoo, MI) implant in 4 patients (30.8%) and polyether ether ketone (PEEK) implant in the other 2 (15.4%) (Figure 2) (Table 1).
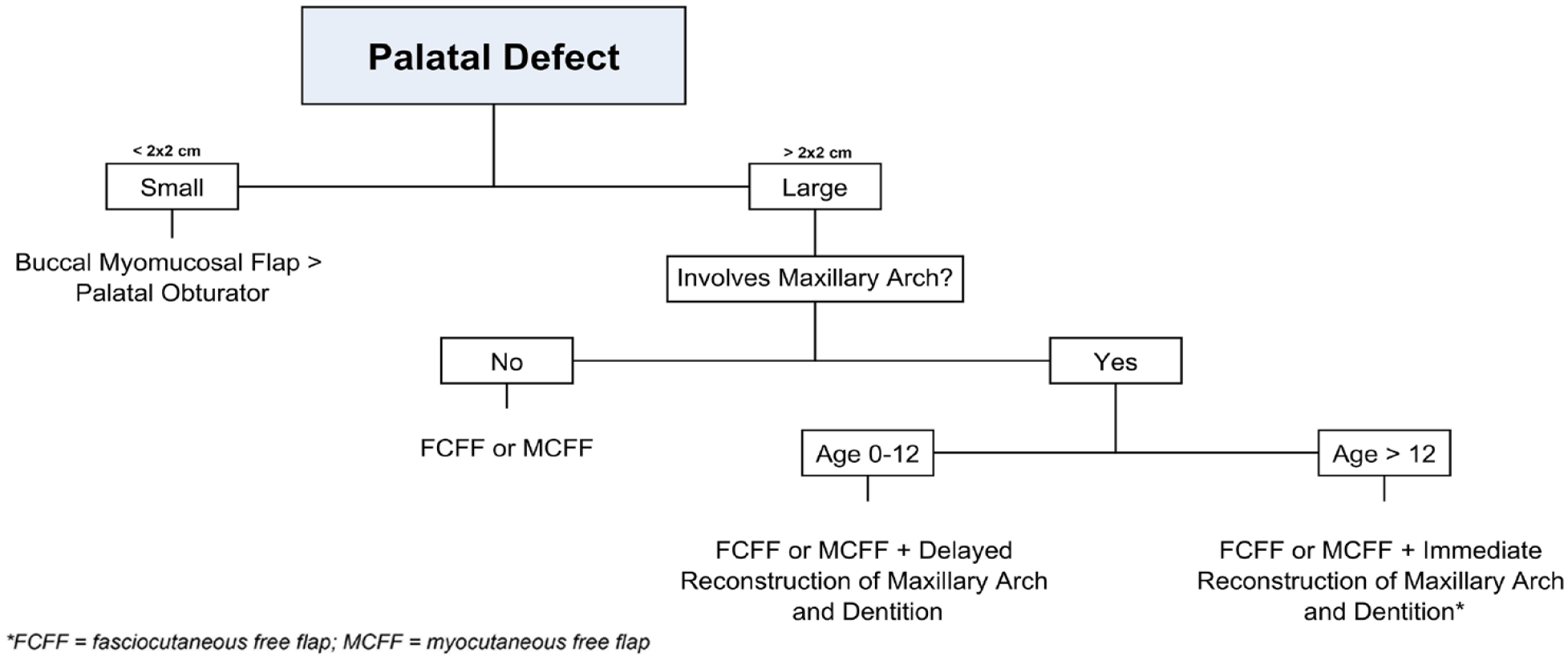
Algorithmic approach to maxillary defects.
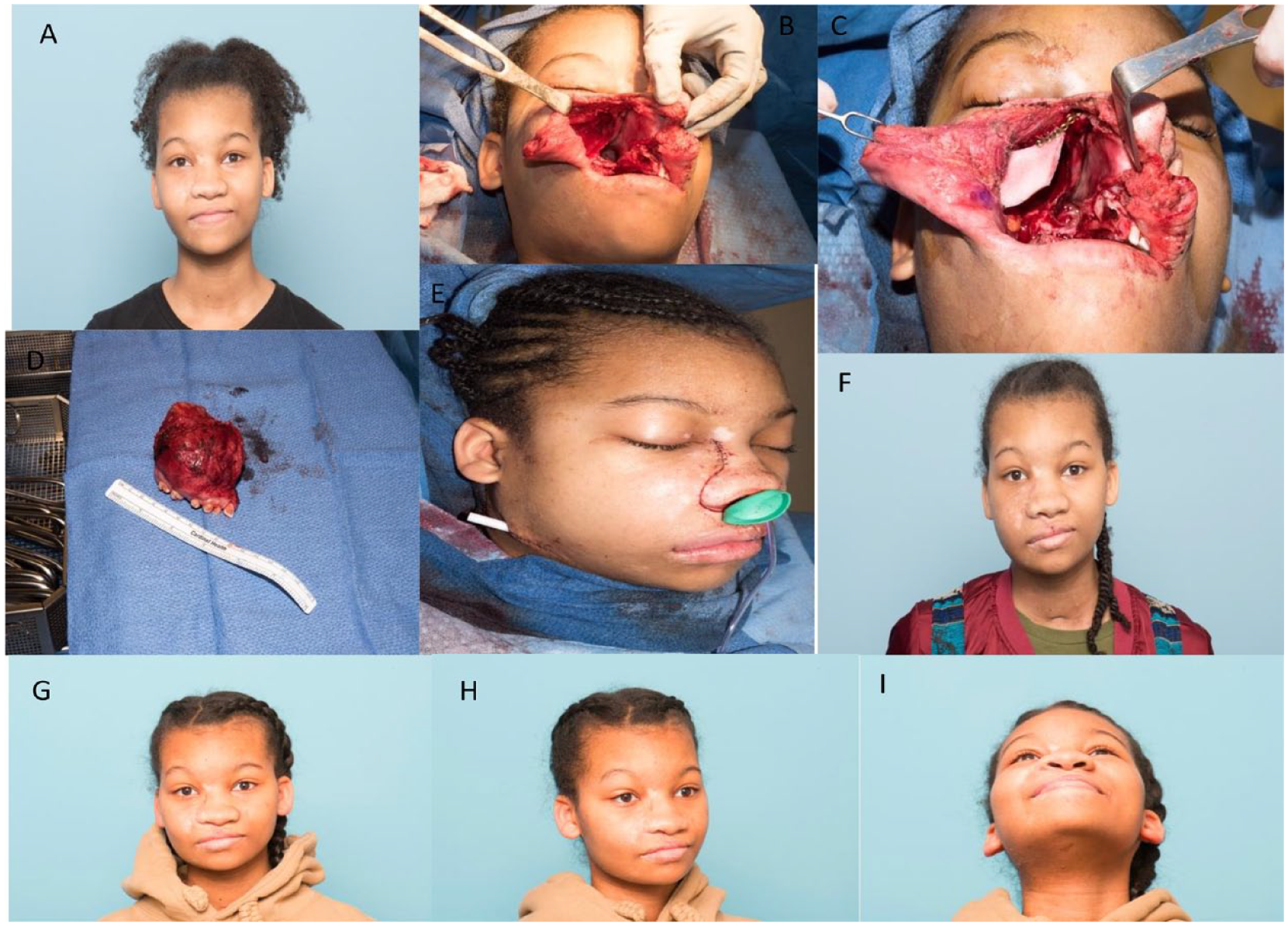
Maxillary reconstruction patient: (A) patient presenting with a right sided maxillary tumor, (B) resultant defect following tumor extirpation, (C) reconstruction using custom PEEK implant and custom titanium plates, (D) gross resection of maxillary tumor, (E) immediate on table result following closure of Weber Ferguson incision, (F) 3 month follow-up, and (G-I) 11 month follow-up from different views.
Mandibular—5 patients (42%) underwent a complete hemimandibulectomy, of which 2 had the anterior mandibular body spared. Seven patients (58%) underwent a subtotal hemimandibulectomy, with sparing of the condyle. Bony reconstruction was performed using an osseous fibular free flap (FFF) in all 12 patients (Figure 3). Customized mandibular titanium plates with fibular reconstruction were used in 7 patients (58%). Dental reconstruction was performed in 3 patients (25%) using osseointegrated dental implants at ages 12, 16, and 17 years.
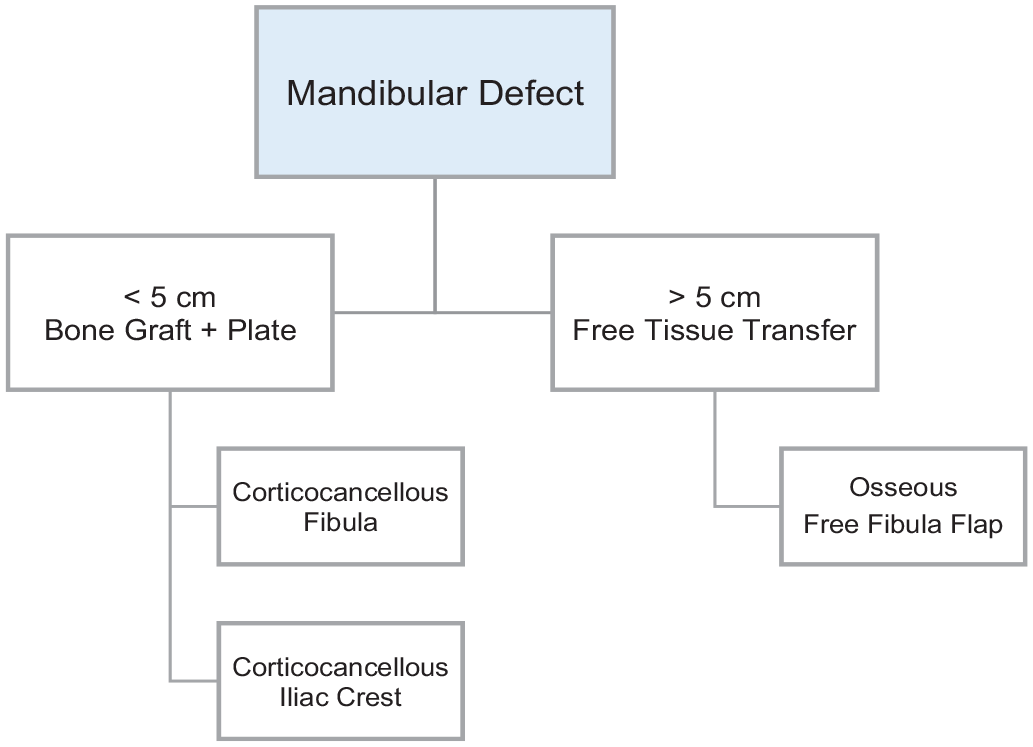
Algorithmic approach to mandibular defects.
Post-Operative Outcomes
Maxillary—Total flap loss was seen in 3 flaps (23%), with 1 patient losing both an ALT and FFF during maxillary reconstruction (Table 1). Both patients had replacement with an additional FTT, one with a VL muscle only flap, and the other with an ALT fasciocutaneous flap. One patient (7.7%) had partial loss of an ALT flap, which required debridement and was left to heal by secondary intention. Two patients (15.4%) developed oronasal fistulas (ONF) postoperatively. Two patients (15.4%) had implant related complications, one infection requiring implant replacement and one exposure following postoperative radiotherapy requiring implant removal. One patient (7.7%) required emergent flap debulking due to compression of the globe secondary to postoperative flap swelling of an ALT flap. No complications regarding FTT donor sites were observed, and all patients achieved satisfactory swallow outcomes following surgery. All 13 patients had greatly improved facial symmetry postoperatively (Figure 2). None of the 13 patients have displayed disrupted facial growth at subsequent follow-up visits.
Mandibular—Mandibular reconstruction complications included hematoma requiring evacuation in 2 patients (16.7%), postoperative mandibular range of motion limitation in 2 patients (16.7%), fibula-mandible non-union in 2 patients (16.7%), and lower lip paresthesia in 1 patient (8.3%). At the donor site of patients reconstructed with a FFF, 1 patient (8.3%) developed regional paresthesia, and 1 patient (8.3%) had minor wound dehiscence. All patients achieved satisfactory swallow outcomes following surgery. Two patients had slight chin deviation with maximal opening. Otherwise, there was no evidence of facial asymmetry or deformity in these patients (Figure 4).
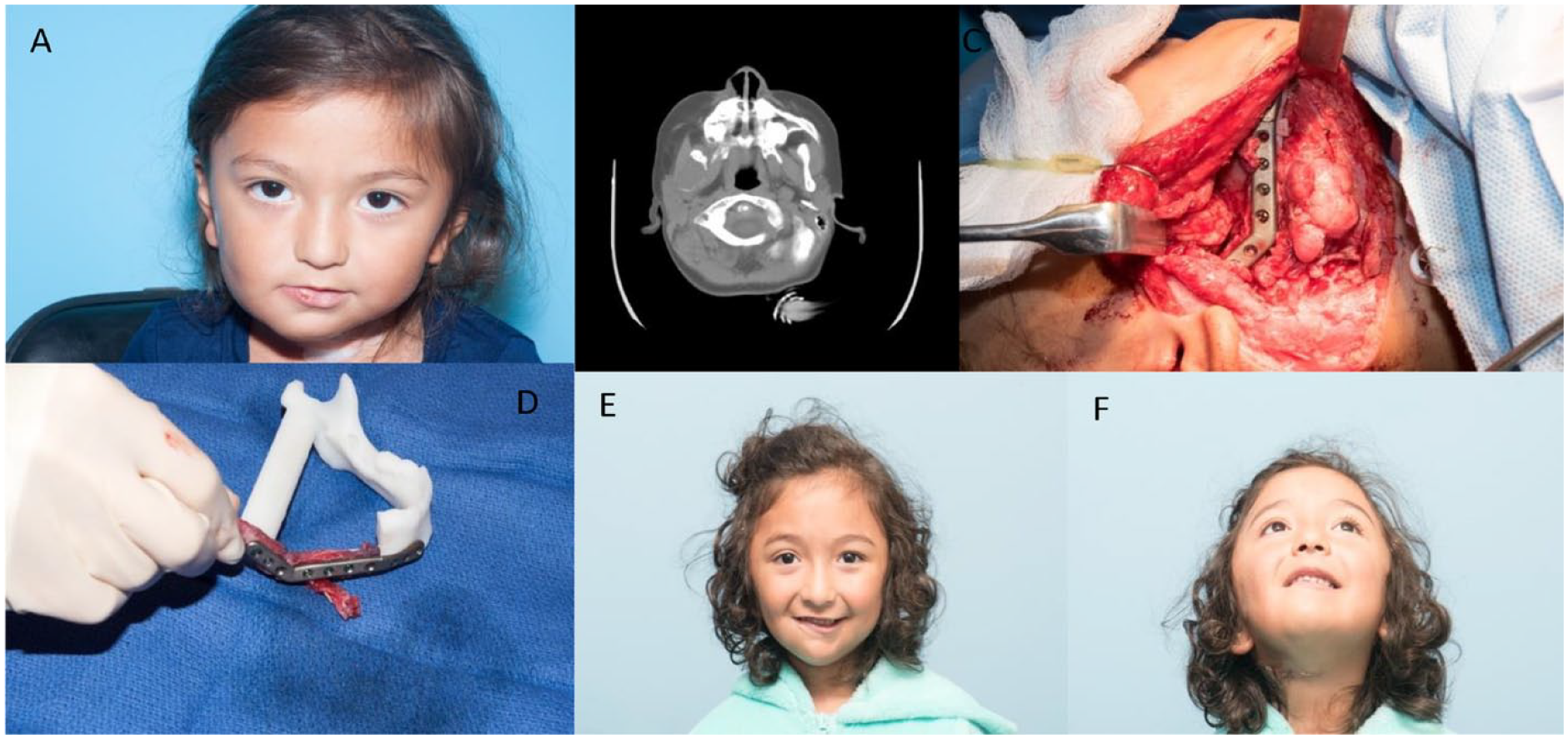
Mandible reconstruction patient: (A) patient presenting with a right sided mandibular tumor, (B) preoperative CT scan demonstrating fibromatosis in the right mandibular ramus and lateral condyle, (C) free flap secured to remnants of mandible and anastomosed to facial vessels, (D) on table reconstruction using osseus fibula free flap secured to custom plate, and (E) 2 years follow up demonstrating complete healing of reconstruction and facial symmetry.
Discussion
Craniofacial reconstruction following tumor extirpation presents with unique challenges in the pediatric population due to difficulty predicting future maxillary and mandibular growth, age dependent differences in skeletal morphology, and concern for surgical impact on functional development.1,10
The most challenging anatomical region for pediatric facial reconstruction is the middle third of the face, which requires consideration of future growth, dentition, oral competence, facial symmetry, and wound closure. Additionally, restoration of the barrier between the sinonasal cavity from the oral and orbital contents, and adequate support and position of the orbital contents, are important functional considerations during maxillary reconstruction.1,11,12 Soft-tissue separation of the oral and sinonasal cavities can be accomplished through palatoplasty techniques or buccal myomucosal flaps in small to medium sized maxillary defects. 1 However, for larger defects (>2 × 2 cm), free tissue transfer with latissimus dorsi, anterolateral thigh, or radial forearm flaps is required for adequate soft-tissue closure (Figure 1).1,12 For hemi-maxillary defects, which are typically greater than 2 × 2 cm even in children, a 2-stage approach is typically utilized. The first stage focuses on reconstruction of mid-face soft tissue envelope, separation of the oral and nasal cavities, reestablishment of midface projection. The second stage is pursued close to skeletal maturity, and centers on dental restoration with an osseous free fibula flap that provides permanent structural support and serves as a scaffold for osseointegrated dental implants. 1
Most of our hemimaxillectomy patients were reconstructed with an anterolateral thigh fasciocutaneous flap or a vastus lateralis myocutaneous flap. These flaps provided adequate coverage both intraorally and over alloplastic or autologous infraorbital implants. All patients achieved satisfactory speech and swallow functional outcomes. Acceptable facial symmetry was achieved in most patients, with only 3 patients displaying mild malar depression/asymmetry on exam. There were no instances of donor site morbidity from the ALT or VL flaps. The 2 instances of flap loss occurred in the setting of both neoadjuvant chemotherapy and adjuvant radiotherapy, however, both patients had successful free tissue transfer with contralateral donor flap replacement. Out of the 11 free flaps, 1 patient developed an oronasal fistula due to partial ALT flap necrosis, which resolved after healing by secondary intention. These outcomes demonstration that free tissue transfer such as the ALT or VL provide adequate separation of intraoral and nasal contents and provide sufficient bulk for postoperative radiotherapy in pediatric patients. While no instances of facial growth disruption in our patients have been recorded to date, further follow-up of these patients through completion of skeletal maturity is necessary to denote long-term craniofacial growth.
As normal maxillary growth is dependent on an intact dental arch, it is difficult to predict growth of the maxilla after hemi-maxillectomy.1,5,13 In hemi-maxillectomy, during which half of the dental arch is removed, the transverse growth of the remaining dental arch, and thus the maxilla, is compromised. 5 These patients tend to develop a large lingual maxillary crossbite that is extremely hard to correct due to resultant postoperative scarring and soft tissue contracture.1,4,5 As such, every attempt during primary reconstruction should be made to preserve normal maxillomandibular occlusal status in these patients.
As with maxillary reconstruction, one of the most challenging aims of mandibular reconstruction is matching the skeletal growth potential of the patient.7,13 The mandible undergoes peak growth during puberty which can be disrupted if resection occurs before adolescence. 14 Additionally, defects from mandibular resection can have a negative effect on oral competence, mastication, speech, and overall facial symmetry.8,13 Mandibular resection involving dentition can affect maxillomandibular occlusion, which must be reestablished in order to prevent subsequent facial growth alterations, maxillary hypoplasia, and malocclusion. An established algorithm based on defect size has been established in adults but does not exist in pediatric patients due to the dynamic nature of their skeletal morphology. Generally, for smaller mandibular defects, nonvascularized corticocancellous grafts are typically used. However, larger mandibular defects require vascularized bone, which increases the likelihood of postoperative neomandibular growth, which is vital in pediatric patients.9,13,15
The vascularized free fibula flap is a reliable method to reconstruct hemimandibular defects and portends minimal donor site complications.11,15-20 The fibular flap can be used to reconstruct the mandibular condyle in complete hemimandibulectomy patients, which preserves the mandibular height for optimal occlusion and restores the temporomandibular joint postoperatively.21,22 The use of virtual surgical planning and 3D custom guides are critical adjuncts in determining appropriate neocondylar length to maximize postoperative occlusion. Custom mandibular plates enhance reconstruction of large defects, as they are made to follow the specific contours and adhere the fibula flap to the mandible in maximal anatomic alignment. Custom plates were used in 7 of our 12 mandible patients and were removed between 6 and 12 months following reconstruction to allow mandibular growth to ensue.
All 12 of the hemimandibulectomy patients included in our study had large resection defects (range 3.1-9.6 cm) that were successfully treated with a vascularized FFF, even in the context of neoadjuvant and adjuvant chemotherapy. Postoperative functional outcomes of normal swallowing and resumption of oral diet were successfully achieved in all 12 patients. Normal occlusion was attained in 9 patients, with 3 patients displaying a mild anterior crossbite. While 1 patient had mild limitations in oral range of motion, 11 patients exhibited good range of motion and adequate maximal opening. Major donor site complications such as flexion contracture of the great toe, ankle instability, and leg length discrepancy were not observed in any patients. These functional outcomes, alongside a very low incidence of donor site complications, demonstrate that the FFF is a great option for large mandibular defects in the developing child.
While postoperative fibula-mandibular synchronous growth is debated, studies endorsing positive growth after at least 3 years report increased height within the mandibular ramus and preserved condyle.21-23 Mechanical stress and tension exerted on the FFF by native mandibular forces has been one of the proposed mechanisms for neomandibular growth postoperatively.9,21 As such, the authors are in favor of removing mandibular plates once evidence of healing is apparent (6-12 months postoperatively) in order to apply native mandibular forces to the FFF and induce resultant growth. All patients had satisfactory facial symmetry at rest. Two patients had minor chin deviation at maximal opening. Of the 5 patients who had condylar resection, 2 achieved adequate condylar height and satisfactory maxillomandibular occlusion with FFF reconstruction.
In pediatric maxillomandibular reconstruction, while esthetic consideration for symmetry should be optimized, the cardinal aim is to achieve a functional oral status. This is accomplished through optimizing maxillomandibular occlusion, separating the oral and nasal cavities, and the creation of a functional scaffold for permanent dentition. The resiliency of pediatric patients is further displayed in the ability of our patients to heal despite chemotherapy, radiation, and potential nutritional preoperative challenges that can accompany such therapies. In effect, the authors propose the use of free tissue transfer for reconstruction of large maxillary and mandibular defects for extirpative defects in pediatric patients.
Another adjunct worth consideration during pediatric reconstruction is alveolar nerve repair at the time of surgery in segmental mandibulectomy patients, which has been shown to enhance functional outcomes.24,25 Damage to the inferior alveolar nerve can result in symptoms of oral incompetence, such as drooling or issues with feeding, due to loss of sensory feedback in the lower lip.24-27 Multiple studies have shown that mental nerve reconstruction was successful in restoring sensation after segmental mandibulectomy, leading to improved functional outcomes with reduced drooling and improved oral competency in adults.24-26 In one of our mandibulectomy patients, the right inferior alveolar and mental nerve were repaired using an interposition nerve allograft at the time of reconstruction. One year postoperatively, the patient did not endorse complaints of numbness or pain in the inferior alveolar/mental nerve distribution, and had resumed normal speech, mastication, and swallowing function. Inferior alveolar nerve repair during pediatric mandibular reconstruction is a relatively straightforward adjunct that could further improve functional postoperative outcomes in patients undergoing mandibular resection that affects continuity of the nerve.
The safety and efficacy of fibula free flap reconstruction for pediatric maxillomandibular defects was recently evaluated in multi-institutional retrospective study by Slijepcevic et al. 28 Pre and post-operative chemotherapy did not appear to affect long-term wound healing in pediatric maxillomandibular reconstruction using a FFF, with minimal instances of hardware exposure reported. 28 Post-operative complications within 30 and 90 days occurred in less than 10% to 20% of patients, with only 1 free flap failure reported, with donor site complications occurring in less than 10% of patients. Dental rehabilitation was completed in a third of patients, and long-term craniofacial growth abnormalities occurred in a quarter of patients. These results suggest that fibula flaps may be harvested safely from pediatric patients without long-term donor site morbidity and minimal risk of free flap failure. However, patients who had dental implantation at the time of fibula reconstruction did not demonstrate long-term craniofacial asymmetries or disturbances of maxillomandibular growth. 28 While functional outcomes such as speech and oral competency are independent of dental implantation, immediate dental implantation may be important for long-term facial growth outcomes and may be indicated before skeletal maturity is realized.
Microsurgical free tissue transfer has facilitated reconstruction of large pediatric maxillary and mandible defects that were formerly believed too difficult to reconstruct.2,16,17,29 Microvascular reconstruction of the pediatric craniofacial region has become much safer, effective, and predictable in recent years, thanks to advances in both microsurgery and virtual surgical planning.15,30 Treating this population is complex, but pediatric functional reserve and enhanced wound healing potential makes microsurgical free tissue transfer an excellent option for maxillomandibular defects in the developing child.11,17,31
Conclusion
Pediatric craniofacial defects can have debilitating, long-term effects on a child’s physical growth, functional development, and esthetic presentation. In this large cohort of patients, maxillary defects were reconstructed utilizing either an ALT fasciocutaneous or VL myocutaneous flap, with no donor site complications and satisfactory postoperative swallowing outcomes. Mandibular defects were reconstructed with a FFF, with minimal donor site complications and satisfactory maximal oral opening and range of motion, with most patients maintaining normal occlusion many years postoperatively. Future research and long-term follow- up on this set of patients is necessary to denote definitive outcomes regarding craniofacial growth following reconstruction in pediatric patients.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
This study was approved by the Baylor College of Medicine Institutional Review Board (H-49937).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
