Abstract
Background:
Rectal administration of antipsychotics is an alternative approach for treating delirium in patients with advanced cancer who are unable to take oral medications.
Objectives:
Characterization of patients treated with quetiapine suppositories (QSP) and determination of its efficacy and safety.
Design:
Retrospective observational study.
Setting/Subjects:
Patients with advanced cancer who died in a Japanese palliative care unit (QSP group, n = 421; non-QSP group, n = 442).
Measurements:
Delirium severity scores (DSSs; sum of scores for items 2–4 of the Nursing Delirium Screening Scale), use of antipsychotics and benzodiazepines, adverse events (AEs) during the last week of life, and overall survival in both groups. DSSs and AEs following QSP administration in the QSP group.
Results:
The QSP group more frequently exhibited delirium on admission than the non-QSP group (22% vs. 15%, p = 0.001). Higher DSSs and frequent antipsychotics (90% vs. 30%, p < 0.001) and benzodiazepines (68% vs. 37%, p < 0.001) use were observed in the QSP group. The mean DSS was 1.8 (95% confidence interval: 1.6–1.9) before QSP administration, dropping to 0.61 (0.47–0.75, p < 0.001) 72 hours after administration. The incidence of hypotension (71% vs. 60%, p < 0.001) and apnea (25% vs. 17%, p = 0.004) was higher in the QSP group. There was no significant difference in median survival (QSP, 14 days; non-QSP, 13 days) after adjustment for patients’ background characteristics using propensity score matching.
Conclusion:
QSP administration alleviated severe delirium in our cohort, and associated AEs did not impact survival.
Introduction
Delirium is a frequent complication in terminal patients with cancer.1,2 Approximately 90% of patients in acute palliative care units (PCUs) experience delirium toward the end of life.3,4 Agitation due to delirium is distressing for patients, their families, and caregivers, and it may compromise their safety.5,6 Sleep disturbance is a common consequence of delirium and can exacerbate it. 7 Although delirium can be mitigated using nonpharmacological approaches by managing the precipitating factors, delirium at the end of life, known as terminal delirium, is difficult to improve. Hence, a major treatment goal is to alleviate the suffering of patients and their loved ones.8,9 Although the use of antipsychotics and benzodiazepines has been proposed as a potential treatment, particularly in hyperactive delirium, research in this area is limited.10–12
Only a few parenteral antipsychotics are available in Japan. Patients frequently have difficulty taking oral medications in their end of life; therefore, rectal administration is considered an alternative route. Rectal administration enables patients to avoid painful injections and is helpful in settings such as home palliative care in which medications cannot always be injected as quickly as necessary. Some Japanese institutions prepare a suppository version of drugs for patients unable to take oral forms.13,14
Quetiapine is a promising antipsychotic for delirium due to its low incidence of extrapyramidal side effects and short half-life, which facilitates dose adjustments.15–20 Its sedative effect might be desirable for terminally ill patients whose delirium is accompanied by sleep disturbances. 7 However, the use of quetiapine in terminal delirium is limited because it is manufactured only in tablet form.
In this context, rectal quetiapine administration is an attractive delirium treatment. A previous pharmacokinetic study revealed that rectal quetiapine produced an area under the drug plasma concentration–time curve of approximately 1.9 times greater than that generated by an equal dose of oral quetiapine, and a Japanese retrospective observational study reported its utility for delirium in terminal cancer.21,22 We used quetiapine suppositories (QSP) in our practice based on these studies; however, research is limited. The previous study was single-armed and did not sufficiently investigate the adverse events (AEs) and the survival effect of QSP.
Investigating clinical courses of QSP-treated patients will provide information on its utility. A comparison between patients treated with QSP and those treated conventionally (i.e., without QSP) will reveal the clinical course characteristics of QSP-treated patients. This study aimed to identify the characteristics of patients treated with QSP and to determine its efficacy and safety for delirium in patients with advanced cancer.
Materials and Methods
Study design, setting, and ethical considerations
This retrospective observational study was conducted in the PCU of Aiwa Hospital, Nagano, Japan. In Japan, the primary role of PCU is to provide symptom relief and end-of-life care for a wide range of patients with cancer, including those with high symptom burden, those who prefer inpatient end-of-life care, and those with home-based care difficulty. According to guidelines of the Japanese Society of Hospital Pharmacists, QSPs were compounded in the Aiwa Hospital pharmacy.23,24 The study protocol and the clinical use of QSP were approved by our institutional review board. All patients or their relatives provided written informed consent for QSP administration. Where possible, written informed consent for study participation was obtained from patients or their families upon admission. An opt-out option for inclusion in research was provided on our website.
Participants
The participants were patients with advanced cancer who died in our PCU following admission between October 2021 and September 2023. When a patient was admitted more than once during this period, only the terminal admission was analyzed. Patients who received QSP were defined as the QSP group, whereas those who did not were defined as the non-QSP group. QSP was largely administered for patients with delirium or at high risk of delirium to improve agitation and sleep disturbances. Therefore, the QSP group comprised patients who were apparently agitated and those who were not agitated but suffered from mild restlessness or confusion who needed nighttime rest. In general, the indication of QSP was dependent on the patients’ subjective distress. Delirium was diagnosed by the attending physicians based on the criteria of the Diagnostic and Statistical Manual of Mental Disorders (5th edition). 25 Patients with a high risk of delirium were identified by the attending physicians. The QSP dosage and the use of additional medications for delirium were at the discretion of the attending physicians. After QSP administration, oral medications were given as appropriate if the patient’s condition allowed. Antipsychotics for symptoms other than delirium were included in our analysis.
QSP preparation
QSP were prepared as previously reported. 22 Briefly, the suppository bases (Vosco® H-15 and Vosco® E-75; Maruishi Pharmaceutical, Osaka, Japan) were melted in a water bath. A pulverized quetiapine fumarate tablet was added to the melted base, and the mixture was stirred and poured into a suppository mold. Each suppository contained 25 mg of quetiapine fumarate, 0.5 g of Vosco H-15, and 0.5 g of Vosco E-75.
Outcomes
In both groups, the delirium severity, use of antipsychotics and benzodiazepines, and occurrence of AEs during the last week of life were evaluated. In the QSP group, changes in delirium severity and AEs following QSP administration were investigated. The overall survival of both groups was assessed.
Data collection
Patients’ medical charts were reviewed using prewritten data extraction sheets. Demographic and clinical data collected upon admission included age, sex, admission type, cancer type, Eastern Cooperative Oncology Group Performance Status (EGOG-PS), Karnofsky Performance Status (KPS), palliative performance scale (PPS), 26 items from the Palliative Prognostic Index (PPI) (delirium, dyspnea at rest, edema, and amount of oral intake), and PPI scores. 27
We reviewed each patient’s chart for the last week of life (days 1–7 before the date of death). Delirium severity, administration of antipsychotics and benzodiazepines, and AEs were recorded for each day. Delirium severity was evaluated using the Nursing Delirium Screening Scale (Nu-DESC). 28 Based on a previous method, 29 the sum of the scores for items 2–4 of the Nu-DESC, which evaluate symptoms of agitation, was used as the delirium severity score (DSS). Each item was scored from 0 to 2, so the range of possible scores was 0–6. DSSs were assessed by PCU nurses every 8 hours. The maximum DSSs on each day were analyzed.
The charts of patients in the QSP group were reviewed for the 72 hours following initial QSP administration. The QSP dose, concomitant use of other antipsychotics or benzodiazepines, DSSs, mortality, and possible AEs were recorded. We gathered DSSs at four timepoints: before QSP administration and 8, 24, and 72 hours after QSP administration.
Because patients with terminal cancer exhibit diverse symptoms, evaluating all possible AEs was difficult. Therefore, we included potentially fatal events, those established as relevant in a previous study, 22 and those listed in the U.S. Food and Drug Administration prescribing information for quetiapine tablets. 30 The resulting list of evaluated AEs included hypertension, hypotension, tachycardia, bradycardia, hypoxia, apnea, fever, depressed level of consciousness, syncope, seizure, gastrointestinal bleeding, hyperglycemia, hypoglycemia, and other remarkable events. AEs associated with vital signs and other remarkable events were assessed using the definitions in Table 1. The other AEs were evaluated using the Common Terminology Criteria for Adverse Events (CTCAE) v5.0. 31 Those classed as grade 3 or above were analyzed.
Definitions of Adverse Events

QSP, quetiapine suppository.
Statistical analysis
Differences between categorical variables were analyzed using chi-square or Fisher’s exact tests. Differences between continuous variables were analyzed using the Mann–Whitney U test. Changes in DSSs were evaluated using a generalized linear mixed-effects model with the Tukey test for multiple pairwise comparisons. We calculated the person-days incidence of AEs using the following formula: (sum of the number of days on which AEs occurred for all patients in the group)/(sum of the number of days evaluated for all patients in the group). Between-group comparisons were performed using the Wald test. Overall survival, defined as the number of days from admission to death, was calculated using the Kaplan–Meier method and compared using the log-rank test.
Propensity score matching adjusting for the effect of patients’ background characteristics at admission was used for the between-group comparison of AEs and prognosis. Propensity scores were calculated using a logistic regression model incorporating age, sex, KPS, the PPI items (delirium, dyspnea at rest, edema, and amount of oral intake), PPS scores, and cancer type. Nearest-neighbor matching was performed (caliper width: 0.20), and a propensity score-matched (PSM) cohort was created.
Two-sided p-values <0.05 were considered statistically significant. All statistical analyses were performed using R v4.3.1 (The R Foundation for Statistical Computing, Vienna, Austria) or EZR v1.63 (Saitama Medical Center, Jichi Medical University, Saitama, Japan), the graphical user interface for R. 32
Results
Patient characteristics
During the study period, 862 patients treated in our PCU were eligible (Fig. 1). They were divided into a QSP group (n = 421) and a non-QSP group (n = 441). On admission, patients in the QSP group had better PS (ECOG PS, KPS, and PPS) and oral intake; however, they exhibited delirium more frequently (22% vs. 15%, p = 0.001) (Table 2). The PSM cohort included 322 matched pairs. The c-statistic of the logistic regression model was 0.66 (95% confidence interval [CI]: 0.62–0.70). There were no significant between-group differences in baseline patient characteristics in the PSM cohort (Table 2).

Flow diagram of the patient selection and recruitment process in this study. QSP, quetiapine suppository; PCU, palliative care unit.
Patient Characteristics

ECOG PS, Eastern Cooperative Oncology Group Performance Status; KPS, Karnofsky Performance Status; n.a., not assessed; n.s., not significant; PPI, Palliative Prognostic Index; PPS, Palliative Performance Scale.
Course of delirium during the last week of life
The mean of the maximum DSSs during the last week of life was significantly higher in the QSP group than the non-QSP group (2.0 [95% CI: 1.9–2.2] vs. 0.86 [95% CI: 0.7–1.0]; p < 0.001). The maximum DSS on each day was also significantly higher in the QSP group than the non-QSP group (Fig. 2). The DSSs in the QSP group decreased gradually as death approached. Consequently, the mean of maximum DSSs on day 1 before the date of death was significantly lower than that on day 7 before the date of death (0.69 vs. 0.82, p < 0.01). Conversely, the DSSs in the non-QSP group increased gradually, and the mean of maximum DSSs on day 1 before the date of death was higher than that on day 7 before the date of death (0.31 vs. 0.20, p = 0.069).

Maximum delirium severity scores (DSSs) on each day during the last week of life in the entire cohort. Data points represent mean values of the maximum DSSs of patients on each day within the last week of life. Error bars indicate 95% confidence intervals. ***p < 0.001, **p < 0.01.
The frequency of antipsychotics (90% vs. 30%, p < 0.001) and benzodiazepines (68% vs. 37%, p < 0.001) administration was significantly higher in the QSP group than in the non-QSP group (Table 3). Supplementary Figure S1shows the types of antipsychotics and benzodiazepines administered. Supplementary Table S1shows the frequency of antipsychotic and benzodiazepine administration in the PSM cohort.
Use of Antipsychotics and Benzodiazepines and Adverse Events During the Last Week of Life for the Entire Cohort
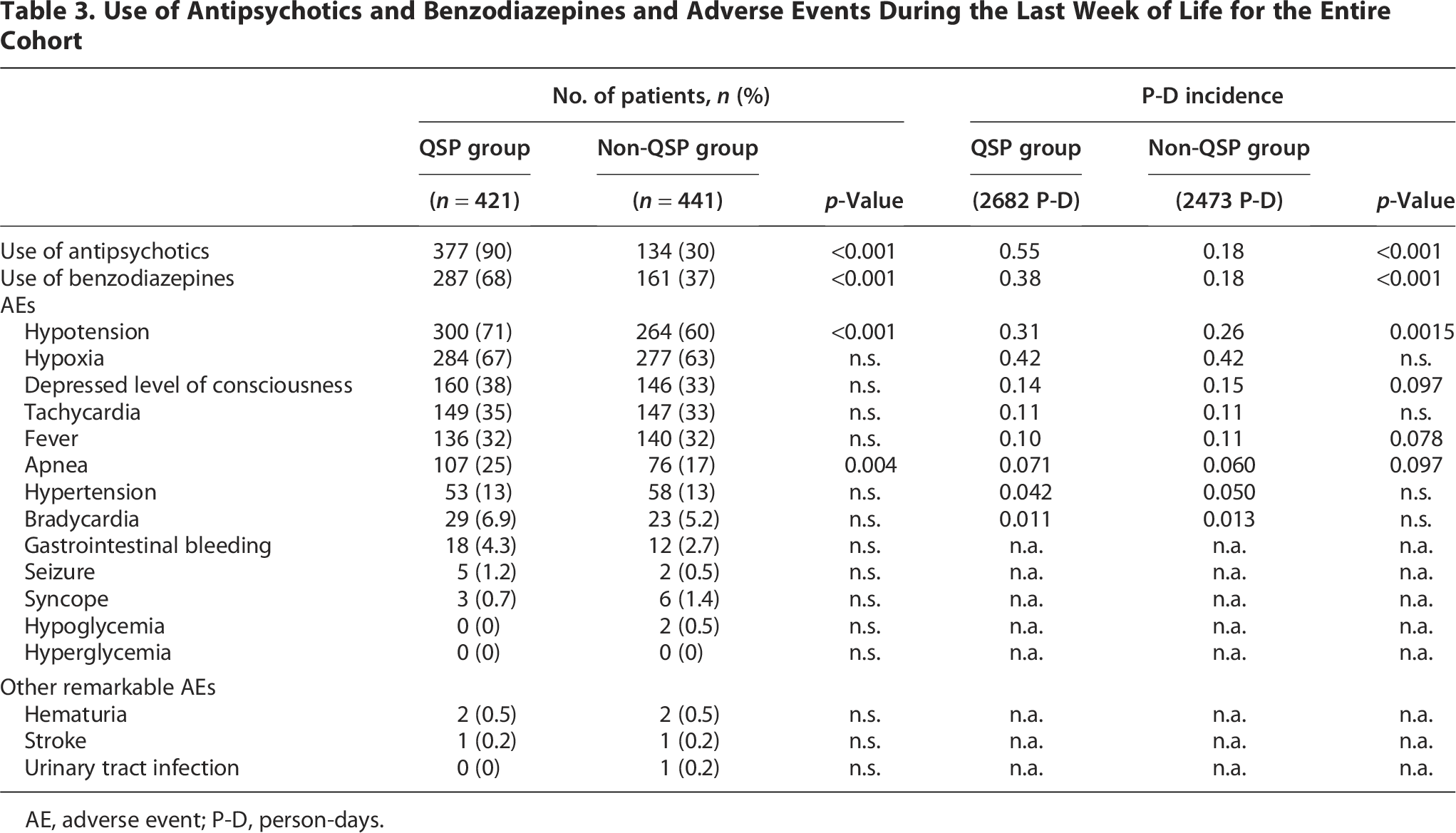
AE, adverse event; P-D, person-days.
Delirium severity following QSP administration
The median day of initial QSP administration was 6 days (range: 0–126) before the date of death. The median QSP dose administered within 72 hours after the initial dose was 75 mg (Table 4). Concomitant use of other antipsychotics and benzodiazepines occurred in 121 (43%) and 178 (64%) patients, respectively, within 72 hours of initial QSP administration. Within 72 hours of initial QSP administration, 108 patients (39%) did not receive additional medication for delirium other than QSP.
Details of Quetiapine Suppository Administration
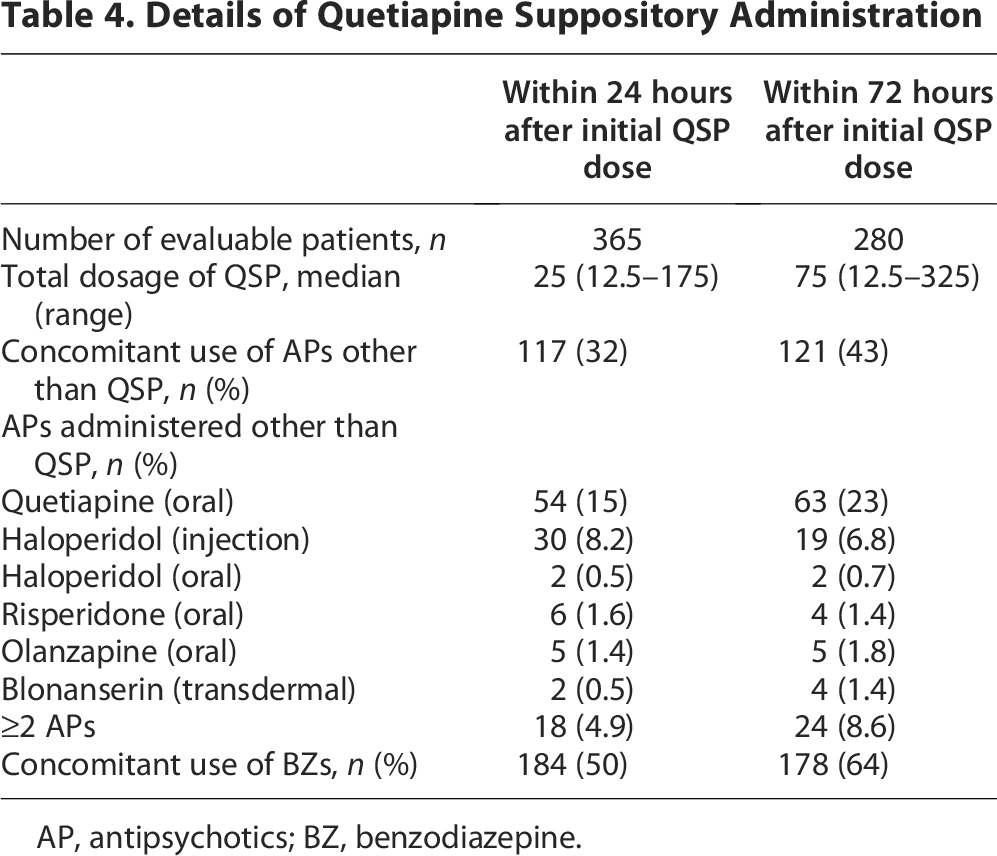
AP, antipsychotics; BZ, benzodiazepine.
Before QSP administration, the mean DSS of the 421 patients in the QSP group was 1.8 (95% CI: 1.6–1.9). After initial QSP administration, it decreased significantly to 0.71 (95% CI: 0.58–0.84, p < 0.001), 0.69 (95% CI: 0.57–0.81, p < 0.001), and 0.61 (95% CI: 0.47–0.75, p < 0.001) at 8, 24, and 72 hours, respectively (Fig. 3A). A separate analysis of those treated with QSP only for delirium determined a mean DSS of 1.4 (95% CI: 1.2–1.7) at baseline. After initial QSP administration, this was significantly reduced to 0.47 (95% CI: 0.26–0.69, p < 0.001), 0.49 (95% CI: 0.28–0.70, p < 0.001), and 0.25 (95% CI: 0.10–0.39, p < 0.001), at 8, 24, and 72 hours, respectively (Fig. 3B). Similarly, analysis of the patients who were first administered QSP during the last week of life revealed significantly decreased DSSs after QSP administration (data not shown).

Changes in delirium severity scores after QSP administration.
AEs following QSP administration
Death occurred in 56 (13%) and 141 (33%) patients within 24 and 72 hours of initial QSP administration, respectively. Among the 280 patients still alive 72 hours after initial QSP administration, frequently observed AEs were hypotension (25%), hypoxia (18%), depressed level of consciousness (14%), fever (13%), and tachycardia (11%) (Table 5). No severe mechanical AEs associated with rectal administration were observed.
Adverse Events Observed Within 72 Hours After Initial Quetiapine Suppository Administration

AEs during the last week of life
During the last week of life, frequently observed AEs included hypotension, hypoxia, a depressed level of consciousness, tachycardia, and fever (Table 3). The incidence of hypotension (71% vs. 60%, p < 0.001) and apnea (25% vs. 17%, p = 0.004) was significantly higher in the QSP group. There was no significant difference in the incidence of depressed levels of consciousness (38% vs. 33%, p = 0.15). Supplementary Table S1 presents AEs observed in the PSM cohort.
Survival
The median survival for all patients was 13 days. The median survival of the QSP group was significantly longer than that of the non-QSP group (16 vs. 11 days, p < 0.01) (Fig. 4A). This significant difference disappeared in the PSM cohort (14 and 13 days, p = 0.20) (Fig. 4B).

Overall survival in the QSP and non-QSP groups.
Discussion
We investigated the clinical course characteristics of QSP-treated patients. QSP-treated patients more frequently exhibited delirium on admission. Delirium severity of QSP-treated patients was higher during the last week of life, whereas it decreased as death approached. Initial QSP administration was observed on a median of 6 days before the date of death, and delirium severity decreased after QSP administration. Hypotension and apnea were more frequently observed during the last week of life. There were no significant between-group differences in overall survival when the effects of patients’ background characteristics were adjusted. Our results were almost consistent with those of the previous study. 22
Overall, the findings mentioned above indicated that patients in the QSP group experienced more severe agitation than those in the non-QSP group, and QSP was selectively administered for patients with comparably severe delirium and reduced its severity. However, several factors such as the retrospective nature of this study and the possible background between-group differences in delirium severity made it difficult to clearly declare the efficacy of QSP.
Although QSP monotherapy reduced delirium severity, 75% of QSP-treated patients received concomitant treatment with another antipsychotic or benzodiazepines. This suggested that QSP was most effective when combined with existing interventions. To investigate the safety of QSP in as many patients as possible, our QSP group included delirious patients without agitation, such as those with mild restlessness. Consequently, the mean DSSs were relatively low. To assess the efficacy of QSP more clearly for delirium-induced agitation, further research only on agitated delirious patients is necessary.
Although there was no significant survival inferiority in the QSP group, the incidence of AEs after QSP administration (52%, including grade 3 and above CTCAE) was higher than that previously reported (11%, including all grades of CTCAE). 22 This may be due to differences in the patient population. Although the previous study did not report the median survival of the patients, that of our patients was only 13 days; some of the AEs might be caused by the natural process of death. In addition, the more severe agitation in the QSP group might have led to the more frequent use of antipsychotics or benzodiazepines, which could have resulted in the higher incidence of hypotension and apnea. There is a similar observation that severe agitation was associated with the serious circulatory or respiratory suppression in the setting of palliative sedation therapy using midazolam. 33 We had a concern about an excessive sedative effect of QSP; however, the incidence of depressed level of consciousness in the last week of life was similar between groups. Nevertheless, it is still possible that QSP could have caused milder levels of disturbed consciousness than were assessed in this study. Further investigations on AEs are warranted.
This study had several limitations. First, we were unable to clearly determine the efficacy and safety of QSP as mentioned above. Contrary to our consideration, there is a possibility that the treatment strategy with QSP resulted in more severe agitation. Several previous studies have provided evidence against the use of antipsychotics for delirium.29,34,35 However, higher DSSs in the QSP group do not always indicate the treatment inferiority of QSP, because the goal of care for terminal delirium should be individually determined and aligned with the preferences and wishes of the patients and their families. The higher DSSs in the QSP group and the equal incidence of a depressed level of consciousness in both groups could be the result of balancing preferences reducing distress while maintaining consciousness even in severe delirium. Second, because many QSP-treated patients received additional delirium interventions, we were unable to determine which intervention contributed to the treatment effect. Considering these limitations, a randomized controlled trial is warranted. Third, although propensity score matching reduces biases associated with predefined covariates, unidentified prognostic factors may still have affected survival. Propensity score-unmatched patients were excluded from the analyses. Regarding the delirium assessment method, a fourth limitation may be our use of Nu-DESC in patients with terminal illness because a previous study has questioned its validity. 36 Nu-DESC enabled a simple and rapid assessment of delirium in daily practice; therefore, our PCU nurses routinely used it. Fundamentally, the true endpoint of delirium interventions is the reduction of distress experienced by patients and individuals around them, rather than merely a decrease in Nu-DESC scores. Similarly, true AEs are those that increase distress, not merely changes in vital signs or events defined by the CTCAE. Future studies should directly evaluate patients’ subjective comfort and distress or identify surrogate endpoints that more accurately reflect these outcomes. Finally, the patients in this study were older than those in previous studies.11,19,20 Caution should be exercised when generalizing our results to younger populations.
Conclusions
In conclusion, our findings suggest that rectal quetiapine was selectively administered for severe delirium in our cohort of patients with advanced cancer, and it reduced delirium severity without a clear impact on survival. Close monitoring for AEs, including hypotension and apnea, is necessary. Further studies are warranted to confirm the efficacy and safety of QSP.
Authors’ Contributions
D.K.: Methodology, formal analysis, visualization, investigation, and writing—original draft. Y.Y.: Conceptualization, methodology, and writing—review and editing. C.Y., K.T., M.H., M.W., K.K., and A.O.: Investigation. K.S.: Investigation and writing—review and editing. M.M. and M.K.: Conceptualization. Y.Y.: Supervision and project administration.
Footnotes
Acknowledgments
The authors thank the patients and their relatives for participating in this study.
Author Disclosure Statement
No competing financial interests exist.
Funding Information
No funds, grants, or other support was received for this article.
Supplemental Material
Abbreviations
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
