Abstract
Reconstruction of large segmental mandibular defects presents a challenge for oral and maxillofacial surgeons, particularly in the skeletally immature pediatric patient. Autogenous bone graft is historically preferred; however, harvest of autograft requires a second surgical site, risking donor-site complications as well as the potential for long-term complications in the growing child. Here, we present the first known report of a pediatric patient who underwent immediate mandibular reconstruction of a 6.5-cm long segmental defect using a cellular bone allograft (VF-CBA) combined with custom-fabricated guides and plates following tumor resection. The use of VF-CBA, along with the custom guides and plates, eliminated the need for autograft harvest in a child, enabled an entirely intraoral approach, avoiding the creation of a cutaneous scar, and reduced the total operative time, resulting in a fast recovery and improved patient satisfaction. By 7 months postoperative, the patient’s mandible was fully healed with solid osseous consolidation. These results support VF-CBA combined with custom intraoral guides and plates as an effective treatment option for reconstruction of large segmental mandibular defects in a pediatric patient.
Keywords
Introduction
Segmental mandibular continuity defects are commonly encountered in oral and maxillofacial surgery and remain a challenge to treat. The primary goal of reconstruction of the mandible is to reestablish the full functionality and esthetics of the lower jaw—in particular, restoration of chewing, phonation, and breathing functions—returning the patient to the pre-diseased state.1,2
Acquired segmental defects of the mandible can be caused by a number of etiologies, but are most commonly a result of tumor ablative surgery. 1 One such benign tumor, juvenile active ossifying fibroma (JAOF), is a rare variant of the conventional ossifying fibroma affecting children and young adults, typically less than 12 years of age.3-5 JAOF is a locally aggressive fibro-osseous lesion that is characteristically well-circumscribed, lacking continuity with adjacent bone. Therefore, surgical excision is most often the treatment of choice, with excellent prognosis given complete resection. However, resection often leads to the creation of a large segmental continuity defect, requiring reconstruction of the jaw.
Depending on the complexity and size of the segmental defect, reconstruction of the mandible can be a formidable task for surgeons. Autogenous bone—commonly taken from the iliac crest—is the historically-preferred graft option because it has the potential to include all 3 essential bone remodeling properties: osteoconductivity, osteoinductivity, and osteogenicity. 6 However, it is associated with various limitations, including limited quality and quantity, donor-site morbidity, and risk of donor-site infection.7,8 A newer alternative to autograft, cellular bone allografts (CBAs), are designed to provide the same bone forming properties of autograft without the associated drawbacks. In particular, an advanced CBA, ViviGen Formable® (VF-CBA; LifeNet Health®, Virginia Beach, VA), was developed to contain viable lineage-committed osteogenic cells in an osteoconductive corticocancellous matrix combined with an osteoinductive demineralized bone matrix, possessing the advantages of autograft while eliminating the risks. VF-CBA meets the criteria set forth by the US Food and Drug Administration (FDA) in 21 CFR Part 1271.10 to be classified as a 361 Human Cells, Tissues, and Cellular and Tissue-Based Product (HCT/P), and therefore its use as a bone void filler in both adult and pediatric patients is a homologous use.9-11 This case report describes the use of VF-CBA to successfully reconstruct a large segmental mandibular defect following JAOF resection in a pediatric patient.
Case Report
This case report is in accordance with the World Medical Association Declaration of Helsinki, as revised in 2013. Written informed consent was obtained for publication of these case details. This case was performed by the first author (DA) at the author’s hospital-based practice.
A 10-year-old male was referred to the Department of Oral and Maxillofacial Surgery, Houston Methodist Hospital, for left mandibular jaw expansion. The expansion was first noted 1 month prior and had continued to slowly increase thereafter. The patient was otherwise asymptomatic with no other pertinent family or medical history. Computed tomography (CT) imaging revealed a large (3.4 × 2.2 × 1.9 cm) mixed hypodense-hyperdense expansile lytic lesion in the left body/angle of the mandible (Figure 1). The lesion was noted to be unilocular and well-circumscribed, characteristic of JAOF. A diagnostic biopsy of the tissue was performed and confirmed JAOF. It was determined that the lesion would be removed by surgical resection.
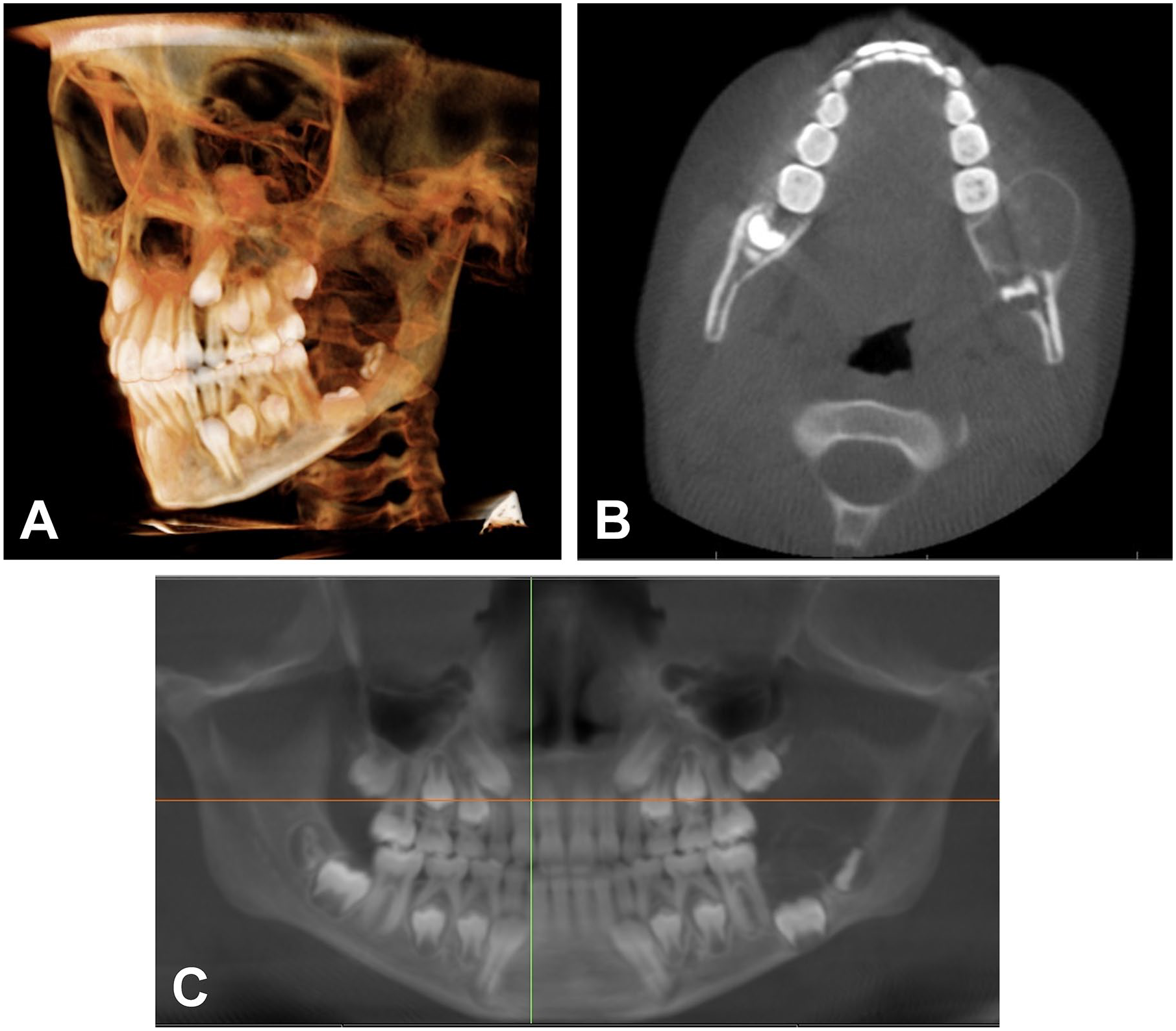
Preoperative computed tomography (CT) image (A), coronal CT section (B), and reconstructed panoramic radiograph (C) showing a large mixed hypodense-hyperdense expansile lytic lesion.
Based on the preoperative CT images, a customized 3-dimensional virtual surgical plan was developed for resection of the JAOF lesion. Correspondingly, custom-fabricated titanium intraoral guides and plates (TRUMATCH® CMF Personalized Solutions, DePuy Synthes, West Chester, PA) were designed and created specifically for this case.
First, an intraoral incision was made to access the left mandible and a supraperiosteal dissection was performed over the area of the tumor and extending anteriorly to the mandibular symphysis. After placement of the anterior cutting guide using 2 fixation screws, the holes were pre-drilled, and the anterior cut was initiated. The guide was then removed, tooth #K was extracted, and the anterior cut was completed.
Next, turning to the left posterior mandible, the temporalis was dissected off the coronoid and the posterior cutting guide was placed with 2 fixation screws. The holes were pre-drilled, the posterior cut was made, and the cutting guide was subsequently removed. The left mandibular segmental resection, including the JAOF lesion and measuring 6.5 × 3.5 × 3 cm (Figure 2), was then completed. Gross evaluation indicated clear margins along all edges of the specimen, which was confirmed by histopathology (data not shown).
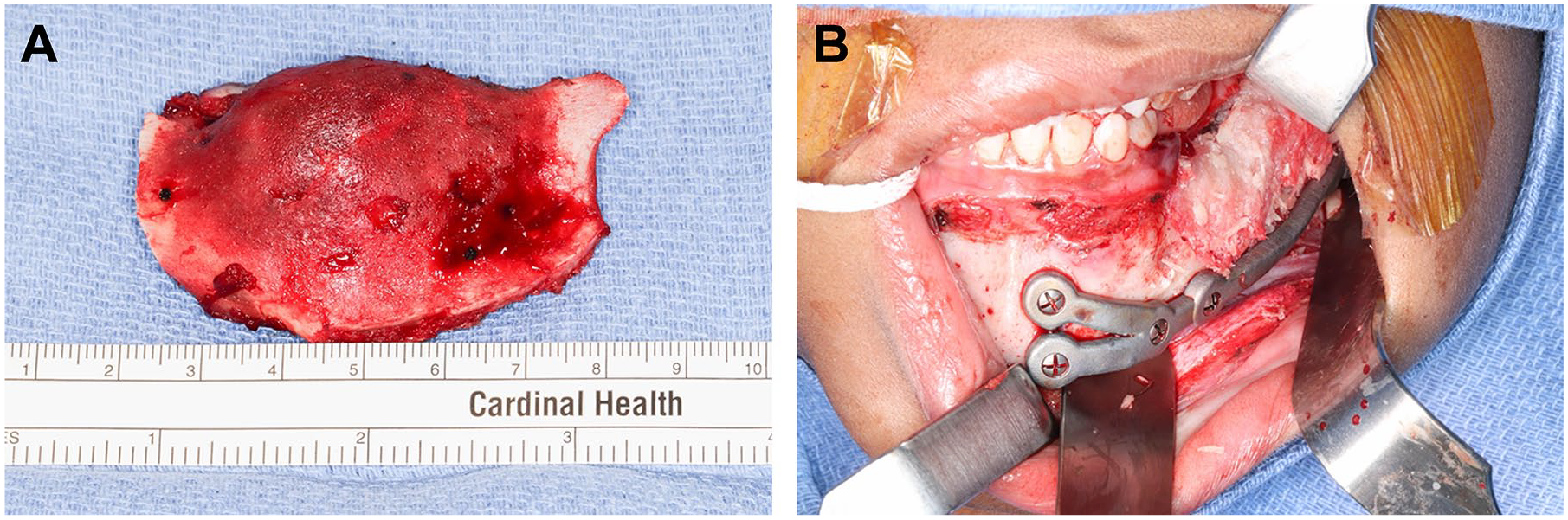
Intraoperative images. (A) Segment resected from the left mandible, including the JAOF lesion. The resected specimen measured 6.5 × 3.5 × 3 cm and had clear margins. (B) VF-CBA implanted in the segmental continuity defect where the JAOF lesion had been removed.
Upon completion of the left mandibular segmental resection, a custom-fabricated plate was fixed to the proximal and distal segments using the pre-drilled holes. The defect was then filled with 5 cc VF-CBA. The demineralized bone fibers contained within VF-CBA result in the graft being moldable making it easy to manipulate and place into desired bone voids, and offering stability within these voids with no appreciable migration. The site was then closed using 4-0 VICRYL® sutures (Ethicon, LLC, Somerville, NJ). The total operative time of the procedure was less than 3 hours. The patient did not experience any postoperative complications and was discharged from the hospital the same day of surgery as an outpatient procedure.
The patient returned at 3 months postoperative and reported doing well with no complications. CT imaging revealed early osseous consolidation where VF-CBA was implanted (Figure 3), evidence of ongoing healing at the defect site. By 7 months postoperative, solid osseous consolidation was noted on CT scans (Figure 4), demonstrating complete healing of the segmental continuity defect where VF-CBA was implanted. Altogether, the patient had a very uneventful recovery.
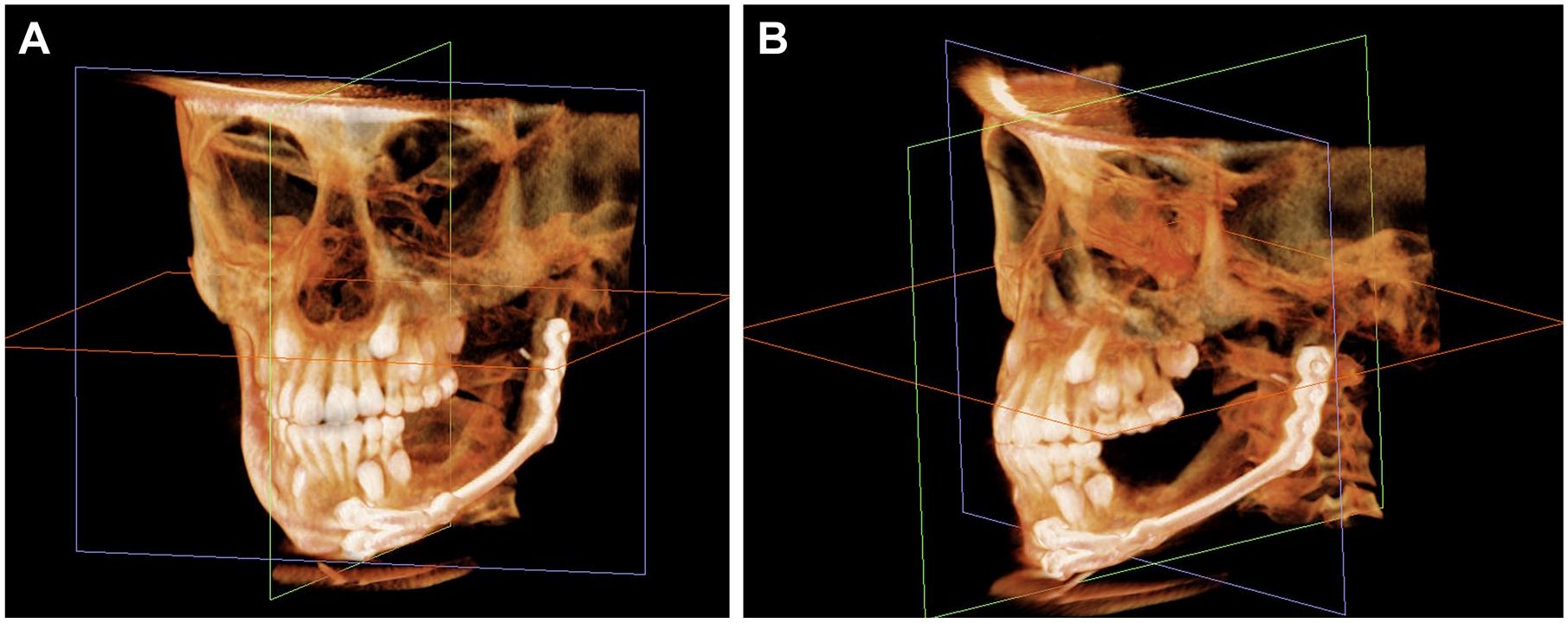
CT images taken at 3 months postoperative showing oblique (A) and lateral (B) views. Early osseous consolidation was noted where VF-CBA was implanted, indicating ongoing healing.
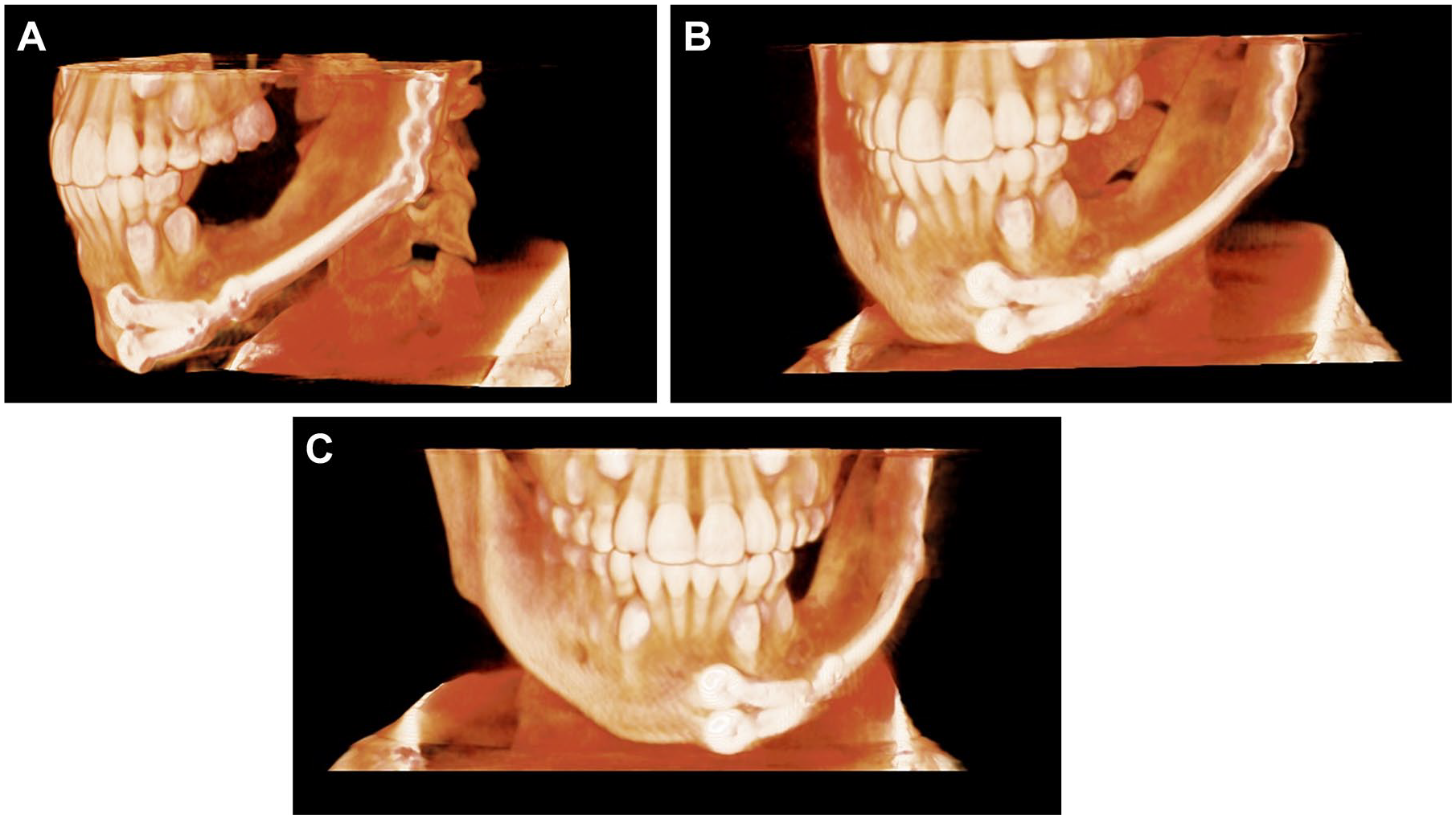
CT images taken at 7 months postoperative showing lateral (A), oblique (B), and anteroposterior (C) views. Solid osseous consolidation was evident where VF-CBA was implanted, indicating successful reconstruction of the segmental continuity defect.
Discussion
Mandibular reconstruction of large segmental defects presents an ongoing challenge for oral and maxillofacial surgeons. This is of particular concern in young children who are still growing, requiring the restoration of functionality and facial esthetics while not restricting growth. 12 Here, we report the successful immediate reconstruction of a large segmental mandibular defect using a viable cellular bone allograft, ViviGen Formable (VF-CBA), in a pediatric patient following JAOF resection. By providing all 3 essential bone remodeling properties, VF-CBA facilitated fusion at the defect site. Although VF-CBA has been used successfully to achieve fusion in spine and orthopedic applications,13-18 its use in oral and maxillofacial surgery is an emerging application,19-21 and in particular, to our knowledge, has not yet been shown in a pediatric patient. Therefore, this is the first known report of a successful immediate reconstruction of a large segmental mandibular defect using VF-CBA in a young child.
Pediatric mandibular reconstruction is typically accomplished using autogenous bone grafts, commonly taken from the iliac crest or fibula. 12 However, autograft harvesting in a growing child is not without potential complications. These include the typical complications associated with donor-site harvest, such as pain and infection, as well as increased operative time due to the complexity of the procedure and, potentially, increased length of hospital stay. 8 Additionally, there are potentially long-term effects related to compromised growth at the donor-sites in the skeletally-immature patient.12,22 Application of VF-CBA eliminated the need for donor-site harvesting of autograft, thereby avoiding these perioperative and potentially long-term complications. At the same time, the custom-fabricated intraoral guides and plates provided an improved efficiency in the procedure, allowing precise placement of the plates and stability to the defect for placement of the VF-CBA graft. The combination of these enabled an entirely intraoral approach, avoiding the creation of a large cutaneous scar, shortening the operative time to under 3 hours, which in turn translates to reduced operative costs, and reducing the risk of perioperative complications known to be associated with both autograft harvest and extraoral approach.7,8,23 Consequently, the patient was able to return home on the same day of surgery, had an uneventful and quick recovery, and ultimately was satisfied with the outcomes, all of which are especially advantageous for a young child.
Reconstruction of larger segmental defects in the mandible has been an area of continued debate for many years. The traditionally held belief is that defects larger than 6 cm should be reconstructed using only a vascularized graft, due to the purported increased failure rates of non-vascularized grafts in defects of this size.24,25 Early on, Pogrel et al, 25 first reported a correlation of failure of non-vascularized grafts (24%) with length of the defect. This was further emphasized by Foster et al, 24 who advocated that defects larger than 5 to 6 cm should only be reconstructed using vascularized grafts. However, use of a vascularized free flap is associated with several limitations, including donor-site morbidity, increased operative time, as well as increased length of hospital stay. 26 An increasing body of evidence is challenging this 6-cm rule, showing success of non-vascularized bone grafts used in reconstruction of larger segmental mandibular defects.21,26-31 A recent meta-analysis found no relationship between non-vascularized grafts and compartment or size of the defect. 30 Schlieve et al 29 demonstrated that, of 9 cases with segmental defects greater than 6.8 cm following resection for benign disease, all were successfully reconstructed using a non-vascularized graft. Nandra et al 28 reported successful reconstructions in 93% of 14 cases in which non-vascularized bone grafts were used for segmental defects greater than 6 cm. In another study, Marechek et al 31 found that 91% of 22 patients with defects greater than 6 cm had successful reconstructions using non-vascularized bone grafts. Finally, in 2 recent publications by Marschall et al, successful reconstruction of large segmental defects using non-vascularized grafts was found following resection for both osteomyelitis 27 (mean defect size of 7.1 ± 2.6 cm for successful grafts, n = 15) and benign disease 21 (mean defect size of 6.5 ± 2.0 cm for successful grafts, n = 42), 2 of which included the use of VF-CBA. 21 In the present case, a non-vascularized graft, VF-CBA, was successful in reconstructing a 6.5-cm long segmental defect following benign tumor resection, further supporting the use of non-vascularized grafts in defects greater than 6 cm. A notable commonality among this and the previous studies is that the patients had not undergone radiotherapy, which can compromise the integrity of the soft tissue bed. 1 Thus, provided adequate soft tissue bed, the use of a non-vascularized bone graft, such as VF-CBA, to reconstruct segmental mandibular defects greater than 6 cm has shown considerable success and is becoming increasingly accepted.
Another area of question among oral and maxillofacial surgeons is the decision for immediate versus delayed, or staged, mandibular reconstruction to treat segmental defects. Historically, delayed reconstruction was the preferred choice, with the primary concern of immediate reconstruction being the risk of contamination and infection of grafts exposed to the oral cavity. 29 For this reason, surgeons have advocated for delaying reconstruction until an oral seal has been achieved. However, unsatisfactory esthetics (eg, facial asymmetry), speech incompetence, and hardware failure are among the drawbacks associated with delayed reconstruction, 29 as well as a prolonged treatment plan with the requirement for a second surgical procedure. A number of reports have demonstrated successful immediate mandibular reconstruction in cases of benign disease, including the present case, as well as in cases of acute or chronic infection.21,27,29,32,33 In one of the earliest and most cited evaluations of simultaneous resection and reconstruction, Obwegeser 33 demonstrated success in intraoral approach for immediate mandibular reconstruction in patients with both benign disease and gross infection. He emphasized that absolute immobilization coupled with well-approximated, tension-free soft-tissue closure, and with antibiotic treatment, as applicable, are essential for success. In this case, rigid internal fixation was achieved with the use of custom-fabricated plates, along with adequate tension-free soft tissue closure, which, combined, provided the conditions needed for a successful immediate reconstruction. Therefore, in the carefully selected patient—that is, no previous radiotherapy, rigid internal fixation, and effective treatment of infection, if applicable—immediate reconstruction in a single surgical procedure is a viable therapeutic option that can offer shorter overall treatment for the patient.
In summary, the present case study is the first known report of the successful use of VF-CBA—an advanced cellular bone allograft—for immediate mandibular reconstruction in a pediatric patient. The use of VF-CBA with custom-fabricated guides and plates eliminated the need for a second surgical site in a pediatric patient and enabled an entirely intraoral approach, the combination of which reduced the total operative time and provided fast recovery and improved patient satisfaction. As a case report, it is important to note that results from individual cases are not predictive of those in other cases. While not readily generalizable, the results from this case support the continued research for VF-CBA use in oral and maxillofacial surgery, consistent with previously published results.19-21
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: E.C.G. is an employee of LifeNet Health, the non-profit organization that processes ViviGen Formable used in this case report. D.M.A., A.H., and S.M.E. report no conflicts of interest.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
