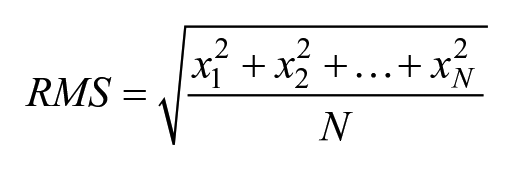Abstract
Background:
Metopic craniosynostosis (MC) represents 10% to 25% of craniosynostosis cases. Endoscopic-assisted strip craniectomy (ESC) with orthotic molding therapy provides a minimally invasive alternative to open fronto-orbital advancement. Herein, we describe a 3D photogrammetry methodology for measurement of regional cranial growth and inter-frontal angle (IFA) as well as examine factors that may contribute to successful outcomes after ESC.
Methods:
A retrospective review was performed on patients with MC treated with ESC and band therapy from 2015 to 2019. Patients obtained 3D photogrammetry preoperatively, postoperatively, and post-helmet therapy. A survey was administered to 14 independent raters, who were asked to determine treatment success based on subjective visual assessment of preoperative and post-banding images. Patients were considered to have successful therapy when ≥75% of raters were in agreeance.
Results:
Thirteen patients met inclusion criteria. Average age at surgery was 3.5 months, with 53.9% of these patients deemed as a successful outcome on aggregate reviewer scores. For patients with successful treatment, there was a trend toward younger at surgery (2.9 months vs 4.2 months, P = .09) and at initiation of helmeting (3.3 months vs 4.8 months, P = .07). Regional root mean square identified significant differences between frontal and temporal (14.04 mm vs 7.39 mm, P = .02) and temporal and parietal (7.39 mm vs 15.37 mm, P = .002) regions. Average postoperative IFA was significantly improved compared to preoperative values (128.19° vs 117.9°, P = .02). Logistic regression modeling found no significant predictors of success.
Discussion:
Though not statistically significant, patients with MC with successful outcomes following ESC and banding were younger at surgery and time of helmeting. Our analysis of regional cranial growth and IFA demonstrated significant differences, which coincide with expected morphologic changes in this patient population. Our results demonstrate the potential for this technology in surgical evaluation and support the importance of early diagnosis and treatment by craniofacial surgeons.
Keywords
Introduction
Metopic craniosynostosis (MC) accounts for approximately 10% to 25% of all craniosynostosis cases. 1 The pathogenesis of MC lies in the premature closure of the metopic suture which, in normal development, begins to ossify as early as 3 months of age and completely fuses by age 9 months in most patients. Premature closure leads to abnormal patterns of cranial growth, which results in phenotypic changes that range from subtle frontal midline vertical ridges to retrusion of the frontal cranium and orbital rims, bitemporal narrowing, and hypotelorism as seen in patients with trigonocephaly.1-3
The surgical treatment of craniosynostosis has evolved from the use of cranial vault remodeling (CVR) and fronto-orbital advancement (FOA) techniques to the use of minimally invasive endoscopic strip craniectomy (ESC) for age-appropriate patients in many centers.1,4,5 ESC relies on removing a strip of calvarial bone that contains the prematurely fused suture. Patients can then be fitted with specialty made orthotic molding helmets to achieve a final skull shape which is closer to normal. The use of ESC is limited, however, by the age of patient presentation, and treatment with this technique is largely reserved for the management of patients <6 months of age. ESC has several benefits when compared to CVR, namely reduced operative and anesthesia times, decreased blood transfusion requirements, and decreased hospital length of stay while providing equivalent outcomes to CVR.1,4,6 Methods for measuring the success of craniosynostosis surgery have historically relied on two-dimensional (2D) metrics, such as the cephalic index (CI) or the cranial vault asymmetry index (CVAI). 2 However, because the CI is a ratio of the anterior-posterior (AP) and lateral dimensions of the skull and MC is not a unilateral pathology that results in large degrees of cranial vault asymmetry, these indices do not translate to reliable measurements of success for patients with MC.
The inter-frontal angle (IFA) has been increasingly used as both a metric for success in MC as well as a diagnostic tool since it was first described.7,8 This metric measures the angle generated by the supra-orbital notches and the most anterior point of the skull. A limitation to this metric is its reliance on computed tomography (CT) data used to generate it, as originally described. In contrast, three-dimensional (3D) photogrammetry, another useful metric, relies on high-definition photographs to generate a 3D representation of a patient’s cranium without the need for infant radiation exposure and has previously been found to be comparable to CT. 9 3D photogrammetry involves the use of 3D photographs taken at regular intervals in order to objectively measure a patient’s progress during orthotic helmet, or band, therapy. Furthermore, given the repeated measurements which are obtained, 3D photogrammetry presents a radiation free method for measuring region-specific cranial growth patterns following surgical intervention and band therapy. However, there is a paucity of data regarding its use as an analog for the calculation of an IFA.
The goal of this study is to assess the outcomes of patients with MC who were treated using ESC with helmet therapy at our institution, measure regional cranial growth patterns, and develop a methodology for the generation of an IFA using 3D photogrammetry.
Methods
Study Design and Data Collection
Following Institutional Review Board (IRB) approval, we performed a retrospective review of patients undergoing treatment for craniosynostosis at our institution. Patients were identified by the International Classification of Diseases (ICD) 10 code for craniosynostosis–Q75.0, as well as by use of the following Current Procedural Terminology (CPT) codes indicating correction of craniosynostosis: 61550, 61552, 615565, 61557, 61558, and 61559. Patients diagnosed with MC and surgically managed at age 6 months or younger with ESC and postoperative band therapy between November 2015 and December 2019 were included in this study. Patients were excluded from analysis if imaging records, needed for generation of our data, were incomplete (Figure 1).
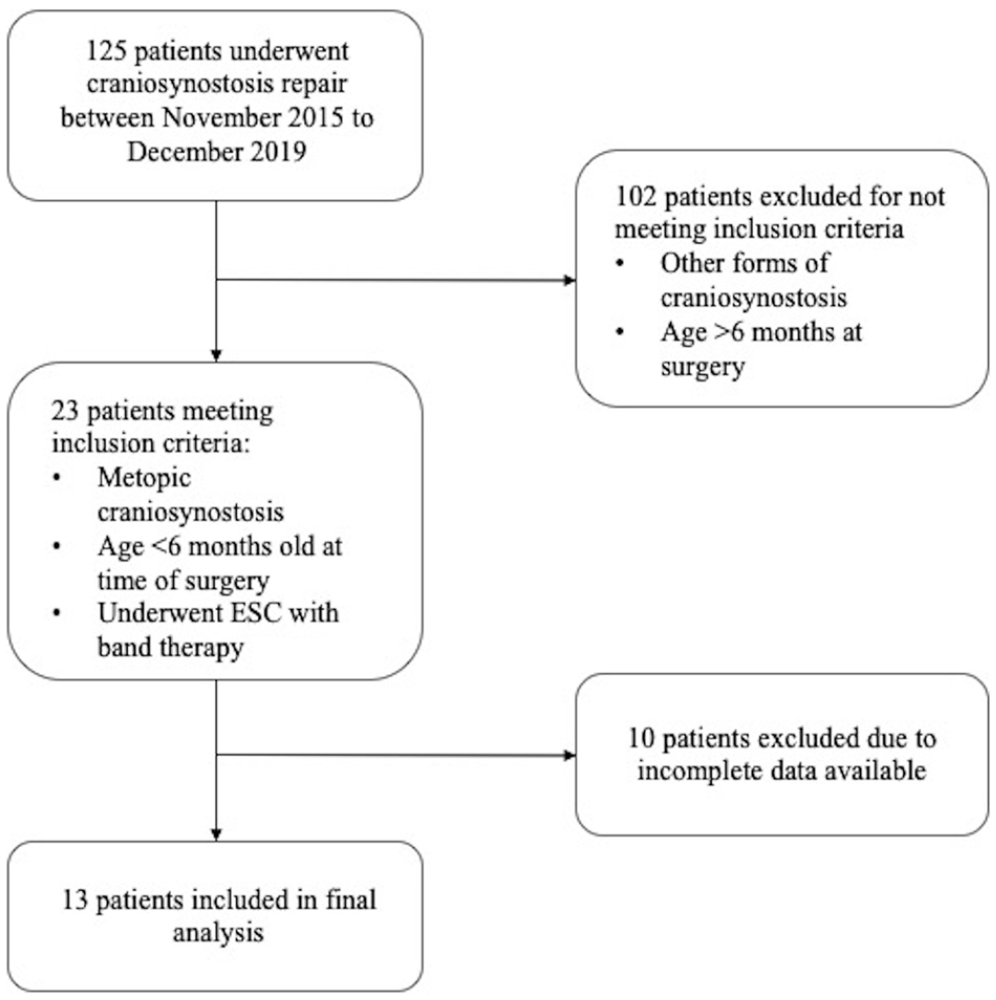
Study design.
A de-identified database of all included patients was created and demographic data collected included the following: age at initial clinical evaluation, at surgical correction, at entry into orthotic helmet therapy, and at exit from orthotic helmet therapy as well as gender and health insurance status. Clinical data gathered included the following: number of orthotic helmets used, duration of helmet therapy, preoperative, postoperative, and post-band 3D photogrammetry images as well as cephalometric measurements and anthropometric landmarks provided by Cranial Technologies™ (Tempe, Arizona).
Generation of Cranial Regions
Anthropometric landmarks were established using the Vectra™ imaging system (Canfield Scientific; Parsippany, New Jersey). This software takes 3D images and generates a computerized model of the patient. Landmarks were placed on the surface of the 3D models using an algorithmic approach for initial registration. Subsequently, quality assurance and review of landmark placement was verified by 3 independent specialists. Cranial regions were specified using established and novel landmarks as indicated below with the use of custom created scripts (Cranial Technologies; Chandler, Arizona) (Figure 2A). Landmarks that were manually registered were kept consistent with our automation process by aligning their x, y, and z coordinates to their appropriate locations using the custom script formula.
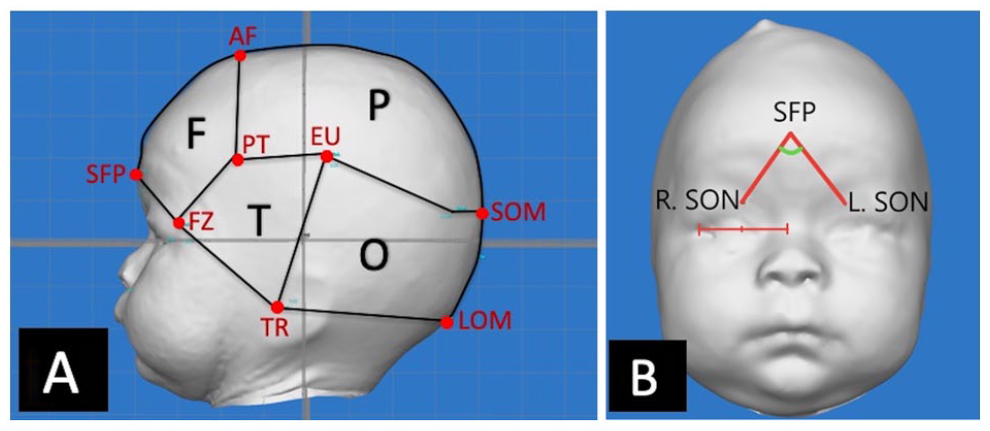
(A) Cranial regions with landmarks. F = frontal; P = parietal; T = temporal; O = occipital. SFP = sagittal frontal prominence; AF = anterior fontanelle; FZ = frontal process of the zygoma; PT = pterion; EU = eurion; TR = tragus; SOM = superior occipital midline; LOM = lower occipital midline. (B) Generation of inter-frontal angle. This was achieved by measuring the angle generated by the right supraorbital notch (R. SON) and the left supraorbital notch (L. SON) with the sagittal frontal prominence (SFP) as the vertex.
Frontal
Bilateral frontal regions were established by designating the sagittal frontal prominence (SFP) as the most anterior aspect and the anterior fontanelle (AF) as the most superior midline point. AF was registered by establishing the mid-point between the tragus (TR) and the frontal process of the zygoma (FZ) laterally and tracking this to its most superior point. The most inferior and posterior points were designated as the FZ and pterion (PT), respectively. The PT was registered in the same fashion as the AF along the same axis as the eurion (EU).
Temporal
Temporal regions were established using the PT as the most antero-superior point, with the FZ as the antero-inferior aspect, the EU as the most posterior, and the TR as the most inferior aspect.
Parietal
Parietal regions were bounded by the AF antero-superiorly, the PT antero-inferiorly, the EU laterally, and the superior occipital midline (SOM) posteriorly. The SOM was identified by establishing the mid-point between the bilateral occipital prominences.
Occipital
The occipital region was created by establishing the most anterior point as the TR with the EU as the lateral aspect, and SOM as the superior-posterior aspect. The inferior-posterior aspect was created by establishing the mid-point between the bilateral tragi, termed the lower occipital midline (LOM).
Generation of Inter-Frontal Angle
The IFA was generated for each patient at the pre-specified time points (preoperatively, postoperatively, and post-band therapy). This was accomplished by calculating the angle generated by the SFP (the most anterior aspect of the cranium when visualized on the lateral view) and bilateral supraorbital notches (SON) (Figure 2B). The bilateral SONs were registered by placing a point on the surface of the 3D image using the y-axis of the FZ. The x-coordinate was defined by finding the midpoint between the lateral canthus and the midline. This was done to approximate the relationship quantified by Ashwini et al, 10 wherein they identified that the SON skews toward midline with the distance from the SON to the nasal midline being 23% to 25% shorter than the distance from the frontozygomatic suture to the SON.
Determination of Cranial Region Root Mean Square
Cranial root mean square (RMS) is a statistical measure of the temporal changes in cranial shape and volume, which can be used to quantify cranial asymmetry. As previously described by Moghaddam et al, 11 RMS is unique to 3D photogrammetry and can be utilized to track changes within each cranial. To determine the RMS for our cohort, preoperative and post-band therapy 3D photogrammetry images were first manually juxtaposed. Then, cranial regions were identified in both images, and the vertical distance between analogous landmark points on the surface of both images were calculated in millimeters (mm). The RMS for each cranial region was calculated using the following equation described by Moghaddam et al, 11 where x represents the vertical distance between the pre- and post-band therapy images and N represents the total number of landmark points used for each cranial region. The RMS for each cranial region was compared to the RMS for other regions.
Determination of Successful Therapy
Although successful therapy can be assessed using quantitative objective metrics, in practice, treatment success is often determined clinically in conjunction with other objective metrics. For this reason, we chose to define “success” based on the clinical assessment of a group of raters. A qualitative survey was administered to a heterogeneous group of 14 independent raters, which included 2 attending craniofacial surgeons, 6 plastic surgery fellows, and 6 Cranial Technologies technicians, in order to determine treatment success. Cranial Technologies technicians are experienced cranial orthotists who are responsible for creating and altering orthotic helmets; they have extensive experience specifically with patients diagnosed with MC and other forms of craniosynostosis.
The survey administered to raters consisted of preoperative, postoperative, and post-band therapy AP and vertex view images of patients who underwent treatment of MC with ESC and helmet therapy. After viewing these images, raters were asked to provide a binary answer to the question “Was therapy successful?.” Raters were asked to choose their answer based on their extensive prior experience with children diagnosed and previously successfully treated with MC. The results of these individual surveys were then aggregated. It was determined a priori that patients with a “successful therapy” designation in ≥75% of raters would be deemed to have undergone successful therapy. This threshold was agreed upon by all investigators prior to data collection as it represents a reasonable majority of the raters. Subsequent analysis of inter-rater reliability was conducted utilizing Cohen’s kappa statistic.
Statistical Analysis
Regional cranial RMS change data was analyzed using one-way Analysis of Variance (ANOVA) with subsequent post-hoc testing to determine significant comparisons. All other continuous and categorical variables were analyzed using student’s t-tests and Chi squared tests, respectively. A multivariate logistic regression model was then constructed to identify factors associated with successful therapy. Data was stored in Microsoft Excel (Excel, Microsoft Corporation), and analyzed using R Software (R Foundation for Statistical Computing, Vienna, Austria).
Results
Demographics and Orthotic Helmeting
Of all patients with MC who had ESC with postoperative band therapy during the study period, 13 had complete records and met our inclusion criteria. Of these, 2 patients (15.4%) were female. The mean age at time of consultation was 1.9 ± 1.4 months (Table 1). Patients subsequently underwent ESC at a mean age of 3.5 ± 1.4 months and started band therapy at age 4.0 ± 1.5 months. These patients had an average time of band therapy of 5.5 ± 1.9 months with an average number of bands prior to completion of 2.1 ± 0.76.
Patient Demographics and Orthotic Helmeting Data.
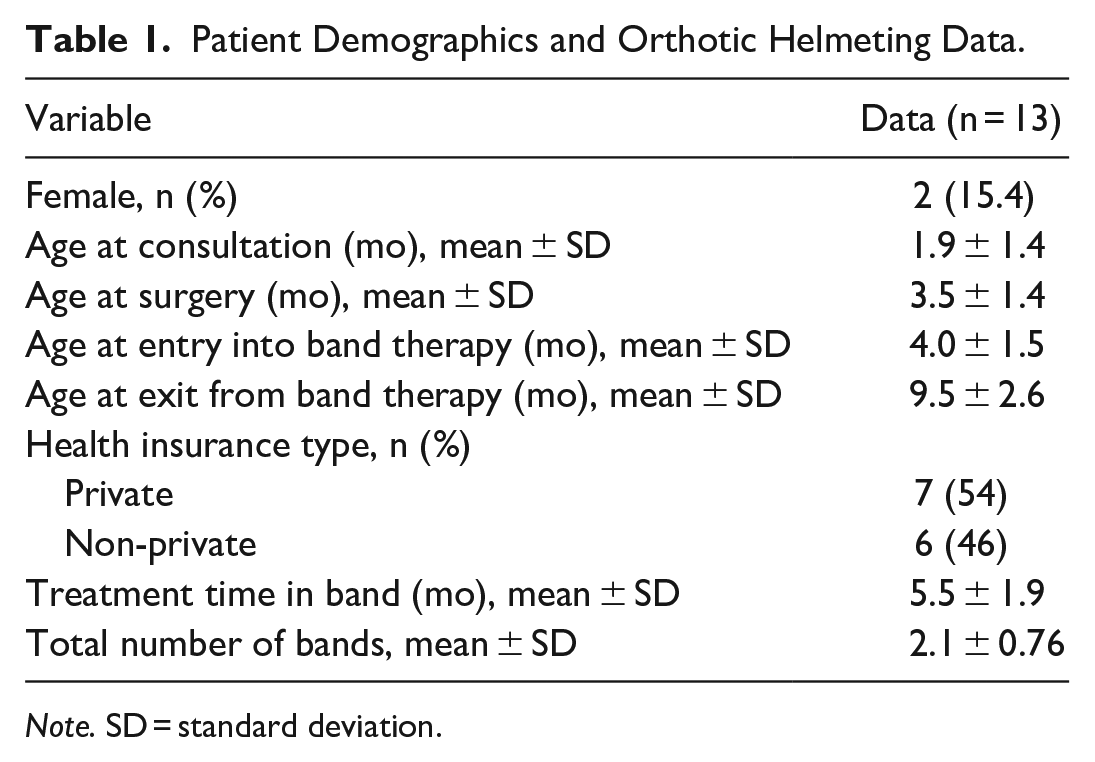
Note. SD = standard deviation.
Regional Cranial Growth
When comparing the RMS changes between the preoperative and post-band therapy 3D photogrammetry data, the greatest changes were seen in the frontal (14.04 ± 5.6 mm) and parietal regions (15.37 ± 4.6 mm), and the least change was observed in the temporal (7.39 ± 3.7 mm) and occipital (10.98 ± 4.9 mm) regions. One-way ANOVA and subsequent post-hoc testing demonstrated that the significant differences were present between the frontal and temporal (14.04 ± 5.6 mm vs 7.39 ± 3.7 mm, P = .02) and temporal and parietal regions (7.39 ± 3.7 mm vs 15.37 ± 4.6 mm, P = .002) (Table 2). No significant differences were identified when comparing the occipital region to frontal, temporal, or parietal regions in post-hoc testing.
Regional Root Mean Square Changes.
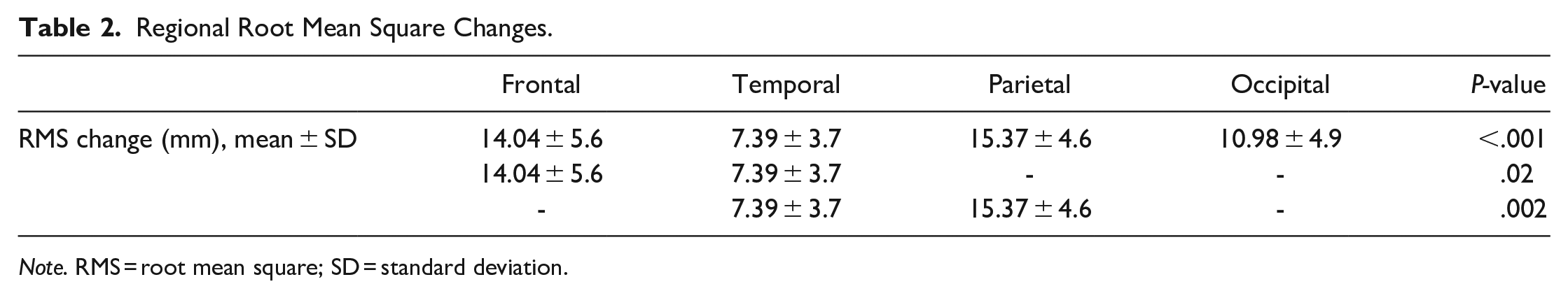
Note. RMS = root mean square; SD = standard deviation.
Inter-Frontal Angle
There was a significant increase in the IFA, from a mean preoperative angle of 117.9° ± 11° to a post-band therapy angle of 128° ± 7.6° (P = .02) (Table 3).
Change in Inter-Frontal Angle.
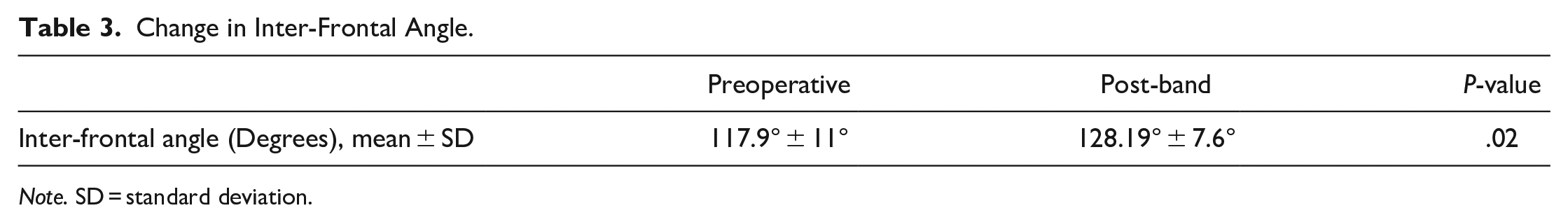
Note. SD = standard deviation.
Qualitative Assessment for Surgical Success
Based on subjective visual evaluation of pre-post photogrammetry results and meeting >75% inter-rater correlation, there was a consensus of 7 out of 13 patients (53.9%) who were deemed to have a successful normalized cranium. Analysis of the inter-rater reliability yielded a Cohen’s Kappa statistic of 0.197. Example vertex photographs of 2 patients from this study, one deemed to have successful and the other found to have unsuccessful treatment, have been provided in the Supplement (Supplemental Figures 1 and 2). When comparing patients with successful therapy to those for whom therapy was unsuccessful, there were no significant differences regarding their gender, age at surgery, age at entry into band, preoperative CI, preoperative CVAI, or preoperative IFA (Table 4). However, there was a trend toward younger age at surgery (2.9 ± 1.5 months vs 4.2 ± 0.98 months, P = .09) as well as initiation of helmet therapy (3.3 ± 1.4 months vs 4.8 ± 1.3 months, P = .07) for patients who had successful therapy. Logistic regression modeling, controlling for gender, insurance status, helmeting clinic site, treatment time, number of bands, and preoperative IFA, did not demonstrate any factors significantly associated with success.
Successful Therapy Craniometric and Demographic Data.

Note. SD = standard deviation; Pre-Op = preoperative; CI = cephalic index; CVAI = cranial vault asymmetry index; IFA = inter-frontal angle.
Discussion
Our study presents a methodology for generating cranial regions to objectively assess the morphological changes seen following ESC and band therapy using 3D photogrammetry. CT data has historically been the gold standard for assessing cranial morphology in patients with craniosynostosis, however in our study we find that 3D photogrammetry stands to be a viable alternative.
Qualitative assessment for perception of normal head shape in craniosynostosis surgery has previously been described.12,13 Al-Shaqsi et al demonstrated that a correction threshold of 70% was required for the general population to deem a corrected craniosynostosis deformity as “normal.” In our study, we deemed success as >75% interrater correlation for success in qualitative assessment.
No significant differences were identified among patients who were deemed to have undergone unsuccessful and successful therapy. However, our results identified a trend toward younger age at surgery and at initiation of helmet therapy in patients who had successful therapy. Given our small sample size, it is possible that our study did not have adequate power to identify a statistical difference between patients who had successful and unsuccessful therapy.
When examining our IFA data, there was a significant difference in the post-band IFA when compared to preoperative. These results are notable given that the preoperative IFA in our study approximates the threshold IFA of 118° for surgical correction set forward by Sisti et al 3 and Kellogg et al, 7 who detailed the generation of this predictive methodology using CT imaging data. Though 3D photogrammetry cannot be directly equivalent to CT imaging due to the inability to resolve osseous structures from overlying soft tissue, we were able to use consistent surface landmarks to generate the IFA using the bilateral SONs. 9 It is also worth mentioning that the mean preoperative IFA was smaller, by almost 10°, for the patients who were deemed to have unsuccessful therapy compared to those with successful therapy. Though this difference was not statistically significant and preoperative IFA was not found to be a significant predictor for success using the logistic regression model, this offers a possible explanation for why therapy was unsuccessful for some patients in our cohort.
When examining the regional cranial growth patterns established by comparing preoperative and post-band therapy 3D photogrammetry data, we found that the frontal and parietal areas demonstrated significantly higher degrees of change when compared to the temporal region, with no significant differences observed when comparing these to the occipital region. These results are consistent with the expected changes associated with removal of the fused metopic suture as well as the restrictive growth pattern driven by orthotic helmeting. In addition, these findings are consistent with prior volumetric assessments of patients undergoing ESC. 13 The phenotypic changes desired with MC correction are directed toward improvement of the trigonocephalic phenotype. As this method of treatment relies on the engine of brain growth to expand, the helmet is more predictive of guidance of growth by providing strategic areas of restriction. Areas of thicker bone, such as the sphenoid wing, may not be as amenable to centripetal brain growth. Herein, we demonstrate that ESC and postoperative helmeting has significant changes to the frontal bone more so than the temporal areas.
Though the ESC technique with postoperative helmeting offers a minimally invasive alternative to open FOA, these results demonstrate the limitations of the technique. By removing the fused metopic strip, we can reliably improve the IFA and trigonocephaly. However, improvement of the pinched temporal areas cannot be reliably predicted as this is dependent on brain growth. A restrictive molding helmet will not be able to correct this deformity as it, by definition, does not expand the cranium. With a qualitative success rate of 53.9% in this study, this may be reflective of the inherent limitations of the surgical technique. The recessed temporal areas are often a residual sequela even after prolonged helmet molding. It has also been shown that more severe objective preoperative metopic phenotypes are associated with worse esthetic dysmorphology, 14 which correlates with our findings.
Limitations
Our study has several limitations, primarily that this was a single center study with a small sample size. Given the relatively low incidence of craniosynostosis, a multi-institutional collaboration may generate a more robust cohort. We are also limited by the surface level data that is provided by 3D photogrammetry when establishing anthropometric landmarks for both IFA and cranial regions. With our technique, our landmarks are placed on the surface of multiple layers of soft tissue as opposed to directly on the target bony landmark, which may decrease the accuracy of our estimates. For example, our methodology of IFA generation is based on that described by Kellogg et al 7 but adapted to surface landmarks identifiable on 3D photogrammetry. Specifically, we identify the most anterior sagittal point and the bilateral SONs to generate this angle, rather than using CT data as previously described.
Additionally, while our regional cranial growth measurements align with expected post-therapy phenotypic changes in patients with MC, our methodology did not correct for age-related head growth. Furthermore, our limited rate of success coupled with a low kappa statistic may be related to our small cohort size as well as the raters’ bias due to the subjective metrics used to determine successful therapy. Long-term follow up may give greater insight into further evolution of fronto-temporal regional changes through cranial maturity. Likewise, future studies may consider querying the parent-reported subjective assessment of treatment success as patient-reported outcomes are important for any type of surgery.
Conclusion
Regional morphologic changes following ESC and helmet molding for metopic synostosis can be well characterized with objective measures. The use of 3D photogrammetry for the generation of an IFA provides a viable radiation-free method of objectively measuring the IFA as well as assessing regional cranial growth. 3D photogrammetry may be a useful tool for providing objective morphologic expectations for both surgeons and patient families and warrant further research and development to increase the ease and accuracy of generation of 3D photogrammetry data. Early diagnosis and evaluation by craniofacial surgeons may provide the best opportunity for successful therapy. If combined with the growing field of machine learning and artificial intelligence, 3D photogrammetry technology may be deployed to screen for craniosynostosis and be used to provide predicted patient specific morphologic changes and guide diagnostic and therapeutic considerations.
Supplemental Material
sj-jpg-1-fac-10.1177_27325016231191638 – Supplemental material for Characterization of Regional Morphological Changes in Metopic Craniosynostosis Following Endoscopic Strip Craniectomy With Postoperative Helmeting: Predictors for Success
Supplemental material, sj-jpg-1-fac-10.1177_27325016231191638 for Characterization of Regional Morphological Changes in Metopic Craniosynostosis Following Endoscopic Strip Craniectomy With Postoperative Helmeting: Predictors for Success by Alfredo Cepeda, Huan T. Nguyen, Chioma Obinero, George N. Washington, Tim Littlefield, Matthew R. Greives and Phuong D. Nguyen in FACE
Supplemental Material
sj-jpg-2-fac-10.1177_27325016231191638 – Supplemental material for Characterization of Regional Morphological Changes in Metopic Craniosynostosis Following Endoscopic Strip Craniectomy With Postoperative Helmeting: Predictors for Success
Supplemental material, sj-jpg-2-fac-10.1177_27325016231191638 for Characterization of Regional Morphological Changes in Metopic Craniosynostosis Following Endoscopic Strip Craniectomy With Postoperative Helmeting: Predictors for Success by Alfredo Cepeda, Huan T. Nguyen, Chioma Obinero, George N. Washington, Tim Littlefield, Matthew R. Greives and Phuong D. Nguyen in FACE
Supplemental Material
sj-jpg-3-fac-10.1177_27325016231191638 – Supplemental material for Characterization of Regional Morphological Changes in Metopic Craniosynostosis Following Endoscopic Strip Craniectomy With Postoperative Helmeting: Predictors for Success
Supplemental material, sj-jpg-3-fac-10.1177_27325016231191638 for Characterization of Regional Morphological Changes in Metopic Craniosynostosis Following Endoscopic Strip Craniectomy With Postoperative Helmeting: Predictors for Success by Alfredo Cepeda, Huan T. Nguyen, Chioma Obinero, George N. Washington, Tim Littlefield, Matthew R. Greives and Phuong D. Nguyen in FACE
Supplemental Material
sj-jpg-4-fac-10.1177_27325016231191638 – Supplemental material for Characterization of Regional Morphological Changes in Metopic Craniosynostosis Following Endoscopic Strip Craniectomy With Postoperative Helmeting: Predictors for Success
Supplemental material, sj-jpg-4-fac-10.1177_27325016231191638 for Characterization of Regional Morphological Changes in Metopic Craniosynostosis Following Endoscopic Strip Craniectomy With Postoperative Helmeting: Predictors for Success by Alfredo Cepeda, Huan T. Nguyen, Chioma Obinero, George N. Washington, Tim Littlefield, Matthew R. Greives and Phuong D. Nguyen in FACE
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Disclosure
Tim Littlefield is the chief scientific officer of Cranial Technologies™, though he did not receive any compensation for this project. Cranial Technologies provided postoperative helmeting and anthropometric raw data. There are no relevant financial disclosures for the remaining authors.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
This study was approved under the University of Texas (IRB HSC-MS-20-0821).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.