Abstract
Introduction:
Investigating sex-related neurophysiological differences across menstrual cycle phases is important. Sex steroid hormones associated with the menstrual cycle influence both brain function and brain structure; therefore, these factors should be considered in studies involving women participants.
Objective:
This study examined the impact of sex-related differences across menstrual cycle phases on stimulus recognition processes in the auditory and somatosensory modalities.
Methods:
Forty-four participants (25 men and 19 women) underwent assessment of auditory and somatosensory evoked potentials under control and simple reaction task conditions. Auditory evoked potentials were elicited using pure tones delivered bilaterally through headphones. Somatosensory evoked potentials were elicited via electrical stimulation of the right index finger. In both task conditions, the interstimulus interval was randomized between 5 and 15 seconds for both sensory modalities.
Results:
In the auditory modality, the N1 amplitude was significantly larger in women during the ovulatory phase under the control condition than in men. In contrast, in the somatosensory modality, the P100 amplitude was significantly larger in women during the menstrual phase under the simple reaction task condition than during the ovulatory phase.
Conclusion:
Sex-related differences modulated by menstrual cycle phases exert distinct effects on stimulus recognition processes in the auditory and somatosensory modalities.
Keywords
Introduction
Sex-related differences in neurophysiological responses are influenced by fluctuations in sex steroid hormones levels across menstrual cycle phases. 1 The levels of sex steroid hormones, including estradiol (E2) and progesterone (P4), fluctuate across the menstrual cycle. Franck et al. (2024) 2 have shown that the circulating E2 concentrations in men are half of those in women of reproductive age. In addition, the E2 levels were significantly higher during the preovulatory than during the menstruation and follicular phases. 3 High E2 levels affect brain function and structure, including peak gray matter volume, a decrease in cerebrospinal fluid volume, 4 an increase in hippocampal gray matter volume, 5 and alterations in neuronal excitatory and inhibitory functions.6,7 Another previous study has reported enhanced auditory functions during ovulation, such as higher distortion-product otoacoustic emission amplitudes, in the auditory modality. 8 Further, data on auditory brain stem response have shown that wave latencies and interpeak intervals during the peri-ovular phase were shorter than those during the luteal phase, 9 indicating that E2 enhances auditory function. 10 Therefore, E2 is major factor underlying sex-related differences. However, a substantial number of basic and clinical studies continue to disregard the effect of menstrual cycle phases in women and the exclusively men group. 11 In other words, it remains unclear whether the observed sex-related differences are due to hormone factor or to other factors.
This issue may help explain the inconsistencies observed in sex-related neurophysiological studies using evoked potentials (EPs). The EPs are bioelectric signals produced by the central nervous system when it is triggered by an explicit external event. 12 Moreover, these event-related potentials (ERPs) can be categorized based on stimulus processing. Short-latency components, which peak within the first 100 ms are referred to as exogenous as they are significantly dependent on the physical parameters of the stimulus.13,14 In contrast, long-latency components, which peak after 100 ms, are called endogenous as they rely on the evaluation and processing of a stimulus.13,14 Previous studies using long-latency auditory EPs (AEPs), such as that conducted by Berchicci et al. (2020), 15 have shown that women exhibit larger N1 and P2 amplitudes than men. Meanwhile, other studies have reported that the N1–P2 complex amplitude was larger in men than in women. 16 N1 and P2 components are commonly interpreted as an index of early sensory-perceptual processing and stimulus salience, reflecting the degree of cortical responsiveness to external stimuli. 17 However, it is unclear whether there is a difference in degree of cortical responsiveness to external stimuli between men and women regarding sex steroid hormonal effects. Importantly, these studies did not take menstrual cycle phases into account. Conversely, previous studies and other studies, which considered the effects of menstrual cycle,6,7,18 have reported that menstrual cycle may change the excitatory and inhibitory function of neurons in the somatosensory modalities using early somatosensory EPs (SEPs) components. However, the effects of sex-related differences on long-latency SEP components (stimulus recognition processes) across menstrual cycle remain unknown. Moreover, previous studies have investigated only a single modality. For these reasons, it is considered important to clarify sex-related neurophysiology differences and properties across multiple modalities and the menstrual cycle phases.
Therefore, this study aimed to investigate the effect of sex-related differences on stimulus recognition processes across menstrual cycle phases. For this purpose, a control condition, and simple reaction task (SRT) condition associated with stimulus recognition,19,20 were applied. Ramchurn et al. (2014) 21 have revealed that faster and slower RTs are related to fluctuations in attentional or executive control. Hence, RT reflects fluctuations in attentional allocation and stimulus processing efficiency. Thus, stimulus recognition processes can be evaluated using the methods used in this study. Moreover, the previous study has been reported that participants in older women with higher estradiol levels had better processing speed, sustained attention, and working memory. 22 In present study, to assess the effect of E2, we evaluated two phases: menstrual phase and ovulatory phase. 3 From the above, we hypothesized that the SRT condition was expected to show a difference of modulation of ERP and behavior compared with the control condition between men and women across the menstrual cycle in both modalities. Specifically, women in the ovulatory phase would exhibit shorter RTs and larger ERPs compared with the menstrual phase and with men, reflecting enhanced stimulus recognition.
Materials and Methods
Participants
In total, 44 (25 men and 19 women) right-handed individuals aged 18–25 years participated in the experiment. This sample size was larger than the minimum of 42 needed for 80% power and a significant level of 0.05 based on an effect size of 0.50. Inclusion criteria were as follows: (1) a regular menstrual cycle with a length of 25–38 days 23 ; (2) no use of hormone contraceptives or other hormone medications 3 ; and (3) no history of neurological or psychiatric disorders (including premenstrual dysphoric disorder). 3 All women participants underwent the experimental procedure during the menstrual phase (2–4 days after the onset of menstruation) and the ovulatory phase (2–4 days after the date of a positive ovulation test).18,24 On the other hand, men were not measured twice because the primary aim of repeated measurements was to assess intra-individual changes across hormonal phases, which are specific to the female menstrual cycle. The current study was conducted in accordance with the Declaration of Helsinki and was approved by the ethics committee of Niigata University of Health and Welfare, Niigata, Japan (19473-250206). Written informed consent was obtained from each participant after the experimental details had been explained.
Recording of the menstrual cycle
Using a basal body thermometer (Citizen Electronic Thermometer CTEB503L; Citizen Systems Co., Tokyo, Japan), the participants were asked to measure basal body temperature upon waking up every morning from approximately 1 month before the start of the experiment. To estimate the ovulation and E2 elevation days, they were also instructed to use an ovulation test kit (Doctor’s Choice One-Step Ovulation Test Clear: Beauty and Health Research, Torrance, CA, the USA) from the day after end of menstruation. Daily basal body temperature and menstrual period were recorded. The results obtained using the ovulation kit were recorded in the conditioning management system (ONE-TAP SPORTS; Euporia Co., Tokyo, Japan). 24
Measurement of sex steroid hormone levels
The method used to measure sex steroid hormone levels was based on previous studies.3,24 The E2 level was determined during each the menstrual and ovulatory phases. After the experiment was completed for each phase, saliva was collected to measure the E2 level using a saliva collection kit (SalivaBio A; Salimetrics, Carlsbad, CA, the USA). The following were prohibited before sample collection: (1) food intake within 60 minutes; (2) alcohol intake within 12 hours; (3) intake of sugary, acidic, or caffeinated beverages; (4) consumption of dairy products within 20 minutes; (5) tooth brushing within 45 minutes. The saliva of the participants was collected by ejecting it into a container (Cryovial, SAL) using a straw (Saliva Collection Aid, SAL). The collected saliva samples were immediately stored in a freezer at a temperature below –80°C. After all samples were collected, the E2 level was analyzed by Funakoshi Corporation (Tokyo, Japan). The E2 level was measured using high-sensitivity salivary 17β-estradiol enzyme immunoassay kit (Salimetrics). The samples were thawed at room temperature, mixed by vertexing, centrifuged at 1500 × g for 15 minutes, and analyzed using the enzyme-linked immunosorbent assay. The dilution factor was uniformly onefold (undiluted). Meanwhile, men participants underwent the same procedure as women participants, but measurements were conducted only on the day of the experiment.
Conditions
AEPs and SEPs were recorded under both the control and SRT conditions. In the control condition, the participants were advised to ignore the stimulus. In the SRT condition, the participants were instructed to press a button using the right index finger as fast as possible after they perceived the auditory or somatosensory stimulus. The participants completed both the control and SRT conditions, which were administered in random order. All female participants underwent ERP recordings in both menstrual phases. We hypothesized that any modulatory effect of estrogen would be reflected in changes in ERP measures. Furthermore, the two experimental sessions were separated by approximately 2 weeks (20.5 ± 15.7 days), which likely minimized potential adaptation effects.
Neurophysiological recording
Neuropack X1 (Nihon Kohden, Tokyo, Japan) was used to facilitate auditory and somatosensory stimulation. AEPs were elicited by pure tone delivered binaurally through headphones. In all participants, the intensity, frequency, and duration of the auditory stimulus were 60 dB, 1000 Hz, and 100 ms, respectively.25,26 SEPs were elicited by a constant current square wave pulse (pulse duration of 0.2 ms) delivered to the index finger of the right-handed using ring electrodes that were attached to the distal interphalangeal joint (anode) and the proximal interphalangeal joint (cathode). The intensity of the somatosensory stimulus was three times the sensory threshold and was not reported as pain and unpleasant sensation.25,27,28 In each stimulus condition, the inter-stimulus interval was randomized between 5 and 15 seconds, and the stimulus was presented 30 times in each modality.
Electroencephalogram recording
The SynAmps amplifier system and scan 4.3 software (Neuroscan, El Paso, TX, the USA) were used for electroencephalogram (EEG) acquisition. The EEG was recorded using five scalp electrodes placed at Fz, Cz, Pz, C3, and C4 according to the 10–20 system, because this waveform is shown to reach a maximum around the centrofrontal region for both auditory 20 and somatosensory 27 modalities. The left earlobe was used as a reference, and ground electrode was placed on Fp1. Electrode impedance was maintained below 5 kΩ. EEG signals were recorded with a notch filter (50 Hz) at a sampling rate of 1000 Hz. Trials with responses exceeding ± 100 μV were excluded from averaging according to our previous studies.26,29 The off-line band-pass filter was set at 0.5–70 Hz. 26 Stimuli were repeatedly presented until 30 artifact-free responses were obtained for each condition. The window of analysis was from 100 ms before (baseline) to 500 ms after stimulus onset.
EEG analyses
The auditory stimulation elicited the N1 and P2 components at all conditions in the auditory modality. The peak latency and the N1 and P2 amplitudes in all electrodes (Fz, Cz, Pz, C3, and C4) were measured at 80–150 and 180–250 ms, respectively. 30 The somatosensory stimulation elicited the P100 and N140 components at all conditions in the somatosensory modality. The peak latency and the P100 and N140 amplitudes in all electrodes (Fz, Cz, Pz, C3, and C4) were measured at 50–120 and 100–190 ms, respectively.26,27,31 We used the peak amplitude from baseline as the measure for analysis, specifically for evaluating the N1, P2, P100, and N140 components, respectively.
Data and statistical analysis
The neurophysiological data of the AEP and SEP amplitudes and behavioral data were obtained from each modality under both the control and SRT conditions. As the Shapiro–Wilk test indicated that ERP and behavior data deviated from a normal distribution, the Kruskal–Walli’s test was used. Because similar patterns were observed across all five electrodes for the auditory N1 and P2 components and the somatosensory P100 and N140 components, amplitudes were averaged across these electrodes for the analysis. 25 The data were used to compare the magnitude of the amplitude between the groups (women during the menstrual phase vs. women during the ovulation phase vs. men). Post-hoc tests (Bonferroni) were performed to assess pair-wise differences in peak amplitude of control-auditory N1 and SRT-somatosensory P100 across menstrual cycle phases. The significance level was set at 0.05 for all tests. Data were analyzed using the Graph Pad Prism 9.1.0 (221).
Results
N1 and P2 amplitudes under control and SRT conditions in the auditory modality
The peak amplitudes of auditory control and SRT N1 waveforms at the five electrode sites Fz, Cz, Pz, C3, and C4 are summarized in Table 1, while Figure 1a shows the grand-averaged waveforms at the Cz position under the control condition across the menstrual cycle in the auditory modality. The N1 amplitude was significantly larger in women during the ovulatory phase than in men under the control condition (H [2] = 3.552, p < 0.05, Fig. 1b). However, no significant differences were observed in the N1 amplitude under the SRT condition or the P2 amplitude under both the control and SRT conditions.

The Peak Amplitude ± SD of Auditory Control and SRT N1
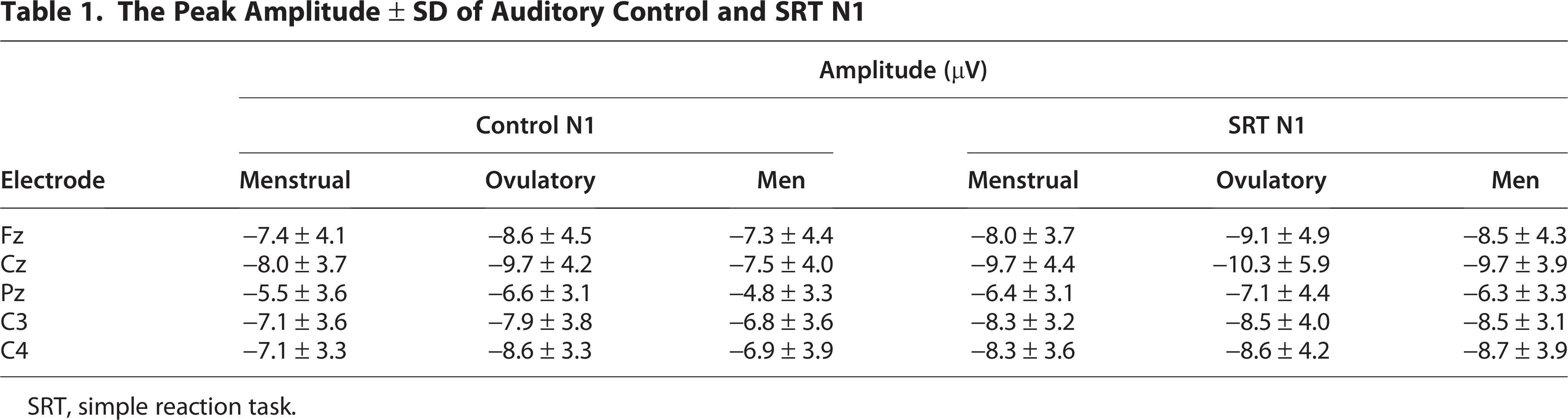
SRT, simple reaction task.
P100 and N140 amplitudes under the control and SRT conditions in the somatosensory modality
The peak amplitudes of somatosensory control and SRT P100 waveforms at the five electrode sites Fz, Cz, Pz, C3, C4 are summarized in Table 2, while Figure 2a shows the grand-averaged waveforms at the Cz position under the SRT condition across the menstrual cycle in the somatosensory modality. Under the SRT condition, the P100 amplitude was significantly larger in women during the menstrual phase than in women during the ovulatory phase (H [2] = 2.717, p < 0.05, Fig. 2b). However, no significant differences were observed in the P100 amplitude under the control condition or the N140 amplitude under both the control and SRT conditions.

The Peak Amplitude ± SD of Auditory Control and SRT P100
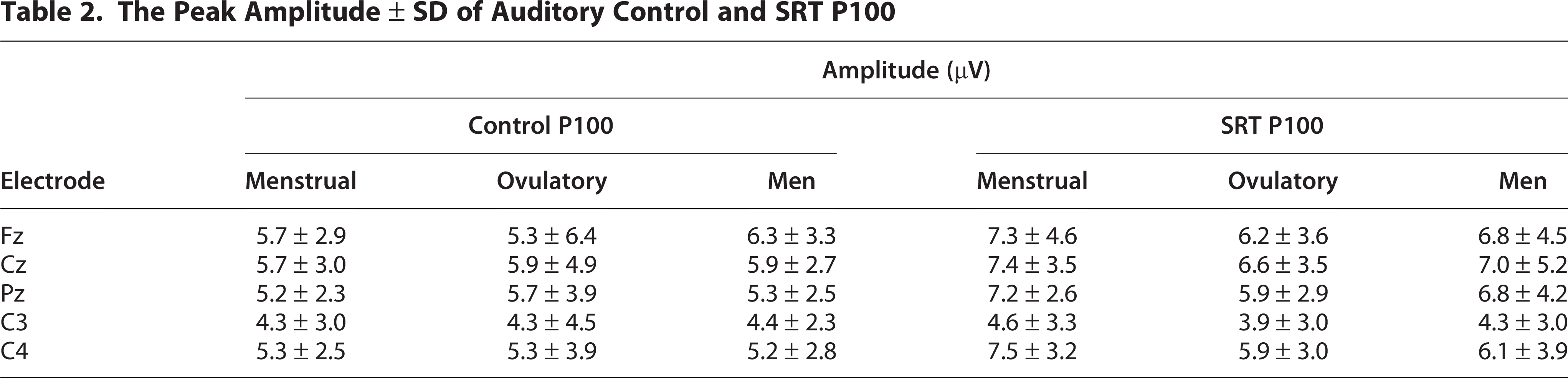
Salivary sex steroid hormone levels
Figure 3 shows the differences in the E2 levels between men and women across the menstrual cycle. The E2 level was significantly higher in women during the menstrual and ovulatory phases than in men, respectively (H [2] = –2.431, p < 0.05, H [2] = –2.752, p < 0.05). However, the E2 level did not significantly differ between women during the menstrual phase and those during the ovulation phase (H [2] = 0.301, p = 1.00).

Salivary sex steroid hormone levels between men and women across the menstrual and ovulatory phases. Created using GraphPad Prism version 9.1.0 (221).
SRT in the auditory and somatosensory modalities
Figure 4 shows the reaction time (RT) during the SRT between men and women across the menstrual and ovulatory phases. The RT data during the SRT did not differ between men and women across the menstrual and ovulatory phases, in both auditory and somatosensory modalities (H [2] = 0.456, p = 0.796, H [2] = 0.430, p = 0.806).

Comparison under the SRT in somatosensory and auditory stimulation between men and women across menstrual and ovulatory phases. Created using GraphPad Prism version 9.1.0 (221). SRT, simple reaction task.
Discussion
This study showed that sex-related differences across the menstrual cycle influenced late ERP components in a modality-specific manner. The following results were obtained: First, the N1 amplitude was larger under the control condition in women during the ovulatory phase than in men. Second, the P100 amplitude was larger under the SRT condition in women during the menstrual phase than in women during the ovulatory phase.
N1 amplitude under the control condition of the auditory modality
In the present study, the N1 amplitude was larger under the control condition in women during the ovulatory phase than in men, and the E2 level was significantly higher in women during the ovulatory phase than in men. In general, a previous study reported that the E2 levels were higher in the ovulatory phase than in other phases. 3 E2 is the most potent and abundant endogenous estrogen during the premenopausal period and is, thus, considered as reference estrogen. 10 Estrogen actions are mediated by three specific receptors: estrogen receptor alpha (ERα), estrogen receptor beta (ERβ), and G protein-coupled estrogen receptor (GPER, also called GPR30). 10 ERα may alter cochlear and vestibular sensory transduction, and ERβ may have a neuroprotective function in the inner ear. 32 Therefore, auditory function may be regulated by fluctuations in E2 levels across the menstrual cycle. In fact, a higher distortion-product otoacoustic emission amplitude during ovulation compared with any other phase of the menstrual cycle, 8 shorter wave latencies and interpeak of auditory brain stem response during the periovular phase than during the luteal phase, 9 and associations between fluctuating estrogen levels and intra-individual variability in auditory function. 10 In addition, Kim et al. (2002) 33 have shown that a lower serum estradiol level possibly impedes hearing sensitivity in postmenopausal women. Moreover, some studies on other sensory modalities that consider the menstrual cycle have also reported that the ovulatory phase involves the disinhibition of neuroactivities in the somatosensory modality6,18 and the visual modality. 7 Therefore, an increase in the N1 amplitude in women during the ovulatory phase could be attributed to enhanced auditory functions and neuronal excitability during this phase. Thus, the increased activities in auditory areas during the ovulatory phase may be influenced by sex steroid hormone levels, a finding supported by previous studies.
The results of previous studies using EPs have been contrasting. Some investigations have found that the EPs amplitudes were greater in women than in men across the AEPs, 15 SEPs, 34 and visual evoked potentials (VEPs).35,36 By contrast, a relevant study using the oddball task revealed that the standard-evoked N100 was higher in women. Meanwhile, the target-evoked N100 was higher in men. 37 Therefore, the authors concluded that a significant association exists between sex and stimulus type. Golgeli et al. (1998) 16 showed that the N1–P2 amplitude was higher in men participants than in women ones. Based on these findings, we hypothesized that the inconsistency among previous studies may be attributed to the lack of consideration of the effect of menstrual cycle. Therefore, the current study considered this factor. Our findings support those of previous reports showing larger N1 amplitude in women than in men under the control condition. Further, the E2 levels were also higher in women during the ovulatory phase than in men. Taken together, these results may contribute to an enhanced activity of auditory cortex in women during the ovulatory phase under the control condition.
P100 amplitude under the SRT condition in the somatosensory modality
In the current study, under the SRT condition, the P100 amplitude was larger in women during the menstrual phase than in women during the ovulatory phase. In contrast, there were no significant changes in the P100 amplitude under the control condition nor in the N140 amplitude under either the control or SRT condition. The response source of the P100 component was located on the upper bank of the Sylvian fissures, corresponding to the secondry somatosensory cortex (S2) in EEG and magnetoencephalography studies.38,39 In general, previous studies have defined S2 as a secondary or higher-level processing area 40 that is involved in attention, 41 sensorimotor integration,42,43 processing of noxious information,44,45 and detection of changes.46,47 Therefore, we observed an increase in the P100 amplitude during menstruation, and this finding may cautiously suggest a possible association with enhanced pain-related and change-detection responses, although the precise origin of this effect remains speculative.
Further, the increase in P100 amplitude under the SRT condition during the menstrual phase may be attributed to the severity of menstruation-related symptoms. Many women experience symptoms associated with menstruation both before and during menstruation, rather than only before or during menstruation. 48 Specially, the prevalence of dysmenorrhea of any severity varies between 16% and 91% among women of reproductive age, 49 and symptoms occur from the first day of menstruation in 31.6%–70.0% of the patients. 48 Recently, Ikarashi et al. (2024) 3 used the menstrual distress questionnaire (MDQ) to assess the severity of menstruation-related symptoms. Result showed that the total menstrual distress questionnaire score was significantly higher during the menstrual phase. Interestingly, Ronca et al. (2025) 50 used visual SRT tasks to examine attention function. They found that visual SRT performance was generally better during the menstrual phase and worse during the luteal phase, while self-reported mood, cognitive, and physical symptoms were all most severe during menstruation. This finding indicates that fluctuations in executive function across the menstrual cycle might be closely related to simple attentional control and processing speeds. Moreover, the menstrual phase is characterized by lower E2 levels. 3 However, present study showed that E2 level was no differences across the menstrual and ovulatory phases. Based on these findings, the increase in P100 amplitude during the SRT condition may be influenced by menstrual-related symptoms rather than the fluctuations of E2 level across the menstrual cycle.
As described above, the P100 component is considered to reflect mainly activity in the S2 and to be involved in change detection46,47 and sensorimotor integration42,43 Accordingly, discomfort associated with the menstrual phase may have movement-specifically enhanced S2 activity during motor preparation, whereas no such modulation was observed in the resting control condition. However, because no questionnaire-based assessments or other subjective measures about menstruation were conducted in the present study, this interpretation remains speculative. Finally, despite the increase in P100 amplitude under the SRT condition, no differences in reaction time were observed between sexes or across menstrual cycle phases, which may be attributable to a ceiling effect in the behavioral measure due to the simplicity of the SRT task.
Limitations
Several limitations exist in the present study. A primary limitation of the present study is that only five electrodes were used, which restricted spatial resolution. Therefore, the interpretation of the present findings regarding differences in P100 amplitude under the SRT condition during menstruation over S2 should be made with caution. Nevertheless, this change likely reflects modulation of cortical activations centered on the S2 region.38,39 Further studies should address the methodological limitations to enable spatially resolved analysis of sex-related differences in EEG components. Furthermore, although the results clearly demonstrated menstrual cycle or sex effects in both measures, their significance remains unclear due to the lack of detailed information on menstrual cycle and MDQ in the present study. Additionally, E2 levels were measured using salivary assays, which may be less accurate or sensitive than serum-based measurements. Nevertheless, salivary hormone measures have been widely adopted in previous studies and have yielded meaningful results, supporting the validity of this approach in research on hormone fluctuations. 24
Conclusion
The present study clarified that the menstrual cycle exerts differential effects on long-latency AEPs and SEPs during control and during the SRT task, respectively. Auditory modality may be more susceptible to the influence of estrogen, whereas its effects in somatosensory modality may be relatively limited.
Footnotes
Acknowledgments
The authors gratefully acknowledge support from the Japan Society for the Promotion of Science (JSPS) KAKENHI (grant number: 25K02998), the Japan Sports Agency Commissioned Project (2023–2025): Establishment of a Regional Sports Medicine and Science Support System, and the Japan Science and Technology Agency (JST) SPRING (grant number: JPMJSP2173).
Authors’ Contributions
Edited the article: S.A., T.M., Y.H., M.E., T.F., G.O., D.S., and K.Y. Conceived and designed the experiments: S.A., K.Y., and D.S. Performed the experiments: S.A., T.M., and Y.H. Analyzed the data: S.A., T.M., and K.Y. Contributed reagent/materials/analysis tools: S.A. and K.Y. Wrote the article: S.A.
Consent to Participate
Informed consent was obtained from all participants involved in the study.
Consent to Publish
The participants provided written informed consent for the use and publication of the data obtained in the experiments.
Data Availability Statement
The data presented in this study are available on request from the corresponding author.
Author Disclosure Statement
The authors declare that they have no conflict of interest.
Funding Information
This study was supported by the Japan Society for the Promotion of Science (JSPS) KAKENHI (grant number: 25K02998), the Japan Sports Agency Commissioned Project (2023–2025): Establishment of a Regional Sports Medicine and Science Support System, and the Japan Science and Technology Agency (JST) SPRING (grant number: JPMJSP2173).
