Abstract
Introduction:
Children with complex chronic conditions (CCC) represent an increasingly prevalent and vulnerable group in pediatric care, accounting for a substantial proportion of hospital admissions and health care expenditures worldwide. These children frequently experience fragmented care and limited coordination during the transition from hospital to home, which may worsen symptom burden and increase the risk of readmission. Early identification of palliative care needs in this population is essential to improving continuity, quality, and equity of care.
Objective:
To describe the clinical and sociodemographic profile of pediatric patients admitted to a Brazilian hospital transition unit and to assess their eligibility for palliative care.
Methods:
We conducted a retrospective cohort study including 249 unique pediatric patients admitted between March 2024 and March 2025. Readmissions were counted as separate events. Data were extracted from electronic medical records and included sociodemographic variables, clinical characteristics, and eligibility for palliative care. Descriptive statistics were complemented by chi-square tests for categorical variables and Kruskal–Wallis tests for nonparametric group comparisons.
Results:
The analysis revealed that almost 80% (n = 198) of patients had at least one CCC; notably, 23.3% (n = 58) required technological support. Longer hospital stays were significantly associated with higher palliative care eligibility (p < 0.01). According to World Health Organization (WHO) criteria, 53.4% of patients met Group 2 eligibility for pediatric palliative care.
Conclusion:
This study highlights the high prevalence of CCC among children admitted to a hospital-to-home transition unit and reveals a persistent gap in care integration within the Brazilian health system. The findings underscore the need for structured protocols to actively screen for palliative needs in transitional pediatric settings. Implementing WHO-based checklists, combined with multidisciplinary team training, may support early identification, appropriate referral, and coordinated, family-centered care during the transition from hospital to home.
Keywords
Introduction
The transition from hospital-based care to home environment is a critical process of modern health care systems, ensuring continuity of treatment, optimizing resource use, and promoting family-centered care. In pediatric populations, this process is inherently complex due to the clinical fragility, technological dependence, and substantial care needs of many children.1,2
Among this population, children with complex chronic conditions (CCC)—defined as serious, long-term illnesses involving multiple organ systems or severe single-organ dysfunction that requires specialized care and recurrent hospitalizations—represent one of the most vulnerable and underserved groups in health care.1–3
These children often experience unpredictable disease trajectories, persistent medical fragility, and significant motor, cognitive, and functional impairments. Many depend on medical and technological support, such as mechanical ventilation or enteral feeding, and most require continuous assistance with basic activities of daily living.1–3
CCC encompasses a wide range of conditions, including neuromuscular disorders, such as cerebral palsy or muscular dystrophies; complex congenital heart diseases (malformations and cardiomyopathies), chronic respiratory diseases (congenital malformations and cystic fibrosis), congenital anomalies and genetic syndromes, as well as oncological diseases and immunodeficiencies.4–6 Ensuring a safe and successful hospital discharge for these children demands careful planning that integrates medical stabilization, caregiver training, and coordination of community-based support.6,7
Pediatric palliative care (PPC), as defined by the World Health Organization (WHO), focuses on relieving health-related suffering and providing interdisciplinary support regardless of diagnosis or prognosis. The WHO framework delineates six patient groups eligible for PPC, including children with acute life-threatening conditions, chronic illnesses with ongoing risk of death, progressive diseases without curative options, and severe nonprogressive neurological conditions with risk of deterioration, as well as newborns with extreme prematurity or severe congenital malformations.7,8 Despite this, many eligible children with CCCs receive late or no palliative involvement, leading to fragmented care and excessive reliance on intensive care services.8,9
Hospital-to-home transition units have emerged as promising models to bridge the gap between acute hospital care and community-based services. These units help ensure medical stability, prevent avoidable readmissions and support caregivers through structured training and coordinated follow-up. By serving as an intermediate level of care, they reduce the risk of unplanned readmissions and adverse events in the immediate post-discharge period.7–9 However, evidence remains limited on how these units can systematically identify palliative care needs and initiate early PPC referrals—especially in low- and middle-income countries (LMICs), where pediatric palliative care infrastructure is still developing.
Brazil illustrates this gap. Although the Unified Health System guarantees universal health coverage, pediatric palliative services remain unevenly distributed and often under-resourced. Standardized screening protocols for PPC eligibility and access to multidisciplinary PPC teams are scarce, especially outside major urban centers.10–12 As a result, children with CCC and their families face inequitable access to coordinated, family-centered care planning.
Furthermore, there is limited research on the use of standardized palliative needs assessment tools, the impact of transition-unit interventions on care quality metrics, and the contextual challenges of implementing PPC in middle-income settings. Few studies examine the unique trajectory of pediatric populations in palliative care, which are marked by uncertainties, multiple professionals involved, and highly individualized needs. Understanding how transition units operate in middle-income contexts can provide valuable insights for designing equitable and efficient care pathways.8,13–15
Therefore, the present study aims to describe the clinical and sociodemographic profile of pediatric patients admitted to a hospital-to-home transition unit in Brazil and to assess their eligibility for palliative care using WHO criteria. By identifying the prevalence of CCC and the proportion of children who meet established palliative indicators, this work seeks to inform the development of proactive screening protocols and staff training programs, ultimately promoting safer and more compassionate transitions for children living with serious illness—an underserved population whose needs remain insufficiently recognized in both national policy and the global health literature.
Materials and Methods
Study design
This was a single-center, historical cohort observational study reported in accordance with STROBE guidelines. We conducted a quantitative analysis combining descriptive and comparative methods, using secondary data extracted from electronic medical records and the institutional database maintained by the pediatric hospital-to-home transition team.
Setting
The study was conducted in a tertiary-care public hospital in Brazil, which hosts a 14-bed pediatric hospital-to-home transition unit. This specialized ward was established to provide medical stabilization, caregiver training, and coordination of community-based resources for patients who require complex care prior to discharge. The unit admits an average of 20 patients per month and represents one of the few structured services of its kind in the country, serving a predominantly underserved population with limited access to specialized health care and PPC resources.
Study period and population
We screened 249 unique patients aged 0–18 years who were admitted between March 2024, the month of its inauguration, and March 2025, representing its first complete year of the unit’s operation. The initial dataset included 268 admissions; however, 19 records were excluded because more than 20% of key variables (demographic data, clinical diagnosis, technology dependence, or palliative eligibility) were missing. Readmissions were counted as separate events but were included only in descriptive analyses of technology-dependent children.
Data collection and variables
Data were extracted from electronic medical records by two trained researchers using a standardized collection form. Variables were grouped into three domains:
Demographic variables: gender, age at admission (categorized according to standard pediatric age groups), race/ethnicity (Brazilian Institute of Geography and Statistics [IBGE] classification), and region of residence. Clinical variables: primary diagnosis, presence of CCC (according to Feudtner classification
15
), length of hospitalization, readmissions, responsible specialty, reason for admission, prior specialty follow-up, technology dependence (tracheostomy, mechanical ventilation, enteral feeding), and functional status, when available.
Functional status was assessed using the Lansky Play-Performance Scale, which ranges from 100 (fully active) to 10 (completely disabled), providing a validated measure of pediatric functional performance.
Palliative care eligibility
Eligibility for PPC was determined according to the WHO criteria, by two independent physicians’ reviewers, with consensus required for final categorization.7,8
Group 1: acute life-threatening conditions with uncertain recovery; Group 2: serious chronic diseases that can be cured or controlled for the long term but still carry an ongoing risk of death; Group 3: progressive diseases for which no curative treatment exists; Group 4: severe, nonprogressive neurological conditions with risk of clinical deterioration and death; and Group 5: extreme prematurity or severe congenital malformations.
Group 6, which refers to bereaved family members after perinatal loss (fetal and neonatal deaths), was not applicable to this setting and was therefore excluded.
Data analysis
Categorical variables were summarized as absolute frequencies and percentages. Continuous variables were described using measures of central tendency (means or medians) and dispersion (standard deviation or interquartile range), according to the normal distribution of data.
Categorical variables were compared using the chi-square test, and continuous variables were compared using the Wilcoxon Rank Sum or Kruskal–Wallis test, as appropriate. Statistical significance was defined as p < 0.05. All analyses were performed using R version 4.3.1.
Ethical aspects
The study was approved by the institution’s Research Ethics Committee, under protocol No. 6,597,366/2024. Because the investigation was retrospective and based exclusively on de-identified secondary data extracted from electronic medical records, informed consent was waived. All ethical principles were strictly followed, including the confidentiality of information and the protection of sensitive patient data.
Results
Population characteristics
During the 12-month study period, 249 pediatric patients were admitted to the hospital-to-home transition unit and met the inclusion criteria. Slightly more than half were male (58.2%), and the majority were identified as white (61.8%), followed by multiracial (30.5%) and black (6.8%) according to IBGE classifications. Age ranged from infancy to late adolescence, with two notable peaks in distribution: school-age (6–10 years, 28.9%) and pre-adolescents (11–15 years, 27.7%), as shown in Table 1 and Figure 1.
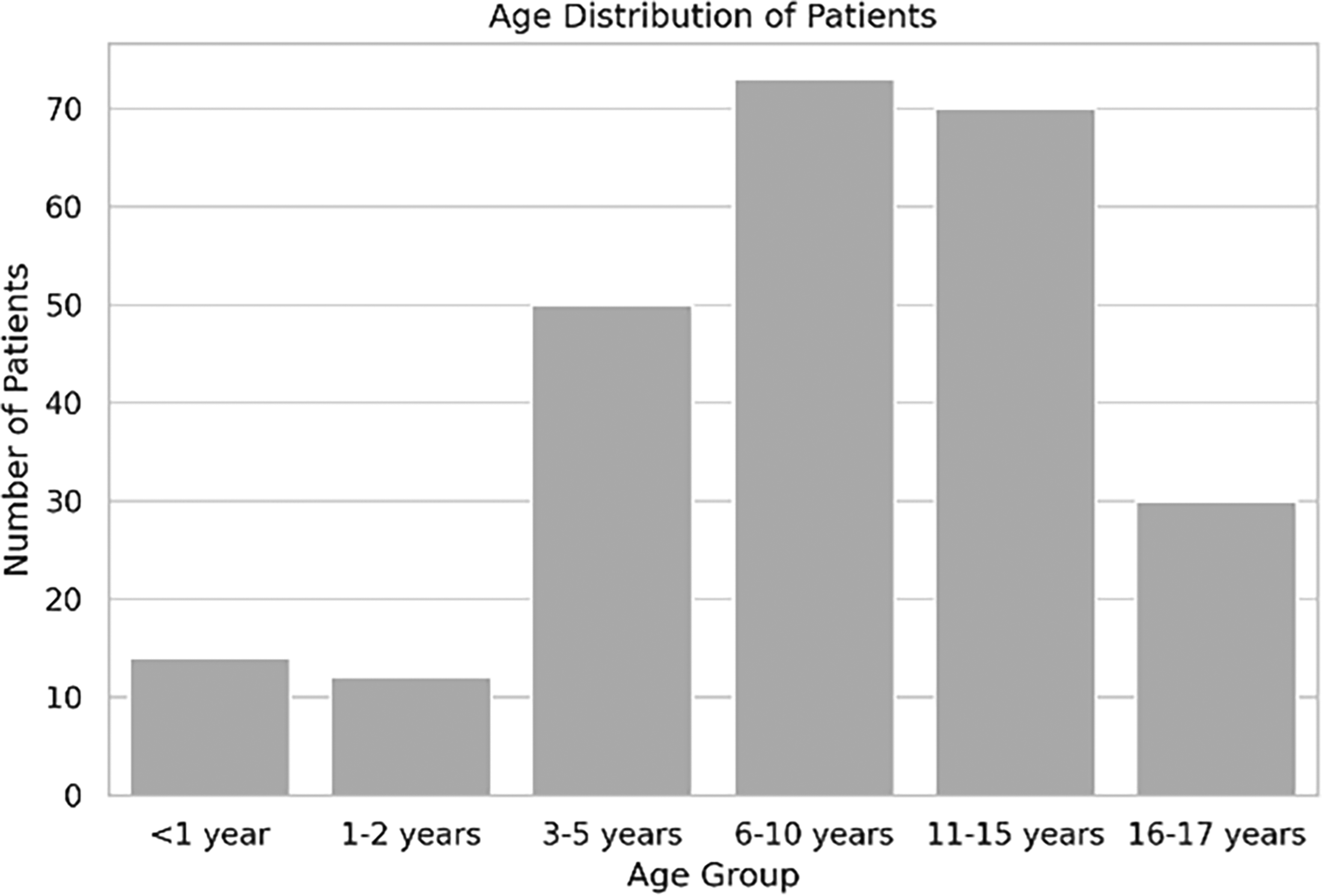
Distribution of pediatric patients admitted to the hospital-to-home transition unit by age group.
Sociodemographic and Clinical Characteristics of the Sample

Complex chronic conditions
A striking 80% of the cohort had at least one CCC, underscoring the clinical vulnerability of this population. Neurological disorders were the most common CCC (18.5%), including chronic encephalopathy, cerebral palsy, and refractory epilepsy. Respiratory conditions—such as cystic fibrosis and bronchiolitis obliterans—accounted for 13.7%, while cardiovascular disorders including tetralogy of Fallot and other congenital heart defects, comprised 7.6%, as demonstrated in Figure 2.

Distribution of pediatric patients admitted to the hospital-to-home transition unit by presence of complex chronic conditions.
Hospitalization patterns
Hospital stays were generally brief. Nearly half of the patients (48.2%, n = 120) were discharged within 6 days, and an additional 30.9% (n = 77) were discharged within 7–10 days. Longer hospitalizations were less common: 13,2% patients (n = 33) stayed for 11–14 days, 3.6% (n = 9) stayed between 15 and 20 days and only 4% (n = 10) remained hospitalized for more than 20 days (Table1).
Technology dependence was present in 23.3% (n = 58) of patients and was associated with significantly different hospitalization characteristics. The most frequent technology-dependent devices included gastrostomy tubes (51.7%), tracheostomy (27.6%), and noninvasive ventilation (6.9%). This subgroup experienced longer hospital stays and higher readmission rates compared with their nondependent peers (p < 0.001), as shown in Table 2.
Demographic and Clinical Profile of the Cohort, Showing Age Distribution, CCC Prevalence by Category, and Hospitalization Patterns

CCC, complex chronic conditions; SD, standard deviation.
The majority of patients (82.3%) were discharged with clinical improvement, 12.4% were transferred to other services, and 5.2% required readmission. Among technology-dependent patients, the primary reasons for admission were rehabilitation and caregiver training (32%); completion of antibiotic therapy (29%), oxygen weaning (16%), and stabilization of chronic conditions (14%).
Palliative care eligibility
Overall, 53.4% of the cohort met WHO criteria for PPC. Eligibility varied markedly by technology status: 48.4% of technology-dependent patients met formal criteria compared with only 8.2% of nondependent patients (p < 0.001). Functional status assessments using the Lansky Play-Performance Scale further emphasized these differences (mean score 61.3 vs. 85.2; p < 0.001).
Using the WHO classification, patients were categorized into Groups 1–5 (Table 3):
Distribution of Patients by Indication Group

Only two patients met the criteria for Group 5 (extreme prematurity or severe congenital anomalies). Conversely, 20.7% of the sample comprised the “Not Applicable” category, most often presenting with acute, self-limiting conditions such as community-acquired infections or elective surgical procedures. Although not formally eligible for PPC, many benefited from symptom relief measures and family guidance, highlighting the value of a person-centered approach across all clinical scenarios.
Hospitalization duration varied significantly across WHO palliative care groups (p < 0.001). Group 1 (acute life-threatening conditions) and Group 4 (severe neurological impairment) require the longest hospital stays and the highest risk of adverse events. This stratification was clinically relevant, as palliative care group assignment strongly predicted discharge outcomes (p < 0.001), indicating that children with greater clinical complexity face worse trajectories.
There was a robust association between the presence of CCC and PPC eligibility (χ2 p < 0.001). Technology-dependent children showed the highest palliative care needs with 35.5% classified in Group 4 and 32.3% in Group 2, highlighting a subgroup requiring multidisciplinary PPC involvement.
Together, these findings demonstrate that children admitted to a hospital-to-home transition unit—particularly those who are technology-dependent—represent a high-risk, underserved population with complex medical and social needs. Technology dependence was consistently associated with lower functional scores, longer hospital stays, and higher eligibility for palliative care. These results underscore the need of structured screening protocols, enhanced caregiver training, and integrated multidisciplinary management to ensure early identification of palliative needs and safe discharge planning.
Discussion
This study provides important insights into the clinical and epidemiological profile of children admitted to a hospital-to-home transition unit in Brazil, offering an important contribution to the limited global literature on pediatric transitional and palliative care in middle-income settings. Three key findings stand out with direct implications for policy and practice: (1) high prevalence of CCC (80% of our cohort) consistent with global trends in rising pediatric medical complexity, (2) the strong association between technological dependence emerges as a powerful predictor of health care utilization, including longer hospital stays, higher readmission rates, and lower functional scores, and (3) the close link between palliative care eligibility, CCC status and technological dependence.
Together, these findings underscore the strategic potential of hospital-to-home transition units as platforms for early identification and coordination of PPC, filling a critical gap in both current Brazilian and international pediatric health care systems and literature.16–18
Early identification of palliative needs
The strong association observed between CCC and PPC eligibility (p < 0.001) aligns with international evidence showing that early recognition of palliative needs improves care continuity and reduces preventable hospitalizations. Studies from high-income countries have similarly demonstrated that children with medical complexity represent up to 60% of PPC referrals and face higher readmission rates when palliative integration is delayed.17–20
In our cohort, nearly one-quarter of patients were technology-dependent, and this group demonstrated longer median lengths of hospital stays and higher readmission risk, findings consistent with those of Feudtner et al. and Berry et al., who identified technological dependence as a marker of medical fragility and care fragmentation.18,20 Incorporating standardized functional tools into routine assessment may facilitate timely referral and ensure the early integration of palliative principles during hospitalization.
Implications of WHO group distribution
The predominance of WHO Group 2 patients (53.4%) in our cohort demonstrates the essential role of “concurrent care,” in which palliative support is provided alongside disease-directed treatments. These findings mirror from European PPC registries, where Group 2 conditions represent the majority of eligible pediatric cases, often overlooked due to the misconception that palliative care is restricted to end-of-life settings.21,22
Although fewer in number, Group 1 patients exhibited the longest hospital stays, illustrating the value of PPC involvement during acute crises may reduce suffering, improve communication and guide shared decision-making for families. These results advocate for universal screening using WHO criteria within 24 hours of admission, an approach already implemented in several high-income pediatric centers to improve early identification and care coordination. 23
Hospitalization patterns and functional stratification
The bimodal distribution of length-of-stay, characterized by rapid discharge for about half the cohort and prolonged stays for a smaller subset, indicates the presence of distinct clinical trajectories requiring tailored approaches. This pattern reflects reports from transition programs in the United States and Canada, which highlight the need for customized discharge pathways based on medical stability and functional status.24,25
The use of the Lansky Play-Performance Scale as a stratification tool may offer a low-cost, practical strategy for prioritizing palliative assessment and transition planning in middle-income settings. Functional thresholds (Lansky <60) could serve as operational triggers for PPC referral, similar to what has been successfully adopted in tertiary children’s hospitals in the United Kingdom and Australia. 26
Technology-dependent subgroup
Technology-dependent children formed a distinct high-risk subgroup, reinforcing international findings that this population experiences the highest rates of adverse events, hospital readmission and caregiver burden.27,28 Evidence suggests that structured “transition bundles” that include caregiver training programs, 72-hour post-discharge telehealth follow-up, guaranteed access to medical supplies and integrated home nursing support are effective strategies for improving safety and reducing unplanned returns to hospital care.22–27
By adapting these evidence-based measures to the Brazilian context, transition units could significantly improve safety and reduce preventable complications and hospital returns, addressing a key gap in current pediatric post-discharge systems.
Strengthening home and community support
Our findings highlight persistent deficiencies in home-care and primary-care integration—a challenge widely reported in other LMICs.26–28 Even with national home-care programs focused on long-term mechanical ventilation or enteral nutrition, fragmented communication between tertiary hospitals and local services undermines continuity of care, long-term follow-up, and family confidence.26–28
Studies consistently show that up to one-third of pediatric discharges in high-income settings experience post-hospitalization care failures, often due to inadequate caregiver preparation or gaps in community support. Evidence from our cohort suggests that these challenges may be even more pronounced in middle-income settings, where socioeconomic disparities, resource limitations, and infrastructural constraints further exacerbate risk.29,30
Policy and system-Level recommendations
At the system level, our data support recognizing pediatric transition units as strategic hubs for integrating palliative care into routine clinical practice. Priority actions include: (1) implementing universal palliative screening using WHO criteria at admission; (2) developing standardized care bundles for technology-dependent children, including structured caregiver competency training and medical equipment supply continuity; and (3) ensuring mandatory involvement of PPC teams for children with high-complexity conditions, particularly those with functional impairment or technological dependence.31,32
Emerging international initiatives, such as the ACEPATH Project, illustrate the effectiveness of co-designed transition pathways that prioritize family engagement, cross-sector coordination, and continuous follow-up.30,31 Adapting these models to LMICs will require addressing systemic barriers such as fragmented funding, limited PPC capacity within community services, and insufficient caregiver support infrastructure.
Family-centered and culturally sensitive care
Effective care transitions extend beyond discharge, and they constitute a longitudinal, family-centered process that begins upon admission and continues until the family achieves full adaptation to home care. Actively involving patients and caregivers in planning, through shared decision-making, personalized discharge plans, and culturally sensitive communication, are associated with improved family satisfaction and reduced readmission rates.30–32 Embedding these participatory strategies within transition units could bridge clinical and social care dimensions, promoting safety, autonomy, and dignity for children and their families.
Limitations
This single-center, retrospective analysis may limit generalizability, and referral bias cannot be excluded. Nevertheless, the cohort is representative of a large public hospital serving an underserved population; it represents one of the first systematic analyses of a pediatric transition unit in Latin America, providing valuable insights for similar contexts in low- and middle-income countries.
Future research should explore prospective, multicenter designs to validate functional cutoffs, strengthen caregiver training models, and evaluate the impact of structured transition bundles on readmissions, family well-being, and health care costs. As the population of medically complex children continues to grow, health systems must adapt to ensure that transition units are served as central to delivering components of integrated, equitable, and family-centered palliative care networks.33,34
Ultimately, this study reinforces the need to expand the concept of pediatric palliative care beyond end-of-life support. Early, proactive PPC integrated from diagnosis and embedded in hospital-to-home transition processes can improve quality of life, optimize health care utilization, and support families navigating the complex journey from hospital to home. Transition units, when equipped with standardized screening, functional assessment, and strong community linkages, hold the potential to close a persistent equity gap in global child health.
Conclusion
This study identified the specific palliative care needs of children with CCC during the hospital-to-home transition, emphasizing the critical role of early and integrated palliative care approaches within this context. The findings demonstrate that pediatric transition units can serve as strategic hubs for the early identification, comprehensive assessment, and coordinated management of palliative needs among medically complex children. Three main conclusions emerge.
First, conventional diagnostic classifications alone are insufficient to capture the multidimensional care demands of this population. The strong association between technology dependence, prolonged hospitalization, and palliative care eligibility underscores the need to combine functional assessment with WHO criteria to accurately identify children who would benefit from integrated palliative approaches, regardless of the controllability of their underlying condition.
Second, our findings highlight three priority domains for health-system improvement: institutional protocols that initiate discharge planning at admission and establish standardized pathways for children with CCC; mandatory involvement of palliative care teams for high-risk subgroups, particularly those with functional impairment or technological dependence, and robust post-discharge monitoring strategies, including telehealth follow-up and guaranteed access to medical equipment and supplies, to reduce readmissions and strengthen family support.
Third, the findings challenge the persistent misconception that palliative care is limited to end-of-life care contexts. More than half of the cohort met WHO Group 2 criteria, illustrating that early, concurrent palliative integration addressing symptom relief, psychosocial support, and sustained disease-directed therapy should be a routine part of care for children with chronic, life-limiting conditions.
Future research should examine determinants of hospital readmissions, prolonged hospitalization, and long-term functional outcomes, as well as the influence of family and community resources in post-discharge success.
In summary, a safe and compassionate transition from hospital to home requires more than the transfer of medical responsibility; it demands individualized therapeutic planning that integrates clinical complexity, family needs, and community resources. By embedding proactive palliative screening and multidisciplinary coordination throughout the transition process can transform hospital-to-home units can become a cornerstone of an equitable pediatric palliative care network, improving both quality of life and health care system performance for one of the most underserved pediatric populations.
Authors’ Contributions
Conceptualization: A.M.d.O.R., P.H.M.M., and D.H.C. Methodology: A.M.d.O.R., L.S.M., and P.H.M.M. Investigation: A.M.d.O.R. and P.H.M.M. Data curation: A.M.d.O.R. and L.S.M. Formal Analysis: A.M.d.O.R. and E.A.H. Writing — original draft: A.M.d.O.R., L.S.M., and P.H.M.M. Writing — review and editing: A.M.d.O.R., E.A.H., and D.H.C. Supervision: E.A.H., D.H.C., and PHMM.
Footnotes
Thank you to the Pediatric Staff at Perdizes Institute.
Author Disclosure Statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding Information
This research received no external funding.
Statements and Declarations
We believe that this declaration covers all relevant interests related to the research presented in our article. We assure you that we have taken utmost care to maintain the objectivity and scientific integrity of our study.
Ethical Approval and Informed Consent Statements
The study was approved by the institution’s Research Ethics Committee, under opinion No. 6,597,366/2024. As this was a retrospective study based on the analysis of secondary data extracted from electronic medical records, there was no direct contact with the participants. All ethical principles were strictly respected, including the confidentiality of information and the protection of sensitive patient data.
Data Availability Statement
In our research, we make the data available on an open-access basis, permitting reuse for other authors after contact with the primary team.
Supplemental Material
Abbreviations
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
