Abstract
Early seizures are common among infants and toddlers hospitalized with mild-moderate traumatic brain injury (TBI) and are associated with unfavorable outcomes. We aimed to develop a risk prediction model for early seizures in this population to support clinical care. Patients <3 years old admitted to our pediatric intensive care unit after mild to moderate TBI (initial Glasgow Coma Scale score 9–15) between 2011 and 2024 were included. The outcome was early electrographic or clinical seizures within the first 3 days of admission. Variable selection was performed using Lasso regression. Variables selected by Lasso were subsequently included in a multivariable logistic regression model, trained using repeated fivefold cross-validation. Model discrimination was assessed using area under the receiver operating characteristic curve (AUROC) and area under the precision-recall curve (AUPRC). Calibration was evaluated with Hosmer–Lemeshow tests and reliability plots. Thresholds for continuous variables were chosen using locally estimated scatterplot smoothing. Coefficients were rounded to the nearest integer to create a point system to facilitate bedside model risk calculation. All values are reported with 95% confidence intervals in parentheses. Of 796 patients who met inclusion criteria, 52 (6%) had early seizures, electroencephalography (EEG) was obtained in 180 (23%), and antiseizure medication prophylaxis was administered to 299 (38%). A 9 variable, point-based prediction model was developed that included (points): age <12 months (+2), presentation concerning for seizure (+2), bruising on physical exam (+0.5), subdural hemorrhage (+1.5), intraparenchymal hemorrhage (+1.5), initial hemoglobin ≤ 7.5 g/dL (+1.5), initial prothrombin time ≥ 15 sec (+1.5), sodium ≥ 145 mmol/L (+2), and glucose ≥ 200 mg/dL (+0.5). AUROC was 0.89 (0.84, 0.95), and AUPRC was 0.46 (0.32, 0.59). The goodness-of-fit test showed acceptable calibration (p = 0.22), as did visualization of a reliability plot. Point thresholds of 1 and 3 had sensitivities of 0.96 (0.87, 0.99) and 0.65 (0.51, 0.78) with specificities of 0.44 (0.40, 0.47) and 0.91 (0.89, 0.93), respectively. This predictive model using nine variables in pediatric patients less than 3 years old with mild to moderate TBI could help identify patients at high risk for early seizures and who should receive seizure prophylaxis or EEG monitoring. This model merits additional validation and refinement.
Introduction
Seizures are common in young children admitted with traumatic brain injury (TBI) of any severity, occurring in 2–48% of these patients. 1–6 Early post-traumatic seizures (EPTS), defined as occurring within the first 3 days after TBI is incurred, have been shown to be preventable with antiseizure drug prophylaxis, but there are no guidelines on prophylaxis or other management for children with mild to moderate pediatric TBI.7–9 Risk factors for EPTS include younger age, higher TBI severity (as defined by lower Glasgow Coma Scale [GCS]), and abusive head trauma (AHT) etiology.2,3 AHT is defined as TBI due to physical abuse, which includes shaking or blunt impact, in patients younger than 5 years old.10–13 Since EPTS are shown to be associated with worse outcomes as well, early recognition and treatment could improve outcomes in this vulnerable population.14,15
Antiseizure drug prophylaxis is recommended in published guidelines on severe pediatric TBI (presenting GCS ≤ 8), but there are no guidelines for seizure prophylaxis for mild to moderate pediatric TBI (presenting GCS 9–15). 8 Certain postinjury findings, such as subdural hemorrhages or ischemia, are known risk factors for seizures, but there is wide variation in antiseizure drug prophylaxis practice.16–19 While antiseizure drug prophylaxis reduces EPTS, the literature is mixed on the drug of choice, contributing to the prescribing variability.20–23 There are also potential risks to antiseizure drug prophylaxis that include side effects such as psychobehavioral cognitive effects, motor instability/ataxia, and concerns for the medications contributing to long-term neurodevelopmental delay, as well as costs to the patients.24–28 Among children with AHT, individualized antiseizure drug prophylaxis may help to prevent seizures while avoiding unnecessary medication initiation in children who are low risk.29 As EPTS are associated with worse outcomes, can be clinically challenging to detect, and require resource-intensive electroencephalography (EEG) to confirm, a well-constructed predictive model to characterize risk for EPTS is a promising approach to accurately identify more at-risk patients while continuing to promote appropriate stewardship of both EEG and antiseizure drug prophylaxis.29–31
In pediatric patients with mild or moderate TBI, as defined by an admission GCS score of 9–15, the decision to obtain an EEG can be challenging. While some children have clearly abnormal neurological exams that warrant EEG monitoring, many appear neurologically intact despite serious injury. In addition, EEG monitoring is a resource-intensive tool that requires technicians, EEG machines, and neurologists, which are not always readily available.32–34 This limitation makes clinical stratification important to ensure patients obtain the appropriate services. In AHT, nonconvulsive seizures are common, affecting 57–65% of these patients.35–37 A 2015 article by O’Neill and colleagues found that 26% of their pediatric TBI cohort presented with a mild or moderate GCS score, and 74% of the patients who seized were diagnosed with AHT. 37 Given limited EEG resources and the high seizure risk in some seemingly well-appearing children, better tools are needed to identify those who would benefit most from EEG monitoring.
Our objective was to create a prediction model to assess the risk of EPTS in patients under 3 years old with mild to moderate TBI, a population that includes a large proportion of AHT. The intended purpose of such a model is to detect occult seizure risk, which could assist clinicians in deciding who should receive antiseizure drug prophylaxis and/or EEGs.
Methods
The University of Pittsburgh Institutional Review Board (IRB) approved this retrospective chart review (STUDY21060042). Procedures were followed in accordance with the Helsinki Declaration of 1975. Patients were included if they were under 3 years old, admitted to the UPMC Children’s Hospital of Pittsburgh pediatric intensive care unit (PICU) between January 1, 2011, and July 31, 2024, with a diagnosis of TBI and a presenting GCS score above 8. The GCS score was the first obtained upon presentation to the emergency department. Mild GCS was defined as a score between 13 and 15 and moderate between 9 and 12. Patients were excluded if they had penetrating head trauma.
Variables abstracted from the electronic health record (EHR) included age, TBI etiology (AHT vs. accidental TBI [aTBI]), history of presenting illness, physical exam findings, type of intracranial hemorrhage, EEG results, presence of antiseizure drug prophylaxis administration, initial laboratory values (hemoglobin [g/dL], platelets [×10e9/L], prothrombin time [sec], partial thromboplastin time [sec], international normalized ratio [INR], fibrinogen [mg/dL], sodium [mMol/L], glucose [mg/dL], blood urea nitrogen [mg/dL], creatinine [mg/dL]), and admission GCS score. An independent radiology review was not carried out, but at our institution, a pediatric neuroradiology attending reads all the neurological images, including those obtained at another hospital.
The primary outcome was the presence of EPTS, defined as documented clinical or electrographic seizures within the first 3 days of admission. Children were classified as having AHT if the Child Protection Team determined the TBI was “highly concerning” or “diagnostic” for AHT. Other blunt TBI etiologies were classified as aTBI. In the history of presenting illness documented in the EMR, a patient was characterized as having a presentation concerning for seizure if a caregiver described an episode concerning for a seizure prior to medical care. Antiseizure drug prophylaxis was defined as any antiseizure drug given before seizure occurrence (clinical or EEG). During this time period, there were no institutional guidelines for mild to moderate TBI regarding which patients to give antiseizure drug prophylaxis or to obtain an EEG. Thus, both giving antiseizure drug prophylaxis and obtaining an EEG were up to the clinician discretion.
Missing laboratory data (<3%) were imputed. Nonparametric tests, including Wilcoxon rank-sum test, Pearson’s chi-square test, and Fisher’s exact test, as appropriate, were used to compare characteristics between patients with and without EPTS. Lasso regression to predict EPTS was conducted to choose variables that would be available at admission. In the Lasso regression, the following variables were inputted: age in months, presentation concerning for seizure, bruising on physical exam, epidural hemorrhage, subdural hemorrhage, subarachnoid hemorrhage, intraparenchymal hemorrhage, no hemorrhage, sodium, glucose, blood urea nitrogen, creatinine, hemoglobin, platelet count, prothrombin time, partial thromboplastin time, and INR.
Thresholds for continuous variables were initially explored using locally estimated scatterplot smoothing (LOESS) to visualize nonlinear associations with EPTS. Candidate thresholds suggested by these visualizations were then evaluated using iterative multivariable logistic regression using repeated k-fold internal cross-validation (fivefolds). The variable correlation coefficients were then rounded to the nearest half point to assign a point value for each variable. Variable points were then added together for each patient to assign a summed point value. The area under receiver operator curve (AUROC) and area under precision-recall curve (AUPRC) were calculated. Goodness of fit was assessed with the Hosmer–Lemeshow test and visualization of reliability plots. All statistical analyses were performed by using RStudio Version 2024.12.1 + 563 (RStudio, Boston, MA) and R Version 4.4.3 (R Foundation for Statistical Computing, Vienna, Austria) with the following packages: readxl, tidyverse, gtsummary, lubridate, glmnet, and mice.
Results
Of 995 patients admitted to the PICU less than 3 years old with TBI, 39 were excluded for penetrating TBI and 160 were excluded for severe TBI (Fig. 1). There were 796 patients less than 3 years old with mild or moderate TBI. The median age of the cohort was 4 months (interquartile range [IQR] 2, 14), with 38% female and 33% with AHT. Most patients were healthy prior to the TBI (83%). The most common past medical history was prematurity (9%). One patient had a history of a seizure, and two patients had a history of TBI. During hospitalization, 65/796 (8%) had seizures, and 52/65 (80%) were classified as EPTS. In the entire cohort, 299/796 (38%) had antiseizure drug prophylaxis given, and 167/796 patients (21%) had EEGs obtained. Of the patients who had seizures, 15/65 (23%) had clinical seizures, and 51/65 (78%) were diagnosed with AHT. There was no difference in the age between the patients with and without seizures. There were 99 patients with a presentation concerning for seizure, 34 (34%) of whom had a seizure. All patients had neuroimaging, with 492/796 (62%) having head CTs only, 49/796 (6%) having brain MRIs only, and 255/796 (32%) having both head CT and brain MRI. The most common type of intracranial hemorrhage was subdural, followed by subarachnoid, epidural, intraparenchymal, and intraventricular hemorrhages (Table 1). In the 65 patients who seized, most had subdural hemorrhage, and only one had no hemorrhage. Antiseizure drug prophylaxis was used in 304/796 (37%) patients, and all patients who seized were given antiseizure drug prophylaxis (Table 1). The median laboratory values that were significantly different between the patients with EPTS versus those with no seizures were hemoglobin, prothrombin time, INR, and glucose.

Patient flowchart shows the flowchart of patients in terms of seizures and seizure timing.
Patient Characteristics by Seizure Timing

Median (Q1, Q3); n (%).
Kruskal–Wallis rank sum test; Fisher’s exact test.
EEG, electroencephalography; TBI, traumatic brain injury.
The following variables had significant coefficients in the Lasso regression: age in months, presentation concerning for seizure, bruising, subdural hemorrhage, intraparenchymal hemorrhage, initial hemoglobin, initial prothrombin time, initial sodium, and initial glucose (Table 2). For the continuous variables, LOESS was conducted for each and found thresholds for predictability of EPTS: age <12 months, hemoglobin ≤ 7.5 g/dL, prothrombin time ≥ 15 sec, sodium ≥ 145 mMol/L, and glucose ≥ 200 mg/dL (Fig. 2).

Locally estimated scatterplot smoothing (LOESS) for continuous variable thresholds. For glucose
Model Creation

After scaling the variable correlation coefficients, the points assigned for each variable were as follows: age <12 months (+2), presentation concerning for seizure (+2), bruising (+0.5), subdural hemorrhage (+1.5), intraparenchymal hemorrhage (+1.5), hemoglobin ≤ 7.5 g/dL (+1.5), prothrombin time ≥ 15 sec (+1.5), sodium ≥ 145 mmol/L (+2), and glucose ≥ 200 mg/dL (+0.5). The AUROC was 0.89 (0.84, 0.95), and the AUPRC was 0.46 (0.32, 0.59) (Fig. 3). The Hosmer–Lemeshow goodness-of-fit test was not significant (Fig. 3). In the Hosmer–Lemeshow plot, predicted probabilities were skewed, with a median value of 0.036 (IQR: 0.008–0.036), and most predictions clustered below 0.05.

Model Performance.
The average points for patients with EPTS were 4.0 (2.0, 4.0) versus 2.0 (0.0, 2.0) in the patients without EPTS (p < 0.01) (Table 3). The reliability plot is available in Supplementary Figure S1. Most patients with EPTS (34/52 [65%]) had 3 points or higher, while most patients without EPTS had less than 3 points (679/744 [91%]). Of the patients with points greater than or equal to 2, 147/468 (31%) obtained an EEG and 232/468 (50%) were given antiseizure drug prophylaxis. Of the patients with points greater than or equal to 3, 83/99 (84%) obtained an EEG, and 91/99 (92%) were given antiseizure drug prophylaxis.
Point System by Seizure
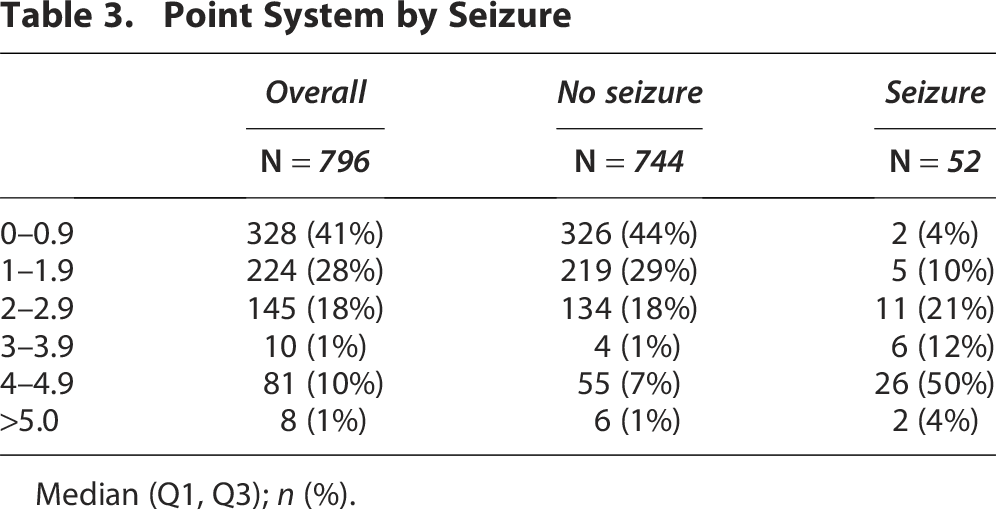
Median (Q1, Q3); n (%).
At a threshold of 1 point in the final model, the sensitivity was 0.96 (0.87, 0.99) and the specificity was 0.44 (0.40, 0.47) with a positive predictive value (PPV) of 0.11 (0.99, 0.12) and a negative predictive value (NPV) of 0.99 (0.98, 0.99). The 1-point threshold would miss 2 patients out of 52 (4%) with EPTS, and there would be 418 false positives. At a threshold of 3 points, the sensitivity was 0.65 (0.51, 0.78) and the specificity was 0.91 (0.89, 0.93) with a PPV of 0.34 (0.28, 0.42) and an NPV of 0.97 (0.96, 0.98). The 3-point threshold would miss 18/52 (34.6%) with EPTS, and there would be 65 false positives.
Discussion
Using more than a decade of data from a quaternary children’s hospital, we developed a high-performing prediction model to identify children with TBI who are at risk for EPTS. Prioritizing parsimony, we ultimately relied on six data elements routinely collected during routine TBI care, enabling straightforward adoption at the bedside. When applied as a point-based system, the model demonstrated strong overall performance, with threshold-based scoring yielding high specificity and NPV.
Our model focuses on patients under 3 years old, a population in which AHT is a leading consideration in the differential diagnosis of TBI and the risk for seizures is correspondingly high.3,4,37–39 We intentionally excluded AHT diagnosis itself as a predictor, as it is rarely established at the time EEG monitoring or seizure prophylaxis decisions are made.40–43 Instead, we selected variables that are both easily obtainable and available at admission, several of which, such as low hemoglobin, bruising, and subdural hematoma, are strongly associated with AHT.11,41,44 Two primary models for predicting AHT exist: PredAHT-2, which incorporates variables from the entire hospitalization, and PediBIRN, which relies on physical exam findings and neuroimaging.45,46 In contrast, our model integrates age, presenting symptoms, laboratory results, and neuroimaging findings, allowing application across a broader range of injury mechanisms and including cases of milder injury severity.
We believe this model can be used as a risk stratification tool to help with clinical decision-making. Lower scores identify patients at low risk for EPTS, while progressively higher scores correspond to increasing seizure risk. A higher score justifies intervention with antiseizure drug prophylaxis or monitoring with EEG, while a patient with a low score may not require anything beyond observation. For example, a 12-month-old infant admitted after a fall with a GCS score of 15, bruising on physical examination, normal laboratory values, and a subdural hemorrhage on imaging would score 2 points, placing them in an intermediate risk category. In this scenario, antiseizure drug prophylaxis without EEG monitoring would be reasonable to prevent EPTS. A 4-month-old presenting with a presentation concerning for seizure, subdural hemorrhage, and a hemoglobin of 7 mg/dL would score 7 points and be considered very high risk for EPTS, warranting both EEG monitoring and antiseizure drug prophylaxis. Further studies are warranted to define thresholds for EEG monitoring and antiseizure drug prophylaxis.
There are no guidelines for antiseizure drug prophylaxis for mild to moderate TBI, and although certain risk factors are recognized, clinical practice varies widely, with reported utilization rates ranging from 9% to 52%.47–52 The most efficacious antiseizure drug prophylaxis is also undefined, making universal prophylaxis challenging given the potential for adverse effects.7,9,20,51,53,54 Older agents, such as fosphenytoin, are associated with ataxia, liver dysfunction, or drowsiness, while the current first-line agent, levetiracetam, commonly causes irritability.55–57 Moreover, prolonged or unnecessary prophylaxis may impair TBI recovery.26,53 Further research is needed to determine optimal prophylaxis strategies, as factors such as drug selection, dosing, and injury endotype likely influence therapeutic efficacy.20,23,58 In our cohort, antiseizure drug prophylaxis was often administered without EEG monitoring, potentially reflecting scenarios where the neurological exam was reassuring but neuroimaging findings were concerning.
In TBI, including AHT, seizures are associated with secondary injury and worse outcomes.16,29,31 The true prevalence of EPTS is uncertain, as many events are nonconvulsive and EEG is not routinely performed in all patients.4,35,36 Based on our model’s thresholds and performance metrics, this tool may aid clinicians in deciding when to obtain an EEG. Key questions arising from our findings include whether the model could guide not only which patients might benefit from EEG but also, given the risk of subclinical status epilepticus, which patients might warrant continuous EEG monitoring. Future investigation is needed, as continuous EEG is resource-intensive and often limited in availability. Current epilepsy guidelines recommend 24/7 real-time supervision by trained personnel with direct access to EEG technologists, nurses, and physicians.59,60 However, a national shortage of nursing and EEG technologists has been documented, and limited or delayed EEG access has been shown to postpone care.60,61 Given that longer seizure duration reduces responsiveness to anticonvulsant therapy and worsens outcomes, prioritizing EEG resources for higher risk patients in this model could be clinically beneficial.62–66 This would target a population who would likely be given antiseizure drug prophylaxis and thus would be more likely to have unrecognized subclinical seizures.
Our study has several limitations. First, the data are from a single center and may not reflect broader clinical practice. Second, the retrospective design raises the possibility that clinical seizures were missed due to incomplete documentation in the EHR or misclassification of antiseizure drug administration as prophylaxis versus treatment. A large proportion of our population received antiseizure drug prophylaxis, which could have altered the rate of seizures. In addition, all patients who had early seizures received antiseizure drug prophylaxis, which could be misclassified as prophylaxis, a signal that prophylaxis is ineffective, and/or that clinicians have a good sense of seizure risk in these patients that was not otherwise documented. Because EEGs were not routinely obtained, nonconvulsive seizures may have gone undetected. We also did not go into detail regarding the number or type of seizure for the patients who did seize or the antiseizure drugs the patients received. Our model also did not leverage the full range of predicted probabilities, which may limit its utility in stratifying patients into distinct high- versus low-risk categories. Finally, although a rationale for model thresholds was provided, these thresholds need to be studied further to determine if they are optimal. Despite these limitations, our model demonstrated excellent predictive performance. Future work should refine using multicenter data and evaluate its utility prospectively.
Conclusion
We developed a predictive model for EPTS in patients under 3 years old admitted to the PICU with mild to moderate TBI. The model demonstrates how to risk-stratify and allows for clinicians to make educated decisions about antiseizure drug prophylaxis and EEG monitoring, with the aim of preventing seizures and enabling early detection and treatment to improve outcomes. Prospective validation will be essential before clinical implementation.
Transparency, Rigor, and Reproducibility Summary
This retrospective cohort study was conducted at a single center and included consecutive patients under 3 years old admitted with mild to moderate TBI from 2011 to 2024. All inclusion and exclusion criteria, as well as outcome definitions, were prespecified and defined in the Methods. STROBE guidelines for reporting observational studies and TRIPOD guidelines for predictive model development were followed. Data were extracted from the EHR using standardized definitions. Due to patient privacy considerations and institutional IRB restrictions, the full dataset cannot be made publicly available. Deidentified summary data and analytic code are available upon request. We used Lasso regression for variable selection followed by multivariable logistic regression to build the predictive model. Thresholds for continuous predictors were determined using LOESS smoothing. Model performance was evaluated using internal 5-fold cross-validation followed by AUROC and AUPRC. Calibration was assessed using the Hosmer-Lemeshow test and reliability plots. This study was approved by the University of Pittsburgh Institutional Review Board (STUDY21060042) and conducted in accordance with the Declaration of Helsinki. Informed consent was waived for this analysis.
Authors’ Contributions
C.R.M. had full access to all the data in the study and takes responsibility for the integrity of the data and accuracy of the data analysis. Concept and design: C.R.M. and C.M.H. Acquisition, analysis, or interpretation of data: All authors. Drafting of the article: C.R.M., P.M.K., and C.M.H. Critical revision of the article for important intellectual content: all authors. Statistical analysis: C.R.M.
Footnotes
Supplemental Material
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
