Abstract
Craniofacial and orthognathic surgery are high risk procedures for surgical blood loss. Significant blood loss leads to increased rates of blood product transfusion which may be associated with increased morbidity to the patient. The use of anti-fibrinolytics such as tranexamic acid has become popular in these procedures. However, the evidence to support its use in pediatric craniofacial and orthognathic surgery is sparse. This review analyzes the current randomized control trials assessing the use of tranexamic acid in craniofacial and orthognathic surgery. The study reviewed published literature up until December 20th, 2020. Six trials were included in this analysis. Pooled data showed that patients who received tranexamic acid during craniofacial or orthognathic surgery have less blood loss compared to those in control groups (mean difference—5.47 ml/kg [CI -7.02-3.82], P value <.05). Hence, rate of blood product transfusion in patients who received tranexamic acid is lower than control group by 2.01 ml/kg (CI 95%, 1.27-2.74, P value <.05). In summary, this review showed that craniofacial and orthognathic surgery patients who receive tranexamic acid might have lower estimated blood loss and receive less volume of blood products transfusion.
Introduction
Craniofacial and orthognathic surgery are high risk for surgical blood loss. Surgical blood loss and subsequent blood transfusion may be associated with increased mortality, morbidity, and length of hospital stay. 1 Studies have reported the rate of blood transfusion in craniofacial and orthognathic surgery to be anywhere between 23% and 100% either intraoperative or postoperatively.1,2 Such detrimental effects are more profound in pediatric surgical patients in which the blood volume is relatively small compared to adults. Strategies to reduce blood loss and transfusion rates are critical principles in surgery and anesthesia. Interventions such as permissive hypotension, pre-operative blood enhancing medications such as iron supplementation, and the use of anti-fibrinolytics have been demonstrated to reduce blood loss and subsequent transfusion rates. 2
Tranexamic acid (TXA) is a synthetic amino acid that inhibits fibrinolysis.3,4 It has been shown to reduce blood loss and the need for blood transfusion in trauma, elective total knee arthroplasty, dental extractions, spine surgery, and cardiac surgery.5 -9 Topical tranexamic acid has been used for treatment of postoperative bleeding in anticoagulated patients after oral and cardiac surgery.10,11 The principal mechanism of action is delayed breakdown of the fibrin matrix leading to increased stability and resistance of the clot to degradation. 12 The high success rate of tranexamic acid use in trauma has precipitated its use in elective procedures with the goal of minimizing blood loss. Use of tranexamic acid has increased in elective craniofacial and orthognathic surgeries. Multiple studies have reported efficacy and safety when used preoperatively and intraoperatively in various plastic surgery procedures.3,4 Despite a substantial rise in the use of tranexamic acid in plastic surgery procedures, there is a paucity of literature examining the effectiveness of tranexamic acid in craniofacial surgery and particularly in pediatric and orthognathic patients. 13 The purpose of this systematic review was to review the current literature as it pertains to the effectiveness of tranexamic acid administration in pediatric craniofacial and orthognathic surgery.
Methods
This review was designed to summarize the current randomized control trials investigating the use of tranexamic acid in elective craniofacial and orthognathic surgery. The review was conducted in accordance with PRISMA guideline for systematic review reporting. The protocol for this systematic review was registered on PROSPERO network (registration number: CRD4202225957) December 12, 2020. The search for the review was conducted using PubMed, MEDLINE, CINHAL, EMBASE, Cochrane Trial Registry, Clinicaltrials.gov, Google Scholar, and Web of Science from inception until December 20, 2020. To investigate the role of TXA in pediatric craniofacial and orthognathic patients, the following search terms were used: “Craniofacial surgery,” “pediatric,” “clinical trials,” “randomized,” “Tranexamic acid,” “plastic surgery,” and “orthognathic surgery” and to further supplement our search, the medical subject headings database for appropriate synonyms were used. The reference lists of included full-text articles were reviewed to ensure that all pertinent studies were included. The search was limited to publications written or translated into English language. The search strategy was formulated and conducted with the assistance of a credentialed medical librarian.
Studies included in this review were randomized controlled trials (RCT) and if they examined pediatric patients (≤18 years of age) undergoing elective craniofacial or orthognathic surgery who were randomized to intravenous tranexamic acid administration, placebo or usual care. Outcome measures of the studies included in this report includes total blood loss, amount of blood transfused, and length of operative time and length of hospital stay. After implementation of the search strategy, 2 reviewers screened all potentially relevant citations by title and abstract independently. Any citations that were deemed relevant by both reviewers proceeded to the abstract detailed screening stage. Subsequently, abstracts that met the inclusion/exclusion criteria underwent full article review. Data extraction and quality assessment were done by the reviewers using standardized data abstraction forms. Data were extracted from included studies: study title, first author, demographic data, and details of the intervention and control groups, outcome data, and risk of bias for each study. Risk of bias was assessed independently and a modified Cochrane risk of bias tool for which each domain is rated as “low,” “probably low,” “high,” or “probably high” was utilized in this review. 14 Risk of bias domains such as sequence generation, sample size discrepancies, allocation sequence concealment, blinding, selective outcome reporting, and other bias (such as the funding sources and termination of studies) were included in the bias assessment of included studies. The overall certainty for evidence for each outcome was determined using the Grading Recommendations Assessment, Development and Evaluation (GRADE) approach. 15 The summary of findings tables were formulated using the Guideline Development Tool (https://www.gradepro.org).
Dersimonian and Laird random-effects methods were used for the statistical analysis in RevMan 5.0 (Cochrane Collaboration, Oxford) software. The study weights were generated using the inverse-variance method. Mean Differences (MDs), Standard Deviations (SD), and Sample Size (SS) for continuous outcomes, all with 95% confidence intervals (CIs) were used. The absolute effects were calculated using the pooled baseline prevalence from the control arm of included trials. Heterogeneity between trials was assessed using visual inspection of the forest plots, the Chi-squared test, and I 2 statistic.
Results
The initial search yielded a total of 322 citations as shown in Figure 1. About 164 duplicates were excluded. Of the remaining 168 articles, 154 were excluded after the title and abstract screening process, and 14 articles underwent full review. Eight studies were excluded during the full text assessment: review articles n = 4; non-randomized trials n = 2; study protocols n = 2. Six studies were included for data extraction, comprising a total of 300 patients.
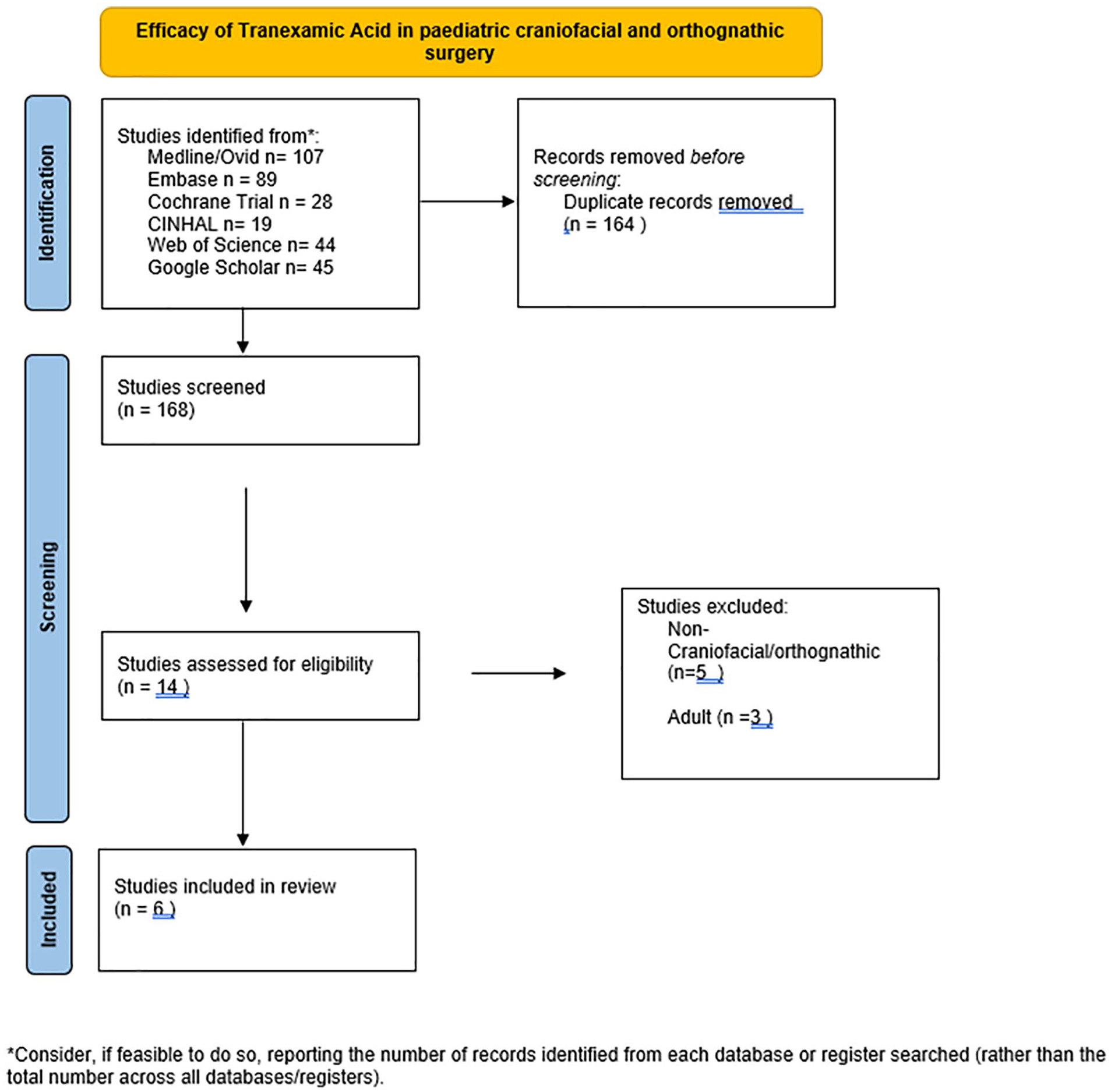
Flow chart for study protocol and outcome.
Description of the Included Studies
Four studies were conducted in a single surgical center while 2 studies included patients from 2 centers. Four studies looked at the use of tranexamic acid in pediatric craniofacial surgery and 2 studies investigated the use of tranexamic acid in orthognathic surgery. The mean age of participants in the craniofacial studies was 1.4 years (range 3 months–2 years). The mean age for the orthognathic surgery study was 16 years (Range: 14–40 years old).
The craniofacial studies included pediatric patients who required open cranial remodeling procedures for syndromic and non-syndromic congenital deformities such as craniosynostosis. The 2 orthognathic studies included patients who required either a single or double jaw surgery for correction of either congenital or post traumatic malocclusion.
All studies excluded patients who were known to have a bleeding diathesis or required an acute cranial operation for traumatic reasons.
Follow up post-intervention for bleeding complications was reported for 24 hours in 1 study, 72 hours in another study, and 1 study followed patients for ten days. The orthognathic studies did not report specifically on postoperative bleeding complications.
Tranexamic acid use protocols in the studies were variable. Fenger-Ericson et al. used a pre-operative TXA of 10 mg/kg bolus at the start of cranial cases, followed by 3 mg/kg infusion for a total infusion time of 8 hours. The control group in this study received equivalent amounts of normal saline. Song et al 16 used a 15 mg/kg bolus infused over 15 minutes at the start of the case, followed by a continuous infusion of 1 mg/kg/h until skin closure.
Goobie et al conducted an RCT in which the aim was to look at the difference between a low dose and a high dose protocol of tranexamic use in craniosynostosis. The low dose protocol was 10 mg/kg bolus followed by 5 mg/kg/h infusion until the end of the case. The high dose protocol was 50 mg/kg bolus followed by 5mg/kg/hr infusion until the end of the case. 17
Sankar et al used a bolus of 10 mg/kg at the start of orthognathic cases followed by 1 mg/kg infusion till the completion of the case. 18 Choi et al 19 used a 20 mg/kg one off bolus at the start of their orthognathic surgeries.
In terms of outcome measurement, all studies reported on blood loss, rate of transfusion, and length of hospital stay. The method of blood loss measurements included, hemodynamics, laboratory values of hemoglobin and hematocrit, estimates by anesthesia team, and estimates by surgical teams. Two studies calculated the cost associated with TXA use per patient versus the cost of blood product transfusions.17,18
The Risk of Bias (RoB) for 5 studies was judged to be low and one was judged to be probably low. The details of the RoB for the studies included are shown in Table 1.
Risk of Bias Assessment for Included Studies.
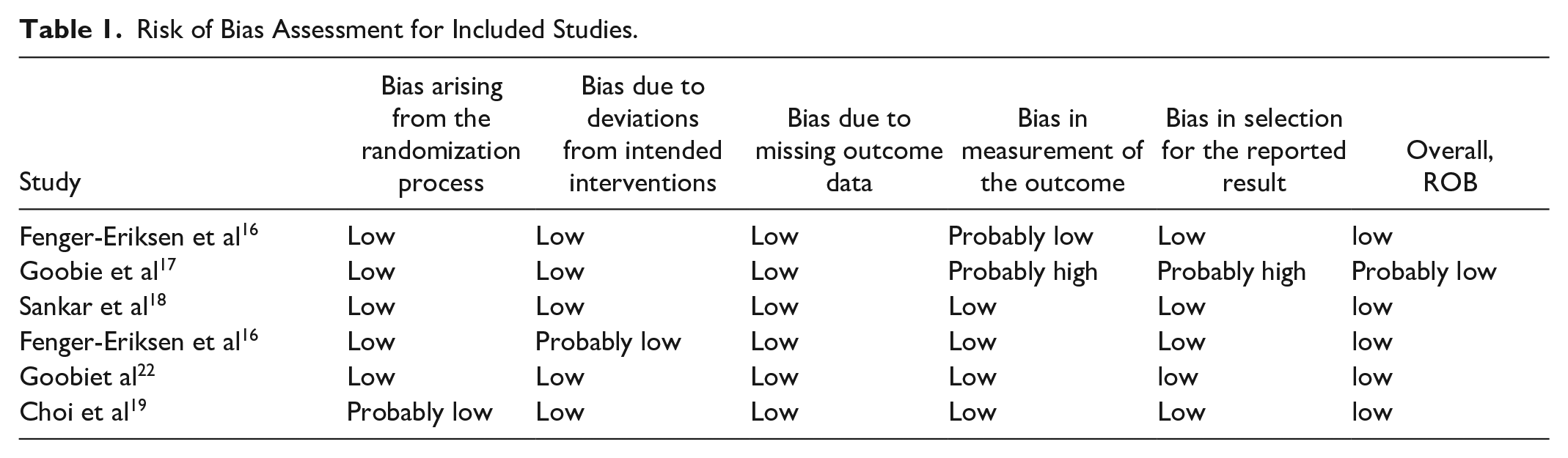
The studies included in this systematic review had a low probability for bias from the randomization process or deviation from the intended measurement as shown in table 2.
summary of Certainty Assessment for Included Studies.
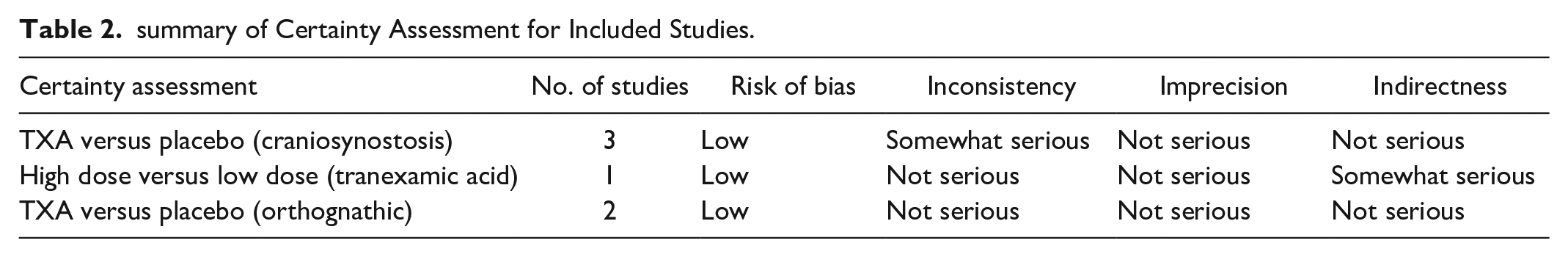
The studies that looked at the use of TXA in craniosynostosis surgery were judged to have somewhat serious inconsistency given that they both utilized different TXA administration and dosing protocols. The other 3 studies were judged to have no serious concerns in inconsistency, impression, or indirectness of outcome measurement.
This study showed that pediatric patients who received TXA had on average 14.7 minutes shorter operative time than children who did not receive TXA (CI 95%: 10.27-19.17, P value <.05). There were 4 studies that reported no difference in length of operative time between the TXA and control groups. Two studies reported that the TXA group had a shorter operative time than the control group. Therefore, in the pooled analysis, the overall length was found to be longer in the control group compared to the TXA group as shown in the frost plot in Figure 2. The pooled studies have a significant heterogeneity of I2 85% as shown in figure 2.
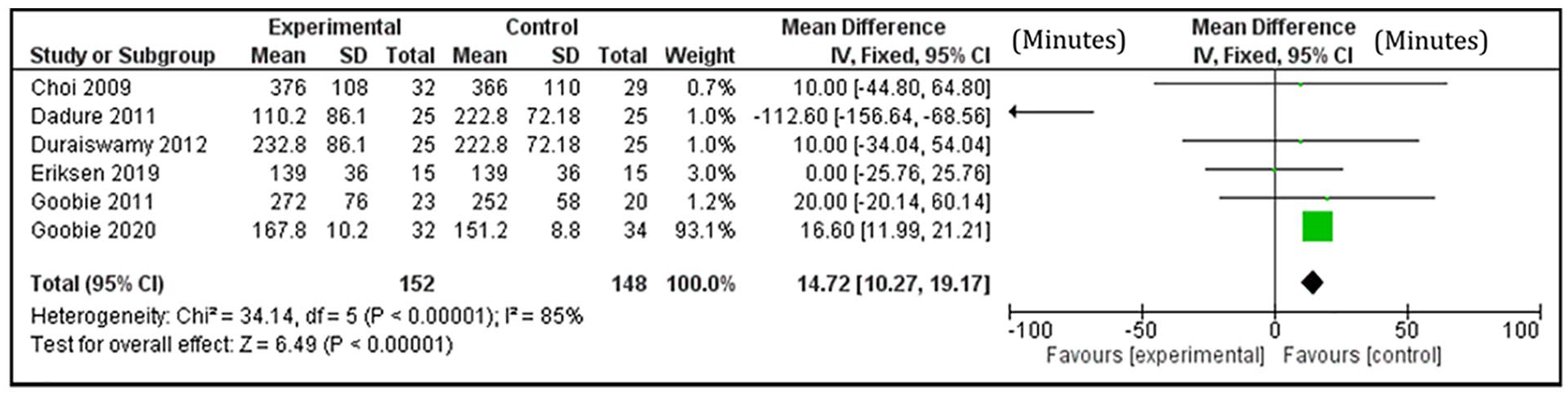
Pooled mean difference in operative time between TXA and control groups (minutes).
This study found that patients who did not receive TXA on average required 2.01 ml/kg (CI 95%, 1.27-2.74) more blood product transfusion compared to those who received TXA. There were 2 studies that did not report an estimate of transfusion rate and therefore the pooled analysis for this outcome was based on 4 studies. Three studies reported higher transfusion rates among the control groups compared to the TXA groups. However, a study by Goobie et al. reported higher overall transfusion rate among TXA groups. The weighted pooled analysis showed bias toward the TXA group having less blood product transfusion than the control group. The heterogeneity in these outcome measures was significant and the I2 is 86% as shown in figure 3.
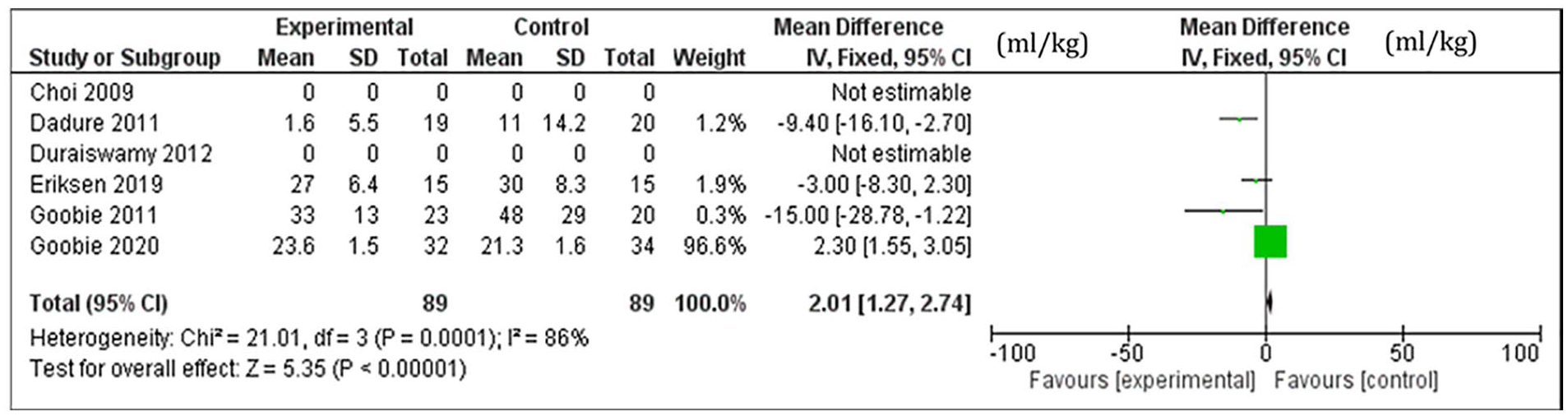
Pooled mean difference in blood product transfusion between TXA and control groups (ml/kg).
Patients who received TXA were found to have 5.47 ml/kg less blood loss than those who did not receive TXA. Four studies reported a significant reduction in estimated overall blood loss in TXA compared to the control group, while 2 studies found inconclusive results. The studies included in the pooled analysis have significant heterogeneity with I2 = 91% as shown in figure 4.
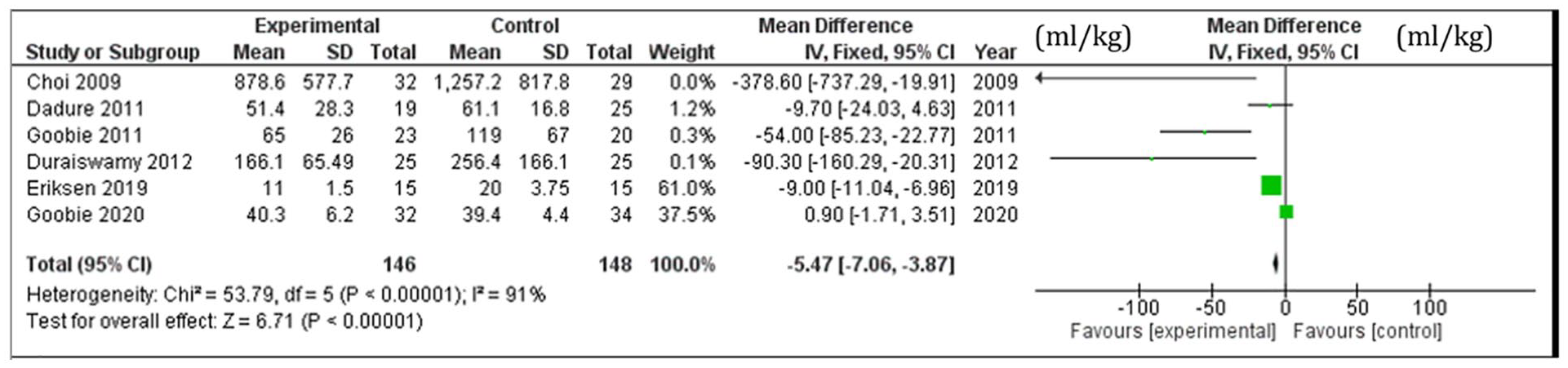
Pooled estimated blood loss in TXA and control groups (ml/kg).
Complications
None of the studies included in this review reported any complications.
Discussion
Craniofacial and orthognathic surgical procedures are at high risk for blood loss and blood product transfusion. Blood transfusion is associated with infective and non-infective complications. Therefore, strategies to reduce transfusion risk are useful in enhancing safety of craniofacial and orthognathic surgeries. Tranexamic acid was introduced in 1957 for postpartum hemorrhage. 3 Since that time and with increased use, it has been shown to have a beneficial impact on blood loss, rate of transfusion, and length of operations in elective orthopedic, gynecology, cardiac, and neurosurgical procedures.6,8,20 -23 This review summarizes the current state of evidence for the use of tranexamic acid in craniofacial and orthognathic surgery.
The number of rigorous RCTs which specifically evaluated the use of TXA in craniofacial and orthognathic surgeries are limited and this review identified only 6 studies. In addition, there was significant heterogeneity of the protocols used to administer TXA in craniofacial and orthognathic surgery. There was no consensus on an optimal protocol for its use in craniofacial and orthognathic surgery. Some studies used an initial bolus dose followed by an infusion dose during surgery while others provided a single bolus dose at the start of the case with no further infusion. The duration of infusion protocol was also variable with some studies providing TXA up to 24 hours beyond the end of surgery. Such variability makes comparative analysis of outcomes from such studies difficult. It also highlights the lack of evidence to guide TXA use and protocol in craniofacial and orthognathic surgery.
Many craniofacial surgeons and pediatric anesthesiologists believe that despite the lack of strong evidence to support the use of TXA in craniofacial surgery, there are minimal side effects.13,22 The studies included in our review did not report any side effects from TXA administration. However, there are many large sample studies from the adult population that reported deleterious sequelae from TXA use. For example, the risk of TXA-induced seizures was noted to be higher in one of the largest studies to investigate the use of TXA in surgery. 24 The Aspirin and Tranexamic Acid for Coronary Artery Surgery trial randomized 4631 patients who were undergoing coronary-artery surgery to either receive TXA or placebo.8,25 The group who received TXA had a higher risk of new-onset seizures postoperatively. This was initially thought to be related to the TXA dosing because the initial protocol was 50 mg/kg bolus and then adjusted to 20 mg/kg with no reduction in overall seizure risk. A subsequent large retrospective study of 11,529 cardiac patients found that TXA was an independent predictor of postoperative convulsive seizures particularly in patients receiving continuous infusion with cumulative doses (>80 mg/kg). 25 TXA is known to cross the blood-brain barrier and cause a hyperexcitability state. The mechanism of seizure activity is postulated to be due to competitive inhibition of glycine and GABA receptors, which subsequently abolishes GABA-mediated inhibition in the central nervous system and led to excitability.26,27 Typically, the seizures occur within 5 to 8 hours after TXA infusion and commonly present as tonic-clonic seizures lasting several minutes, but rarely progress to status epilepticus state. There was only 1 case report of a TXA-induced clinical seizure in an infant who underwent an open cardiac surgery and TXA infusion for 6 hours of circulatory support. 10 The most rigorous examination of the safety profile of tranexamic acid was from a multicenter collaborative study that evaluated perioperative complications in craniosynostosis surgery for patients receiving tranexamic acid (n = 1638). 13 In that study, the authors reported a 0.3% rate of seizures and no thrombotic events related to tranexamic acid. The rate of seizures was similar to the group those did not receive tranexamic acid. 13 Nevertheless, it is possible that the rate of reported seizure complications is underestimated given that most children receiving TXA while paralyzed and anesthetized therefore, seizure could be undetected. Future studies should be performed to examine the efficacy of tranexamic acid use in different clinical scenarios and identify the associated risks.
There are several limitations inherent to this study. The quality of a systematic review relies on the quality of the assessed studies. The heterogeneity of the individual studies included in this review reduces the generalizability of the study. Although TXA is effective in decreasing blood loss in trauma, orthopedic, and cardiac adult surgery, its efficacy in pediatric plastic surgery is not proven. Furthermore, there is no standardization regarding the administration route or dosage in pediatric populations. The studies included in our analysis utilized variable protocols of TXA dosing and administration. Analyses such as funnel plot analyses were not performed, and the potential for publication bias was not analyzed in our review. Considering the limitations of the present study, large, multicenter, randomized control trials are conducted specifically in pediatrics craniofacial and orthognathic surgery. Despite these limitations, this review summarizes the current literature about the use of TXA in pediatric and orthognathic surgery.
Conclusions
This review summarizes the current literature on tranexamic acid use in pediatric craniofacial and orthognathic surgery. There is a paucity of information on TXA for craniofacial and orthognathic use. The available evidence has variable administration and dosing protocols. This calls for larger, multi-center randomized trials with a standardized administration protocol to provide definitive evidence regarding the efficacy of TXA use in pediatric craniofacial and orthognathic surgery. Although the role of TXA in plastic surgery is promising, further investigations are needed to standardize the optimal administration, route, and dosage.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
